Abstract
Background:
With the development and wide application of next-generation sequencing (NGS), multiple-gene mutations related to lung cancer are detected using this technology. Notably, even multigene concomitant mutations (co-mutations), which occur at a relatively low incidence, can be detected more effectively using NGS. It is well-known that the percentages of non-small cell lung cancer (NSCLC) in the elderly lung cancer population are also gradually increasing, while its prognosis is hard and the quality of long-term survival is poor. This study aimed at investigating the common clinicopathologic features of multigene co-mutations for better evaluating the prognosis of elderly NSCLC patients.
Methods:
A total of 464 NSCLC patients were divided into 3 groups according to the types of gene mutation, whose clinical data were retrospectively analyzed.
Results:
In total, 38.36% (178/464) of NSCLC patients were in the nonmutation group, 50% in the single-gene mutation group, and 11.64% in the multigene co-mutation group. Nonmutation, single-gene mutation, and co-mutation groups were all prone to occur in male adenocarcinoma patients (P < .05). EGFR gene mutation rates were the highest in the single-gene mutation and co-mutation groups (54.31% and 24.35%). In the co-mutation group, the incidence of EGFR/PIK3CA, LK/KRAS, and EGFR/MET co-mutations was the highest (16.67%, 11.11%, and 7.41%). ALK/HER2/MET, EGFR/HER2/MET, and EGFR/HER2/MET/ROS1 multiple-gene co-mutations were, respectively, found in 1 case, and the multigene co-mutation patients commonly had a worse median progression-free survival (PFS) than that of single-gene mutation (7.13 vs 12.34 months, P = .013).
Conclusion:
With the application of NGS, the detectable rates of gene co-mutation are increasingly high in elderly patients with NSCLC, which mainly occurs in male adenocarcinoma patients commonly with poor PFS. It will be critically necessary to conduct multigene detections by NGS for directing targeted therapy of elderly NSCLC patients.
Keywords
Introduction
With aged tendency of population increasing year by year, the incidence of lung cancer is also increasingly higher in the elderly population. 1 Especially NSCLC, it accounts for 80% to 85% in lung cancer patients in China. 2 Most diagnosed NSCLC patients miss the optimal timing for treatment due to concealment of this disease and lacking typical clinical features and effective diagnosis. 2 In recent years, the strategies of diagnosis and therapy for NSCLC have obtained significant improvements with the progression of tumor pathogenesis studies and translational medicines. A recent study showed that the clinical application of tyrosine kinase inhibitors (TKIs) improved the PFS and overall survival of NSCLC patients. Therein, gefitinib, erlotinib, and afatinib are the first-line therapeutic options for EGFR mutation-positive NSCLC patients. 3 However, in clinical work, we found that NSCLC patients showed obvious individual differences in the benefits of EGFR-TKIs, and the degree of drug resistance was also different.
Basic studies confirmed that NSCLC was mainly caused by single-gene mutation, 4 while a part of NSCLC patients with multigene co-mutations is hard to be diagnosed. With the development and application of NGS technology, multigene co-mutations also excited in NSCLC patients, such as EGFR/TP53, EGFR/ALK, EGFR/KRAS, and EGFR/PIK3CA. However, these co-mutations may only account for a unique subset of NSCLC patients associated with prognosis.5-7 In addition, some studies showed that multigene co-mutations were associated with EGFR-TKI resistance. 8 According to the specific conditions of co-mutations, combined targeted therapy may become a new therapeutic way to treat NSCLC patients with multigene co-mutations. The National Comprehensive Cancer Network recommended routine detection of EGFR, KRAS, ALK, BRAF, ROS1, MET, RET, ERBB2, and other genes in NSCLC patients. This study aimed at investigating the incidence of typical multigene co-mutations, and analyzing the impacts of these co-mutations on the prognosis of elderly NSCLC patients, and providing theoretical directions for individualized and precise treatments of elderly NSCLC patients.
Data and Methods
The general information
A total of 464 NSCLC patients who were admitted to the Department of Geriatric Respiratory and Critical Care Medicine at the First Affiliated Hospital of Anhui Medical University between May 2016 and April 2021 were collected into this clinical retrospective study. All enrolled patients accepted NGS detection using tumor tissues. Specific detected genes included ALK, BRAF, EGFR, HER2/ERBB2, KRAS, MET, PIK3CA, RET, and ROS1 gene. According to the results of gene detection, the patients were divided into 3 groups: 178 nonmutation patients, 232 patients with single-gene mutations, and 54 patients with multigene co-mutations. Inclusion criteria were as follows:
(a) aged 60 years or older, and
(b) having been diagnosed with NSCLC by histology or cytology.
