Abstract
Background:
Abdominal obesity (AO) is associated with hyperuricemia (HU) in people with type 2 diabetes (T2DM), but most evidence comes from Asian studies and Latin American data are scarce. This study addresses that gap in a Peruvian setting. We aimed to evaluate the association between AO and HU in adult Peruvians diagnosed with T2DM.
Methods:
We carried out a cross-sectional observational study in adults with T2DM in a private clinic in Lima – Peru. We defined HU as serum uric acid ⩾ 6 mg/dL and defined AO as waist circumference ⩾ 94 cm in men and ⩾88 cm in women. In addition, we estimated crude and adjusted prevalence ratios (PRs) with 95% CIs for the AO–HU association, adjusting for sex, glycosylated hemoglobin, estimated glomerular filtration rate, and systolic/diastolic blood pressure.
Results:
We evaluated 815 adults with T2DM. 62.6% were male, and the average age was 57 years. AO was present in 82.9% and HU in 22.7%. The prevalence of HU in adults with AO was 24.3%, while the prevalence was 15.1% in those not obese. AO was associated with HU in the crude analysis (cPR: 1.60 95% CI 1.05-2.43) and remained statistically significant after adjustment for sex, glycosylated hemoglobin, estimated glomerular filtration rate, and systolic/diastolic blood pressure (aPR: 1.95 95% CI 1.31-2.90).
Conclusion:
In Peruvian adults with T2DM, abdominal obesity was associated with nearly double the prevalence of hyperuricemia. Because waist circumference is simple and low-cost to measure, integrating it into routine diabetes visits, along with targeted uric acid screening and management, may enhance cardiometabolic risk stratification and guide timely interventions.
Plain Language Summary
People with type 2 diabetes often gain fat around the waist, sometimes called ‘belly fat’. This type of fat can raise uric acid levels in the blood, which may lead to gout and other health problems. Most research connecting belly fat and high uric acid has focused on Asian populations, so we wanted to understand whether this pattern also appears in Latin America. We reviewed information from 815 adults with type 2 diabetes who received care in Lima, Peru, noting each person’s waist size and uric acid test. Many had a large waist, and a sizeable number had high uric acid. Those with more fat around the middle were about twice as likely to have elevated uric acid as those without it, even after we accounted for sex, and other health factors. For Peruvians living with type 2 diabetes, a growing waistline seems to be a clear warning sign for high uric acid. Paying attention to waist size, and taking practical steps like healthy eating, regular physical activity, weight management, and seeking medical support when needed, may help lower uric acid and reduce related health risks.
Introduction
Obesity is a global public health problem due to its high prevalence and associated comorbidities. 1 In 2016, the World Health Organization (WHO) estimated a global prevalence of 13%, with projections exceeding one-fifth of the population by 2030. 1 In 2017, Mexico and Bolivia had the highest obesity prevalence in Latin America (32.2% each). 2 In Peru, prevalence was 19.7% in 2019, with 2030 projections similar to global estimates. 3 Obesity is linked to multiple noncommunicable diseases, including hypertension, dyslipidemia, nonalcoholic fatty liver disease, type 2 diabetes mellitus (T2DM), and hyperuricemia (HU). 4 Because obesity clusters with T2DM and other cardiometabolic conditions, clarifying its relationship with HU is clinically relevant for risk stratification and care planning.
HU is common in cardiometabolic conditions, and its prevalence increases with adiposity. This overlap motivates examination of how adiposity influences urate metabolism. Like obesity, HU is associated with several noncommunicable chronic diseases,5,6 and although multiple determinants contribute to HU, adiposity is a principal risk factor.7 -10 The pathophysiology linking obesity to HU remains incompletely understood 11 ; proposed mechanisms include greater exogenous purine intake, increased endogenous purine production, and reduced renal urate excretion. 12 In Peru, population-based estimates of HU are scarce; available reports are largely limited to clinical cohorts, with prevalence around 23.2% among patients with T2DM and 37.9% among those with cardiorenal involvement.13,14
Individuals with T2DM often present coexisting obesity and HU, conditions linked by complex, and possibly bidirectional, pathophysiologic pathways.6,15 -17 Although the mechanisms are not fully elucidated, adipose tissue expansion in obesity, together with hyperinsulinemia, appears to perturb uric acid metabolism.10,11 Adipokines (also termed adipocytokines), bioactive mediators produced by adipose tissue, including adiponectin, leptin, resistin, TNF-α, and IL-6, regulate energy balance, insulin sensitivity, and inflammation, and may influence both uric-acid production and renal excretion 18
Evidence on the obesity–HU association comes largely from Asian cohorts,16,17,19 with limited evaluation in Hispanic populations, where waist circumference (WC) cutoffs for abdominal obesity (AO) differ from other ethnic groups, as recommended by the Latin American Diabetes Association. 20 Moreover, many prior studies defined obesity by body mass index (BMI), whereas WC is a superior predictor of cardiometabolic risk.21,22 In Peru, the prevalence of AO (38.9%) markedly exceeds BMI-defined obesity (23.2%), underscoring the relevance of WC-based definitions. 23 Accordingly, we aimed to evaluate the association between AO and HU among Hispanic adults with T2DM in Lima, Peru.
