Abstract
Introduction:
The associations between the clinical characteristics of non–small cell lung cancer (NSCLC) and mutations in telomerase reverse transcriptase (TERT) gene remain unclear. In this study, we used next-generation sequencing (NGS) to investigate the incidence rate and clinical correlates of TERT mutations in patients with NSCLC.
Methods:
In total, 283 tumor samples from patients with NSCLC were tested using an NGS panel from September 2017 to May 2020. The genetic testing results and clinical data of all patients were collected.
Results:
TERT mutations were found in 30 patients, which were significantly associated with age, smoking history, sex, and metastasis (P < 0.05). Survival analyses showed that patients who carried TERT mutations had a poorer prognosis. Of the 30 TERT-mutation carriers, 17 harbored epidermal growth factor receptor (EGFR) mutations, which were significantly associated with sex, histopathology type, and metastasis (P < 0.05; overall survival [OS], 21 months; 95% confidence interval [CI], 8.153-33.847 months). Three TERT mutation patients harbored Kirsten rat sarcoma virus (KRAS) mutations, which were significantly associated with metastasis risk (P < 0.05), KRAS mutations carriers had a worse prognosis, with an OS of 10 months (95% CI, 8.153-33.847 months). Multivariate Cox regression analyses showed that age, cancer stage, and TERT mutation carrier status were independent risk factors for NSCLC, and the TERT mutation was 2.731 times higher than that without TERT mutation (95% CI, 1.689-4.418, P < 0.001).
Conclusions:
TERT mutations were present in 11% of patients with NSCLC. TERT mutations were associated with age, smoking history, sex, and distant metastasis. Co-mutations in TERT and EGFR/KRAS indicated a poor prognosis. The co-mutations of TERT and EGFR differed according to sex, histopathology type, and metastasis, whereas TERT and KRAS co-mutations were only associated with patient metastasis. Age, cancer stage, and TERT mutation carrier status were independent risk factors for poor prognosis in patients with NSCLC.
Introduction
Lung cancer is the second most common cause of morbidity worldwide and the leading cause of cancer-related death worldwide for both sexes. 1 According to the histological classification, 85% of lung cancers are non–small cell lung cancers (NSCLCs). 2
It is estimated that up to 69% of patients with advanced NSCLC have treatable mutations in multiple genes, including epidermal growth factor receptor (EGFR), Kirsten rat sarcoma virus (KRAS), anaplastic lymphoma kinase (ALK), mesenchymal-epithelial transition factor receptor (MET), c-ROS oncogene 1 (ROS1), human epidermal growth factor receptor (HER2), and V-RAF mouse sarcoma oncogene homolog B1 (BRAF). 3 Telomerase reverse transcriptase (TERT) mutations have been detected in various tumors, including lung cancer. However, mutation rates vary among studies.4-6
Telomerase is a key enzyme for cell survival, and the basis of tumorigenesis is the infinite proliferation of malignant cells, which in most cases is achieved by activating telomerase. 7 Inhibition and/or shortening of telomerase in human cells is considered a natural evolutionary strategy against cancer, 8 and telomerase activation and telomere damage are related to the occurrence of human lung cancer.9,10
TERT is the main factor that controls telomerase activity. 11 TERT point mutation, amplification, rearrangement, and epigenetic modification through the TERT promoter can regulate human TERT (hTERT) expression level, activate telomerase, maintain telomere length, and promote tumor growth. 12
Studies have found that TERT co-mutation with other genes mutations can increase the degree of malignancy of tumors, consistent with an anticipated poor prognosis. 13 Co-occurring genomic alterations have emerged as core molecular and clinical heterogeneity determinants in oncogene-driven lung cancer subtypes. 14 In vitro studies have suggested that EGF upregulates the expression of RFPL3 and hTERT proteins in NSCLC cells through the MEK pathway, which promotes cell proliferation and inhibits apoptosis. 15 KRAS gene mutations can increase telomerase activity and TERT expression through RAS/MEK signaling, thus increasing NSCLC aggressiveness. 16 However, the prognostic significance of TERT co-occurrence with EGFR/KRAS in NSCLC is unknown and warrants further study.
Our study investigated the presence of TERT mutations in NSCLC tissues and delineated correlations with other clinical parameters. The clinical importance of TERT co-mutations with EGFR or KRAS in patients with NSCLC was also examined.