In addition, the exclusion criteria were also shown in below:
(a) other malignant tumors;
(b) severe disease of the heart, brain, kidney, or other organs;
(c) incomplete records of clinical and pathological data; and
(d) patients with non-neoplastic death.
The authors accounted for all aspects of the work, and ensure that questions related to the accuracy or integrity of all data are appropriately investigated and resolved. The study was proceeded in accordance with the Declaration of Helsinki (as revised in 2013). We only used the de-identified data retrieved from electronic medical records which were used in clinical retrospective studies, not necessary for patient’s consents and signatures. The retrospective study was approved by the Ethics Committee of the First Affiliated Hospital of Anhui Medical University.
NGS detection
Lung tissues of NSCLC patients were obtained by puncture lung biopsies according to standard clinical procedures. In addition, NGS detection samples were collected using a DNA extraction kit (Tiangen Biochemical Technology Co., Ltd., Beijing, China). DNA collection process was carried out strictly according to the manufacturer’s instructions from Tianjin Novogene Bioinformatic Technology Co, Ltd’s Gene Library Building kit (Tianjin, China) for targeted drug use in solid tumors (for scientific research only). The detection sites were sequenced using an on-board high-throughput sequencing kit (Illumina, San Diego, CA, USA). In the detection reports, mutation abundance was the variant allele frequencies (VAF), which can be calculated using the following formula:
Taking the fields in the VAF files as an example, where DP represents total depth and AD represents allele depth.
Follow-up aiming at enrolled patients
The patients with single-gene mutations and multigene co-mutations who received TKIs’ treatment were followed up until April 1, 2021, either through consultation with the electronic medical record system or by telephone return visiting. According to the follow-up results, 24 cases were in the single-gene mutation group and 11 were in the co-mutations group. PFS of all patients were recorded; in general, PFS was regarded as the period from receiving TKIs’ treatment to tumor progression or death.
Statistical analysis
