Abstract
Background:
It is controversial whether primary tumour resection (PTR) and the sequencing of chemotherapy and PTR are associated with the survival of patients with incurable stage IV colorectal cancer. This study aimed to explore the effects of PTR and the sequencing of chemotherapy and PTR on asymptomatic colorectal cancer with synchronous unresectable metastases (asmCRC).
Patients and Methods:
Patients with asmCRC were retrospectively identified from a single centre and categorised into 3 groups: PTR followed by chemotherapy (POC), upfront chemotherapy followed by PTR (UFC), and palliative chemotherapy (PC). The primary end points included median overall survival (OS) and progression-free survival (PFS). Clinical features were analysed using χ2 test, while survival curves were generated using the Kaplan-Meier test. Propensity-score matching (PSM) was performed when comparing survival between POC and UFC groups.
Results:
From 2008 to 2014, 255 patients were identified and included into the POC (n = 101), UFC (n = 40), and PC (n = 114) groups. The UFC and POC groups had significantly better median OS compared with the PC group (40.7 vs 16.3 months, P < .0001; 39.7 vs 16.3 months, P < .0001). Before PSM, the UFC group had better median PFS than the POC and PC groups (18.5 vs 9.7 months, P = .038; 18.5 vs 6.1 months, P < .0001). After PSM, UFC has better PFS than POC (P = .038). And the UFC group did not have higher postoperative or preoperative morbidity compared with the POC group (P = .235).
Conclusions:
Primary tumour resection could improve the survival of patients with asmCRC. Compared with POC or PC, UFC was associated with a better median PFS without significantly increasing preoperative or postoperative morbidity.
Introduction
Colorectal cancer (CRC) is the third most common malignancy and the second leading cause of cancer-related death globally. 1 On initial diagnosis, 20% of patients with CRC exhibit metastatic disease. 2 Primary tumour resection (PTR) is inevitable for patients with primary tumour–related symptoms or complications (obstruction, perforation, haemorrhage, etc) and those with resectable metastases. However, the necessity of PTR in patients with asymptomatic colorectal cancer with synchronous unresectable metastases (asmCRC) remains controversial. 3 Moreover, treatment strategies aimed at improving the survival of patients with asmCRC in the presence of systemic chemotherapy 4 have long been a topic of debate among surgeons and oncologists due to the unique dilemma of balancing operative timing and strategy.5 -7
Primary tumour resection before chemotherapy has been found to prevent primary tumour-related complications (obstruction, perforation, haemorrhage, etc) and improve the patient’s quality of life. Accordingly, Faron et al, 8 who synthesised data from 4 randomised trials involving patients with CRC having unresectable metastases, showed that PTR improved overall survival (OS) by 10% to 2% and reduced the risk of death by 40%. Furthermore, several authors have recommended PTR to improve quality of life and OS, typically for more than 5 months.9 -11 Conversely, a national population-based epidemiological study carried out by Hu et al 12 showed an increase in survival rate following a decrease in PTR rate. Moreover, a study from Korea 13 demonstrated no significant differences between resection and non-resection groups after matching. Recently, a randomised clinical trial (JCOG1007, iPACS study) discovered that PTR before chemotherapy does not carry any advantages over chemotherapy alone; surprisingly, however, up to 13% of patients who received chemotherapy alone required palliative surgery due to severe intestinal complications. 14 Unfortunately, the mentioned study had been riddled with several limitations, including insufficient statistical power, as well as poor patient accrual and quality, which had negatively affected the final results. Thus, we are convinced that the controversy regarding PTR remains unresolved. The most critical issue regarding the utility of PTR could perhaps be the method for identifying patients who can benefit therefrom. Recently, upfront chemotherapy (UFC) has been advocated to improve OS in patients with asmCRC without promoting surgical morbidity for emergent complications related to the primary tumour.15,16 Stelzner et al 17 also concluded that UFC was the only treatment-related factor associated with better survival in patients who received PTR. Conversely, a small sample research revealed that PTR followed by chemotherapy did not promote better 2-year OS compared with UFC, although it did facilitate better 2-year cancer-specific survival. 3 Thus, there is a pressing need to determine whether UFC followed by PTR carries any benefit for patients with asmCRC.