Materials and Methods
Patients and samples source
From September 2017 to May 2020, 283 samples from patients with NSCLC were collected from the First Affiliated Hospital of Yangtze University (Jingzhou, China). The First Affiliated Hospital of Yangtze University Ethics Committee approved the study (IRB: KY202204), and all the participants provided written informed consent. Clinicopathologic features and prognostic information were collected. Clinical characteristics are shown in Table 1. The deadline for follow-up was November 1, 2021.
Demographics and clinicopathologic features of 283 patients with non–small cell lung cancer.

Abbreviations: ADC, adenocarcinoma; SQ, squamous carcinoma; TERT, telomerase reverse transcriptase.
Next-generation sequencing
Next-generation sequencing detected the hot spot region of 65 genes, including the whole exon region of 10 genes and 14 fusion genes, which could detect single nucleotide polymorphism (SNP), insertion or deletion (InDels), fusions, and copy number variations in tissue samples: Genomic DNA from formalin-fixed paraffin-embedded samples was extracted using the GeneRead DNA FFPE Kit (Qiagen, Hilden, Germany). Nucleic acid fragmentation and terminal repair results in a specific fragment size.
Library construction was performed using the SureSelect XT Low Input kit (Agilent Technologies, Santa Clara, USA), according to the manufacturer’s protocol. The captured DNA fragments were amplified with 12 to 13 cycles of PCR using 1 × KAPA HiFi Hot Start Ready Mix. In addition, Agilent Bioanalyzer (Agilent Technologies, Santa Clara, USA) quantified the library fragment size. NGS libraries were sequenced on Illumina NextSeq CN500 platform (Illumina Inc., San Diego, CA, USA). 17
Raw image data obtained by high-throughput sequencing were transformed into raw sequences by base calling. Then, clean data were obtained by filtering adapter, low-quality reads, and reads with length < 36 bp and aligned to the reference genome (UCSC HG19) using Burrows Wheeler Aligner (BWA) to obtain BAM initial alignment result. The Genome Analysis Toolkit (GATK) containing Picard was used for local realignment, base quality recalibration, etc. Thus, the result in the BAM format and quality statistics, such as mapped reads, mean mapping quality, and mean coverage were obtained. Finally, the HaplotypeCaller module of GATK was used to detect SNP/InDels.
Statistical analyses
The chi-square test (sample size ⩾ 40, theoretical frequency < 5, Fisher’s exact test) was used to analyze the association between mutations and clinical and biological characteristics. Overall survival (OS) was defined as the time from diagnosis to death or the last follow-up date for living patients. The Kaplan-Meier method was used to draw the survival curve, and the difference in survival rate was tested and analyzed using the log-rank test for statistical significance. The Cox proportional risk regression model was constructed using patients’ pathological characteristics and gene mutations for multi-factor analysis. In univariate Cox regression analysis, variable screening criterion was set as A = 0.1, and variables with P < 0.1 were included in multivariate Cox regression analysis. Multivariate Cox regression analysis (P < 0.05) was set as an independent prognostic factor. SPSS 25.0 software (IBM Corp., Chicago, IL, USA) was used for data analysis.
Results
Patient characteristics
The sample included 283 patients with NSCLC (average age, 62 years), the majority (n = 164;58%) of whom were male and nonsmokers (n = 178, 63%). Among 283 patients, 262 (93%) were diagnosed with adenocarcinoma and 21 (7%) were diagnosed with squamous cell carcinoma. Regarding tumor staging, the numbers of patients with cancer stages I to IV was 27 (9.5%), 41 (14.4%), 43 (15.2%), and 172 (60.7%), respectively (Table 1).
TERT mutation screening in patients with NSCLC
Using NGS, we found the TERT mutation rate of 11% (30/283), the KRAS mutation rate of 11% (27/283), and the EGFR mutation rate of 55% (156/283). We identified the TERT gene mutation point using the sequencing data of these patients (Table 2).
TERT gene mutation type and composition ratio.

Abbreviations: TERT, telomerase reverse transcriptase.
The clinicopathologic characteristics of patients with TERT promoter mutations are presented in Table 1. TERT mutation carrier status was associated with sex (P < 0.0001), age (P < 0.0001), smoking history (P < 0.0001), and metastasis (P < 0.0001).
