Abstract
Most individuals with ventricular septal defect survive to adulthood which allows time for other complications such as pulmonary arterial hypertension to gradually develop over a period of time. When there are other associated cardiac conditions that also contribute to the development of pulmonary hypertension such as valvular heart disease, the pulmonary hypertension may be exaggerated. Because these different etiologies of the pulmonary hypertension have different mechanisms, their coexistence can complicate patient management. We present a 26-year-old man with a large ventricular septal defect and rheumatic mitral valve disease who developed severe pulmonary hypertension that became complicated by atrial fibrillation and later sudden cardiac death.
Keywords
Introduction
Ventricular septal defect (VSD) is the commonest congenital heart disease (CHD) at birth with a prevalence of 0.3% in children 0 to 6 years old. 1 Unrepaired VSD carries a higher risk of death than repaired VSD; however, most children with unrepaired VSD survive to adulthood. 2 A common complication of CHD is pulmonary hypertension which occurs in 4.2% of CHD cases in general and 6.1% of patients with septal defects 1 with up to 22% in patients with VSD in particular. 3 Half of those with VSD who develop pulmonary hypertension later develop Eisenmenger syndrome. 4 VSD may be isolated or may occur with other congenital heart diseases with up to half of patients with VSD having associated congenital mitral valve incompetence. 5 Mitral valve disease associated with VSD may also be acquired following VSD repair which occurs in 13% irrespective of the method used, be it surgical or transcatheter repair. 6 Acquired mitral valve disease in VSD may also be of rheumatic etiology, mostly mitral stenosis7,8 although up to 20% of cases of rheumatic mitral valve disease in general may present with only isolated mitral regurgitation. 7 Regardless of the etiology of the mitral regurgitation, significant mitral valve regurgitation can lead to pulmonary hypertension with pulmonary artery systolic pressure (PASP) >50 mmHg occurring in 23% of cases. 9
Patients with VSD who again develop mitral valve disease are at dual risk of developing severe pulmonary hypertension from both precapillary and post capillary physiologies increasing the risk of adverse outcomes. In sub-Saharra Africa where rheumatic heart disease is common, the association of rheumatic mitral stenosis with atrial septal defect (Lutembacher syndrome)10 -14 and that of VSD have also been reported. 14 However, the occurrence of VSD and rheumatic mitral valve disease with isolated regurgitation has not been reported. We report a case of a young man with a large VSD and rheumatic mitral regurgitation which became complicated with very severe pulmonary hypertension, paroxysmal atrial fibrillation and death.
Case Presentation
Twenty-six-year-old man was referred for cardiac evaluation at a private cardiology clinic in Kumasi, Ghana with complain of palpitation, progressive shortness of breath and easy fatiguability over 15 years which; had worsened over the 3 months prior to presentation. He mentioned he had been diagnosed with “whole in heart” 15 years earlier at age 11 at another hospital when he presented there with similar symptoms. He had however defaulted follow up and no repair of the “whole in heart” was done. There was orthopnea and paroxysmal nocturnal dyspnea but no chest pains or syncope. He had been started on oral bisoprolol 2.5 mg daily and digoxin 125 µg daily a week earlier when he visited another hospital because of worsening of his symptoms.
On examination, he looked ill, afebrile, anicteric, not pale, not cyanosed, had no clubbing or any peripheral stigmata of infective endocarditis but had minimal pitting bipedal edema. His pulse rate was 110 beats per minute (bpm), regular, weak volume and non-collapsing with blood pressure of 110/72 mmHg. His jugular venous pressure was raised to 12 cm of water. His apex beat was normal but had right parasternal heave and a thrill in the left upper sternal border. He had normal S1 with fixed split S2 with loud P2 component. There was also a grade 3 holosystolic murmur best heard at the left lower sternal border and an early diastolic mummer best heard at the left upper sternal border and had bibasal fine crackles. He had ascites and a tender hepatomegaly which was non-pulsating. His neurological and musculoskeletal systems examination were normal.