The current propensity-score-matched cohort study therefore sought to demonstrate the effectiveness of PTR and compare the effectiveness between POC and UFC in patients with asmCRC.
Patients and Methods
Patients
We retrospectively investigated patients diagnosed with stage IV CRC at West China Hospital, Chengdu, China, between June 2008 and December 2014. A total of 255 patients were included based on the following criteria: (1) histological diagnosis of adenocarcinoma of the colon or rectum; (2) age between 18 and 85 years; (3) Eastern Cooperative Oncology Group performance status (ECOG-PS) ranging from 0 to 2; (4) resectable primary tumour and unresectable metastases evaluated by an experienced surgeon; (5) absence of severe symptomatic disease related to the primary tumour, such as perforation, obstruction, or bleeding; and (6) absence of clinically significant cardiac disease, including kidney failure or hepatic failure. Eligible patients were then divided in 3 groups according to initial treatment patterns by the multidisciplinary team consisting of experienced surgeons and oncologists: PTR with postoperative palliative chemotherapy (POC group), UFC followed by PTR (UFC group), and only palliative chemotherapy without PTR (PC group). And the UFC group includes patients who first receive PC and need to receive PTR because of the presence of primary tumour. Decisions regarding treatment were determined by a multidisciplinary team comprising experienced surgeons and oncologists. The demographic traits (age, sex, ECOG-PS score, primary location, differentiation, T stage, N stage, metastatic sites, and the level of carcinoembryonic antigen [CEA], white blood cell [WBC], platelet [PLT], alkaline phosphatase [ALP], lactate dehydrogenase [LDH]), treatment strategies (surgery, chemotherapy, radiation, bevacizumab treatment, and cetuximab treatment), and survival information were assessed from the Hospital Information Manage System. We retrospectively recorded all the data, including the stage and laboratory data, prior to the initial treatment. The reported T, N, and M stages of tumour were clinically evaluated based on surgical findings or imaging techniques. This retrospective study was approved by the Medical Ethical Committee of West China Hospital. And all methods were performed in accordance with the relevant guidelines and regulations. Our chemotherapy regimens, including UFC and palliative chemotherapy, heterogeneously included oxaliplatin-based chemotherapy (FOLFOX or XELOX) and irinotecan-based chemotherapy (FOLFIRI) with or without bevacizumab and cetuximab. The interval between UFC and surgery ranged from 2 weeks to 2 months. Radiotherapy was used in neoadjuvant chemoradiotherapy or used to relieve local symptoms and improve quality of life. For example, some patients in the PC group underwent radiation due to tumour-related compression symptoms.
PSM method
To minimise possible confounding effects and establish well-matched cohorts, a 1:1 propensity-score matching (PSM) method was used to match patients with UFC and POC through the nearest neighbour method with a calliper size of 0.02. Variables used for matching included age, sex, ECOG-PS, T stage, N stage, primary tumour location, and number of metastatic organs, which were important prognosis factors of asmCRC.9,18
Survival outcomes
The primary end points included OS and progression-free survival (PFS). OS was recorded as the time from the date of diagnosis of smCRC to the date of death, whatever its cause, or the last follow-up. Progression-free survival was recorded as the interval from the date of diagnosis of smCRC to the date of the first disease progression or death, whatever its cause, whichever came first. For all patients, follow-up assessments were performed until progression, death, or date of last follow-up. Primary tumour–related morbidity and surgical complications were collected from the database of West China Hospital. The last follow-up was conducted on January 10, 2019.
Statistical analyses
Categorical data were analysed using the χ 2 test, with the Fisher exact test being applied as required. Kaplan-Meier analyses were performed to analyse PFS and OS. Propensity-score matching was performed using the nearest neighbour matching method with a calliper size of 0.02 on the propensity scale with logistic regression. Potential prognostic characteristics were analysed using univariate and multivariate analyses. Variables having a P value <.05 on univariate analysis were included during multivariate analysis using the Cox model. All statistical analyses were performed using SPSS version 22.0 (IBM Inc., Chicago, IL) and R version 4.0.3 (http://www.R-project.org), with a P value of less than .05 indicating statistical significance.
