Abstract
Background:
Evidence of efficacy and safety of digital ecosystems in Latin America is scarce, which has limited their implementation. The objective of this study is to evaluate the safety and efficacy of the use of a digital ecosystem (Zutrics) in people with Type 2 Diabetes (PwT2D) treated with insulin.
Methods:
Analytical observational prospective cohort study in PwT2D, treated with insulin and oral or injectable antidiabetics, with HbA1c >8%, and followed up with a digital ecosystem (Zutrics). HbA1c and derived time in range (TIRd 70-180 mg/dL) were evaluated at baseline and at 3-month follow-up. Additionally, hypoglycemia events were evaluated during the follow-up.
Results:
We analyzed 69 patients (age 62 ± 12.5 years, 56.6% female), 45.1% had chronic kidney disease (CKD) and 25.4% coronary artery disease. About 45.1% were on multiple dose insulin treatment. Median HbA1c levels decreased from a baseline value of 9.1% (interquartile range, IQR 7.5-11.4) to 7.0% (IQR 6.3-8.08) at 3-month follow-up (P = .044). The median of HbA1c changes over time was −1.3% (IQR −0.13, −4.2). The mean TIRd of 70 to 180 mg/dL increased from 74% at baseline to 76.1% at the end of follow-up. The TBRd of <70 mg/dL did not change significantly, going from 0.5% at baseline to 0.94% The incidence density of hypoglycemia episodes was 0.009 events/patient-day during the follow-up. About 95.6% of patients met the goal of TBRd <70 mg/dL less than 4%
Conclusion:
This study suggests that the use of a digital ecosystem in the follow-up of PwT2D allows better glycemic control without increasing the risk of hypoglycemia.
Plain language summary
This study evaluated the efficacy and safety of a digital ecosystem called Zutrics for the outpatient management of type 2 diabetes mellitus in patients using insulin in Colombia. Researchers conducted a prospective observational cohort study in 69 patients to determine the impact of Zutrics on HbA1c levels and the incidence of hypoglycemia during a three-month follow-up period. The results showed a significant reduction in HbA1c and a low rate of hypoglycemic events, suggesting that this type of digital platform can improve glycemic control without increasing the risk of hypoglycemia in this patient population. The study highlights the need for future research with larger cohorts and longer follow-up to confirm these findings in other settings.
