Abstract
Background:
Liraglutide can effectively reduce the weight of patients with type 2 diabetes. Nonetheless, its weight loss effect was highly heterogeneous in different patients in the clinical practice.
Objective:
To identify the factors most associated with the weight loss effect of liraglutide in obese or overweight patients with type 2 diabetes with poorly controlled oral medication in northeast China.
Design:
A prospective study.
Methods:
A prospective study was performed in subjects with type 2 diabetes who were taking oral medication and had a body mass index (BMI) of ⩾24 kg/m2. Liraglutide was administered for at least 12 weeks, while the original hypoglycemic regimen was kept unchanged (Phase I). Later, liraglutide treatment was continued or stopped as necessary or as subjects thought fit in the 13–52 weeks that followed (Phase II), and the potential factors affecting the effect of weight loss of liraglutide were analyzed.
Results:
Of the 127 recruited subjects, 90 had comprehensive follow-up data at week 12. In Phase I, the subjects’ blood sugar levels and weight decreased significantly(P < 0.001). Among all the significant factors, the gastrointestinal adverse reactions score (GARS) was more correlated with BMI change (ΔBMI; r = 0.43) and waist circumference change (ΔWC; r = 0.32) than the baseline BMI (BMI0) and WC (WC0). At week 12, linear regression showed that BMI0 independently affected ΔBMI and ΔWC, whereas WC0 only affected ΔWC. The GARS was significantly associated with ΔBMI and ΔWC, and this association continued until week 52, even after most subjects had discontinued liraglutide treatment.
Conclusion:
The degree of obesity and gastrointestinal adverse reactions were the most promising predictors of weight loss in liraglutide treatment.
Keywords
Introduction
Overweightness and obesity are closely linked to insulin resistance and type 2 diabetes mellitus (T2DM).1–5 Weight loss has been demonstrated to improve glucose metabolism in patients with T2DM.5–8 However, diet control and exercise are seldom sufficient for losing weight. 8 Many hypoglycemic drugs, including insulin, can cause weight gain as a side effect. Therefore, weight control has emerged as a concern for treating overweight or obese patients with T2DM.
In recent years, an increasing number of hypoglycemic drugs have focused on controlling weight and reducing blood sugar levels. Representative agents, such as glucagon-like peptide–1 (GLP-1) analog or GLP-1 receptor agonists, reduce weight by acting on the nervous system causing loss of appetite,9,10 gastrointestinal tract by delaying gastric emptying to reduce food intake,11,12 and white fat browning for reducing weight. 13 Liraglutide, a widely used long-acting GLP-1 analog, has been confirmed to aid weight loss in the Liraglutide Effect and Action Diabetes (LEAD) study14–19 as well as in the clinical practice.20–27 Nonetheless, its weight loss effect was highly heterogeneous in different patients and had no effect in some cases in the clinical setting. Therefore, investigating the clinical characteristics enabling efficient weight loss in patients through liraglutide treatment is necessary.
The principal clinical factors influencing potential weight loss through liraglutide treatment include baseline obesity status (body mass index, BMI), baseline degree of abdominal obesity (waist circumference, WC), degree of gastrointestinal adverse reactions (GAR), liraglutide dosage, and age of the patients.28–30 Exploring the factors having a high impact on liraglutide-mediated weight loss is of the utmost significance. In this study, we identified the determinant factors in obese or overweight patients with T2DM with poorly controlled oral medication in northeast China to provide better evidence for predicting the weight loss effect of liraglutide treatment.
Methods
A prospective study was performed at the Shengjing Hospital of China Medical University between January 2019 and July 2020. In this 52-week study, we enrolled overweight or obese patients with T2DM with poor glycemic control in northeast China. Liraglutide was administered for at least 12 weeks (Phase I) and the treatment was continued or discontinued in the next 13–52 weeks (Phase II). Subjects were visited at week 12, followed by a telephonic interview at week 52. All potential factors affecting weight loss were recorded and followed up to observe their effects on changes in BMI (ΔBMI) and WC (ΔWC) from baseline to weeks 12 and 52.