Results
Patients’ clinical characteristics
In total, 255 eligible patients with asmCRC were herein analysed. The median age was 57 years, with 101 (39.6%), 40 (15.7%), and 114 (44.7%) patients receiving POC, UFC, and only PC treatment, respectively. A total of 14 (9.9%) patients underwent liver metastasis resection due to chemotherapy following PTR. Baseline characteristics are listed in Table 1. Patients in the PC group were more likely to have more well-differentiated primary tumours (P < .0001), more metastatic organs (P = .017), higher WBC counts (P = .013), higher ALP levels (P = .038), and higher rates of abnormal LDH levels (P = .003) compared with those in the POC and UFC groups. Moreover, the POC group had more patients diagnosed with high T-stage disease compared with the other groups (P < .0001). Moreover, 13.3% and 18% of the patients received bevacizumab and cetuximab, respectively, with no significant difference in the number of patients receiving each treatment among the groups.
Baseline characteristics in the unmatched cohort.

Abbreviations: ALP, alkaline phosphatase; CEA, carcinoembryonic antigen; ECOG-PS, Eastern Cooperative Oncology Group performance status; LDH, lactate dehydrogenase; PC, palliative chemotherapy; PLT, platelet; POC, postoperative chemotherapy; UFC, upfront chemotherapy; WBC, white blood cell.
Survival analysis
To determine the OS and PFS, Kaplan-Meier survival analysis was performed. The median follow-up was 25.4 months (95% confidence interval [CI], 21.8-29.0). The POC and UFC groups had significantly better median OS compared with the PC group (40.7 months vs 16.3 months, P < .001; 39.7 months vs16.3 months, P < .001, respectively) (Figure 1A). However, no difference in OS was noted between the 2 PTR groups (P = .1). In addition, the UFC group had better median PFS compared with the POC and PC groups (18.5 months vs 9.7 months, P = .012; 18.5 months vs 6.1 months, P < .001, respectively), whereas the POC group had a longer PFS than the PC group (P = .001) (Figure 1B).
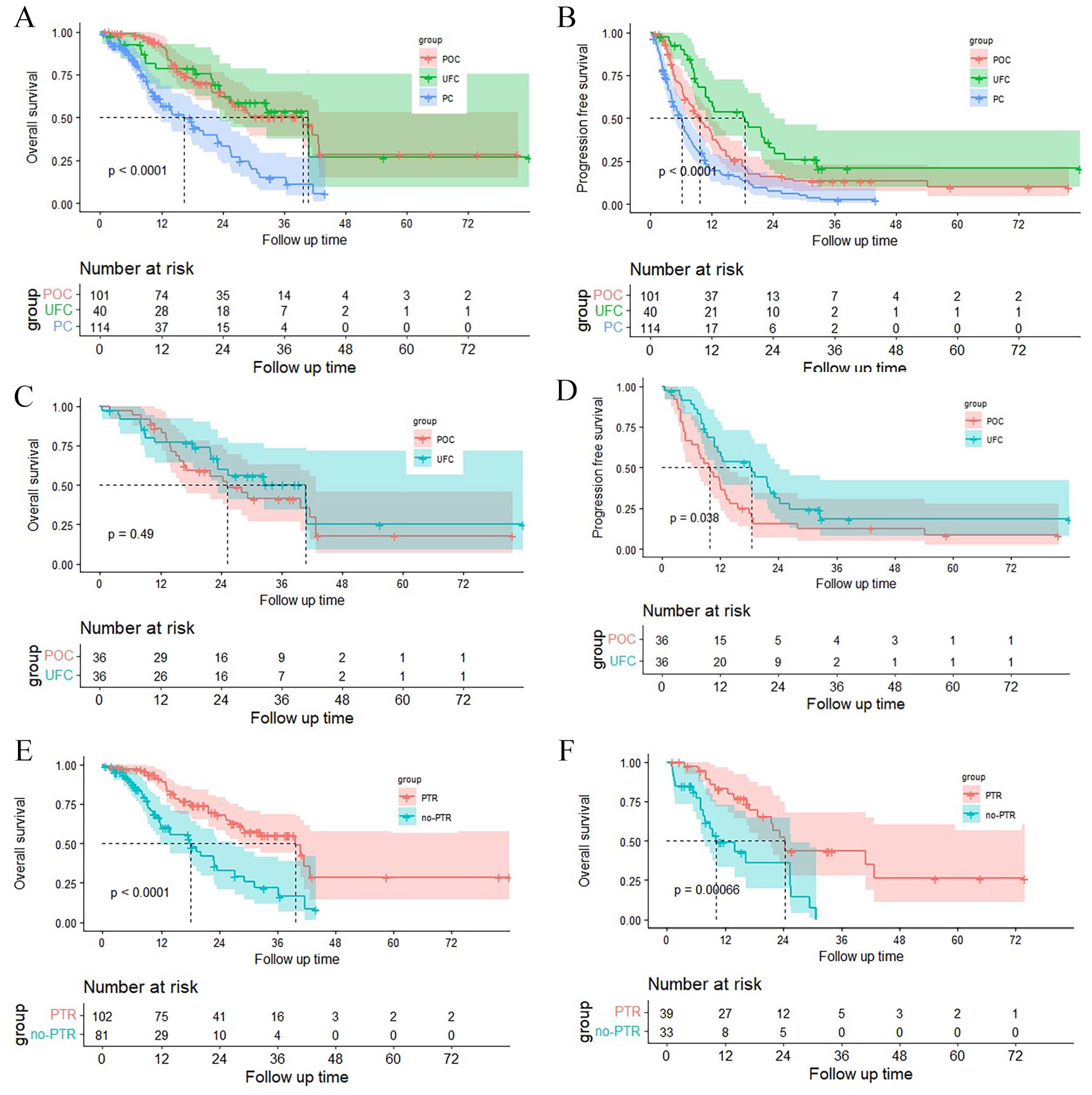
