Abstract
Background:
This post hoc analysis assessed continuous glucose monitoring (CGM)-based metrics with basal insulin icodec (icodec) versus once-daily (OD) comparators in insulin-naive and insulin-experienced Japanese individuals from the global, phase 3a ONWARDS 1, 2, and 4 trials.
Methods:
Double-blinded CGM data were collected from Japanese adults (aged ≥20 years) with type 2 diabetes (T2D) during prespecified assessment periods (weeks 0–4 [ONWARDS 1, 2, and 4], weeks 22–26 [ONWARDS 1, 2, and 4], weeks 48–52 [ONWARDS 1], and weeks 74–78 [ONWARDS 1]). For all assessment periods, percentages of time in range (TIR; sensor glucose [SG] 70–180 mg/dL), time above range (TAR; SG >180 mg/dL and >250 mg/dL), and time below range (TBR; SG <70 mg/dL and <54 mg/dL) were analyzed. Proportions of participants achieving internationally recommended CGM targets were calculated for all assessment periods except weeks 0–4. Duration of overall hypoglycemic episodes (SG <70 mg/dL for ≥15 min) was assessed.
Results:
Across trials, during all assessment periods, mean percentages of TIR, TAR, and TBR were broadly similar between treatment arms. In ONWARDS 1, the proportion of participants achieving >70% TIR was numerically higher with icodec versus OD comparator during all assessment periods; the opposite was observed in ONWARDS 2 and 4. TBR (SG <70 mg/dL and <54 mg/dL) remained below the recommended targets (<4% and <1%). Across trials, in both arms, the median duration of overall hypoglycemic episodes was ≤45 min; most hypoglycemic episodes included no level 2 hypoglycemic periods (SG <54 mg/dL for ≥15 min).
Conclusions:
In Japanese individuals with T2D, CGM-based metrics were similar for icodec and OD comparators. Overall duration of hypoglycemic episodes was ≤45 min for icodec and OD comparators and most hypoglycemic episodes included no or <15 min with SG <54 mg/dL. These data support icodec use across the spectrum of T2D in Japan.
Introduction
The use of continuous glucose monitoring (CGM) technology in randomized clinical trials is growing substantially.1,2 In line with the 2023 international consensus statement on CGM data collection and reporting in clinical trials, the standardized assessment of CGM and subsequent reporting of CGM metrics is highly encouraged. 1 Furthermore, Japanese consensus guidelines also support the use of real-time CGM in investigative studies and clinical practice.3,4
The efficacy and safety of insulin icodec (icodec), a once-weekly basal insulin analog, 5 were evaluated across a spectrum of clinical scenarios in the ONWARDS phase 3a development program, which comprised six trials (ONWARDS 1–6).6–11 ONWARDS 1–5 included participants with type 2 diabetes (T2D) who were insulin-naive (ONWARDS 1, 3, and 5), receiving basal-only insulin treatment (ONWARDS 2), or following a basal-bolus regimen (ONWARDS 4).6–10 All five trials were randomized, multinational trials, with a treatment period of 26 weeks (ONWARDS 2–4), 52 weeks (ONWARDS 5) or 78 weeks (ONWARDS 1).6–10 In these trials, noninferiority (ONWARDS 1–5) and superiority (ONWARDS 1, 2, 3, and 5) of icodec versus once-daily (OD) basal insulin comparators (insulin glargine U100 [glargine U100], insulin glargine U300 [glargine U300], or insulin degludec [degludec]) were demonstrated in terms of HbA1c reduction.6–10
CGM data were collected during prespecified periods of ONWARDS 1, 2, and 4. Post hoc analyses of CGM-based metrics and the duration of CGM-derived hypoglycemic episodes from the global populations of ONWARDS 1, 2, and 4 have been published previously, providing detailed insights into CGM-based glycemic control during icodec initiation, ongoing treatment, and during the discontinuation of icodec treatment.12,13 Given the well-documented differences in the pathophysiology of T2D between individuals of Western (i.e., European and American) and East Asian descent that may impact the efficacy and safety of basal insulin therapy, conducting a similar post hoc analysis of CGM-based metrics in the East Asian populations of ONWARDS 1, 2, and 4 is warranted. 14 Indeed, as opposed to increased insulin resistance, a predisposition toward impaired insulin secretion exists among East Asian populations with T2D, including Japanese individuals, making this the predominant factor in T2D pathogenesis in these populations.14,15 Furthermore, significantly lower fasting serum insulin levels have been reported in Japanese individuals with T2D versus Chinese and Korean individuals with T2D, suggesting that pathophysiological differences in T2D also exist between East Asian populations. 16 Notably, ONWARDS 1, 2, and 4 included Japanese individuals. 17
In the global population of ONWARDS 1, the percentage of time in range (TIR, 70–180 mg/dL [3.9–10.0 mmol/L]) during the end of the main phase (weeks 48–52) was statistically significantly higher with icodec compared with glargine U100 (estimated treatment difference: 4.41% [95% confidence interval: 1.92, 6.90], confirming the superiority of icodec. 6 In ONWARDS 2 and 4, percentage of TIR was comparable between treatment groups during the last 4 weeks of treatment (weeks 22–26).7,9 In the Japanese subpopulation of these trials, similar HbA1c reductions were observed in each trial for icodec and comparators, and the percentage of TIR was comparable between treatment groups during weeks 48–52 (ONWARDS 1) and weeks 22–26 (ONWARDS 2 and 4). 17
The aim of this post hoc analysis was to explore CGM data collected from Japanese individuals with T2D who participated in the global ONWARDS 1, 2, and 4 trials, and to evaluate the effect of icodec and OD comparators on CGM-based metrics and CGM-derived hypoglycemia duration in this subpopulation.