Introduction
The global prevalence of diabetes in adults aged 20 to 79 years is estimated to be 11.1% (approximately 1 in 9) based on the 2025 IDF Diabetes Atlas. This translates to 589 million adults living with diabetes worldwide. 1 Glycemic control is the cornerstone of the treatment of people with Type 2 Diabetes (T2D), however, only 36% of patients achieve a Glycated Hemoglobin (HbA1c) <7.0%. 2 There are barriers that make it difficult to achieve metabolic control goals, including lack of adherence to management, educational failures, restricted access to assessments by specialized personnel, and limited access to health services. 2
The implementation of telemedicine and the use of applications on mobile devices (mHealth) has reduced the barriers of access between patients and specialized professionals in different health systems. 3 A 2019 review found 328 mHealth, 23 of them focused on diabetes management. The use of mHealth applications allows remote monitoring of blood glucose readings, carbohydrate intake, physical activity, and makes it possible to implement reminders for medication intake and application. 4
Zutrics is an ecosystem of digital platforms that brings together a variety of disease-related data in 1 place. Patients can access advice on diet, physical activity, and calorie calculation. The application also allows the configuration of alarms and the automatic upload of data from the glucometer via Bluetooth. In addition, it generates glycemic control reports, following the Ambulatory Glucose Profile (AGP) recommendations through the Gluco® Vision platform (Figure 1), allowing the healthcare team to access the data. 5 The use of this type of platform favors early changes in therapy, achieving improvement in metabolic control. 6
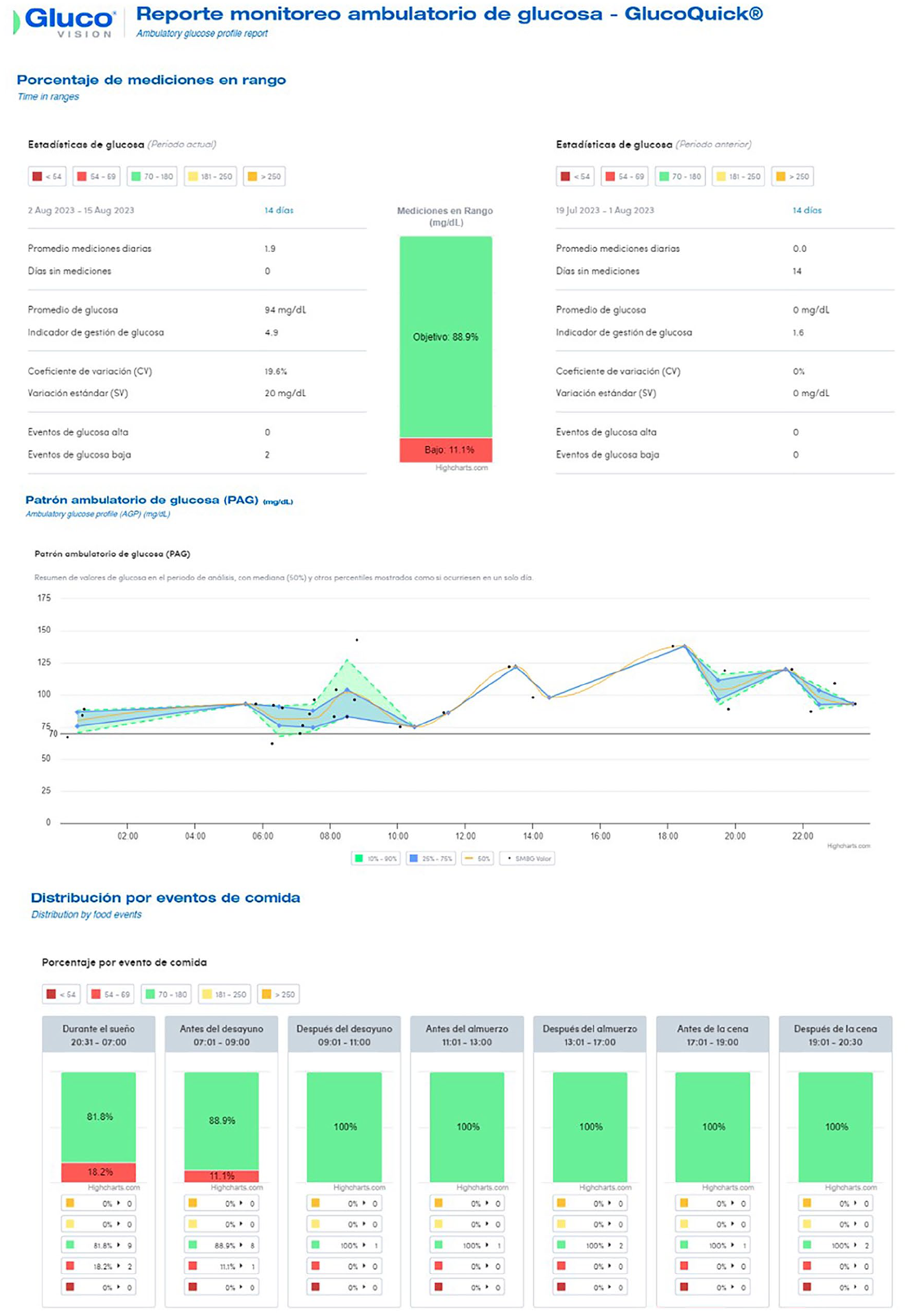
Ambulatory glucose profile (AGP) through the Gluco® vision platform.