Kaplan-Meier survival estimates for patients with asmCRC. (A) OS for all patients with asmCRC in POC, UFC, and PC groups. (B) PFS for all patients with asmCRC in POC, UFC, and PC groups. (C) OS after PSM for matched patients with asmCRC in POC and UFC groups. (D) PFS after PSM for matched patients with asmCRC in POC and UFC groups. (E) OS for left-sided patients with asmCRC in PTR and non-PTR groups. (F) OS for right-sided patients with asmCRC in PTR and non-PTR groups. asmCRC indicates asymptomatic colorectal cancer with synchronous unresectable metastases; OS, overall survival; PC, palliative chemotherapy; PFS, progression-free survival; POC, postoperative chemotherapy; PSM, propensity-score matching; PTR, primary tumour resection; UFC, upfront chemotherapy.
Considering some differences in baseline characteristics between patients receiving UFC and POC, PSM was performed a with 1:1 ratio of UFC to POC to reduce potential biases and achieved comparable baseline characteristic between both groups. A total of 35 patient pairs with comparable baseline characteristics, except for N stage, were subsequently obtained (Table 2). Thereafter, Kaplan-Meier survival analysis post-PSM was performed to determine PFS and OS. Although the UFC group showed no significant advantage in OS over the POC group (40.7 vs 25.2 months, P = .49) (Figure 1C), the matched UFC group still exhibited better PFS compared with the POC group (18.5 vs 9.7 months, P = .038) (Figure 1D).
Baseline characteristics of POC and UFC patients after match.

Abbreviations: ALP, alkaline phosphatase; CEA, carcinoembryonic antigen; ECOG-PS, Eastern Cooperative Oncology Group performance status; LDH, lactate dehydrogenase; PC, palliative chemotherapy; PLT, platelet; POC, postoperative chemotherapy; UFC, upfront chemotherapy; WBC, white blood cell.
The location of the primary tumour has an impact on the effect of treatment of mCRC. So, we analyse the difference of OS in the context of PTR group and non-PTR group. The PTR treatment significantly benefitted both left-sided tumours (39.7 vs 18.0 months; P < .001; Figure 1E) and right-sided tumours (24.4 vs 10.2 months; P < .001; Figure 1F).