Statistical description measurement data for normally distributed data are expressed as Mean ± SD. One-way analysis of variance was used for comparison between the 3 groups after normal and homogeneity of variance test, F value was obtained, least significant difference (LSD) t test was used for pairwise comparison, a chi-square test was used for statistical analysis, and survival analysis was performed using the Kaplan–Meier method. Cox multivariate analysis was performed for determining independent prognostic factors related to PFS, and the hazard ratio (HR) and its 95% confidence interval were also calculated. For this study, statistical significance was set at P < .05.
Results
Clinical baseline data among the 3 groups were compared
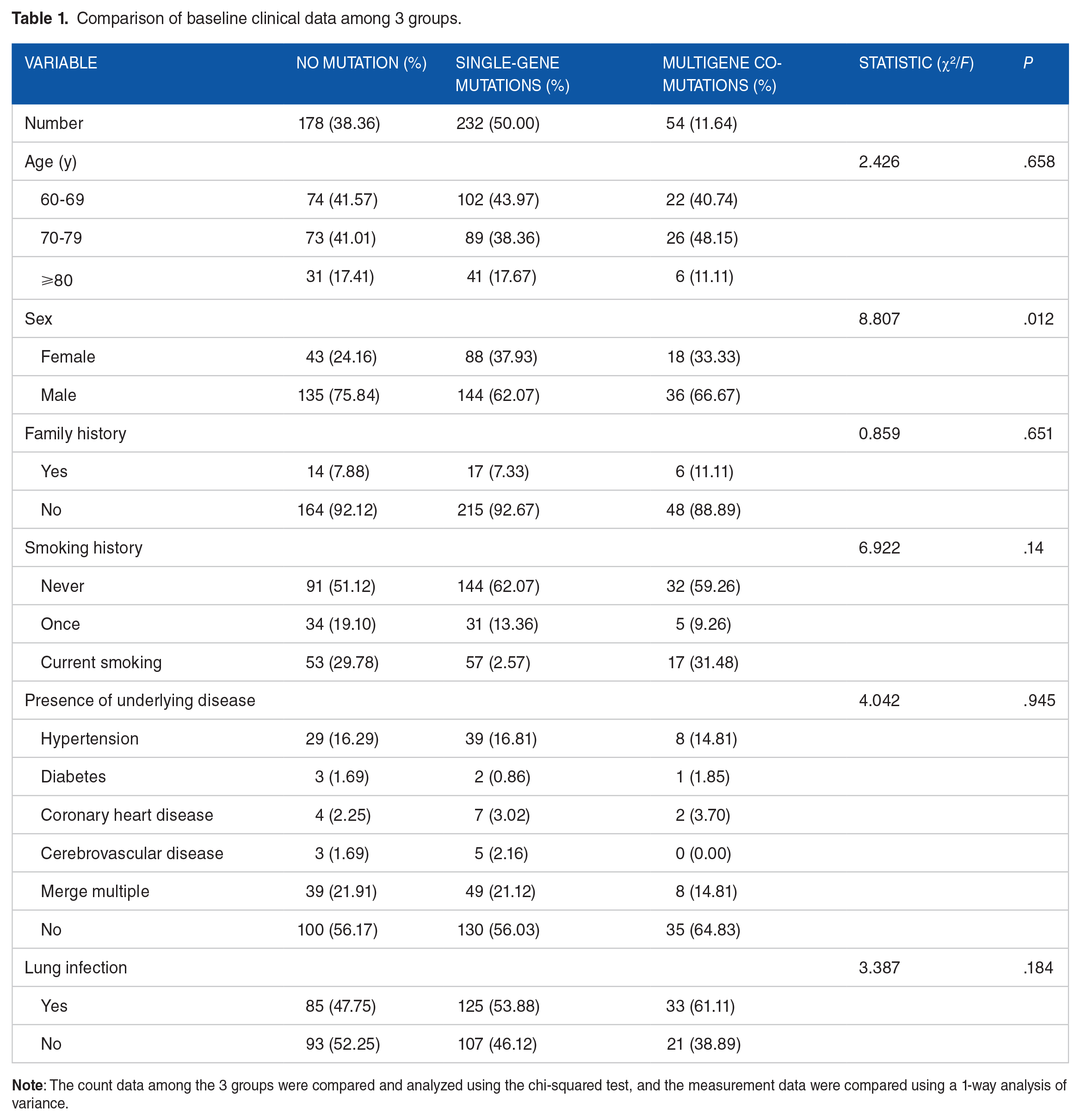
According to the NGS detection results, the enrolled patients were divided into a nonmutation group, single-gene mutation group, and multigene co-mutation group. The average ages of patients in the nonmutation group were 72.49 ± 7.48 years; the ages of the single-gene mutation group were 71.65 ± 7.28 years; and the ages of the co-mutation group were 71.04 ± 7.52 years. There were no significant differences among the 3 groups (P > .05). There were 43 females (24.16%) and 135 males (75.84%) in the nonmutation group, 88 females and 144 males in the single-gene mutation group, and 18 females and 36 males in the co-mutation group. There were significant differences in sex among the 3 groups (P < .05), whereas no significant differences in family histories, smoking histories, combined underlying diseases, or pulmonary infection (P > .05), as shown in Table 1.
Comparison of baseline clinical data among 3 groups.
Comparison of pathological data among the 3 groups
The pathological data in each group, respectively, have 106 cases, 195 cases, and 39 cases with adenocarcinoma; 46 cases, 31 cases, and 14 cases with squamous cell carcinoma, respectively; and 26 cases, 6 cases, and 1 case with other types. There were significant differences in pathological types among 3 groups (P < .05). However, there were no significant differences in the clinical stages and the proportion of distant metastasis among 3 groups (P > .05), as shown in Table 2.
Comparison of pathological data among groups.

Adenosquamous carcinoma, large cell lung carcinoma, and pulmonary sarcomatoid carcinoma are included in other types.