His ECG (Figure 1) showed fast atrial fibrillation with heart rate of 122 beats per minute. Chest X-ray (Figure 2) showed cardiomegaly with upper lobe diversion and prominent pulmonary trunk with dilated pulmonary arteries suggestive of pulmonary hypertension. Transthoracic echocardiogram (Figures 3-6) showed a large perimembraneous VSD of 2.8 cm with left to right shunting, severe pulmonary hypertension (right ventricular (RV) systolic pressure 112.5 mmHg), with dilation of the pulmonary trunk (3.6 cm) and pulmonary arteries, RV hypertrophy with dilatation, and right atrial dilatation. Echocardiography also showed reduced RV systolic function with tricuspid annular plane systolic excursion of 1.3 cm. The mitral valve leaflets were thickened and calcified with severe mitral regurgitation (no stenosis) and dilated left atrium. The left ventricle (LV) had normal wall thickness but was dilated (LV internal diameter, diastole of 7.5 cm) with moderate LV systolic dysfunction (LV ejection fraction of 31%). He had paroxysmal atrial fibrillation with rapid ventricular response at the time of the echocardiography.

ECG of the patient showing fast atrial fibrillation (heart rate of 122/minute).

Postero-anterior and lateral Chest X-ray of the patient showing cardiomegaly and upper lobe blood diversion.

Epical 4 chamber view 2-dimension echocardiogram showing a large perimembranous ventricular septal defect of diameter 2.8 cm (arrows) and biatrial dilatation.
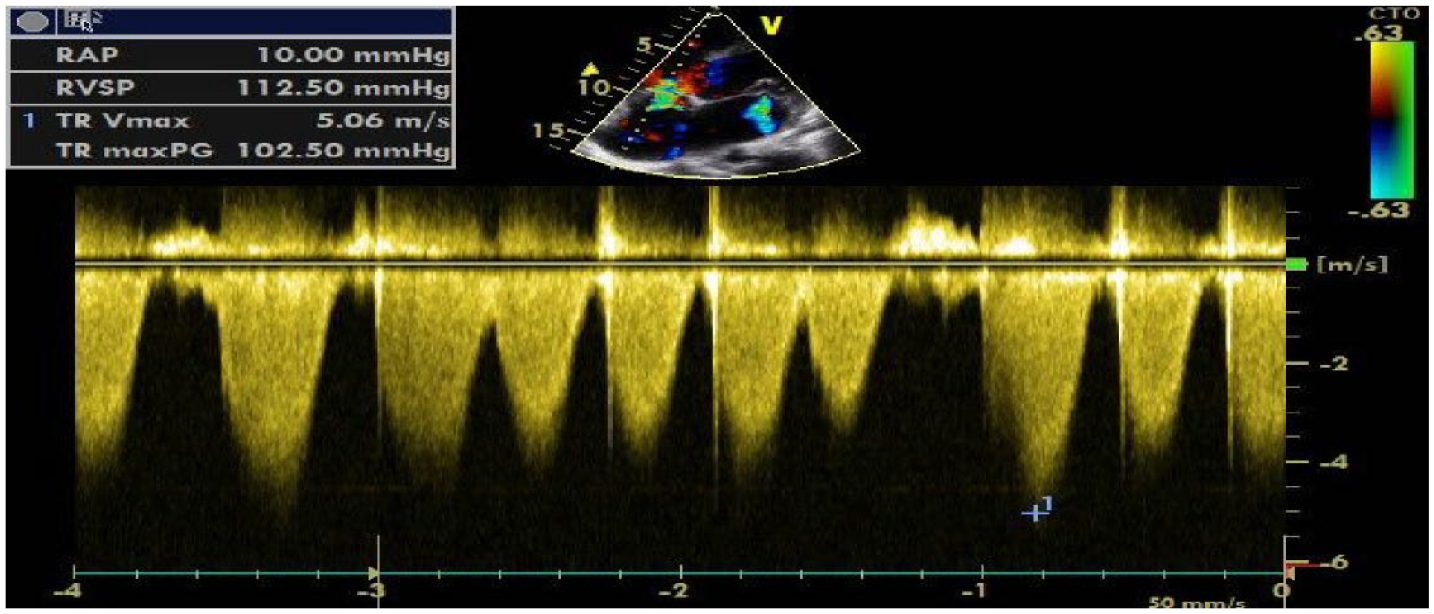
Continuous wave spectral doppler echocardiogram across the tricuspid valve giving peak tricuspid regurgitant velocity (TR max PG) of 102.5 mmHg and estimated right ventricular systolic pressure (RVSP) of 112.5 mmHg giving a right atrial pressure of 10 mmHg.

Parasternal short axis view 2-dimension echocardiogram showing dilated pulmonary trunk of diameter 3.6 cm (long arrow) and pulmonary arteries (short arrows).

Parasternal long axis view 2-dimension echocardiogram showing thickened mitral valve leaflets with mild calcifications (short arrow) a large perimembranous ventricular septal defect (long arrow), dilated left atrium both ventricles dilated.