Inclusion and exclusion criteria
Subjects in the 18- to 70-year age group, BMI ⩾ 24 kg/m2, and diagnosed with T2DM in ⩽10 years were enrolled in the study. Eligible subjects were provided with a specific diet and exercise regimen. A stable dosage of antidiabetic drugs of two or more types, including metformin (mandatory), sulfonylureas, meglitinides, α-glucosidase inhibitors, and thiazolidinediones, was perorally administered for at least 3 months. Subjects had stable body weight with glycated hemoglobin level (HbA1c) values between 7.5% and 11.0% during the screening process. Subjects with the following characteristics were excluded: changes in antidiabetic medication regimen in the past 3 months (except short-term treatment for intercurrent illness), treatment with insulin, GLP-1 analogs, GLP-1 receptor agonists, or dipeptidyl peptidase-4 enzyme inhibitors in the past 3 months, treatment with systemic corticosteroids for more than 7 days, hypoglycemia unawareness or recurrent severe hypoglycemia, pregnancy or lactation, family or personal history of multiple endocrine neoplasia type 2 or familial medullary thyroid carcinoma, history of nonfamilial medullary thyroid carcinoma, history of chronic gastrointestinal disease or pancreatitis, increased levels of aspartate aminotransferase or alanine aminotransferase (⩾2.5 times upper normal range), chronic kidney disease (estimated glomerular filtration rate < 60 mL/min/1.73 m2), and congestive heart failure (New York Heart Association grade IV).
Study design
Phase I
Previous treatment with oral antidiabetic drugs was a prerequisite to begin Phase I. After their enrollment, subjects were injected with subcutaneous liraglutide (Novo Nordisk, Denmark) in the abdomen once daily at no specific time using a Prefilled Pen of Liraglutide with 4- or 5-mm needles. They were encouraged to inject liraglutide at the same time each day. Liraglutide doses were increased every week from 0.6 to 1.2 mg or to 1.8 mg. The maximum liraglutide dose was determined by the blood glucose level and tolerance degree of adversely affected patients. Liraglutide was required to be used for a minimum of 12 weeks unless an unacceptable adverse event or severe hypoglycemia occurred. Self glucose monitoring (measured during fasting, 2 h after the meal, and bedtime) was encouraged to identify hypoglycemia.
The medical history of the subjects, including family history, previous medication history, and diabetes comorbidity, was collected in the Shengjing Hospital information system. Baseline body weight, WC, and vital signs were recorded by a trained nurse and measured again at week 12. The gastrointestinal adverse reactions score (GARS) was recorded under the guidance of the investigators. GAR included nausea, vomiting, anorexia, abdominal distension, diarrhea, and other symptoms, and the GARS ranged from 0 to 10 points. The GARS was set as 0 when no GAR occurred during the 12 weeks and 10 when the GAR was extremely severe. Biochemical indicators and HbA1c were detected at the baseline and week 12. Laboratory analyses were performed by the central laboratories of the Shengjing Hospital. Subjects possessed the right to withdraw from the study for any reason or due to poor glucose control (determined by the physician).
Phase II
The physicians and the subjects decided whether to continue using liraglutide or shift to other medications, including insulin, between weeks 13 and 52. To determine the effect of liraglutide treatment duration on weight loss, subjects were divided into two groups: the no-stop group (n = 12), which contained patients who continued liraglutide treatment until week 52, and the stop group (n = 78), which contained patients who discontinued treatment between weeks 13 and 52. The original plan of the study was to conduct outpatient visits at 52 weeks to measure the weight and WC of subjects. However, given the sudden coronavirus epidemic, medical work and the general movement of people were greatly limited. Thus, for the visit at week 52, we guided the subjects to measure their weight and WC via video and obtained reliable measurement data.
Statistical analysis
The study subject screening and progress flow chart is shown in Figure 1. We estimated sample size according to the relevant literature 31 and the principle of 10 outcome events per variable. We assumed a weight loss of 2.5 kg after 52 weeks of liraglutide treatment with a standard deviation of 4, an efficacy of 90%, and a drop-out rate of 40%. Therefore, at least 80 subjects were calculated to be required for this study. The baseline characteristics of the study population were expressed as absolute and relative frequencies for categorical variables, mean ± standard error for approximately normally distributed continuous variables, and median (interquartile range) for non-normally distributed continuous variables. We used the two-sample t-test, Mann–Whitney U-test, and chi-square test for comparing two independent groups, and a one-sample t-test for comparison between the follow-up (week 12 or week 52) and baseline data. Moreover, we used the Pearson correlation coefficient for analyzing the correlation between continuous variables. Non-normal distribution data were converted before analysis.

Study subject screening and progress flow chart.