Details of gene mutations in the enrolled patients
Through analyzing the 9 mutant genes obtained from NGS detection in mutation groups, we found that the proportion of EGFR mutation was the highest among all groups. In the single-gene mutation group, the proportion of EGFR mutation was 54.31% with the mutation abundance of 12.69% (2.90-26.04). In the co-mutation group, it was 24.35% and 15.09% (7.12-33.83). In addition, the proportion of KRAS mutation was second (13.36%) in the single-gene mutation group with the mutation abundance of 10.55% (3.45-23.82). The proportion of PIK3CA mutation was second (17.39%) in the co-mutation group with the mutation abundance of 6.14% (3.14-9.80). The gene with the lowest mutation rate in the single-gene mutation group was MET, accounting for 1.29%, and the gene in the co-mutation group was BRAF accounting for 5.22%, as shown in Table 3.
Proportion and abundance of 9 gene mutations in the groups.

The main classification of co-mutant genes
Among the 54 patients with co-mutations, the proportion of EGFR and PIK3CA co-mutations was the highest (16.67%), followed by ALK and KRAS co-mutations (11.11%), and EGFR and MET co-mutations (7.41%); other types of co-mutations were all lower than 7%. The proportion of co-mutations with 3 genes was 5.55%, including ALK + HER2/ERBB2 + MET, EGFR + HER2/ERBB2 + MET and ALK + EGFR + ROS1. The proportion of the co-mutation with 4 genes was 1.85% (EGFR + HER2/ERBB2 + MET + ROS1), as shown in Figure 1.

Details of the proportion of gene mutations in 54 patients with co-mutation (the highest proportion of EGFR + PIK3CA was 16.67%, followed by 11.11% of ALK + KRAS mutation, and 7.41% of EGFR + MET mutation).
Analysis of prognostic factors
The ages, stages, and pathology of the follow-up patients were included in the multivariate Cox analysis. The results confirmed that ages and stages were not independent risk factors for PFS. In addition, the pathology was associated with the decrease in PFS. The patients with squamous cell carcinoma showed a more decreased PFS compared with adenocarcinoma (HR, 5.772; 95% confidence interval, 1.094-30.454; P < .05), as shown in Table 4.
Multivariate Cox regression analyses of prognostic variables for PFS.

Comparison of prognosis of patients with different types of mutations
In comparing the follow-up PFS of patients, the average PFS of patients with a single-gene mutation were 12.34 (11.73-12.95) months, which were significantly higher than 7.13 (2.41-11.85) months for patients in the co-mutation group. There was a significant difference between the 2 groups (P < .05), as shown in Figure 2.

Comparison of progression-free survival between the 2 groups. The median survival time of the single-gene mutation group was 12.34 months, and the median survival time of the co-mutation group was 7.13 months. Log Rank (Mantel-Cox). χ2 = 6.236, P = .013.
Discussion
Non-small cell lung cancer is the most common malignant tumors in elderly lung cancer patients.2,9 Recent studies showed that most lung cancer patients are more than 60 years in China, including NSCLC. 10 Most elderly NSCLC patients have been in the advanced stage when diagnosed. The targeted therapy has become the main strategy for elderly NSCLC patients with typical gene mutation. Due to individual differences, different NSCLC patients showed heterogeneous clinical outcomes when receiving typical targeted drugs. Studies have confirmed that the genetic co-mutations were important factors affecting the therapeutic effects of NSCLC patients closely associated with worse clinical outcome.11-13 With the wide application of NGS, more mutant genes regulating NSCLC progression were detected. This study retrospectively analyzed the clinical pathological features and prognosis of elderly NSCLC patients with single-gene and multigene co-mutations, exerting underlying directing values for targeted therapy and prognosis assessment.
Previous clinical studies confirmed that EGFR mutations were common in female adenocarcinoma patients, and had potential correlation with smoking histories. 14 ALK positivity is common in adenocarcinoma, never-smoking, and young patients, 15 and ROS-1 fusion mutations often occur in adenocarcinoma, female, and nonsmoking patients. 16 However, other studies showed that the clinical and pathological features of patients with co-mutations were not significantly different from those patients with single EGFR, ALK, or ROS1 mutation. 5 In contrast, Wu et al 17 reported that multigene co-mutations were more common in women, adenocarcinoma patients, and patients without a history of smoking. Chen et al 7 detected gene mutations using NGS in 36 EGFR mutation-positive NSCLC patients and found that co-mutations were significantly correlated with smoking histories. The study results showed that the male proportion in the 3 groups was higher compared with females, and the males in the nonmutation group were the highest. This may be due to smaller sample sizes and biases in our statistics. In addition, it may also be related to region, ethnicity, or sociology. Further exploration requires research evidence from others. The pathological types in gene mutant NSCLC patients were mostly adenocarcinoma, and the proportion of multigene co-mutations was higher in male adenocarcinoma patients. There were some differences between this study and previous literature reports, as this study was a nonrandomized controlled study with certain statistical bias, and the specific mutation subtypes were not studied.