A diagnosis of VSD and rheumatic mitral regurgitation with severe pulmonary hypertension, and heart failure with reduced ejection fraction complicated by paroxysmal atrial fibrillation was made. He was sent back to the primary physician with a recommendation that he should be started on a parenteral furosemide 40 mg twice daily, lisinopril 5 mg once daily orally, eplerenone 25 mg once daily orally, warfarin 5 mg once daily orally, tadalafil 20 mg once daily orally, bisoprolol was increased to 5 mg once daily orally and digoxin was discontinued. These medications were started and the patient was scheduled for review in a week. Patient however died from sudden cardiac death on day 4 after she was seen.
Discussion
VSD occurs as a result of defect in embryogenesis leading to an interruption in the interventricular septum. 15 Based on the location of the defect and the structures along its borders, VSD is classified into 4 main types; perimembranous, muscular, inlet and outlet VSD. 16 Perimembranous is the commonest type of VSD accounting for more than 80% of cases. 17 Most VSD close spontaneously before adulthood but larger defects may persist, however with most patients surviving into adulthood albeit with complications. 18 These complications include pulmonary arterial hypertension (which can lead to Eisenmenger syndrome), arrhythmias and heart failure as a common endpoint.3,4,17
Pulmonary arterial hypertension is defined as mean pulmonary arterial pressure >20 mmHg or pulmonary vascular resistance (PVR) of >2 woods units (WU) with pulmonary capillary wedge pressure (PCWP) of <15 mmHg according to the 2022 European society of cardiology and the European respiratory society pulmonary hypertension guidelines. 19 Up to 42% of patients with CHD who develop pulmonary hypertension have VSD. 4 Pulmonary hypertension due to VSD is classically characterized by elevated pulmonary arterial vascular resistance and therefore classified under group 1 pulmonary hypertension by world symposium on pulmonary hypertension. 20 However, the cause of pulmonary hypertension in VSD and other congenital heart diseases may be multifactorial. Pulmonary hypertension may be due to volume and pressure overload secondary to left-to-right shunting of blood through the septal defect. 21 The size of the defect has been found to be a key factor for developing pulmonary hypertension in this group of patients. Small-to-moderate defects, with diameter <1.5 cm, lead to Eisenmenger syndrome in only 3% of cases. However, larger defects (diameter > 1.5 cm) almost always progressively lead to the syndrome if no intervention are instituted. 22
The etiology of pulmonary hypertension in patients with CHD may also be attributable to genetic mutations. 22 There is evidence to suggest that 6% of patients with CHD have genetic mutations in the bone morphogenetic protein receptor type 2 (BMPR2), a mutation associated with pulmonary hypertension. 23 BMPR2 gene mutation is found in 70% of patients with familial pulmonary hypertension and 10% to 20% of those with idiopathic pulmonary hypertension. 24 Among patients with CHD who have pulmonary hypertension, 7.5% have mutations in this gene compared to 1.2% in those with CHD without pulmonary hypertension. 25 Newer genetic mutations have also been identified in cases of pulmonary arterial hypertension including the SRY-related high-mobility-group (HMG) box transcription factor 17 (SOX17). 26 A recent study that looked at 4241 patients with pulmonary arterial hypertension cases from the National Biological Sample and Data Repository for pulmonary hypertension identified other possible genetic mutations including fibulin 2 and platelet-derived growth factor D. 27 These genetic mutations may have roles to play in patients with CHD related pulmonary hypertension. This presupposes that the precapillary pulmonary hypertension that is caused in VSD may not entirely be as a result of the volume overload but also the manifestation of primary pulmonary hypertension as a result of mutations in the above-mentioned genes.