Methods
Study Design
We conducted a cross-sectional analytical observational study through a secondary analysis of data from a previously published study. The secondary analysis was carried out between November and December 2020 using fully de-identified electronic health records from Clínica Internacional (Lima, Peru), a private clinic. The records analyzed correspond to health records belonging to the year 2018, as part of the primary study that evaluated the association between glycemic control and albuminuria in patients with T2DM. 24
Population, Sample, and Statistical Power
We collected data using non-probability (convenience) sampling. The target population comprised patients who attended the ‘Take Care’ Health Program at Clínica Internacional (Lima, Peru) in 2018. ‘Take Care’ is a disease-management program for individuals with diabetes mellitus, bronchial asthma, arterial hypertension, and dyslipidemia. Benefits provided through patients’ insurance policies include laboratory tests, procedures, and medical consultations according to patient comorbidities. Clinic staff record all patient information in the program database to enable personalized follow-up and appropriate treatment.
We included adults ⩾ 18 years with physician-diagnosed T2DM. Exclusion criteria were current allopurinol use and incomplete key data. The initial sample comprised 907 patients; 86 were excluded for allopurinol use and 6 for missing data, leaving 815 participants for analysis (Figure 1). A sample of 815 provided ≈81% power to detect prevalence ratios ⩾1.6 at a two-sided α = .05. Power calculations were performed using Epidat v4.2

Participant selection flowchart.
Variables and Measurements
The dependent variable was HU, defined as serum uric acid (UA) ⩾ 6 mg/dL, measured using a colorimetric method, following Kidney Disease: Improving Global Outcomes (KDIGO) guidance. 25
The independent variable was AO, defined as waist circumference (WC) ⩾94 cm in men and ⩾88 cm in women, per the Latin American Diabetes Association. 18 WC was measured by trained health professionals using a non-stretchable tape at end-expiration, with participants standing, abdomen relaxed, and the tape positioned horizontally at the umbilical level, ensuring no skin compression.
Covariates included age (years); sex (male/female); glycated hemoglobin (HbA1c, %); fasting plasma glucose (mg/dL); serum creatinine (mg/dL); 24-hour urine albumin (mg/dL); estimated glomerular filtration rate (eGFR) calculated with the Cockcroft–Gault formula (mL/min/1.73 m2); and systolic and diastolic blood pressure (mmHg). All tests formed part of the program’s annual evaluation and were obtained after an overnight (⩾8-hour) fast.
Statistical Analysis
We conducted all analyses in Stata v16 (StataCorp LLC, College Station, TX, USA). Continuous variables were summarized as mean (SD) or median (IQR) according to distribution, and categorical variables as n (%). Distributional assumptions were examined visually and with the Shapiro–Wilk test. Group comparisons by HU and by AO status used Student’s t test for normally distributed continuous variables, Mann–Whitney U for skewed variables, and χ2 or Fisher’s exact tests for categorical variables, as appropriate.
We made a multivariate analysis to determine the association between AO and HU. Confounding variables were added using a generalized linear model of the Poisson family with robust variance. Variables from the crude analysis with P-values <.2 were entered into the adjusted model. Using P < .20 at the bivariate screening stage follows the purposeful selection approach to avoid excluding potential confounders that might not meet a stricter threshold (P < .05) but can still materially influence the exposure–outcome estimate. In practice, this reduces residual confounding and the risk of type II error when deciding on adjustments. Collinearity relationships between variables were verified using the variance inflation factor (VIF).