Methods
Trial design and participants
This post hoc analysis used data from the ONWARDS 1, 2, and 4 trials; the trial designs, methods, and statistical analyses have been published previously.5–7,9 Key points are summarized below.
ONWARDS 1, 2, and 4 were randomized, open-label, treat-to-target, phase 3a trials that investigated the efficacy and safety of once-weekly icodec in individuals aged ≥18 years (≥20 years for Japanese individuals) with T2D (Supplementary Fig. S1). ONWARDS 1 comprised a screening period (up to 2 weeks), a 78-week treatment period (52-week main phase and 26-week extension phase), and a 5-week follow-up period after completion of trial treatment. Insulin-naive adults with a screening HbA1c value of 7.0%–11.0% (53.0–96.7 mmol/mol) were randomized 1:1 to icodec or glargine U100. ONWARDS 2 and 4 were 26-week trials with 5-week follow-up periods. In ONWARDS 2, adults previously treated with once- or twice-daily basal insulin, with or without noninsulin glucose-lowering agents, who had a screening HbA1c value of 7.0%–11.0%, were randomized 1:1 to icodec or degludec. In ONWARDS 4, adults with T2D treated with a basal-bolus insulin regimen (screening HbA1c value of 7.0%–10.0%) were randomized 1:1 to basal-bolus regimens with icodec or glargine U100, both in combination with 2–4 daily injections of insulin aspart (aspart).
ONWARDS 1, 2, and 4 were conducted in accordance with the principles of the Declaration of Helsinki, and in compliance with the Good Clinical Practice guidelines of the International Conference for Harmonization. All Japanese individuals who participated in the ONWARDS 1, 2, and 4 trials were included in this post hoc analysis.
Treatments
Across trials, participants randomized to the icodec arm received insulin pens with 700 U/mL of icodec (Novo Nordisk; prefilled pen injector) to be used subcutaneously. Icodec and OD comparator doses were titrated weekly based on three prebreakfast self-measured blood glucose (SMBG) values (measured 2 days before titration and on the day of the titration) to achieve a target of 80–130 mg/dL [4.4–7.2 mmol/L]).
In ONWARDS 1, once-weekly icodec was initiated at a starting dose of 70 U/week, while the OD comparator basal insulin (glargine U100; Sanofi-Aventis) was initiated at a starting dose of 10 U/day. In ONWARDS 2 and 4, for individuals switching from daily basal insulin to once-weekly icodec, the weekly icodec dose was calculated by multiplying the pretrial daily basal insulin dose by seven. For the first injection only, a one-time additional 50% dose of icodec was also administered in ONWARDS 2 and 4. In ONWARDS 4, in both treatment arms, the switch from the participant’s previous bolus insulin to aspart was done unit-to-unit per meal. Adjustments in aspart dose during the first 8 weeks were only permitted for safety reasons. Thereafter, aspart dose adjustments were made at intervals of 3–4 days based on preprandial and bedtime SMBG values. Across trials, background therapy with noninsulin glucose-lowering medications was permitted during the trial, except for sulfonylureas and glinides, which were discontinued at randomization.
CGM assessments
In ONWARDS 1, 2, and 4, participants wore a blinded CGM device (Dexcom G6; Dexcom, Inc., San Diego, CA, USA) during the following prespecified trial periods: weeks 0–4 (ONWARDS 1, 2, and 4), weeks 22–26 (ONWARDS 1, 2, and 4), weeks 48–52 (ONWARDS 1), and weeks 74–78 (ONWARDS 1). For a participant to be included in the summary of CGM glycemic control endpoints, ≥70% of the planned CGM measurements had to be available for the period assessed. 18
Post hoc evaluation
CGM-based metrics during weeks 22–26 (ONWARDS 2 and 4), weeks 48–52 (ONWARDS 1) and weeks 74–78 (ONWARDS 1) have been previously published. 17 In this post hoc analysis, CGM-based metrics collected during treatment initiation (weeks 0–4) in ONWARDS 1, 2, and 4 and during the mid-trial period of ONWARDS 1 (weeks 22–26) were analyzed. Duration of CGM-derived hypoglycemic episodes was also evaluated across all CGM assessment periods.
Post hoc evaluation: CGM-based glycemic management
For all prespecified assessment periods in each trial, the CGM-based percentage of TIR (defined as sensor glucose 70–180 mg/dL [3.9–10.0 mmol/L]), time in tight range (TITR; sensor glucose 70–140 mg/dL [3.9–7.8 mmol/L]), time above range (TAR; sensor glucose >180 mg/dL [>10.0 mmol/L] and >250 mg/dL [>13.9 mmol/L]), and time below range (TBR; sensor glucose <70 mg/dL and <54 mg/dL [<3.9 mmol/L and <3.0 mmol/L]) were analyzed. Additionally, the proportion of participants achieving the TIR target (>70%), the TAR (sensor glucose >180 mg/dL [>10.0 mmol/L]) target (<25%), the TAR (sensor glucose >250 mg/dL [>13.9 mmol/L]) target (<5%), the TBR (sensor glucose <70 mg/dL [<3.9 mmol/L]) target (<4%), the TBR (<54 mg/dL [<3.0 mmol/L]) target (<1%), and the double composite CGM target {>70% TIR and <4% TBR [<70 mg/dL (<3.9 mmol/L)]} were determined for all prespecified assessment periods in each trial, except for weeks 0–4 (since steady-state kinetics were not yet reached).
