Abstract
Sinus of Valsalva aneurysm is a rare defect that can present with fatal complications in case of rupture. Heart failure symptoms are common due to high fraction of the resultant shunt. Multimodality imaging and invasive hemodynamic assessment is essential for comprehensive evaluation of the defect and guiding surgical planning. We describe the case of a 40-year-old woman who presented with heart failure symptoms and was found to have ruptured sinus of Valsalva aneurysm on transthoracic echocardiogram. Cardiac computed tomography angiography further characterized the defect and the associated anomalies. Right heart catheterization assessed the hemodynamic significance of the left to right shunt. Intraoperative findings highlighted the associated congenital anomalies including supracristal ventricular septal defect. The use of intraoperative transesophageal echocardiography proved essential in detecting worsening of the right ventricular outflow track infundibular dynamic obstruction post repair thus delineating the importance of maintaining adequate cardiac preload. This case highlights a stepwise approach in the anatomical characterization of sinus of Valsalva aneurysm using multimodality imaging and the use of hemodynamic assessment and intraoperative imaging to guide surgical planning.
Keywords
Introduction
An aneurysm of the sinus of Valsalva (SoV) is a rare acquired or congenital anomaly predominantly affecting the right and the noncoronary sinuses, with an estimated prevalence of 0.09% of the population.1,2 Although usually asymptomatic when intact, it can present as acute heart failure, cardiac tamponade, acute coronary syndrome, or sudden cardiac death in case of a rupture.1,3 Most cases of ruptured SoV aneurysms require urgent surgical repair; however, transcatheter closure has been described. 4 We present a case of ruptured SoV aneurysm into the right ventricular outflow tract (RVOT) presenting as a large left to right shunt. The case highlights the complexity of preoperative planning and the intraoperative hemodynamic challenges that could arise after closure.
Case Presentation
A 40-year-old woman with a history of a childhood murmur presented with exertional dyspnea and decreased exercise tolerance over 1 month. She also reported palpitations and progressive orthopnea. There was no pedal edema. Cardiac examination revealed a continuous “machinery” murmur best heard in the left second intercostal space radiating to the carotid arteries, and a laterally displaced apical impulse associated with a thrill. The patient also displayed water hammer pulses and a “pistol shot” sign in bilateral femoral arteries.
Laboratory data including complete blood count, metabolic profile, and thyroid function test were unremarkable. Brain natriuretic peptide was elevated at 1192 pg/mL (<100 pg/mL). A 2-dimensional transthoracic echocardiogram (TTE) was obtained and showed a severely dilated left ventricle (left ventricular end-diastolic diameter of 6.6 cm) with a low normal ejection fraction of 50% to 55%. In parasternal long-axis views, a dilated right coronary SoV was noted with evidence of systolic and diastolic left to right flow by color Doppler imaging (Figure 1A, B, and D). In parasternal short-axis view, a ruptured SoV aneurysm connecting into the RVOT was visualized with re-demonstration of color flow across the defect. Additionally, a calcified area was noted in the supracristal portion of the interventricular septum immediately inferior to the aortic valve and RVOT (Figure 1B). Doppler echocardiography showed peak systolic and diastolic velocities of 4.2 m/s and 3.6 m/s, respectively, across the defect (Figure 2A). Diastolic flow reversal in the descending thoracic aorta was noted (Figure 2B). A note was made of a dynamic RVOT gradient with a peak of 30 mm Hg. A cardiac computed tomography angiography (CCTA) was obtained to further delineate aortic root anatomy, RVOT anatomy, assess for associated congenital deformities, and rule out coronary artery disease. CCTA confirmed normal coronary arteries, tricuspid aortic valve, and ruptured right SoV aneurysm (with windsock deformity also noted on TTE) into the RVOT just below a quadricuspid pulmonic valve (Figure 3). Additionally, there was evidence of calcifications in the supracristal region of the interventricular septum without definitive evidence of a ventricular septal defect (VSD). Right heart catheterization revealed a mean pulmonary artery pressure of 24 mm Hg with equalization of left ventricular and pulmonary end-diastolic pressures, as well as an aortic to pulmonic diastolic gradient of 25 mm Hg (Figure 4). There was significant oxygen step up from the right atrium to the right ventricle with a shunt fraction (Qp/Qs) of 7.3.