A previous study in our center compared the use of mHealth with usual care in patients with T2D treated with insulin at hospital discharge, demonstrating a significant improvement in Hb1Ac levels and adherence to self-monitoring, without increasing hypoglycemia.7,8 However, despite the demonstrated advantages, the use of this technology is low in both developed and developing countries, where the presence of socioeconomic disparities generates additional barriers. 9
The aim of this study is to describe the change in HbA1c levels and incidence of hypoglycemia events in a cohort of patients diagnosed with T2D treated with insulin and followed up through the Zutrics digital ecosystem.
Methods
Analytical observational prospective cohort study evaluating patients with a diagnosis of T2D, treated on an outpatient basis with insulin, and followed up in the endocrinology clinic of the Hospital Universitario San Ignacio in Bogotá, Colombia, between July 2023 and July 2024. Adult patients were included, with basal or multiple daily injections (MDI) and stable treatment in the 3 months prior to enrollment, with HbA1c >8%, and with availability of access to a computer and internet for data downloading. Patients were excluded if they had presented any of the following conditions in the 3 months prior to the start of the study: acute decompensation of diabetes (diabetic ketoacidosis, hyperosmolar state), acute myocardial infarction, stroke, uncontrolled psychiatric disease, as well as pregnant, or breastfeeding women. Additionally, those on glucocorticoid treatment, with renal disease on stages 4 and 5, Child Pugh C liver disease, proliferative retinopathy, severe peripheral neuropathy, or with cognitive impairment that would prevent them from using the application were excluded. Glucose monitoring users were also excluded from the study. All patients signed the informed consent form to participate. The study was approved by the ethics committee of the Hospital Universitario San Ignacio and the Pontificia Universidad Javeriana FM-CIE-0689-19.
Patients who met the inclusion criteria were identified and invited to participate. At the first visit, demographic data, baseline clinical data, history of severe hypoglycemia in the last month, and HbA1c were collected. Subsequently, a 14-day screening phase was carried out during which adherence to self-monitored blood glucose (SMBG) was evaluated, which had to be greater than 80% of the frequency recommended by their healthcare professional in order for the patient to be included in the study. In a second visit (baseline visit), the included patients were instructed on the use of the Zutrics platform (DIABETRICS HEALTHCARE SAS, Barranquilla - Colombia) according to the user manual and the study protocol. In addition, they received education about insulin titration, importance of adherence to self-monitoring and management of hypoglycemia events. Virtual check-ups were carried out by telephone every 2 weeks by a trained doctor who reviewed the data uploaded by the patients into the application, adherence to SMBG, the incidence of hypoglycemia, and adjusted the insulin dose in case of hypoglycemia events (<54 mg/dL and <70 mg/dL) or recurrent hyperglycemia events (>180 mg/dL), according to the insulin used.10,11 Finally, a third site visit was made at the end of 12 weeks of follow-up, during which data on the number and values of glucose values were collected, in addition to information on hypoglycemia events. During the follow-up, there were a total of 3 face-to-face visits and 6 virtual check-ups. All patients performed SMBG with Glucoquick GD50 (DIABETRICS HEALTHCARE SAS, Barranquilla—Colombia).
Baseline and 3-month HbA1c measurements were performed according to the guidelines of the National Glycohemoglobin Standardization Program (NGSP). Additionally, derived Time In Range (TIRd) was calculated from SMBG profiles using the values obtained from the 7-point profile of blood glucose measurements (before and 2 hours after meals such as breakfast, lunch and dinner, and before going to bed) taken during the first week after enrollment and the week before the end of follow-up. 12 The TIRd was calculated, corresponding to the percentage of blood glucose measurements in range (70-180 mg/dL) over the total number of blood glucose measurements taken, according to the ranges suggested by consensus.13,14 Level 1 (<70 mg/dL), level 2 (<54 mg/dL) and level 3 or severe hypoglycemia events were classified according to the definitions of consensus.15,16 Diabetic nephropathy was defined according to the KDIGO classification. 17
The sample size was calculated based on the study performed by Lim et al 18 to detect a difference in means, assuming a reduction in HbA1c level of 0.5%, calculating a power of 80%, and an alpha error of .05, with a standard deviation of 1. The calculated sample size was 70 patients.