The proportion of participants achieving the glycemic variability target (within-participant variability ≤36%), evaluated using the coefficient of variation (CV%), was assessed during all prespecified assessment periods.
Post hoc evaluation: CGM-derived duration of hypoglycemia
CGM-derived overall hypoglycemic episodes (level 1 and 2) were defined as a sensor glucose value <70 mg/dL (3.9 mmol/L) for ≥15 consecutive minutes, ending when sensor glucose returned to ≥70 mg/dL (≥3.9 mmol/L) for ≥15 consecutive minutes (Supplementary Fig. S2). A clinically significant (level 2) hypoglycemic period was defined as sensor glucose <54 mg/dL (<3.0 mmol/L) for ≥15 consecutive minutes, ending when sensor glucose returned to ≥54 mg/dL (≥3.0 mmol/L) for ≥15 consecutive minutes. Multiple periods with sensor glucose <54 mg/dL (<3.0 mmol/L) may occur within a single CGM-derived overall hypoglycemic episode.
The duration of CGM-derived overall hypoglycemic episodes (sensor glucose <70 mg/dL [3.9 mmol/L]) was assessed for all prespecified CGM assessment periods. Overall hypoglycemic episodes (sensor glucose <70 mg/dL [3.9 mmol/L]) were classified based on the time spent with sensor glucose <54 mg/dL (<3.0 mmol/L) (no time, >0 to <15 consecutive minutes, or ≥15 consecutive minutes).
Statistical analysis
Descriptive statistical summaries were provided for percentages of TIR, TAR, and TBR. The percentage of time spent in a given range was defined as the number of recorded measurements in a given range, divided by the total number of recorded measurements, multiplied by 100. Descriptive summaries were also provided for the proportion of participants meeting CGM targets. For hypoglycemia episode duration, median and interquartile range (IQR) were calculated across all episodes for each assessment period.
Results
Participants
This post hoc analysis included the Japanese participants from ONWARDS 1 (icodec, n = 78; glargine U100, n = 86), ONWARDS 2 (icodec, n = 51; degludec, n = 49), and ONWARDS 4 (icodec, n = 44; glargine U100, n = 41).
Baseline characteristics for the Japanese subgroups of ONWARDS 1, 2, and 4 have been described previously. 17 Briefly (all given as mean [SD]), in ONWARDS 1, the age was 58.8 (10.2) years, diabetes duration was 12.6 (7.2) years, and baseline HbA1c was 8.03% (0.83%). In ONWARDS 2, the age was 62.4 (9.7) years, diabetes duration was 17.2 (9.0) years, and baseline HbA1c was 8.04% (0.72%). In ONWARDS 4, the age was 60.7 (11.3) years, diabetes duration was 19.2 (9.8) years, and baseline HbA1c was 8.02% (0.77%).
CGM-based glycemic metrics
CGM-based metrics from weeks 0–4 of ONWARDS 1, 2, and 4 and weeks 22–26 of ONWARDS 1 are summarized in Figure 1, alongside those previously published for weeks 22–26 (ONWARDS 2 and 4), weeks 48–52 (ONWARDS 1), and weeks 74–78 (ONWARDS 1). 17 CGM-based metrics by treatment day after icodec injection or OD comparator titration during weeks 0–4 (ONWARDS 1, 2, and 4), weeks 22–26 (ONWARDS 1, 2, and 4), weeks 48–52 (ONWARDS 1), and weeks 74–78 (ONWARDS 1) are summarized in Figure 2. Summary ambulatory glucose profiles of CGM measurements during weeks 0–4 (ONWARDS 1, 2, and 4), weeks 22–26 (ONWARDS 1, 2, and 4), weeks 48–52 (ONWARDS 1), and weeks 74–78 (ONWARDS 1) are shown in Supplementary Figure S3.