Univariate and multivariate Cox regression analyses
Univariate Cox analysis (Table 3) of all patients showed that POC and UFC were positively associated with OS (P < .001 and P < .001, respectively). The following factors, including ECOG-PS, primary tumour location, primary tumour histological differentiation, N stage, CEA level, WBC count, PLT count, ALP level, LDH level, and radiotherapy, were significantly associated with OS (P < .05). Multivariate Cox analysis of the aforementioned variables (Table 3) found that POC and UFC were significantly associated with better OS compared with PC (P = .001; P = .027, respectively). Based on multivariate analysis, ECOG-PS, primary tumour differentiation, CEA level, PLT level, and LDH level were identified as independent predictors of OS.
Univariate and multivariate analyses for the prognostic variables of overall survival for the treatment of synchronous mCRC (N = 255).

Abbreviations: ALP, alkaline phosphatase; CEA, carcinoembryonic antigen; CI, confidence interval; ECOG-PS, Eastern Cooperative Oncology Group performance status; LDH, lactate dehydrogenase; PC, palliative chemotherapy; PLT, platelet; POC, postoperative chemotherapy; UFC, upfront chemotherapy; WBC, white blood cell.
Morbidity and mortality
The overall tumour-related complication is composed of all the complications that occurred in the disease process, including preoperative and postoperative complications. The overall tumour-related complication rates in the POC, UFC, and PC groups were 7.9%, 15%, and 9.6%, respectively (Table 4). None of the patients died within 30 days after PTR. No significant difference in overall morbidity was observed between the PTR (POC and UFC) and PC groups (22.9% vs 9.6%, P = .6). Among those who underwent PTR, 1 patient in the UFC group and 5 patients in the POC group suffered postoperative ileus during the subsequent chemotherapy course. In the POC group, 1 case developed an anastomotic fistula, 1 developed infection, and 1 exhibited poor wound healing. No significance difference in morbidity existed between the POC and UFC groups (7.9% vs 15%, P = 0.235).
Tumour-related morbidity and surgical morbidity according to treatment features.

Abbreviations: PC, palliative chemotherapy; POC, postoperative chemotherapy; PTR, primary tumour resection; UFC, up-front chemotherapy; P*: P value between PTR and non-PTR groups in overall tumour-related complication; P**: P value for the POC group and UFC group in postoperative tumour-related complication.
Discussion
In our study, eligible patients were divided into 3 groups by the multidisciplinary team consisting of experienced surgeons and oncologists: POC group, UFC group, and PC group. Our study showed that patients with asmCRC who underwent PTR had significantly longer OS and PFS compared with those who received PC alone. Moreover, UFC followed by PTR was significantly associated with better median PFS compared with POC in pre-PSM and post-PSM.
In our study, the median PFS of UFC group was evidently higher than that reported by a previous study (18.5 vs 10-11 months), which may attribute to the definition of PFS.14,18 We calculate PFS from the date of smCRC diagnosis to disease progression or death, whereas the previous study recorded PFS from the data of initial treatment. Notably, we discovered that PTR treatment significantly benefited both left-sided tumours and right-sided tumours, which is in accordance with the previous study. 18 Manuel Benavides et al also explored the efficacy of PTR in the context of different mutational status and found PTR may benefit RAS/BRAF wild-type asmCRC. 18 Unfortunately, we did not study the influence of gene mutations due to the absence of these data, which need more studies to explore in the future.
