Abstract
Background:
Graves’ disease (GD) ranks as a primary cause of hyperthyroidism across all age demographics. The clinical presentation, treatment approaches, and overall therapeutic objectives can differ among various age groups. This study aimed to describe the clinical, laboratory, and radiological features, as well as the outcomes of the management of GD within the Saudi pediatric population.
Methods:
A cross-sectional, multicenter study across 2 tertiary care centers in Saudi Arabia (2010-2021). Clinical, biochemical, and imaging data were collected for children diagnosed with hyperthyroidism under the age of 18, utilizing an electronic medical records system. Data were analyzed using the Statistical Package for the Social Sciences (SPSS) version 21.
Results:
We enrolled 93 patients with hyperthyroidism (Mean age at diagnosis = 11.5 years; females = 68, 73.1%). Notably, 40/93 patients (43%) had a significant family history. The primary etiologies were GD (N = 60, 64.5%) and hashitoxicosis (N = 10, 10.8%). The commonest presentations were goiter (57%) and tachycardia (55%). GD was predominantly associated with exophthalmos, lid lag, sweating, tremors, and weight loss (Odds Ratios = 3.71, 3.8, 2.77, 2.34, and 2.28, respectively). An increase in thyroid radioactive iodine uptake was observed in 29/93 patients (48.3%; P-value = .029), in contrast to non-sensitive thyroid ultrasound results (P-value = .228). Thyroid Stimulating Immunoglobulin (TSI; N = 9.1, P-value = .000), Anti-thyroglobulin (TG; N = 537.5, P-value = .018), and Anti-thyroid peroxidase (TPO; N = 366.5, P-value = .017) were significant alongside FT4 and FT3 (P-value = .000) in diagnosing GD. Most patients were treated exclusively with methimazole (MMT), showing good compliance (N = 44, 73%) and minimal adverse effects (N = 56, 93.3%). Radioactive iodine (RAI) ablation was performed in 14 patients (23.3%), and thyroidectomy in 5 patients (8.3%).
Conclusion:
In our cohort, GD emerged as a leading cause of hyperthyroidism among children and adolescents. Pediatric endocrinologists in Saudi Arabia tend to underutilize RAI and prefer alternative treatment methods.
Plain Language Summary
Graves’ disease (GD) is one of the main causes of overactive thyroid (hyperthyroidism) in children. This study investigated how common GD is, how physicians in Saudi Arabia treat it, and the treatment of children diagnosed with the disease. We reviewed the medical records of 93 patients under 18 years old who were diagnosed with hyperthyroidism between 2010 and 2021 at two centers in Saudi Arabia. Most of the patients were girls (73%), and the mean age at diagnosis was about 11 years. About half of the children had a family history of thyroid disease. Graves’ disease was the most frequent cause (about two-thirds of cases), followed by hashitoxicosis. The most common symptoms were an enlarged thyroid gland (goiter) and fast heartbeat. Children with GD were more likely to have symptoms such as bulging eyes, eyelid lag, sweating, trembling, and weight loss. Blood tests measuring thyroid antibodies and hormones were very helpful in confirming the diagnosis. Most patients were treated with medication methimazole, which worked well and caused few side effects. Only a small number needed radioactive iodine treatment or surgery. Overall, the study identified that Graves’ disease is the main cause of hyperthyroidism in Saudi children and adolescents, and that physicians in Saudi Arabia tend to use medication rather than radioactive iodine to manage the condition.
Introduction
Hyperthyroidism, is a rare condition in children characterized by excessive activity of the thyroid gland, resulting in elevated levels of thyroid hormones that accelerate metabolic processes in peripheral tissues. 1 Graves’ disease (GD) is the leading cause of hyperthyroidism in pediatric population. 2 The hypermetabolic symptoms associated with GD arise from the synthesis and release of thyroid hormones, which is stimulated by the interaction between stimulating thyrotropin receptor antibodies (TRAb), also known as thyroid-stimulating immunoglobulins (TSI), and thyroid-stimulating hormone receptors (TSH-R). 3 While the symptoms of GD in children are largely similar to those observed in adults, children may present with non-specific signs that can easily be overlooked or mistaken for normal developmental changes, such as nervousness, sleep disturbances, fatigue, or behavioral and learning difficulties. 4
Currently, there are 3 primary treatment modalities for GD in children: anti-thyroid drugs (ATDs) such as methimazole (MMT) and propylthiouracil (PTU), radioactive iodine (RAI) ablation, and thyroidectomy. 5 Although ATDs are not typically curative for GD, they serve to alleviate the symptoms of hyperthyroidism until the condition either undergoes spontaneous remission or the patient receives definitive treatment through RAI or surgery. 6
Information regarding the patterns and outcomes of GD in Saudi Arabia is limited. However, the collection of such clinical data, along with insights into the attitudes of treating physicians, has become increasingly important in light of recent advancements in therapies, genetic testing, imaging techniques, and other diagnostic approaches for GD.