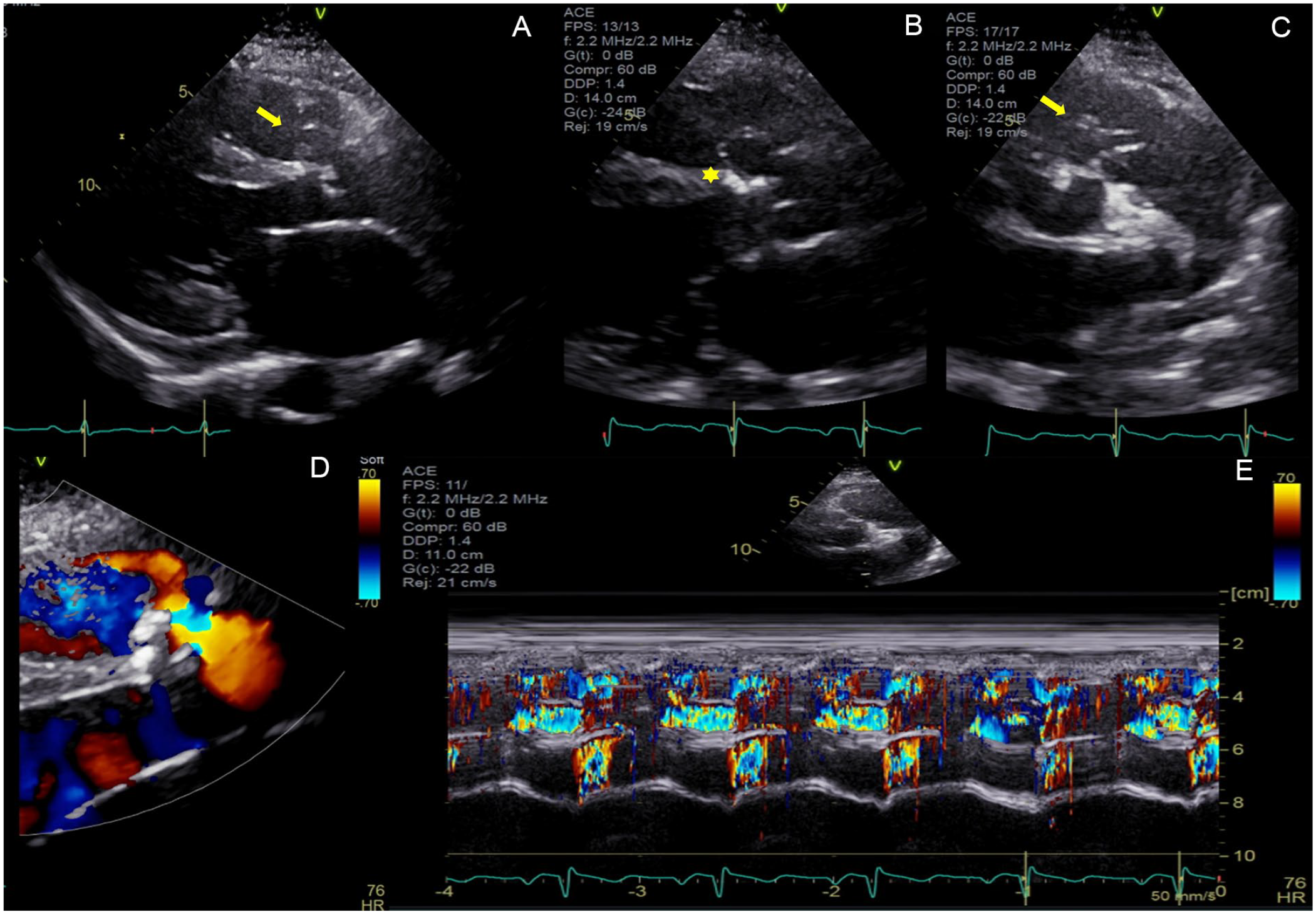
Transthoracic echocardiogram demonstrating right sinus of Valsalva (SoV) defect in parasternal long-axis view (Panels A and B). Subaortic area of calcification noted in the interventricular septum (asterisk, Panel B). Short-axis view demonstrates windsock deformity representing ruptured right SoV into the right ventricular outflow tract (Panel C). Color flow Doppler demonstrates left-to-right flow (Panel D). Color M-Mode through the ruptured SoV aneurysm demonstrates systolic and diastolic flow (Panel E).