Quantitative variables were described by means and standard deviations, or medians and interquartile ranges, depending on whether or not the normality assumption was met. The Shapiro-Wilk test was used to evaluate this assumption. Categorical variables were described by means of absolute numbers and proportions.
For the analysis of the primary outcome (change in HbA1c levels between the initial visit and 3 months after the start of the study), a Wilcoxon test was used considering that this change did not have a normal distribution and that these were repeated measurements in the same patient. Finally, the proportion of patients who met the consensus recommendations was analyzed: TIRd (70-180 mg/dL) >70%, time above range (TBRd >180 mg/dL) <25%, TBRd >250 mg/dL <5%, time below range (TBRd <70 mg/dL) <4%, and TBRd 54 mg/dL <1%.13,14 Statistical analysis was performed using the statistical program STATA 16® (Stata, College Station, Texas, USA).
Results
Of a total of 200 patients invited to participate, 71 patients formed the cohort followed with Zutrics and 69 patients completed the follow-up time. Two patients were excluded from the analysis due to loss to follow-up. The mean age was 62 ± 12.5 years, 56.6% were female, the main complications associated with diabetes were chronic kidney disease (45.1%) and coronary artery disease (25.4%). About 45.1% of the patients included were treated with MDI. The baseline demographic and clinical characteristics are described in Table 1.
Demographic and clinical characteristics of patients at study entry.

Abbreviations: BMI, body mass index; DPP4, dipeptidylpeptidase type 4; DTSQ, Diabetes Treatment Satisfaction Questionnaire; GLP1, glucagon-like peptide type 1; IQR, interquartile range; SD, standard deviation; SGLT2, glucose cotransporter type 2.
Median HbA1c levels decreased from a baseline value of 9.1% (Interquartile range, IQR 7.5, 11.4) to a value of 7.0% (IQR 6.3, 8.08) at 3-month follow-up (P = .044). The median of HbA1c changes over time was −1.3% (IQR −0.13, −4.2).
The mean TIRd of 70 to 180 mg/dL increased 2.1% from 74% at baseline to 76.1% at the end of follow-up. The incidence density of hypoglycemia episodes was 0.009 events/patient-day during the follow-up. The TBRd of <70 mg/dL did not change significantly, going from 0.5% at baseline to 0.94% at the end of follow-up (Figure 2). There was no significant change in insulin requirement during follow-up.