This study aims to describe the clinical, laboratory, and radiological features, as well as the outcomes of the management of GD within the Saudi pediatric population.
Methods
This study included all children under the age of 18 years who were diagnosed with hyperthyroidism that includes GD and non-GD cases at King Abdulaziz Medical City (KAMC) in Jeddah and King Abdullah Specialized Children’s Hospital in Riyadh, Saudi Arabia, between January 2010 and December 2021.
We conducted a retrospective chart review of Saudi children diagnosed and treated for hyperthyroidism at KAMC Jeddah and Riyadh. Data were extracted from the patients’ electronic medical records using the Best Care system EMR for a diagnosis coded as thyrotoxicosis, hyperthyroidism, or GD and compiled into a data collection sheet for analysis. The inclusion criteria encompassed Saudi children (under 18 years of age) with a diagnosis of hyperthyroidism (GD and other causes of hyperthyroidism). Collected data included patient demographics (age, gender), clinical characteristics (presentation, disease course, duration), investigations (biochemical, hormonal, serological, and radiological data), and treatment parameters (medications used, duration of treatment, and adverse events related to the medications). Patients with incomplete data were excluded from the study. Height and weight were measured and converted to percentiles using the WHO growth charts for children aged 2 to 19 years, with Z-scores calculated accordingly
Patients were categorized based on clinical presentation, biochemical profiles, imaging, and response to therapy. GD was diagnosed with suppressed TSH, elevated free T4/T3, positive TSH receptor antibodies (TRAB), and characteristic ultrasound features. Hashitoxicosis was diagnosed clinically as a transient hyperthyroid state characterized by positive thyroid autoantibodies (antithyroglobulin and/or antithyroid peroxidase antibodies) with negative TRAB, in the absence of histological confirmation. The diagnosis was based on the clinical course, antibody profile, and ultrasound findings, as histology was not routinely performed. Thyroiditis was considered in cases with tender thyroid glands, elevated inflammatory markers, and ultrasound features, often without antibody positivity. Thyroid nodules were identified via ultrasound, with functional status determined using scintigraphy—hot or cold nodules. Fine-needle aspiration was not performed routinely unless indicated. Subclinical hyperthyroidism is a hormonal status that defined by a decreased serum TSH level, with normal free T4 and total or free T3 levels.
Ultrasonography criteria included vascularity, echogenicity, and gland size. Scintigraphy was requested when ultrasound findings were inconclusive, when precise nodule functional status was necessary, or when surgical planning required localization, in accordance with standard protocols.
Precautions for RAI therapy include ensuring euthyroidism before treatment, recommending isolation post-therapy, and discussing effects on growth and development; parental counseling and follow-up protocols were adhered to.
Ethical approval for conducting this study was obtained from the Institutional Review Board (IRB) of King Abdullah International Medical Research Center (KAIMRC) under the reference number IRB RJ19/038/J.
Statistical Analysis
Numerical values from the data collection sheet and demographic data were analyzed using descriptive
statistical methods: mean and SD used for continuous variables and frequency and percentages presented for categorical variables. The χ2 test and unadjusted OR test were applied to nonparametric variables, and unpaired t test was applied for continuous variables. with a P-value of <.05 considered statistically significant. All collected data were entered and analyzed using IBM Statistical Package for the Social Sciences (SPSS) version 21.