Ethical Aspects
The study protocol was approved by the Ethics Committee of Universidad Peruana de Ciencias Aplicadas (CEI 527-10-20 PI 221-20). This is a secondary analysis of a primary study approved by the same committee (CEI 023-03-20-PI 113-18). The clinic from which data were obtained is not affiliated with the university; for the primary study, the clinic provided formal authorization to use de-identified electronic health records for research (Authorization Letter No. 006-TI-D-UDID-CI-2019). No author is employed by or affiliated with the source clinic, and none has previously held such roles or conducted research on its behalf. Data access was granted under a data-use agreement, and the research team analyzed a fully de-identified dataset. Given the retrospective design and de-identification, informed consent was waived. No direct patient contact occurred. All procedures complied with the Declaration of Helsinki and applicable national regulations.
Results
General Characteristics of the Population
We analyzed 815 patients, of which 62.6% were male with an average age of 57 years. The mean HbA1c was 6.7%, and the median eGFR was 109.2 ml/min/1.73 m2. Of the patients evaluated, 22.7% presented HU and 83% AO. The rest of the characteristics are shown in Table 1.
General Characteristics of Study Population.

Abbreviations: DBP, diastolic blood pressure; eGFR, estimated glomerular filtration rate; HbA1c, blood glycosylated hemoglobin; SBP, systolic blood pressure.
Mean (Standard deviation), Median (Interquartile range), and Number (Percentage).
Bivariate Analysis
We made 2 bivariate analyzes separately of the factors associated with AO and HU. In the first bivariate analysis, the factors associated with AO were HU (P = .019), fasting glucose (P < .001), HbA1c (P = .025), albuminuria (P < .001), eGFR (P < .001), SBP (P < .001) and DBP (P < .001; Table 2). In the second bivariate analysis, the factors associated with HU were AO (P = .019); sex (P < .001), fasting glucose (P < .001), HbA1c (P < .001), creatinine (P < .001), eGFR (P < .001), and SBP (P = .016; Table 3).
Bivariate Analysis According to Abdominal Obesity Among the Participants.

Abbreviations: DBP, diastolic blood pressure; eGFR, estimated glomerular filtration rate; HbA1c, blood glycosylated hemoglobin; SBP, systolic blood pressure.
Mean (Standard deviation), Median (Interquartile range), and Number (Percentage).
Bivariate Analysis According to Hyperuricemia Among the Participants.
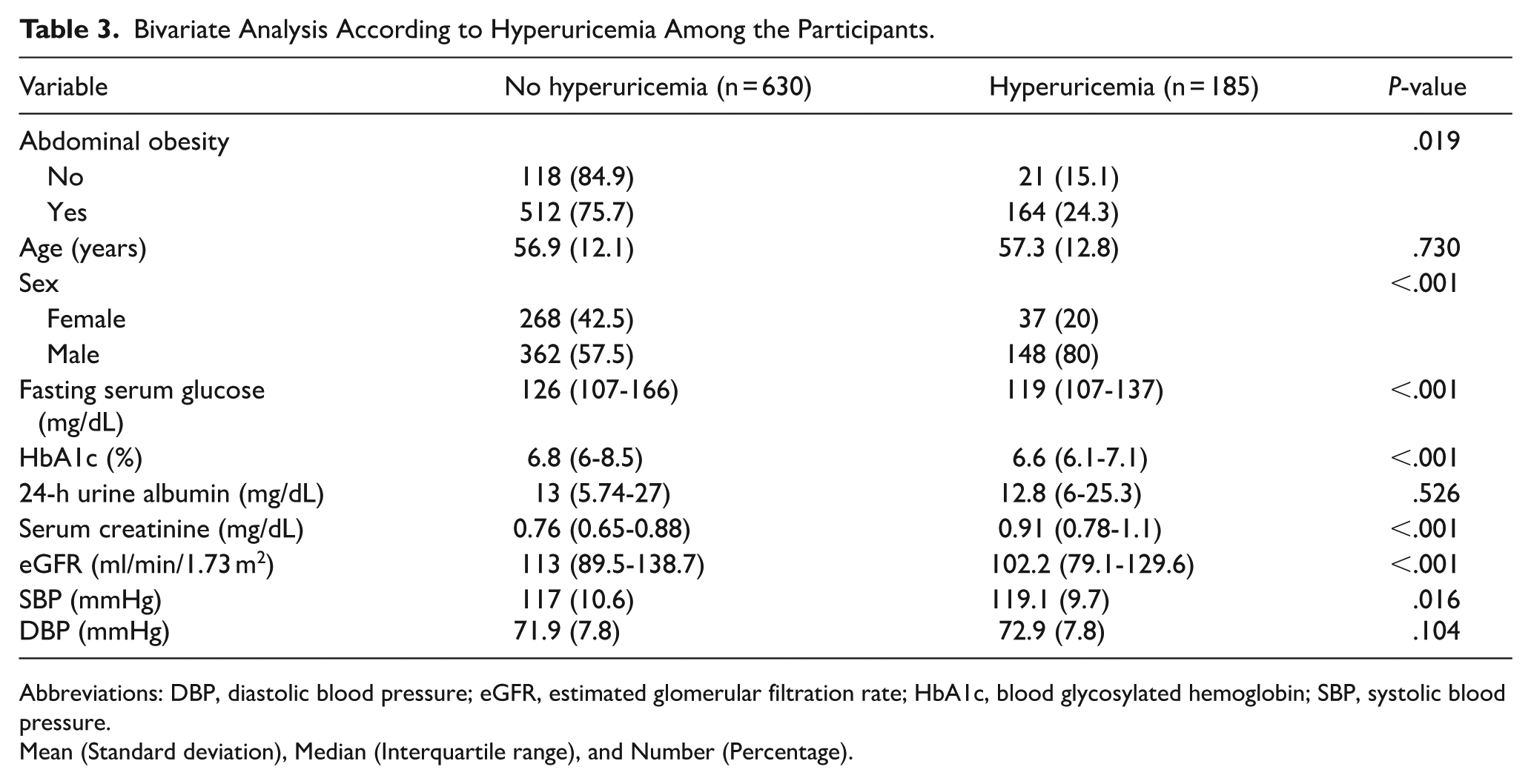
Abbreviations: DBP, diastolic blood pressure; eGFR, estimated glomerular filtration rate; HbA1c, blood glycosylated hemoglobin; SBP, systolic blood pressure.
Mean (Standard deviation), Median (Interquartile range), and Number (Percentage).
Regression Analysis
In the crude analysis, there was a statistically significant association between AO with HU, male sex, fasting glucose, HbA1c, creatinine, and SBP (Table 4). In the adjusted analysis, the statistically significant association between AO and HU was maintained (PR: 1.95 95% CI 1.31-2.90), with the male sex (PR: 2.45 95% CI 1.77-3.38), eGFR (PR: 0.99 95% CI 0.98-0.99) and HbA1c (PR: 0.76 95% CI 0.69-0.84; Table 4).
Crude and Adjusted Regression Model of Abdominal Obesity and Hyperuricemia Association.

Abbreviations: DBP, diastolic blood pressure; eGFR, estimated glomerular filtration rate; HbA1c, blood glycosylated hemoglobin; SBP, systolic blood pressure.
Variable did not enter the adjusted regression model due to collinearity with Hb1Ac.