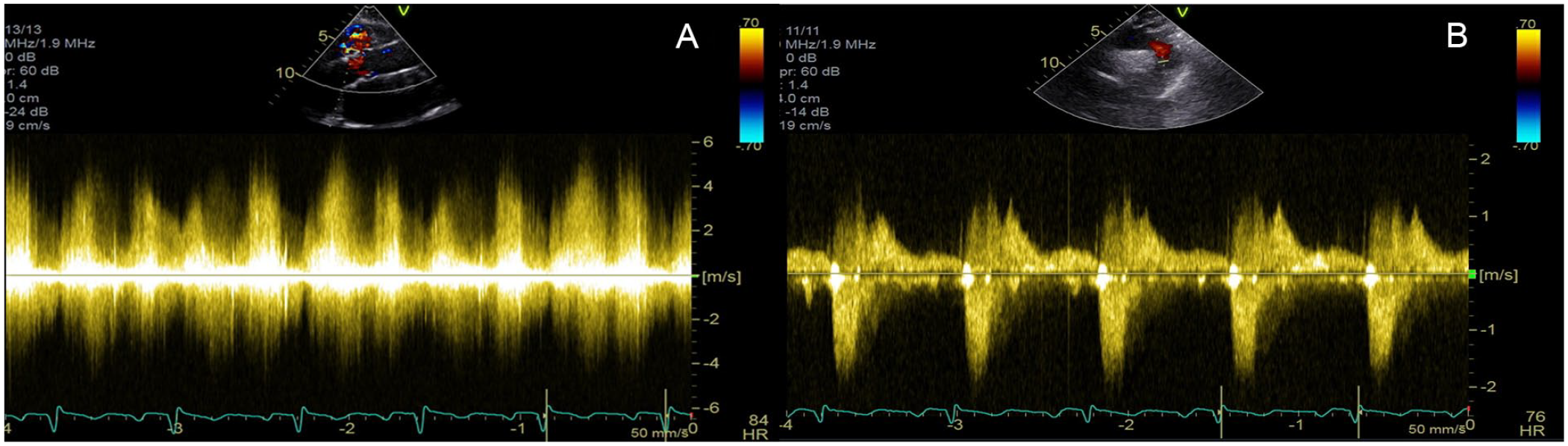
Doppler echocardiography demonstrates flow across ruptured right sinus of Valsalva in systole and diastole (A). Flow Doppler in the descending thoracic aorta shows diastolic flow reversal (B).

Multi-planar reconstruction of cardiac computed tomography angiography. Ruptured right sinus of Valsalva aneurysm into the right ventricular outflow tract in sagittal projection (arrow, Panel A). Membranous interventricular septal calcification (asterisk, Panel A). Quadricuspid pulmonary valve depicted in axial view (Panel B).

Right and left heart catheterization simultaneous hemodynamic tracings demonstrate equalization of end-diastolic left and right ventricular pressures (A) and reduced diastolic gradient between the aorta and the pulmonary artery (B).