In this group, the co-mutated rate of the 9 genes was 11.64%. EGFR was the most common mutant gene in the single-gene mutation group and the co-mutation group, and the mutation frequencies were, respectively, 54.31% and 24.35%. We also showed that the EGFR mutation rates in elderly NSCLC patients were consistent with that in Asian NSCLC patients. 18 KRAS mutation rate was the second in a single genome (13.36%), and PIK3CA mutation was the second in the co-mutation group (17.39%). In NSCLC patients receiving first-line EGFR-TKI therapy, the median PFS of patients with high EGFR mutation abundance was significantly longer than that of patients with low EGFR mutation abundance. 19 Won et al 20 reported that the efficacy of targeted drugs was related to gene mutation abundance of NSCLC patients with EGFR/ALK co-mutations. However, the relationship between the mutation abundance, clinical characteristics, and prognosis of NSCLC patients is still unclear, and further studies about analyzing these questions have been to be done.
Recent studies showed that TP53 was the most common co-mutation gene of EGFR in NSCLC patients commonly with poor prognosis.13,21 In addition, in EGFR-mutated NSCLC patients, TP53, KRAS, and PIK3CA were the most common combined-mutant genes. 7 Other studies showed that EGFR/ALK was the most common co-mutation type (31.7%), secondly ALK/KRAS (17.5%) in 3774 NSCLC patients using the amplification block mutation system. 5 In our results, the most common co-mutation types were EGFR/PIK3CA, ALK/KRAS, and EGFR/MET (16.67%, 11.11%, and 7.41%). The molecular mechanism of gene co-mutation may be related to tumor heterogeneity. Different mutations may coexist in the same tumor cells and also exist in different tumor regions. Some NSCLC patients with multigene co-mutations with TKIs’ treatment were commonly with worse PFS, which was consistent with previous studies.13,22 These results suggest that multigene co-mutations play an important role in tumor progression and drug resistance affecting clinical outcomes.
The Cox multivariate analysis shows that PFS is only related to pathology, and the prognosis of squamous cell carcinoma is worse, while it is not related to ages or stages. In the later stages of this study, only the PFS of some patients was followed up, the effect of co-mutation on prognosis still needs further studies. In addition, this retrospective study belonged to nonrandomized controlled study and had potential statistical biases which may cause a higher proportion of co-mutations in elderly male NSCLC patients. In addition, due to limited patient follow-up, no adverse events were mentioned in this article. In future study, increasing samples and effective follow-up are necessary for clarifying NSCLC patients.
Conclusion
Multigene co-mutations are not rare in elderly NSCLC patients and may represent a special subset, and the proportion of co-mutations is higher in elderly male adenocarcinoma patients. EGFR/PIK3CA, ALK/KRAS, and EGFR/MET are the most common co-mutations types, and patients with co-mutations have a poor PFS. This study has important reference values for targeted therapy of elderly NSCLC patients. It will be critically necessary to conduct multigene detection by NGS for directing targeted therapy of elderly NSCLC patients.
Footnotes
Acknowledgements
The authors thank all the patients and investigators involved in this study.
Funding:
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Colleges and Universities in Anhui Province Natural Science Research Projects (grant no. KJ2018A0208).
Declaration of conflicting interests:
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Author Contributions
Study concepts: HZ, FC, and WL. Study design: YZ, HZ, and FC. Data acquisition: HZ, FC, and WL. Quality control of data and algorithms: HZ, FC, WL, FM, and ZK. Data analysis and interpretation: HZ, FC, WL, FM, and ZK. Statistical analysis: MX and FX. Article preparation: HZ, FC, and WL. Article editing: HZ, FC, and WL. Article review: YZ.
Ethics Approval and Consent to Participate
The study was approved by the Ethics Committee of the First Affiliated Hospital of Anhui Medical University (approval ID: PJ2023-10-49), and the informed consents were obtained from all enrolled NSCLC patients.