This finding was consistent with that reported in previous studies, which indicated that PTR was associated with prolonged survival. 9 ,19 -22 A population-based and propensity-score-adjusted trend analysis, including 37 793 patients with metastatic colorectal cancer, concluded that palliative PTR was associated with improved OS and cancer-specific survival. 20 Another recent study based on the ARCAD database also identified PTR as a prognostic factor, with patients receiving PTR having better survival compared with those who did not. 22 Moreover, a meta-analysis reported that PTR was correlated with improved survival and perhaps better response to postoperative chemotherapy. 21
Whether UFC followed by surgery or PTR followed by chemotherapy provides better prognosis among patients with asmCRC remains controversial. In our study, UFC was associated with significantly better PFS compared with POC both before and after PSM, whereas no difference in OS had been observed after PSM. Those results were consistent with previous studies.15,23,24 Indeed, a retrospective study of 103 patients with asmCRC found that although OS did not differ significantly between the UFC and POC groups, UFC may prevent unnecessary surgery and consequently high morbidity rates associated with PTR. 15 Similarly, a multicentre randomised controlled trial founded no significant differences in 2-year OS between POC and UFC groups, although this clinical trial included a small sample (total 48 patients, each group 20 patients) and used a short follow-up duration (only 2 years), which made it difficult to determine differences in survival. 3 However, Kim et al 25 found that patients who received PTR first had longer OS compared with those receiving UFC (17.2 vs 13.6 months, P = .002). It should be noted that most patients (221 of 252) in the UFC group did not receive PTR, which would have promoted a negative effect on survival outcomes in this treatment arm.
Consistent with the results of multivariate analyses of prognostic factors associated with OS in patients with asmCRC presented in a review article, 9 patients who underwent PTR in the present study had better survival, which might have been attributed to their lower tumour burden (lower level of CEA, PLT, and LDH) and single-organ limited metastases. Considering the survival benefits and comparative morbidity, we suggest performing PTR in critically selected patients with asmCRC who have lower tumour burden. The optimal time for conducting surgery may be after chemotherapy, with only those responding to UFC being considered.
Surgery-related morbidity was acceptable in this study, with only 3 patients in the PTR groups suffering from operative complications, including anastomotic fistula, infection, and poor wound healing. These patients promptly recovered from their illnesses without the need for surgical interventions. And our study found no significant difference in tumour-related complications between the PTR group and PC group (22.9% vs 9.6%, P = .6). And UFC did not significantly increase tumour-associated complications compared with upfront surgery (15% vs 7.9%, P = .235). This result was consistent with previous studies, where only 11% of patients needed intervention due to tumour-related complications.15,26 Given that morbidity and mortality were not primary end points in our study, chemotherapy-related adverse effects were not particularly determined. More valid evidence from well-designed prospective studies is still needed.
Some limitations of the present study are worth noting. First, selection bias could not be eliminated due to the retrospective nature of our study. Second, heterogeneities existed in chemotherapy regimens and other treatment models, such as radiation and target therapy. Third, we only enrolled single-centre patients, and the sample size was small. To further determine the optimal treatment option or patients with asmCRC, 2 Chinese trials (NCT02149784 and NCT02291744) aiming to determine the impact of PTR timing in patients with asmCRC have been in progress, with future results being expected.
Conclusions
In this study, we retrospectively summarised the clinical characteristics, survival outcome, and prognostic factors of patients with asmCRC who received heterogeneous treatment from West China Hospital databases in China. We found that patients with asmCRC who underwent PTR combined with chemotherapy had significantly longer OS and PFS compared with those patients who received palliative chemotherapy alone. Moreover, those receiving UFC followed by PTR showed better survival compared with those receiving PTR followed by chemotherapy. The results of this study suggest that UFC followed by PTR can be considered an appropriate treatment strategy for patients with asmCRC, especially in patients with lower tumour burden.
Footnotes
Acknowledgements
The authors thank professors in the Department of Abdominal Oncology, West China Hospital, Sichuan University, for valuable data on all patients in this study.
Funding:
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by 1·3·5 Project for Disciplines of Excellence, West China Hospital, Sichuan University (ZYJC21017).
Declaration of conflicting interests:
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Author Contributions
L.H. collected data, reviewed the literature, and wrote the manuscript. G.X.W. collected data and wrote and revised the manuscript. N.C. collected data and rechecked the manuscript. J.W.L. assisted in drawing. Z.Q.W., Y.Y.Y., and M.Q. designed and revised the manuscript. All authors contributed to the article and approved the submitted version.
Availability of Data and Materials
If you want to request for data, please contact Meng Qiu (E-mail:
Ethical Approval
This retrospective study was approved by the Medical Ethical Committee of West China Hospital (2013(17)), registered 30 May 2013.
Principal Investigator
Meng Qiu.