The factors affecting BMI and WC reduction after liraglutide treatment were evaluated using multiple linear regression considering baseline BMI (BMI0) or baseline WC (WC0), age, gender, liraglutide dosage, and GARS as independent variables and the changes in BMI and WC in 12 weeks (ΔBMI12w and ΔWC12w, respectively) as dependent variables. Similarly, the factors influencing ΔBMI and ΔWC were analyzed at week 52. In addition to the above independent variables, the duration of liraglutide therapy, insulin, or sulfonylureas treatments were also added as independent variables because insulin and sulfonylureas cause weight gain. As BMI and WC were co-linear, they were not included in the same regression models. The significance level was set at 5%, and the two-sided P-values were reported. Data were analyzed using the SPSS 22.0 software (Statistics for Windows; SPSS Inc., Chicago, IL, USA).
Results
Of the 127 recruited subjects, 90 who provided comprehensive follow-up data at week 12 were included in the analysis (Figure 1). After 12 weeks, 53 subjects (58.9%) continued the liraglutide treatment for 24 weeks, and 12 subjects (13.3%) for 52 weeks or longer. Seventy-eight subjects discontinued the treatment before week 52 for the following reasons: unwillingness to continue subcutaneous injections (n = 30, 38.5%), economic reasons (n = 26, 33.3%), inefficient weight loss (n = 18, 23.1%), or intolerable GAR (n = 4, 5.1%).
Effect of liraglutide on blood sugar in Phase I
During the 12 weeks of liraglutide treatment (Table 1), the proportion of men subjected to maximum liraglutide dose (1.8 mg/d) was smaller than that of women (46.9% versus 69.2%; P = 0.055). After liraglutide treatment for 12 weeks, HbA1c levels decreased by 1.75 ± 0.29% (P < 0.001) on average, and the reduction was comparable in both men and women, although women showed higher HbA1c levels than men at baseline (P < 0.001). Fasting plasma glucose (FPG) decline was more pronounced in men (P < 0.05); hence, the HbA1c improvement in women was speculated to be mainly due to postprandial blood glucose decline.
The clinical characteristics of the subjects in baseline and follow-up.

Diabetic macrovascular complications include arteriosclerosis, stroke, and coronary heart disease; microvascular complications include diabetic retinopathy, diabetic nephropathy, and diabetic peripheral neuropathy.
BMI, body mass index; DBP, diastolic blood pressure; FPG, fasting plasma glucose; GARS, gastrointestinal adverse reaction score; NAFLD, nonalcoholic fatty liver disease; SBP, systolic blood pressure; WC, waist circumference.
Only the subjects who completed liraglutide treatment for 12 weeks are included (n = 90).
P < 0.001 versus baseline.
Effect of liraglutide on GARS in Phase I
Major GAR during liraglutide treatment included anorexia, nausea, vomiting, diarrhea, and flatulence (Figure 2(a)). Of these, anorexia and nausea were the most common symptoms, occurring in 55% and 47% of subjects (n = 98), respectively. In the 90 subjects who could tolerate the GAR during 12 weeks of liraglutide treatment, the reactions were relieved after an average of 3.1 weeks; 15 (16.7%) of these subjects continued to lose weight after the GAR disappeared. The percentages of various major GAR are shown in Figure 2(a). In addition, more than 50% of the subjects had a very low GARS (0–2) after liraglutide treatment. Eight subjects (8.2%) had a high GARS (9–10); however, three of them discontinued liraglutide treatment and withdrew from the study because of intolerable side effects (Figure 2(b)).