TERT and EGFR or KRAS co-mutation genomic alterations in NSCLC
We found that 17 of the patients with TERT mutations had EGFR mutations, and 3 had KRAS mutations (Table 3). TERT and EGFR co-mutations status was significantly correlated with sex (P = .0105), histopathology type (P = .009), and metastasis (P = .006). TERT and KRAS co-mutations were associated with metastasis (P < .0001).
Clinical baseline table of co-mutations of TERT and EGFR or KRAS.

Abbreviations: ADC, adenocarcinoma; EGFR, Epidermal growth factor receptor; KRAS, Kirsten rat sarcoma virus; SQ, squamous carcinoma; TERT, Telomerase Reverse Transcriptase.
Clinical outcomes
The follow-up period ended on November 1, 2021. From a total of 283 patients, 161 had died at study end-point. The median survival times of the TERT-mutant vs TERT wild-type group was 14 vs 30 months (Table 4). There was a significant difference in OS between TERT mutation carriers with vs without co-mutations (P = 0.0061, Figure 1A).
The association between TERT mutation carrier status and overall survival.

Abbreviations: CI, confidence interval; OS, overall survival; TERT, telomerase reverse transcriptase; SE, standard error.

Overall survival for 283 patients with non–small cell lung cancer. (A) Kaplan-Meier (K-M) curve of TERT mutant and wild-type OS shows their association (P = 0.0061). (B) K-M curves of OS under different mutation states of TERT and EGFR. There were significant differences in OS (P = 0.0056) among the four groups in NSCLC. (C) K-M curves of OS under different mutation states of TERT and KRAS shows significant differences in OS (P = 0.0012) among the four groups in NSCLC.
The median OS was 21 months in the TERT and EGFR co-mutated griou; 10 months in the TERT mutated and EGFR wild-type group; 33 months in the TERT wild-type and EGFR mutated group; and 27 months in both the TERT and EGFR wild-type group (Table 5). There was significant difference in OS among the four groups, suggestting that different groups had different effects on prognosis (P = 0.0056, Figure 1B).
Overall survival of TERT and EGFR in different alteration status.

Abbreviations: 1, TERT and EGFR co-mutation; 2, TERT mutation and EGFR wild-type; 3, TERT wild-type and EGFR mutation; 4, TERT wild-type and EGFR wild-type; CI, confidence interval; EGFR, epidermal growth factor receptor; OS, overall survival; SE, standard error; TERT, telomerase reverse transcriptase.
The median OS was 10 months in the co-mutated TERT and KRAS group, 15 months in the wild-type TERT and mutant KRAS group, 15 months in the wild-type TERT and KRAS mutation group, and 30 months in the wild-type TERT and KRAS group (Table 6). A significant difference was found in the OS among the four groups (P = 0.012, Figure 1C). The patients with TERT and KRAS co-mutations have the worst survival.
Overall survival of TERT and KRAS in different alteration statuses.
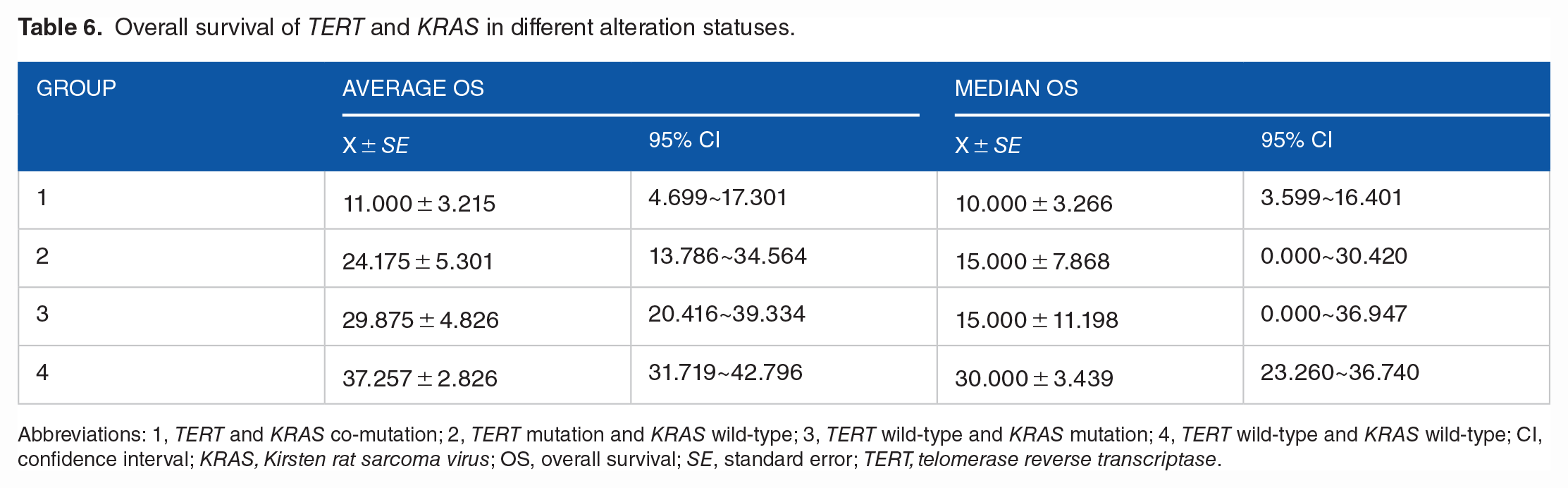
Abbreviations: 1, TERT and KRAS co-mutation; 2, TERT mutation and KRAS wild-type; 3, TERT wild-type and KRAS mutation; 4, TERT wild-type and KRAS wild-type; CI, confidence interval; KRAS, Kirsten rat sarcoma virus; OS, overall survival; SE, standard error; TERT, telomerase reverse transcriptase.
Overall survival predictors