Continuous glucose monitoring (CGM) metrics in Japanese participants from the
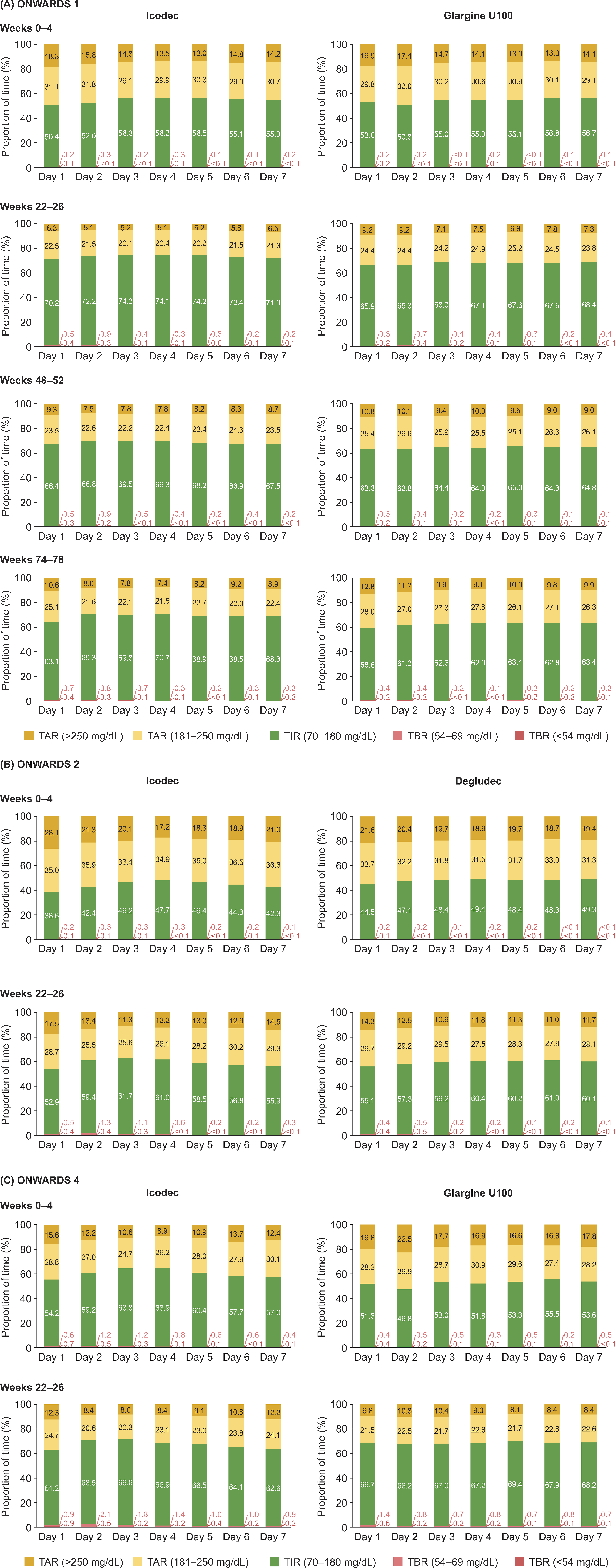
CGM metrics by day after icodec injection or once-daily (OD) comparator titration in Japanese participants from the
ONWARDS 1 (insulin-naive participants)
During all prespecified CGM assessment periods, mean percentage of TIR, TAR, and TBR were similar between treatment arms (Fig. 1A). Mean percentage of TIR was above the recommended target (>70%) during weeks 22–26 with icodec, but not with glargine U100. The mean percentage of TIR was beneath the recommended target of >70% in both treatment arms during weeks 0–4, weeks 48–52, and weeks 74–78. Mean percentage of TAR was above the recommended target (<25%) during all CGM assessment periods in both treatment arms. In all CGM assessment periods, mean percentage of TBR (<70 mg/dL and <54 mg/dL [<3.9 mmol/L and <3.0 mmol/L]) remained below the recommended target (<4% and <1%) in both treatment arms. The mean percentage of TITR was numerically higher with icodec than with glargine U100 during all CGM assessment periods.
During all CGM assessment periods, mean percentage of TIR remained relatively stable throughout the week after icodec injection or OD comparator titration (Fig. 2A). In both treatment arms, minor fluctuations in mean percentage of TAR were observed during all CGM assessment periods; however, mean percentage of TBR remained relatively stable throughout the week. During all CGM assessment periods, the median CGM measured glucose level tended to be above the target range (>180 mg/dL [>10.0 mmol/L]) for fewer h throughout the day with icodec than with glargine U100 (Supplementary Fig. S3A).
ONWARDS 2 (insulin-experienced [basal-only] participants)
In ONWARDS 2, during weeks 0–4 and weeks 22–26, the mean percentage of TIR, TAR, and TBR were broadly similar between treatment arms (Fig. 1B). During both CGM assessment periods, in both treatment arms, the mean percentage of TIR was beneath the recommended target of >70%; however, the mean percentage of TBR (<70 mg/dL and <54 mg/dL [<3.9 mmol/L and <3.0 mmol/L]) was below the recommended targets of <4% and <1%, respectively. The mean percentage of TAR was above the recommended target of <25% during both CGM assessment periods in both treatment arms. The mean percentage of TITR was numerically lower with icodec than with degludec during weeks 0–4, but was similar between arms during weeks 22–26.
During weeks 0–4 and weeks 22–26, the mean percentage of TIR remained relatively stable throughout the week in both treatment arms (Fig. 2B). In both CGM assessment periods, there were minor fluctuations in the mean percentage of TAR and TBR (<70 mg/dL and <54 mg/dL [<3.9 mmol/L and <3.0 mmol/L]) in both treatment arms, with the mean percentage of TBR (<70 mg/dL [3.9 mmol/l]) tending to be slightly higher on days 2 and 3 following icodec injection compared with other days of the week during weeks 22–26. During both CGM assessment periods, the median glucose level was above the target range (>180 mg/dL [>10.0 mmol/L]) for several h throughout the day in both treatment arms (Supplementary Fig. S3B).