Results
A total of 93 patients were included in the study. Table 1 outlines the basic characteristics of the patients diagnosed with hyperthyroidism. Among the 93 patients analyzed, 68 (73.1%) were female, and 40 (43.0%) reported a family history of GD. The findings indicated that 60 out of 93 patients (64.5%) had GD. Most patients exhibited normal height and weight for their age. Preliminary data indicate that approximately 15% had tall stature (above the 97th percentile), while the majority (around 70%) were of normal weight. The mean age at the time of data collection was 15.4 years (SD = 4.8), while the mean age at diagnosis was 11.5 years (SD = 5.3). The overall average duration of the disease was 3.2 years (SD = 2.1).
Basic Characteristics and Etiology of Hyperthyroidism in Our Patients.

The most common clinical presentations among our patients included goiter, tachycardia, palpitations, sweating, and weight loss. The least frequently observed clinical signs were eye pain, increased deep tendon reflexes (DTR), and ptosis (Figure 1). The distribution of etiologies for hyperthyroidism revealed that 64.5% had GD, while 10.8% had hashitoxicosis (Table 1). Thyroid nodules were detected in 9.7% of patients.
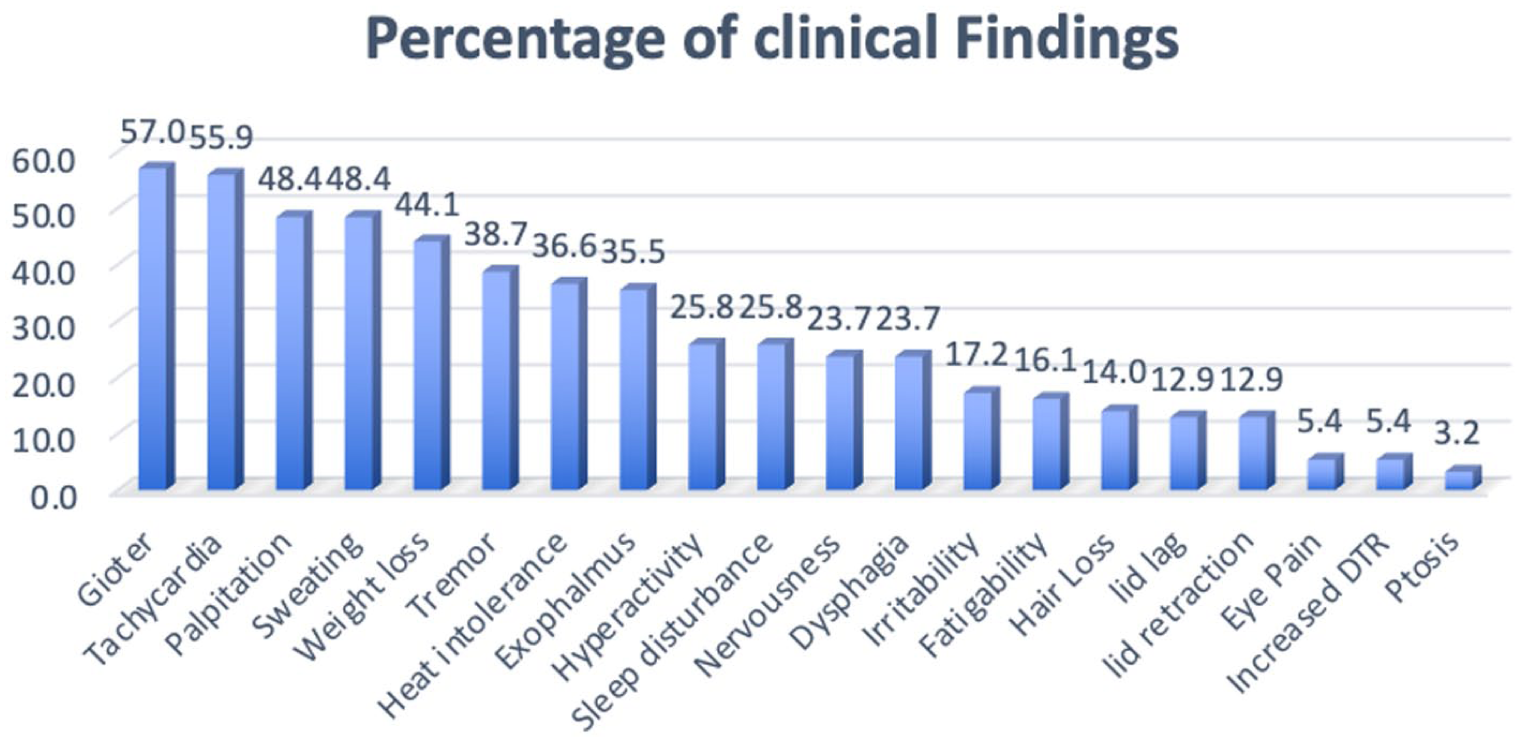
Clinical presentation and examination findings in our patients.