Pulmonary hypertension arising from mitral valve disease occurs as a result of volume and pressure overload building up in the left atrium as a result of mitral regurgitation or stenosis that is transmitted to the pulmonary veins, capillaries and then the pulmonary arteries.28 -30 This type of pulmonary hypertension belongs to the group 2 pulmonary (venous) hypertension. 20 Although, both mitral valve stenosis and regurgitation can elevate left atrial volumes and pressure, stenosis tends to be a commoner etiology of pulmonary hypertension with PASP > 50 mmHg occurring in 30% to 40% of those with severe mitral stenosis compared to 12% to 32% of those with moderate to severe mitral regurgitation. 30 When septal defects co-exist with valvular heart disease, there is an increases risk of developing pulmonary hypertension and symptoms of heart failure tend to occur at an earlier age. 14
Our patient had severe mitral regurgitation which contributed to the pulmonary hypertension caused by the VSD that was further worsened by the moderately reduced left ventricular systolic function.28 -30 Volume or pressure overload in the left atrium leads to left atrial dilation which increases the risk of supraventricular arrhythmias 31 as seen in our patient who developed paroxysmal atrial fibrillation. In patient with valvular heart disease, the presence of pulmonary hypertension increases the risk of mortality.32,33 Severe pulmonary hypertension, regardless of the cause can lead to progressive right ventricular systolic dysfunction and death. Sudden cardiac death (SCD) can also occur in severe pulmonary hypertension as in the case of our patient. SCD may occur as result of arrhythmias, pulmonary artery rupture, pulmonary dissection, myocardial infarction from compression of the left main coronary artery (coronary compression syndrome), pulmonary artery dissection, pulmonary artery rupture, and severe anemia from massive hemoptysis. 34
The volume and pressure overload in the lung vasculature as a result of VSD and left sided heart pathology (valvular heart disease and left ventricular systolic dysfunction in this case) leads to corresponding elevation of the pulmonary pressures initially, as a result of the volume overload. 28 However, in the long term, the pulmonary vasculature in a bit to compensate, undergoes changes by causing both intimal and medial thickening in the pulmonary arteries and veins which further propagates the hypertension.35,36 This probably is due to the release of inflammatory cytokines such as interleukin-1β which may lead to activation and proliferation of myofibroblast, and leukocyte infiltration which can lead to pulmonary vascular muscularization and fibrosis thereby causing pulmonary hypertension.37,38 Platelet activation and thrombosis also play roles in vascular narrowing contributing to the pulmonary hypertension.39,40 These may explain why genetic mutations in genes coding for fibroblast and platelet activation have been identified in patients with pulmonary hypertension.
VSD is diagnosed with 2-dimensional Doppler echocardiography which helps identify the anatomic features of the defect in terms of its location, dimensions and number. Furthermore, it helps determine the hemodynamic status including the direction of flow across the defect, flow magnitude, peak gradient and also evaluate for other congenital abnormalities such as coaptation of the aorta, valvular pathologies and others that may be associated with the VSD. 41 Our patient had VSD together with rheumatic mitral regurgitation that was diagnosed with a transthoracic echocardiography. Cardiac catheterization also provides additional information concerning the hemodynamics, and gives accurate pulmonary artery pressure measurements including the PCWP42,43 which would have been important in our case as it may have objectively shown the contribution of the left heart dysfunction (from the severe mitral regurgitation and systolic dysfunction) to the pulmonary hypertension. However, cardiac catheterization could not be done because the service is not available at our clinic. Cardiac computed tomography and cardiac magnetic resonance imaging offer additional imaging options in cases where echocardiography is limited.43,44 The size of the VSD should be evaluated together with direction of flow to determine whether Eisenmenger has developed which has management and prognostic implications. Imaging is also needed to evaluate markers of poor prognosis such as right heart strain, other associated congenital anomalies and complications such as infective endocarditis and atrial fibrillation. 17 Auxiliary investigations such as electrocardiogram (ECG) may show evidence of right heart strain 45 and arrhythmias, as in the case of our patient who had right ventricular hypertrophy and right axis deviation on ECG.
Most small VSD may not pose any significant hemodynamic impairment and may therefore not require any immediate treatment. However, VSD closure is indicated when the ratio of pulmonary-to-systemic blood flow is >1.5:1 which can be done surgically or via transcatheter repair. When Eisenmenger develops, the goal will be to reduce the PVR to less than 5 WU with medications (including endothelin receptor blocker such as bosentan, phosphodiesterase inhibitors like tadalafil and others) and then consider defect closure. Surgery is however deferred if PVR remains very high (5 WU) despite optimal medical therapy. 19 Our patient was started on medical therapy including tadalafil with the aim of reducing the pulmonary hypertension and consider the possibility surgical intervention.
Conclusion
Pulmonary hypertension is a common complication of CHD including VSD. Other conditions such as valvular heart diseases can exacerbate this complication through other pathological pathways. Early diagnosis and management is recommended to avoid complications and needless death. Also, epidemiological studies looking at the burden of the combined effect of congenital and acquired heart disease in Africa is recommended to deepen our understanding of this condition.
Footnotes
Author Contributions
All authors made a significant contribution to this paper, whether that is in examination and management of this case. All authors also took part in the drafting, revising, and gave approval for the publication of this manuscript.
Funding:
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests:
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Consent
Mother of the patient provided written informed consent for the publication of this case.