The univariate and multivariate Cox regression analyses showed that TERT mutation carrier status was an independent prognostic marker in patients with NSCLC (Table 7). The results showed that age, metastasis, cancer stage, TERT mutation, and TERT and KRAS co-mutations were associated with worse prognosis. Meanwhile, age, stage, and TERT mutation exhibited potential as independent risk factors for OS.
Univariate and multivariate Cox regression analysis in NSCLC patients.

Abbreviations: CI, confidence interval; EGFR, Epidermal growth factor receptor; HR, hazard ratio; KRAS, Kirsten rat sarcoma virus; NSCLC, non–small cell lung cancer; TERT, Telomerase Reverse Transcriptase.
Discussion
NSCLC is a common and highly heterogeneous malignancy, accounting for approximately 85% of all lung cancer cases, with a very low 5-year survival rate. 18 However, targeted therapy has improved 5-year survival. 19 NGS can effectively identify the status of various important lung cancer-related gene changes. 20
TERT mutations are considered a low mutation type in NSCLC. 21 In a cohort study of 103 patients with NSCLC, using DNA gene sequencing, Yuan et al 22 suggested that the mutation rate of the TERT region was approximately 5.8%. Whereas Jung et al 23 reported showed that the TERT promoter mutation rate was 2.2% (4/188). However, our study showed that the TERT mutation rate in patients with NSCLC was 11%. Although the mutation rate of TERT was slightly higher than that reported in previous studies, the mutation rate was still low.
Our study also showed a correlation between TERT gene mutation carrier status, smoking history, age, sex, and distant metastasis. This result is consistent with those of a previous meta-analysis which confirmed that TERT gene mutations were associated with age, sex, and distant metastasis. 23
The median survival time of the TERT-mutated group was 14 months, and that of the TERT wild-type group was 30 months. Similarly, there was a significant difference in OS between TERT mutation carriers with vs without co-mutations (P = 0.0061). Our results are consistent with those of the study performed by Jung et al. 23
In an analysis of the clinicopathologic and genetic prognostic markers, we found that age, stage, and TERT mutation carrier status were independent risk factors for poor OS in patients with NSCLC. Among them, there were 2.731 times as many TERT mutation cases as there were TERT wild-type cases (95% CI, 1.689-4.418, <0.001). The median survival time of the TERT-mutant group was 14 months, and that of the TERT wild-type group was 30 months.
In the context of cancer, TERT mutations can co-occur with other risk-associated mutations in genes, such as BRAF, EGFR, KRAS, and IDH in cancer.24-26 TERT mutations alone or in combination with other genetic mutations can be used to identify different tumor types and predict prognosis and outcome. In thyroid cancer, especially papillary thyroid cancer, TERT and BRAF co-mutation suggest a poor prognosis. 27 In high-grade gliomas, TERT and IDH co-mutation was associated with worse outcomes than IDH only. 25 KRAS and EGFR mutations are common in NSCLC. In our study, an analysis of the clinical correlates of TERT and EGFR co-mutation showed that they were related to the patient’s sex, histopathology type, and metastasis. Whereas TERT and KRAS co-mutations were found to be correlated with metastasis alone.