Radiological findings from thyroid ultrasounds and uptake scans were evaluated according to the status of GD in the patients (Table 2). In our analysis, 43.6% of ultrasound studies in GD patients indicated diffuse enlargement, while 33.3% showed signs of thyroiditis. Thyroid uptake scans demonstrated increased activity in 93.5% of children with GD who underwent the scan, compared to 86% in Non-Graves’ cases.
Biochemistry and Imaging Findings in Patients With Graves’ Disease and Patients With Other Causes of Hyperthyroidism.

Mean ± SD; n (%)
Significant at 5%
Significant at 1%
The likelihood of having GD based on clinical presentations was assessed using unadjusted odds ratios for the association between GD status and patient presentations. Exophthalmos, sweating, tremors, lid lag, and weight loss were significantly associated with GD status, as detailed in Table 3.
The Likelihood of Graves’ Disease By Clinical Presentation & Findings a .

Reference category is non-presentation for all variables.
Unadjusted odds ratio of clinical presentation and findings by Graves’ disease status.
Significant at 5%.
Significant at 1%.
The results of testing and significant differences in diagnostic parameters between patients with Graves’ disease and those with Non-Graves’ conditions are summarized in Table 2. Parameters such as FT4, FT3, TSI, Anti-thyroglobulin, Anti-thyroid peroxidase, and creatinine levels were found to be significant differentiators. In GD patients, the mean FT4 level was 32.6 pmol/L and the mean FT3 level was 20.3 pmol/L, compared to FT4 levels of 23.6 pmol/L and FT3 levels of 8.3 pmol/L in those with hashitoxicosis.
The treatment modalities employed in our cohort and their outcomes were analyzed according to the status of GD, as shown in Table 4. The initial therapeutic approach for all subjects involved the administration of anti-thyroid drugs (ATDs). The majority of GD patients were treated exclusively with ATDs (73%), while 23.3% of the cohort received radioactive iodine therapy, and 8.3% underwent thyroidectomy. Four cases (6.6%) in our cohort experienced adverse effects from medications: 1 patient developed elevated liver enzymes with agranulocytosis, 1 had agranulocytosis alone, 1 had a methimazole allergy, and 1 developed elevated liver enzymes. There was a significant association between the medication used and the diagnosis of GD. However, no significant associations were found between GD and variables such as RAI, duration of medication, side effects, compliance with medications, or surgery.
Treatment Used and Outcome of Graves’ Disease Patients.

Abbreviations: IQR, interquartile range; NA, not available; RAI, radioactive iodine.
Discussion
GD is the predominant cause of hyperthyroidism, in children and adolescents within our cohort. Hashitoxicosis ranks as the second most common cause of hyperthyroidism in this young population. Notably, a family history of GD was identified in slightly less than half of our cases. In Saudi Arabia, treating physicians appear to underutilize radioactive iodine (RAI), with less than a quarter of our cohort receiving RAI treatment, favoring instead the prolonged use of anti-thyroid drugs (ATD). It is essential to encourage greater utilization of RAI in the management of pediatric GD among our physicians. Long-term follow-up studies over 35 years for children and adolescents treated with RAI have demonstrated its efficacy and safety, with no reported increases in thyroid cancer or leukemia.