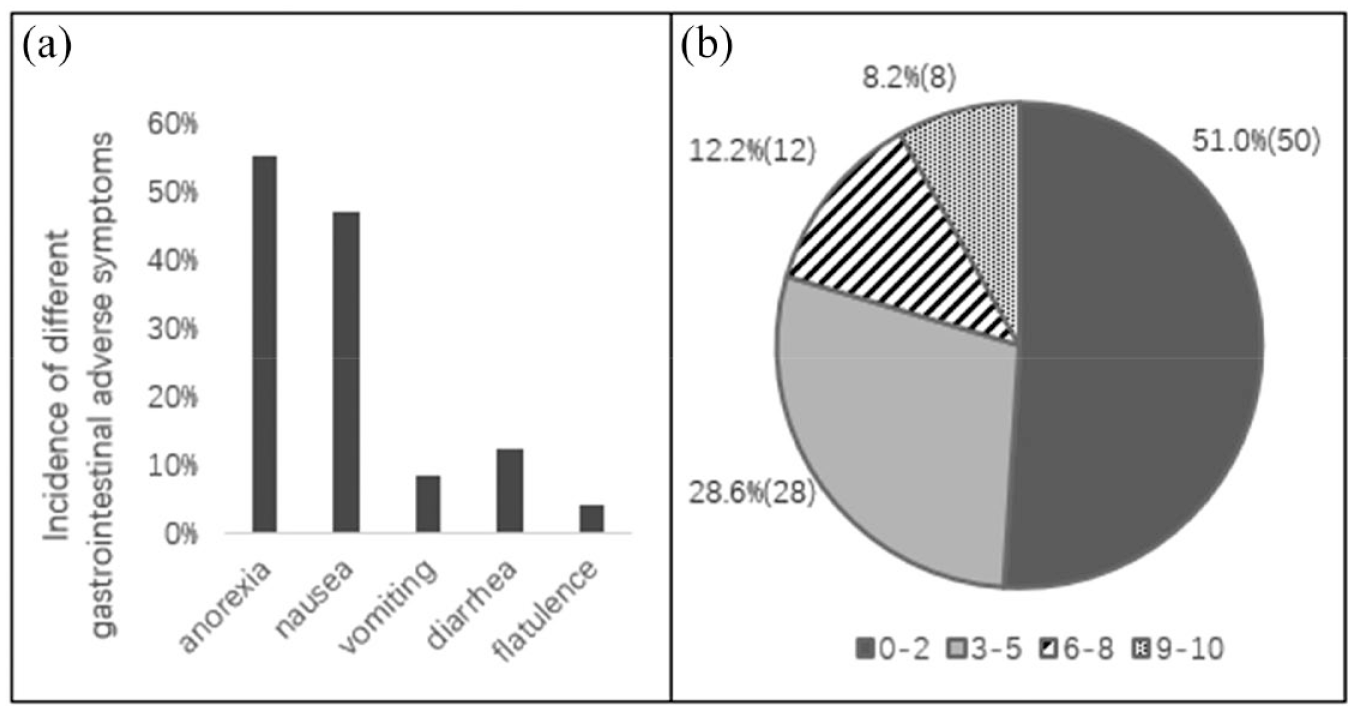
(a) Incidence of different gastrointestinal adverse symptoms in all subjects (n = 98). (b) Proportions of subjects showing different levels of gastrointestinal adverse reaction scores (GARS) (n = 98).
Effect of liraglutide on body weight, BMI, and WC in Phases I and II
Changes in body weight, BMI, and WC
The subjects who underwent liraglutide treatment for 12 weeks (n = 90) had a significantly lower body weight within 12 weeks (3.76 ± 0.48 kg) than at baseline (Figure 3(a)). In the first 12 weeks, 41.1% of subjects lost 5% or more of their baseline body weight, and only 7 of the 26 (29%) of subjects who did not lose their body weight gained weight from the baseline (Figure 3(d)). At week 52, the no-stop group maintained the original weight loss-promoting effect. The degree of weight loss was higher in the no-stop group than in the stop group. This may have been because the dose of liraglutide in the no-stop group was higher than that in the stop group (P < 0.05). This result may also be related to the overall trend of GARS, BMI0, and WC0 in the no-stop group being slightly higher than that in the stop group (no significance; this may be due to the small sample size of the no-stop group). In addition, subjects in the stop group who switched to other glucose-lowering regimens exhibited some weight loss at week 52, but the change was lower than the weight loss of the no-stop group (1.99 ± 0.57 kg vs 5.75 ± 1.63 kg; P < 0.05; Figure 3(a)). Importantly, the most significant weight loss occurred in the first 12 weeks. In the stop group, the effect of weight loss was maintained during liraglutide treatment; however, when liraglutide was discontinued, 72 (92.3%) of these subjects gradually regained their weight (Figure 3(a)). Furthermore, BMI and WC showed the same trend as that of body weight at weeks 12 and 52 (Figure 3(b) and (c)).

The changes in body weight, BMI, and waist circumference (WC) of the 90 subjects who continued liraglutide for 12 weeks. Changes in (a) body weight, (b) BMI, and (c) WC from baseline. The stop group refers to the 78 subjects who discontinued liraglutide after week 12 and before week 52. The no-stop group refers to the 12 subjects who continued liraglutide for 52 weeks. **P < 0.001 versus baseline. # P < 0.05 versus week 12. & P < 0.05 versus stop group. (d) Percentage of weight change in subjects after 12 weeks of liraglutide treatment.