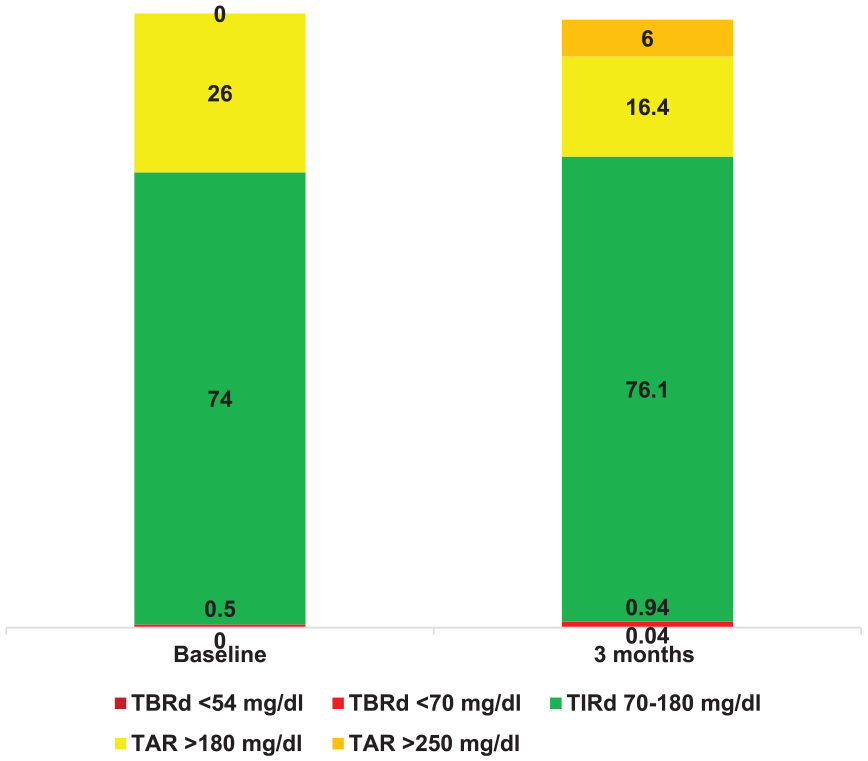
Metrics of glycemic control at baseline and 3-month follow-up.
The time above the derived range (TARd >180 mg/dL and >250 mg/dL) and time below the derived range (TBRd <70 mg/dL and <54 mg/dL) at baseline and at the end of the follow-up are shown in Figure 2. When analyzing the percentage of patients who met the metabolic control goals according to the consensus, it was found that 73.9% achieved TIRd greater than 70%. The proportion of patients who met the goal of TBRd <70 mg/dL less than 4% was 95.6%. The median coefficient of variation was 28.1% (IQR 19.8-46).
Discussion
The present study evaluated the results of the use of the Zutrics digital ecosystem for the follow-up of patients with T2D treated with insulin, achieving better glycemic control, without increasing the frequency of hypoglycemia, strengthening the evidence of the implementation of technology in the management of patients with diabetes in Latin America.
When evaluating the efficacy of the follow-up strategy, this study found a clinically and statistically significant decrease in Hb1Ac. These findings are similar to those reported in other centers in Latin America and developing countries in other regions.19 -23 This may be associated with the fact that the use of technology favored frequent contact with the health care team, achieving faster treatment adjustments, without increasing the number of hypoglycemia events, which in the long run could favor adherence to follow-up. In fact, other studies have shown that the effect on glycemic control is maintained in the long term when this type of strategy is used. 24
A previous study developed in our center showed a decrease in Hb1Ac of up to 2% in patients using mHealth at the time of transition from inpatient to outpatient care and who received weekly monitoring 8 a result similar to that found in this study. Our data suggest that the effect on glycemic control is associated with the frequency of follow-up, and not only with the education received for diabetes self-care by expert staff. Several meta-analyses have highlighted the importance of close follow-up of patients for better glycemic control. 25 In addition, the reduction in HbA1c was not associated with an increase in insulin requirement, which could favor the cost-effectiveness of this type of intervention.26,27
Hypoglycemia is a strong marker of morbidity and mortality in patients with T2D, therefore the reduction of hypoglycemia events is a therapeutic objective.28,29 When evaluating the safety of follow-up with the Zutrics ecosystem, a very low incidence of hypoglycemia events during the follow-up was found. A meta-analysis showed that this type of intervention can decrease hypoglycemia by 40%, as well as improve HbA1c control. 30 Even reductions of up to 86% of hypoglycemia events in patients with type 1 diabetes, who are at increased risk for hypoglycemia events have been reported in the literature. 31 These findings suggest that the implementation of technology in the follow-up of insulin-treated patients can reduce the risk of hypoglycemia, without deterioration of metabolic control.