In our study, we found that the female-to-male ratio in cases of hyperthyroidism is approximately 2.7:1, comparing with ratios reported in other regions, such as Turkey (3.4:1), Sweden (4:1), Denmark (4.3:1), and China (6:1).7-10 There is a notable linear correlation between the child’s age and a greater incidence of hyperthyroidism in females. 11 GD emerges as the primary etiological factor in childhood and adolescent hyperthyroidism, comprising two-thirds of the cases in our cohort. This prevalence contrasts with previous reports from Denmark (86.8%), the United Kingdom (85%), and Sweden (97%).8,9,12
Hashitoxicosis constitutes the second most prevalent cause of hyperthyroidism in our population, accounting for 10.7%, while corresponding figures in the United Kingdom and Turkey are 12% and 14.9%, respectively.7,12 Our data also revealed an elevated incidence of toxic nodules, which accounted for 10% of cases, significantly diverging from prevalence rates reported in the United Kingdom (1.8%), Turkey (0.8%), and Sweden (0%).7,8,12 Alarmingly, 30% of our cases presented with short stature, a manifestation not previously reported at such a high frequency in hyperthyroidism. A family history of GD was noted in 43% of our cases, markedly higher than the 0.8% prevalence documented in a prior Turkish study. 7 Furthermore, we observed a heightened incidence of thyroiditis at 5.4%, in stark contrast to the less than 1% prevalence seen in the United Kingdom. 12 The combination of a heightened familial predisposition and an increased prevalence of autoimmune thyroid diseases in our study likely correlates with the elevated rate of consanguinity observed. 13 Similar findings were noted in another local study focusing on goiter patients, where a family history was present in 43% of cases. 14
Analysis of clinical data from our study shows that the initial clinical symptoms largely align with previously documented findings. However, a notable discrepancy was found in the incidence of goiter, which was lower in our cohort compared to previous studies of GD in adolescents.7,8,12,15 This difference may be partly due to our predominant reliance on manual palpation for assessing thyroid volume. Léger and Carel et al highlighted significant variability in thyroid volume among pediatric cases of GD, a pattern similar to findings from UK and French registries.4,11,12 Nonetheless, our study supports previous research in identifying key manifestations associated with GD, such as exophthalmos, sweating, lid lag, tremors, and weight loss. Ocular manifestations were observed in nearly a third of our patients, consistent with findings in the UK, although this frequency is notably higher than in previously reported series of GD.7,12 Exophthalmos was less frequent in our patients compared to those in 2 similar European studies.16,17 This discrepancy may be attributed to clinically unrecognized mild cases within our cohort. A recent study (2023) conducted in Al-Madinah province, KSA, by Alradadi et al reported that exophthalmos was the most common clinical manifestation among their GD patients, followed by goiter, palpitations, and tremors. 18
A characteristic feature of GD is the diffuse increase in parenchymal vascularization. Ultrasound examinations are routinely performed for GD patients presenting with thyroid gland asymmetry or palpable nodules. If a nodule is confirmed, fine-needle aspiration biopsy is recommended, alongside I-123 or Tc-99 scans. 19 In our study, while most ultrasound findings in GD revealed diffuse enlargement and fewer exhibited signs of thyroiditis, there was no significant association between ultrasound findings and GD status. Thyroid uptake was notably associated with GD status, showing slightly increased activity in children with GD compared to those with other causes of hyperthyroidism.