Variable did not enter the adjusted regression model due to collinearity with GFR.
Discussion
Our principal finding is that AO was independently associated with HU among Peruvian adults with T2DM: participants with AO had nearly twice the prevalence of HU compared with those without AO. We also observed higher HU among men, whereas greater HbA1c and higher eGFR were inversely associated with HU.
In our study, individuals with T2DM and AO had nearly twice the prevalence of HU compared with those without AO, consistent with a Nigerian study reporting an approximately 1.9-fold higher probability of HU among diabetic patients with AO. 26 The mechanisms linking AO to HU are multifactorial. Dietary factors likely contribute: consumption of purine-rich foods (eg, red meat, certain seafoods) increases urate production, and high intake of saturated fats may impair renal urate excretion.8,27,28 Alcohol ingestion elevates serum lactate, which competitively inhibits renal urate transport, thereby reducing urate excretion.8,29 Fructose (prevalent in sugar-sweetened beverages) induces rapid hepatic ATP depletion with AMP accumulation, which is catabolized to UA via xanthine oxidase, increasing urate generation.8,30 These pathways may be particularly relevant in Hispanic Latin American settings, where a substantial share of dietary energy comes from carbohydrates, fats, processed foods, and sugar-sweetened beverages. 31 On the other hand, the role of adipocytes of obese and diabetic people in HU has been suggested. 32 The increase in adipose tissue produces an alteration in adipocytokines, increasing leptin and decreasing adiponectin, associated with insulin resistance and subsequent development of T2DM.33,34 In addition, hyperinsulinemia increases sodium and UA reabsorption in the renal tubules, decreasing urinary UA excretion and consequently causes HU in these patients.32,35
In our study, male sex was associated with a higher prevalence of HU. Likewise, an Ethiopian study reported that men with T2DM aged ⩾ 45 years had approximately twice the prevalence of HU as women. 36 This difference may be partly explained by higher estrogen levels in women of reproductive age: estrogens increase renal urate clearance and inhibit tubular urate reabsorption, conferring protection against HU, an effect that wanes after menopause. 37 Estrogen signaling may also modulate urate transporter expression and function, thereby lowering serum UA 38 ; key transporters include ABCG2 and GLUT9/SLC2A9.39,40 Additional mechanisms likely contribute to the pronounced sex differences in serum UA and remain to be fully elucidated. Moreover, men typically have greater visceral adiposity, a phenotype linked to insulin resistance (as in metabolic syndrome and T2DM) and to elevated serum UA, in part via hyperinsulinemia-mediated reductions in renal urate clearance5,6
Conversely, higher HbA1c and greater eGFR were associated with a lower prevalence of HU. Our HbA1c result is consistent with findings from Iran and Nigeria.41,42 A plausible explanation is that glycosuria, when the renal threshold is exceeded, promotes uricosuria by competing with tubular urate reabsorption, thereby lowering serum UA. 41 Although most participants had glucose values below classic glycosuria thresholds, intermittent/postprandial glycosuria or a higher filtered glucose load could exert similar effects; this warrants confirmation in prospective studies. The inverse association with eGFR aligns with prior reports 43 and likely reflects greater renal urate clearance at higher eGFR, whereas lower eGFR reduces clearance and favors higher serum UA, even within ranges above conventional chronic kidney disease thresholds. 44