Linear correlation analysis between ΔBMI12w, ΔWC12w, and various clinical characteristics
The change in BMI in 12 weeks (ΔBMI12w) was related to BMI0 and GARS (r = 0.33 and 0.43, respectively), and the change in WC in 12 weeks (ΔWC12w) was related to BMI0, WC0, and GARS (r = 0.29, 0.27, and 0.32, respectively). GARS had the highest correlation coefficient with ΔBMI12w and ΔWC12w(Figure 4).
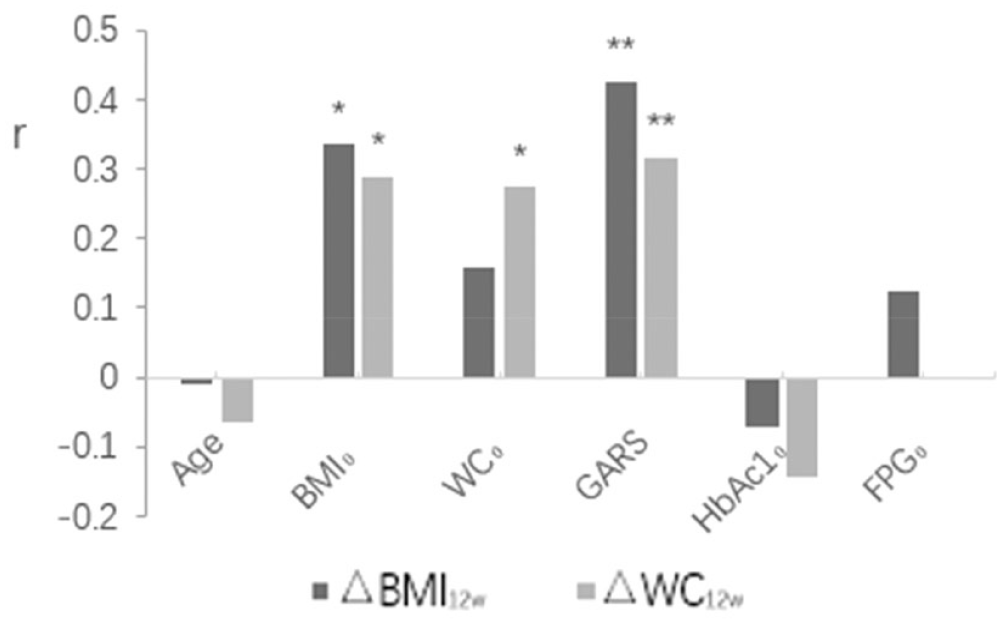
Linear correlation between △BMI12w and △WC12w and various clinical characteristics. Only the subjects who completed liraglutide treatment for 12 weeks are included (n = 90).
Multivariate linear-regression analysis of the factors associated with changes in BMI and WC
BMI0 significantly affected ΔBMI12w, which was retained after adjusting for multiple potential confounders, such as gender, age, and liraglutide dosage (adjusted regression coefficient, β = 0.303–0.352; P < 0.05), and also ΔWC12w (adjusted β = 0.245–0.295; P < 0.05; Figure 5(a)). Similarly, WC0 independently affected ΔWC12w after adjustment for confounders (adjusted β = 0.226–0.3; P < 0.05; Figure 5(b)).

Multivariate regression analysis of the association of baseline BMI (BMI0) (a), baseline waist circumference (WC0) (b), and GARS (c), with changes in BMI and WC within 12 weeks and 52 weeks. The regression was performed after adjusting for other confounding factors. (a)–(b) Model 1: Single factor linear-regression analysis; Model 2: Model 1 + age + gender; Model 3: Model 2 + liraglutide dosage; Model 4: Model 3 + GARS; Model 5: Model 4 + treatment of insulin + treatment of sulfonylureas; Model 6: Model 5 + liraglutide treatment duration. (c) Model 1: Single factor linear-regression analysis; Model 2: Model 1 + age + gender + liraglutide dosage; Model 3: Model 2 + BMI0; Model 4: Model 2 + WC0; Model 5: Model 3 + treatment of insulin + treatment of sulfonylureas + liraglutide treatment duration; Model 6: Model 4 + treatment of insulin + treatment of sulfonylureas + liraglutide treatment duration.
Consistent with the correlation analysis, the most obvious factor during liraglutide treatment was the GARS, which independently and significantly affected BMI and WC changes (Figure 5(c)), regardless of adjustment for confounders (adjusted β = 0.397–0.416 and 0.279–0.311, respectively, P < 0.05). This independent effect persisted up to week 52 even after liraglutide withdrawal (adjusted β = 0.385–0.456 and 0.164–0.256, respectively), suggesting a lasting effect of the GARS on BMI and WC reduction.