There is considerable variability in the investigations conducted and their reporting, making the differentiation between hashitoxicosis and Graves’ disease challenging based on immunological or radiological results. Relying solely on the presence or absence of thyroid autoantibodies for distinguishing these conditions may lead to misleading conclusions. 20 In our study, patients with GD exhibited a higher prevalence of positive Anti-TPO and Anti-TG antibodies compared to those without GD. Biochemical investigations demonstrated a greater consistency, with suppressed TSH values in all cases. Levels of Free T4 and Free T3 were significantly elevated in GD patients compared to those with hashitoxicosis. The median FT4 level reported by Alradadi et al in children with GD was 24.89 pmol/L, significantly lower than that observed in our patients. 18 Additionally, serum creatinine levels were reduced in hyperthyroid patients (both Graves and Non-Graves) in our cohort, reflecting an inverse relationship with thyroid function and indicating decreased total muscle mass in the body. 21
In terms of management, the initial therapeutic intervention for all subjects involved the administration of anti-thyroid drugs. The majority of patients with GD were treated solely with ATD, while fewer received radioactive iodine therapy or underwent thyroidectomy. This highlights the cautious approach taken by pediatric endocrinologists in Saudi Arabia regarding the adoption of radical treatments and the underutilization of RAI therapy within our cohort. Identified predictive indicators at disease onset, which signify a potential need for prolonged or extended ATD therapy or early radical interventions, include factors such as young age (children under 13 years), the presence of a markedly enlarged goiter, pronounced biochemical hyperthyroidism, elevated TRAb levels, and non-Caucasian heritage.4,22,23 It is noteworthy that RAI treatment has demonstrated efficacy in children with hyperthyroidism, achieving remission rates of 95%. 22 However, the utilization of this modality varies significantly across countries, ranging from 0% in Denmark to 33% in Scotland. This variation is partly due to concerns regarding potential thyroid malignancies, further compounded by limited access to this treatment, contributing to its relatively infrequent use in the pediatric population with GD.9,24 The treatment modalities reported in the cohort studied by Alradadi et al in Madinah also suggested underutilization of RAI therapy in KSA. 18
Read et al reported on GD patients under 20 years of age who received radioiodine therapy and were followed for 36.2 years. 25 None of their patients developed thyroid cancer or leukemia, suggesting that treating children and adolescents with GD using radioiodine is both safe and effective in the long term. Moreover, according to the European Thyroid Association Guidelines for the Management of Graves’ Hyperthyroidism, confidence in the safety of RAI has increased in recent years, particularly for post-pubertal younger individuals, with RAI being considered when the child reaches 16 years or older. 26 Additionally, disparities in the treatment practices for GD are evident, not only in the utilization of radioactive iodine but also in the prevalence of thyroidectomy, which ranges from 3.8% in Denmark to approximately 59% in Turkey and Sweden, compared to 8.3% in our cohort.7-9 These variations underscore the current absence of an international consensus regarding the selection of definitive treatments for pediatric cases of GD, a sentiment echoed in existing literature on this medical condition.4,27,28 iodine deficiency was unlikely to significantly influence our findings and that any temporal variation over the study period was minimal.
The latest European Thyroid Association (ETA) guideline provides updated recommendations for pediatric GD, noting that it can manifest with hyperthyroid symptoms as well as behavioral issues or declining school performance. It recommends testing for TSH receptor antibodies in all affected children. Methimazole or carbimazole are advised as first-line antithyroid drugs for at least 3 years, using a dose-titration approach. Propylthiouracil is discouraged due to safety concerns. These 2 recommendations by ETA 2022 guidelines were practiced and followed in this cohort diagnosis and initial management. For definitive treatment, total thyroidectomy or radioactive iodine (RAI) is suggested by ETA 2022, with RAI dosed for complete thyroid ablation and not recommended for children under 10 years. Surgery is preferred for those with a large goiter. 29 The definitive management used in our cohort also reflect the ETA 2022 recommendations for RAI use in children.
While our data primarily reflect established patterns of pediatric hyperthyroidism, our study offers valuable contemporary data specific to Saudi Arabia, encompassing recent trends in clinical presentation, diagnostic approaches, and management practices at 2 major tertiary centers. This localized data can inform future national guidelines and highlight areas needing further research. While our data are informative, cautious extrapolation to the entire country is advised due to regional variations in healthcare access and possibly iodine status in some areas. Future multicenter studies are recommended for broader representation.
As a limitation, no power analysis for sample size calculation was conducted.
Conclusion
In our cohort, GD was the most common cause of hyperthyroidism in children and adolescents, followed by hashitoxicosis. Pediatric endocrinologists in Saudi Arabia tend to underutilize RAI therapy, preferring instead the prolonged use of anti-thyroid drugs. Given the well-established long-term safety of RAI, there should be a concerted effort to encourage its increased use in children with hyperthyroidism as per the ETA 2022 guidelines, particularly in refractory cases.
Footnotes
Author’s Note
Mohammed Al Dubayee is now affiliated with King Abdullah Children’s Hospital, Ministry of National Guard Health Affairs, Al Qassim, Saudi Arabia.
Ethical Considerations
IRB Approval obtained from King Abdullah International Medical Research Center, Saudi Arabia. The IRB No: RJ19/038/J.
Consent to Participate
This study waived from consent since the data is going to be collected retrospectively.
Authors Contributions
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
The data of the present study will be available upon request.