ONWARDS 4 (insulin-experienced [basal-bolus] participants)
In ONWARDS 4, during weeks 0–4 and weeks 22–26, the mean percentage of TIR, TAR, and TBR were broadly similar between treatment arms (Fig. 1C). During both CGM assessment periods, in both treatment arms, the mean percentage of TIR was beneath the recommended target of >70%; however, the mean percentage of TBR (<70 mg/dL and <54 mg/dL [<3.9 mmol/L and <3.0 mmol/L]) was within the recommended targets of <4% and <1%, respectively. In both treatment arms, the mean percentage of TAR was above the recommended target of <25% during both CGM assessment periods. The mean percentage of TITR was numerically higher with icodec than with glargine U100 during weeks 0–4, but numerically lower with icodec than with glargine U100 during weeks 22–26.
During weeks 0–4 and weeks 22–26, the mean percentage of TIR remained relatively stable throughout the week in both treatment arms (Fig. 2C). In both CGM assessment periods, there were minor fluctuations in the mean percentage of TAR and TBR (<70 mg/dL and <54 mg/dL [<3.9 mmol/L and <3.0 mmol/L]) in both treatment arms, with the mean percentage of TBR (<70 mg/dL [3.9 mmol/l]) tending to be slightly higher on days 2–4 following icodec injection compared with other days of the week during weeks 22–26. During weeks 0–4, the median CGM-measured glucose level was above the target range (>180 mg/dL [>10.0 mmol/L]) for fewer h throughout the day with icodec compared with glargine U100; in contrast, during weeks 22–26, the median glucose level remained within range (70–180 mg/dL [3.9–10.0 mmol/L]) throughout the day in both treatment arms (Supplementary Fig. S3C).
Proportion of participants achieving CGM targets
For ONWARDS 1, 2, and 4, the proportions of participants achieving CGM targets are summarized in Table 1.
Proportion of Participants Meeting Criteria for TIR, TBR, TAR, the Double Composite CGM Target, and Within-Person Variability ≤36%

For a participant to be included in the summary of CGM glycemic control endpoints, ≥70% of the planned CGM measurements had to be available for the period assessed; the number of participants meeting this criterion was used as the denominator to calculate the proportions of participants achieving the CGM targets. 18
CGM, continuous glucose monitoring; CV, coefficient of variation; degludec, insulin degludec; FAS, full analysis set; glargine U100, insulin glargine U100; icodec, insulin icodec; TAR, time above range; TBR, time below range; TIR, time in range.
ONWARDS 1 (insulin-naive participants)
The proportions of participants achieving >70% TIR were numerically higher with icodec than with glargine U100 during weeks 22–26, 48–52, and 74–78 (60%, 44%, and 55% with icodec, and 45%, 38%, and 34% with glargine U100, respectively). During weeks 22–26, 48–52, and 74–78, the proportions of participants achieving the recommended target of <25% TAR were 55%, 39%, and 43% with icodec, and 35%, 24%, and 25% with glargine U100, respectively. Most participants (>95%) in both treatment arms achieved <4% TBR (<70 mg/dL [3.9 mmol/L]) and <1% TBR (<54 mg/dL [<3.0 mmol/L]). During all CGM assessment periods, the proportion of participants achieving the double composite CGM target {>70% TIR and <4% TBR [<70 mg/dL (<3.9 mmol/L)]} was numerically higher with icodec than with glargine U100. The proportion of participants achieving a CV% of ≤36% for within-participant glycemic variability was broadly similar between treatment arms during all CGM assessment periods.
ONWARDS 2 (insulin-experienced [basal-only] participants)
The proportion of participants achieving >70% TIR during weeks 22–26 was numerically lower with icodec than with degludec (28% and 35%, respectively). During weeks 22–26, in both treatment arms, ≤22% of participants achieved <25% TAR; meanwhile, ≥96% and ≥96% of participants achieved the recommended targets of <4% TBR (<70 mg/dL [3.9 mmol/L]) and <1% TBR (<54 mg/dL [<3.0 mmol/L]), respectively. The proportion of participants achieving the double composite CGM target {>70% TIR and <4% TBR [<70 mg/dL (<3.9 mmol/L)]} during weeks 22–26 was numerically lower in the icodec arm than in the degludec arm (26% and 35%, respectively). During weeks 22–26, most participants achieved a CV% of ≤36% for within-participant glycemic variability in both treatment arms (icodec: 80%, degludec: 90%).
ONWARDS 4 (insulin-experienced [basal-bolus] participants)
The proportion of participants achieving >70% TIR during weeks 22–26 was 38% with icodec and 60% with glargine U100. During weeks 22–26, the proportion of participants achieving <25% TAR was 33% with icodec and 48% with glargine U100; however, ≥83% and ≥85% of participants achieved the recommended targets of <4% TBR (<70 mg/dL [3.9 mmol/L] and <1% TBR (<54 mg/dL [<3.0 mmol/L]), respectively. The proportion of participants achieving the double composite CGM target {>70% TIR and <4% TBR [<70 mg/dL (<3.9 mmol/L)]} during weeks 22–26 was numerically lower with icodec than with glargine U100. Furthermore, the proportion of participants achieving a CV% of ≤36% for within-participant glycemic variability was numerically lower with icodec than with glargine U100 (icodec: 63%, glargine U100: 75%).