Discussion
GLP-1 analogs (such as liraglutide) and GLP-1 receptor agonists can both lower blood sugar levels safely and control body weight,32–34 possibly making them a better choice for obese patients with T2DM. The weight loss effect of liraglutide is achieved by suppressing appetite, delaying gastric evacuation, controlling food intake by directly affecting the nervous system,10,35–38 and browning of white fat.13,39,40. In the LEAD series of studies, LEAD-2 15 and LEAD-4 17 reported that weight loss caused by liraglutide treatment was dose-dependent. In addition, the LEAD-3 study found that the weight loss due to liraglutide was closely related to nausea and was not dose-dependent. 16 In contrast, the LEAD-5 study showed that weight loss was independent of GAR, although a few patients with sustained nausea seemed to lose more weight. 18 Based on the results of the LEAD series of studies, we can conclude that liraglutide provides a significant weight loss advantage over other oral hypoglycemic drugs; however, whether the weight loss depends on the degree of GAR is unclear. In addition, none of the LEAD studies included Chinese subjects; hence the conclusions on the weight loss effect of liraglutide may not be applicable to Chinese people. Therefore, we conducted this study in Chinese patients with T2DM to observe the weight loss effect on obese or overweight subjects and its influencing factors.
In this study, 41.11% and 33.33% of subjects lost 5% or more of their baseline weight in 12 and 52 weeks, respectively, after starting liraglutide treatment. The weight loss effect of liraglutide was closely related to the GARS. Although numerous subjects experienced GAR, the average time of symptom relief was 3.1 weeks. During the 12 weeks, after the GAR disappeared, 16.67% of subjects reported further weight loss. In addition, the weight loss mainly occurred in the first 4 weeks, and this period coincided with the duration of gastrointestinal discomfort, further confirming the significance of a high GARS on weight loss. With regard to the continued weight loss-promoting effects observed in the subjects after the disappearance of GAR, it was speculated that GLP-1 may affect weight loss by modulating the browning of white fat, in addition to its role in the gastrointestinal tract.
Thirty-two of the 78 subjects who discontinued liraglutide treatment before week 52 regained their weight; 23 of them even exceeded their original weight, possibly because they switched to hypoglycemic drugs with weight gain side effects, indicating that liraglutide should be used continuously for a long period to achieve sustained weight loss. Furthermore, the main reason subjects discontinued liraglutide treatment was not associated with GAR intolerance but the cost and daily inconvenience of subcutaneous injection. This indicates that GAR due to liraglutide could be a good predictor of weight loss.
In addition, our research also showed that, during liraglutide treatment, both BMI0 and WC0 had an independent association with WC reduction, but only baseline BMI was associated with BMI decline. However, these effects disappeared at week 52 because most subjects discontinued liraglutide treatment, suggesting that the effects of liraglutide on BMI and WC may considerably weaken after drug withdrawal.
To the best of our knowledge, this was the first prospective study specifically designed to evaluate the weight loss effect of liraglutide using BMI and WC changes rather than body weight change in patients with T2DM. In addition, this study is the first to demonstrate self-reported GARS of subjects for assessing GAR and to explore the factors influencing the weight loss effect. However, this study did have a few limitations. First, the sample size was insufficient as all subjects were selected from a single center in northeast China. Second, the weight and WC were not measured by physicians at week 52; this information was collected through telephonic interviews.
In conclusion, we found that among all the potential factors, the baseline BMI and GARS were the most important determinants affecting ΔBMI and ΔWC during the first 12 weeks of treatment. These effects were independent of other confounding factors. In particular, GARS was independent of baseline obesity status and lasted even after liraglutide was discontinued.
Because of the single-centered design and the limited amount of participants enrolled in this study, a prospective study with a larger sample size from multiple centers will be more helpful to reveal the effect of liraglutide on weight loss.
Conclusion
This study preliminarily confirms that adding liraglutide to the oral hypoglycemic therapy regimen for 12 weeks can reduce BMI and WC in overweight or obese patients with T2DM in northeast China. Baseline BMI and the degree of GAR were significant and independent predictors for weight loss; however, the effect of GAR was more long-lasting. Altogether, liraglutide should be considered for long-term retention in the hypoglycemic prescriptions of patients to achieve long-term weight maintenance.