We further analyzed the effect of TERT and EGFR co-mutations on the prognosis of patients with NSCLC. The results showed that the TERT-mutated and EGFR wild-type groups had the shortest OS, followed by the co-mutation group. In NSCLC, KRAS mutations increase TERT expression and telomerase activity through RAS/MEK signaling, hence contributing to the expression of more aggressive phenotypes of NSCLC. 16 Our study showed that median OS was significantly different among the TERT and KRAS co-mutation group; TERT mutation and KRAS wild-type group; TERT wild-type and KRAS mutation group; and TERT and KRAS wild-type group (P = 0.012). These results indicated that different groups had different prognosis. Moreover, the co-mutation of TERT and KRAS resulted in worse survival outcomes.
Our study has several limitations. First, the database had limitations in terms of investigating other potential sources of bias. TERT, EGFR, and KRAS mutation rates were all slightly higher in our study than in others. KRAS mutations are a major cause of NSCLC, accounting for over 30% of cases in Western countries, but less than 10% in Asia. 28 The KRAS mutation rate in patients with NSCLC was 11% in our study. The frequency of EGFR mutations in NSCLC in the People’s Republic of China ranges from 28.2% to 43%. 29 In our study, the EGFR mutation rate was 55%. Difference in TERT mutation incidence between our study and others may be related to the choice of sample size and region. Due to financial constraints, not all patients attending the clinic underwent genetic testing. Second, our patients were mainly recruited from rural areas and had limited access to treatment, which affected their OS. We found worse survival in the TERT and EGFR co-mutation group than in the EGFR mutation and TERT wild-type group. We hypothesized that this might be related to the use of tyrosine kinase inhibitor preparations in most EGFR mutation-positive patients; however, because not all patients were treated in the same way, we did not include the treatment of patients in the study, which may have influenced the results. Therefore, prospective studies are needed to confirm this finding.
Conclusions
In conclusion, we have illustrated the incidence and clinicopathologic correlates of TERT mutations in patients with NSCLC. We also demonstrated that age, stage, and TERT mutations were independent risk factors for poor prognosis in patients with NSCLC. This result provides a reference for the clinical evaluation of prognosis in patients with NSCLC.
Research Data
sj-xlsx-1-onc-10.1177_11795549221140781 – Supplemental material for TERT Mutations in Non–Small Cell Lung Cancer: Clinicopathologic Features and Prognostic Implications
sj-xlsx-1-onc-10.1177_11795549221140781 for TERT Mutations in Non–Small Cell Lung Cancer: Clinicopathologic Features and Prognostic Implications by Liu Yang, Meng Wang, Na Li, Lu-Da Yan, Wen Zhou, Zhi-Qiong Yu, Xiao-Chun Peng, Jun Cai and Yong-Hua Yang in Clinical Medicine Insights: Oncology
Footnotes
Acknowledgements
We thank all the patients and investigators involved in this study.
Funding:
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by Hubei Province Health and Family Planning Scientific Research Project (grant no.WJ2015MB212, WJ2018H201) to JC, and the National innovation and entrepreneurship training program for College Students (grant no. 202010489017) to XCP.
Declaration of Conflicting Interests:
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Author Contributions
XCP, YHY, and JC contributed to this paper with the design and reviewed the submitted version of the manuscript. YHY, LY, WZ, and LDY worked on the acquisition, analysis and interpretation of the data. XCP, JC, YHY, NL, and LY worked on the statistical analysis. LY, MW, and NL, YZQ contributed to tables and figures. LY, JC, YHY, and XCP wrote the manuscript. XCP and JC acquired funding. All authors contributed to the article and approved the submitted version.
Ethics Approval and Consent to Participate
The study was approved by the Ethics Committee of the First Affiliated Hospital of Yangtze University and complied with Good Clinical Practices (IRB: KY202204). The patients/participants provided their written informed consent to participate in this study.
Availability of Data and Materials
The original data sheet of our experiment results can be found in the supporting information.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