CGM-derived hypoglycemia
ONWARDS 1 (insulin-naive participants)
In ONWARDS 1, during all CGM assessment periods, the median durations of overall hypoglycemic episodes were within the range of 30–40 mins for both treatment arms (Fig. 3A). Furthermore, during all CGM assessment periods, most overall hypoglycemic episodes in both treatment arms did not include any time spent with sensor glucose <54 mg/dL (<3.0 mmol/L) (Fig. 4A). The percentage of overall hypoglycemic episodes with ≥15 consecutive minutes spent with sensor glucose <54 mg/dL (<3.0 mmol/L) with icodec versus glargine U100 was 15.3% versus 17.2% during weeks 0–4; 23.3% versus 23.7% during weeks 22–26; 17.5% versus 30.9% during weeks 48–52; and 22.2% versus 26.0% during weeks 74–78.

Duration of overall CGM-derived hypoglycemic episodes (sensor glucose <70 mg/dL) from the (A) ONWARDS 1, (B) ONWARDS 2, and (C) ONWARDS 4 trials. Duration of overall CGM-derived hypoglycemic episodes was assessed during weeks 0–4 (ONWARDS 1, 2, and 4), weeks 22–26 (ONWARDS 1, 2, and 4), weeks 48–52 (ONWARDS 1), and weeks 74–78 (ONWARDS 1). Plots show median (IQR). CGM, continuous glucose monitoring; degludec, insulin degludec; glargine U100, insulin glargine U100; icodec, insulin icodec; IQR, interquartile range.

Classification of CGM-derived overall hypoglycemic episodes (sensor glucose <70 mg/dL) by time spent with sensor glucose <54 mg/dL (no time, <15 consecutive minutes, or ≥15 consecutive minutes) from the (A) ONWARDS 1, (B) ONWARDS 2, and (C) ONWARDS 4 trials. CGM-derived overall hypoglycemic episodes were classified during weeks 0–4 (ONWARDS 1, 2, and 4), weeks 22–26 (ONWARDS 1, 2, and 4), weeks 48–52 (ONWARDS 1), and weeks 74–78 (ONWARDS 1). CGM, continuous glucose monitoring; degludec, insulin degludec; glargine U100, insulin glargine U100; icodec, insulin icodec.
ONWARDS 2 (insulin-experienced [basal-only] participants)
In ONWARDS 2, during weeks 0–4 and 22–26, the median durations of overall hypoglycemic episodes were within the range of 30–45 mins for both treatment arms (Fig. 3B). Furthermore, during both CGM assessment periods, most overall hypoglycemic episodes in both treatment arms did not include any time spent with sensor glucose <54 mg/dL (<3.0 mmol/L) (Fig. 4B). The percentage of overall hypoglycemic episodes with ≥15 consecutive minutes spent with sensor glucose <54 mg/dL (<3.0 mmol/L) with icodec versus degludec was 28.1% versus 11.3% during weeks 0–4, and 21.0% versus 36.9% during weeks 22–26.
ONWARDS 4 (insulin-experienced [basal-bolus] participants)
In ONWARDS 4, during weeks 0–4 and 22–26, the median durations of overall hypoglycemic episodes were within the range of 30–40 mins for both treatment arms (Fig. 3C). Furthermore, during both CGM assessment periods, most overall hypoglycemic episodes in both treatment arms did not include any time spent with sensor glucose <54 mg/dL (<3.0 mmol/L) (Fig. 4C). The percentage of overall hypoglycemic episodes with ≥15 consecutive minutes spent with sensor glucose <54 mg/dL (<3.0 mmol/L) with icodec versus glargine U100 was 27.4% versus 18.5% during weeks 0–4, and 22.6% versus 20.8% during weeks 22–26.
Discussion
This post hoc analysis evaluated CGM data from Japanese individuals with T2D in the ONWARDS 1, 2, and 4 trials. In all three trials, during all CGM assessment periods, percentages of TIR, TAR, and TBR were broadly similar between treatment arms, consistent with findings from the respective global populations.12,13 When assessed by day, trends in mean percentages of TIR, TAR, and TBR throughout the week after icodec injection or OD comparator titration were also broadly comparable to those observed in the respective global populations.12,13 In all three trials, across all CGM assessment periods and in both treatment arms, the proportion of participants achieving >70% TIR and <25% TAR was relatively low (≤60% and ≤55%, respectively). Reassuringly, across the three trials and during all CGM assessment periods, percentage of TBR (sensor glucose <70 mg/dL and <54 mg/dL [<3.9 mmol/L and <3.0 mmol/L]) remained very low and well beneath the recommended targets (<4% and <1%). 1 In addition, mean TBR (sensor glucose <54 mg/dL [<3.0 mmol/L]) was between 0 and 3 min per day in all three trials during all CGM assessment periods for both icodec and OD comparators. Across trials, the median duration of overall hypoglycemic episodes was ≤45 min. Moreover, in all three trials, the majority of overall hypoglycemic episodes did not include any level 2 hypoglycemic periods (sensor glucose <54 mg/dL [<3.0 mmol/L] for ≥15 consecutive minutes).