The digital ecosystem displays metabolic control metrics, including TIRd and TBRd, to the patient, and their healthcare team. In evaluating this cohort, time in range was only assessed at baseline and in the final week. However, the targets for TIRd, TARd and TBRd were met. Although not clinically significant in this study, TIRd increased at the end of follow-up with a reduction in TARd without an increase in hypoglycemia. TBRd values <70 mg and <54 mg/dL were low at baseline and final follow-up, which may be because hypoglycemic events, especially at night, may be missed with SBMG in comparison to continuous glucose monitoring. 32 Williamson et al recently published the results of a telemedicine program and described that integrating time-ranging data can improve glycemic control and self-management skills in patients with T2D administering insulin. 33
When assessing adherence to self-monitoring, the average number of measurements was 2 glucose readings/day compared to other publications where the average number of measurements was 6 glucose readings/day. 34 However, a significant percentage of patients in this study were basal insulin users only, so they did not require more than 2 glucose measurements per day, suggesting that patients with simple treatment regimens in real-life conditions could benefit from this technology.
This study evaluates the use of a digital ecosystem in the follow-up of patients with T2D in a structured education program in Latin American patients, demonstrating that the clinical benefits do not differ from those presented in other countries with better access to technology. In addition, the evidence of the use of this technology in a real-life Latin American population is increasing. However, several limitations of the study should be recognized. First, the number of patients included is relatively small, and from a single center of excellence in diabetes management, so new studies should be conducted in other Latin American settings with larger sample size to confirm our findings. Second, we excluded patients with advanced diabetes-related complications and did not conduct a usability assessment of the platform. Additional studies are needed to assess the impact of the platform in different populations, and the impact on quality of life and long-term device engagement. In addition, the lack of a control group limits the assessment of whether the effects are related to the ecosystem or only to more frequent follow-up, which is not standard practice. Finally, the number of contacts between health care providers and patients was higher in this study. This does not reflect standard clinical practice in Latin America, where patients typically receive only 1 face-to-face visit per month or quarter. However, the changes reported in HbA1c demonstrate the improvement in metabolic control, and the data reported in this study are similar to those reported previously in the literature.
Conclusion
The present study suggests that in patients with T2D treated with insulin, the use of a digital ecosystem such as Zutrics can improve glycemic control without increasing hypoglycemia events, favoring closer contact with the health care team, and rapid treatment adjustments. Further studies with larger sample sizes, less intensive contact schedules and longer follow-up are needed to confirm our findings and to support recommendations for the use of this type of application in our setting.
Footnotes
Acknowledgements
This research was supported as an investigator-initiated study. The glucometers and supplies to perform self-blood glucose monitoring were donated by DIABETRICS HEALTHCARE SAS, Barranquilla—Colombia.
Abbreviations
HbA1c: glycated hemoglobin; CV%: coefficient of variation; GMI: Glucose management indicator; TARd: Derived Time above the range; TBRd: Derived Time below the range; TIRd: Derived Time in range; T2D: Type 2 Diabetes; AGP: Ambulatory Glucose Profile; DTSQ: Diabetes Treatment Satisfaction Questionnaire; SD: standard deviation; IQR: interquartile range; SGLT2: glucose cotransporter type 2; DPP4: dipeptidylpeptidase type 4; GLP1: glucagon-like peptide type 1; BMI: body mass index,
Ethical Considerations
The study was approved by the ethics committee of the Hospital Universitario San Ignacio and the Pontificia Universidad Javeriana FM-CIE-0689-19.
Consent for Publication
Informed consent for analysis and data publication was signed by all patients enrolled in the study.
Author Contributions
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: No source of funding was received for the preparation of this study. The glucometers were donated by DIABETRICS HEALTHCARE SAS, Barranquilla - Colombia.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Ana M. Gómez reports speaker’s fees from NovoNordisk, Elli Lilly, Boeringher Ingelheim, Abbott, and Medtronic. Diana C. Henao reports speaker’s fees from Novo Nordisk, Sanofi, Servier, and Abbott. No other potential conflicts of interest are reported.
Data Availability Statement
Not applicable.