We recommend implementing AO screening in resource-limited clinics by adding waist circumference to triage vitals at every diabetes visit (or at least quarterly). When AO is documented, staff should provide brief lifestyle counseling (reduce dietary sodium and sugar-sweetened beverages; set physical-activity goals) and review potential contributors (eg, diuretics). Serum UA can be measured annually, or targeted to patients with AO or suboptimal metabolic control.
Our study has limitations. First, the cross-sectional design precludes causal inference and prevents establishing temporality between AO and HU in adults with T2DM. Second, participants were recruited by non-probability (convenience) sampling from a single private clinic in urban Lima during routine care; selection bias is therefore possible. Attendees may differ from the broader Peruvian T2DM population (eg, urban residence, insurance/socioeconomic status, health-seeking behavior, disease severity), and exclusions for missing data may have further shaped the analytic sample. Third, we could not determine the timing of onset of obesity, HU, or T2DM. Fourth, we lacked data on some comorbidities and medications that influence urate metabolism, so residual confounding cannot be excluded. Despite these limitations, to our knowledge this is the first study in a Hispanic T2DM population to evaluate the AO–HU association. Prospective cohorts in diverse settings, with detailed comorbidity and medication data, are warranted to clarify temporality and causation.”
Conclusion
Among adults with T2DM, abdominal obesity was independently associated with a higher prevalence of hyperuricemia after adjustment for sex, age, systolic blood pressure, HbA1c, and eGFR. Integrating waist-circumference screening and brief lifestyle counseling into routine diabetes visits, coupled with annual or targeted uric acid testing, may facilitate earlier detection and follow-up of hyperuricemia, particularly in resource-limited settings
Footnotes
Ethical Considerations
Ethical approval for this study was obtained from the Ethics Committee from Universidad Peruana de Ciencias Aplicadas with the CEI code 527-10-20 PI 221-20.
Consent to Participate
Informed consent was not sought for the present study because is a secondary analysis of a primary study. The private clinic extracted the data from its electronic records and provided it in a de-identified format. Only the researchers had access to the data.
Consent for Publication
Not applicable.
Author Contributions
F. Peralta-Vera: Conceptualization, Data curation, Methodology, Software, Writing - original draft, Writing - review & editing. Enzo Castillo-Céspedes: Conceptualization, Data curation, Methodology, Software, Writing - original draft, Writing - review & editing. Lucero Collazos-Huamán: Conceptualization, Methodology, Software, Writing - original draft, Writing - review & editing. Camila Guerreros-Espino: Conceptualization, Methodology, Software, Writing - original draft, Writing - review & editing. Percy Herrera-Añazco: Conceptualization, Methodology, Software, Writing - original draft, Writing - review & editing. Vicente Benites-Zapata: Conceptualization, Formal analysis, Methodology, Software, Supervision, Writing - original draft, Writing - review & editing.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
Data are available upon reasonable request to the corresponding author.