In a database study of unblinded CGM data from 1695 Japanese individuals with T2D using intermittently scanned CGM devices, percentage of TIR did not meet the recommended target (>70%) in individuals receiving multiple daily insulin injections (MDIs) (68.8%), but exceeded the target in those receiving basal-only insulin treatment (71.2%). 19 Although direct comparison between studies is not feasible due to differences in design, treatment modalities, and CGM devices, these findings are broadly in line with those observed in the Japanese subgroup of ONWARDS 4 (basal-bolus population), wherein the percentage of TIR was also beneath the recommended target (>70%). This observation may be expected in individuals with long-standing T2D who are receiving MDIs, as loss of pancreatic β-cell secretory function is a key feature of advanced T2D, resulting in a reduction in endogenous insulin and increased glycemic variability.20,21 As observed in the Japanese subgroups of ONWARDS 2 and 4, the percentage of TAR (>180 mg/dL [>10.0 mmol/L]) reported by Ogawa et al. (2024) was above the recommended target (<25%) in individuals with T2D receiving MDIs (27.1%) and basal-only insulin treatment (25.7%). 19 As the trend in percentage of TAR exceeding the recommended target (<25%) was observed in the Japanese subgroups of ONWARDS 2 (basal-only population) and ONWARDS 4 (basal-bolus population), and in the previous study, it may be attributed to pathophysiological factors (e.g., comorbidities), or lifestyle and socioeconomic characteristics (e.g., education, diet, exercise), rather than a specific intervention. Indeed, in ONWARDS 1, 2, and 4, relatively lower weekly icodec doses were observed across the Japanese subgroups during the last 2 weeks of treatment compared with the respective global populations, which may be a result of higher insulin sensitivity and residual insulin production in Japanese individuals with T2D.6,7,9,17 It may also be that Japanese clinicians place a greater focus on the prevention of hypoglycemia (i.e., by reducing an individuals’ percentage of TBR) than on maintaining an optimal percentage of TAR, especially in individuals with T2D who are at greater risk of negative outcomes following a hypoglycemic episode (i.e., due to age or comorbidities). Concordantly, Japanese guidelines recommend that glycemic targets be individualized in elderly individuals, and that the lower limit of the HbA1c target should be applied to those who are at risk of severe hypoglycemia. 22 Indeed, in the Japanese trial populations, no severe hypoglycemic episodes were reported in ONWARDS 1 and 2, and only a single severe hypoglycemic episode was reported in the icodec arm of ONWARDS 4. 17
In ONWARDS 1, across all CGM assessment periods, a numerically higher proportion of participants achieved the double composite CGM target (>70% TIR and <4% TBR) in the icodec arm versus the glargine U100 arm. These data are consistent with findings previously reported for the global population of ONWARDS 1, 12 and demonstrate that achievement of the double composite CGM target is maintained in the Japanese subgroup, regardless of the pathophysiological differences in T2D between Western (i.e., European and American) and East Asian populations. 14 However, the opposite trend can generally be seen in ONWARDS 2 and 4 for the double composite CGM target; across both CGM assessment periods, in both arms, numerically lower proportions of participants achieved the target with icodec than with the OD comparator. The double composite CGM target was not evaluated for the global populations of ONWARDS 2 and 4. 13
Across the three trials, during all CGM assessment periods and in both treatment arms, a majority of participants achieved a CV% ≤36% for within-person glycemic variability, indicating that most Japanese individuals receiving icodec had stable glucose levels per the international consensus definition. 18 This finding suggests that icodec may be an effective treatment option for maintaining glycemic control in Japanese individuals.
In ONWARDS 1, 2, and 4, across all CGM assessment periods, the median duration of overall hypoglycemic episodes reported for the Japanese subgroup was ≤45 min and thus was not considered prolonged compared with the OD comparators, and well below the international consensus definition (120 min). 18 Furthermore, in all three trials, and across all CGM assessment periods, the median duration of overall hypoglycemic episodes was the same as or similar to that reported for the respective global populations.12,13 These findings suggest that hypoglycemia duration was generally similar between Japanese and Western individuals receiving icodec in the ONWARDS 1, 2, and 4 trials, despite the pathophysiological differences that have been reported between individuals with T2D in East Asian and Western populations. 14
In all three trials, during all CGM assessment periods, most overall hypoglycemic episodes in both treatment arms did not include any periods with sensor glucose <54 mg/dL for ≥15 consecutive minutes and were therefore not considered clinically significant per the international consensus statement definition. 1
A key strength of this analysis is the inclusion of data from three global, randomized phase 3a trials that enrolled people across the spectrum of T2D, including individuals with T2D who were insulin-naive (ONWARDS 1), previously treated with a basal insulin (ONWARDS 2), or previously treated with a basal-bolus insulin regimen (ONWARDS 4). A further strength of this analysis is that CGM data from all three trials were double-blinded, reducing the potential bias that could have resulted from its use. However, this was a post hoc analysis that was not prespecified.
Conclusions
In this post hoc analysis of insulin-naive and insulin-experienced Japanese individuals with T2D from the ONWARDS 1, 2, and 4 trials, percentage of TIR, TAR, and TBR were broadly similar between treatment arms across CGM assessment periods. In addition, the median durations of overall hypoglycemic episodes were within the range of 30–45 min for once-weekly icodec and OD comparators, and most overall hypoglycemic episodes included no level 2 hypoglycemic periods (sensor glucose <54 mg/dL [3.0 mmol/L] for ≥15 consecutive minutes). Overall, findings from the Japanese subgroup broadly align with those from the global trial populations, providing support for the use of icodec across the spectrum of T2D in Japan.
Authors’ Contributions
R.N., T.M.P.R., Y.H., T.K., T.N., T.Y., and H.W. were involved in the conception, design, and conduct of the study, the interpretation of the results, and the writing of the article. All authors edited, reviewed, and approved the final version of the article.
Footnotes
Acknowledgments
Medical writing support was provided by Chloe Fletcher, MSc, of Oxford PharmaGenesis, Oxford, UK, with funding from Novo Nordisk.
Author Disclosure Statement
R.N. received consulting/lecture fees from Abbott Japan LLC, Astellas Pharma Inc., AstraZeneca K.K., Eli Lilly Japan K.K., Kissei Pharmaceutical Co. Ltd, Kowa Company Ltd., Medtronic Japan Co. Ltd, Nippon Boehringer Ingelheim Co. Ltd, Novo Nordisk Pharma Ltd, Ono Pharmaceutical Co. Ltd, Sanofi K.K., Sumitomo Pharma Co. Ltd, and Teijin Pharma Ltd; and research funding/grants from Abbott Japan LLC, Mitsubishi Electric Corporation, Nippon Boehringer Ingelheim Co. Ltd, Ono Pharmaceutical Co. Ltd, Sumitomo Pharma Co. Ltd, and Taisho Pharmaceutical Co. Ltd. T.M.P.R. is an employee of Novo Nordisk A/S and holds stock options in Novo Nordisk. Y.H. and T.N. are employees of Novo Nordisk Pharma Ltd. T.K. has received honoraria for lectures from Eli Lilly, Boehringer Ingelheim, Novo Nordisk Pharma, MSD, Sanofi, Astellas Pharma, Abbott Japan, Teijin Pharma, Kowa, Terumo, Mitsubishi Tanabe Pharma, Sumitomo Pharma, Taisho Pharmaceutical, Ono Pharmaceutical and Fujifilm Toyama Chemical; article fees from Boehringer Ingelheim, Abbott Japan, Novo Nordisk Pharma, Teijin Pharma and Fujifilm Toyama Chemical; scholarship grants from Eli Lilly, Boehringer Ingelheim Japan, Mitsubishi Tanabe Pharma, Taisho Pharmaceutical, and Sumitomo Pharma; and an affiliation with an Endowed Department at Asahi Life Insurance. T.Y. has received honoraria for lectures from Abbott Japan, ARKRAY, Astellas Pharma, AstraZeneca, Bayer, Boehringer Ingelheim, Cosmic Corporation, Covidien Japan (Medtronic Japan Co. Ltd), QueDaiichi Sankyo, Dojindo Laboratories, Eli Lilly, FUJIFILM Toyama Chemical Co. Ltd, Kissei, Kowa Company Ltd, Kyorin Pharmaceutical, Kyowa Kirin Company Ltd, LifeScan Japan, Mitsubishi Tanabe Pharma, Merck & Co., Musashino Foods Co. Ltd, Nippon Becton Dickinson, Novartis, Novo Nordisk, Ono, Roche DC Japan, Sanofi, Sanwa Chemistry Laboratory, Sumitomo Dainippon, Taisho, Takeda Pharmaceutical Company, Teijin Healthcare, and Terumo Co.; and scholarship grants from Aero Switch Therapeutics Inc., Asahi Mutual Life Insurance Company, Astellas Pharma, Bayer, Daiichi Sankyo, EA Pharma, Eli Lilly, Kowa, Kowa Company Ltd., Kyowa Kirin Company Ltd, Minophagen, Mitsubishi Corporation Life Sciences, Mitsubishi Tanabe Pharma, Mitsubishi Tanabe Pharma Corporation, MSD, Nippon Boehringer Ingelheim Co. Ltd, Nipro Corporation, Nitto Boseki Co. Ltd, Novartis, Novo Nordisk, Novo Nordisk Pharma Ltd, Ono, Sanofi, Shionogi & Co. Ltd, Sumitomo Dainippon Pharma Co. Ltd, Taisho Pharmaceutical Co., Takeda Pharmaceutical Company, Tosoh Corporation, and Viatris Pharmaceutical Japan Inc. H.W. has acted as an advisory board member for Novo Nordisk, as a speaker for Astellas Pharma, AstraZeneca, Eli Lilly Japan, FUJIFILM Pharma, Kissei Pharmaceutical, Kowa Pharmaceutical, Merck Sharp & Dohme, Mitsubishi Tanabe Pharma, Nippon Boehringer Ingelheim, Novartis, Novo Nordisk, Ono Pharmaceutical, Sanofi, Sanwa Kagaku Kenkyusho, Sumitomo Dainippon Pharma, and Takeda Pharmaceutical, and has received grants from Astellas Pharma, AstraZeneca, Daiichi Sankyo, Eli Lilly Japan, Johnson & Johnson, Kowa, Kyowa Hakko Kirin, Merck Sharp & Dohme, Mitsubishi Tanabe Pharma, Mochida Pharmaceutical, Nippon Boehringer Ingelheim, Novartis Pharma, Novo Nordisk Pharma, Ono Pharmaceutical, Pfizer Japan, Sanofi, Sumitomo Dainippon Pharma, Taisho Toyama Pharmaceutical, Takeda Pharmaceutical, and Terumo.
Funding Information
This study was funded by Novo Nordisk.
Supplemental Material
Supplemental Material
Supplemental Material
Supplemental Material
Abbreviations
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