The patient was taken for surgical repair of the ruptured SoV aneurysm. Intraoperative transesophageal echocardiogram (TEE) done for intraoperative monitoring further delineated the anatomy of the defect and the RVOT. Additionally, the previous finding of calcifications in the subaortic interventricular septum was noted without any evidence of color flow across the septum (Figure 5). Intraoperative findings demonstrated severe and bulky exophytic calcifications of right aortic annulus at the right cusp hinge and at the membranous septum just below the right coronary cusp associated with a VSD (Figure 6A and B). A surgical repair of the ruptured SoV aneurysm and the underlying VSD was performed by primary closure. Aortic valve could not be salvaged. Hence, it was replaced with a 21-mm mechanical prosthesis. Post-repair intraoperative TEE images showed no residual shunt but a finding of a hyperdynamic infundibular portion of the RVOT and right ventricular hypertrophy. The patient had an unremarkable postoperative course. She showed evidence of systemic hypertension for which she was initiated on an antihypertensive regimen with angiotensin converting enzyme inhibitor and a β-blocker. She was discharged home on postoperative day 4. At 1 and 3 months clinic follow-up, the patient reported near resolution of her symptoms.
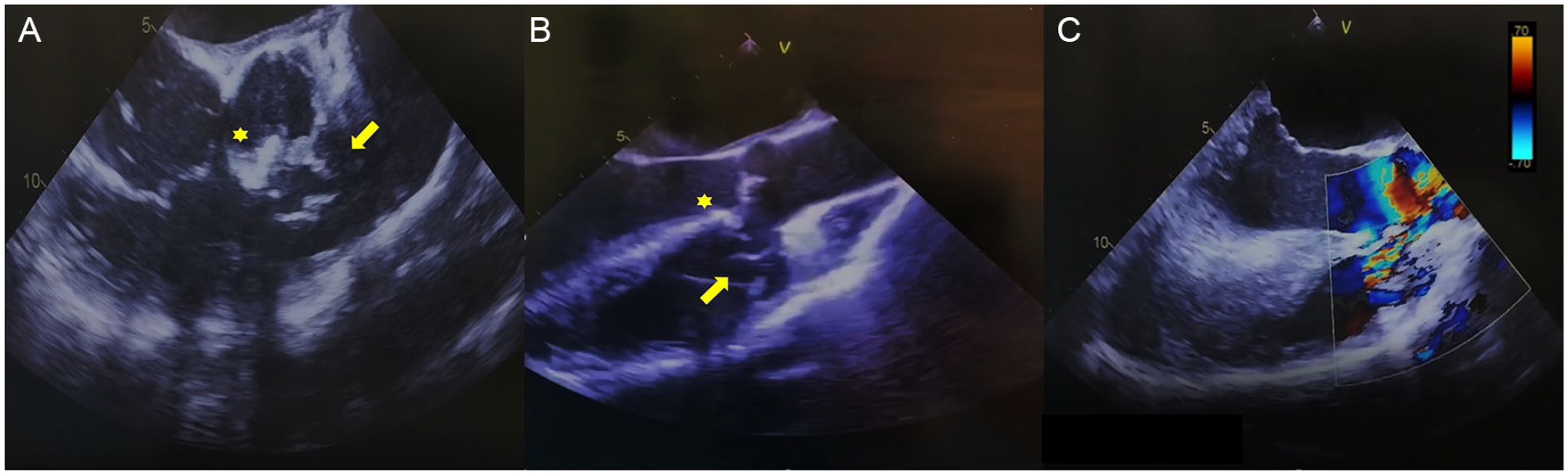
Intraoperative transesophageal echocardiogram re-demonstrates the ruptured right sinus of Valsalva defect into the right ventricular outflow tract in 60° (A) and 120° (B). Color Doppler echocardiography demonstrates flow across the defect in 120° (C).
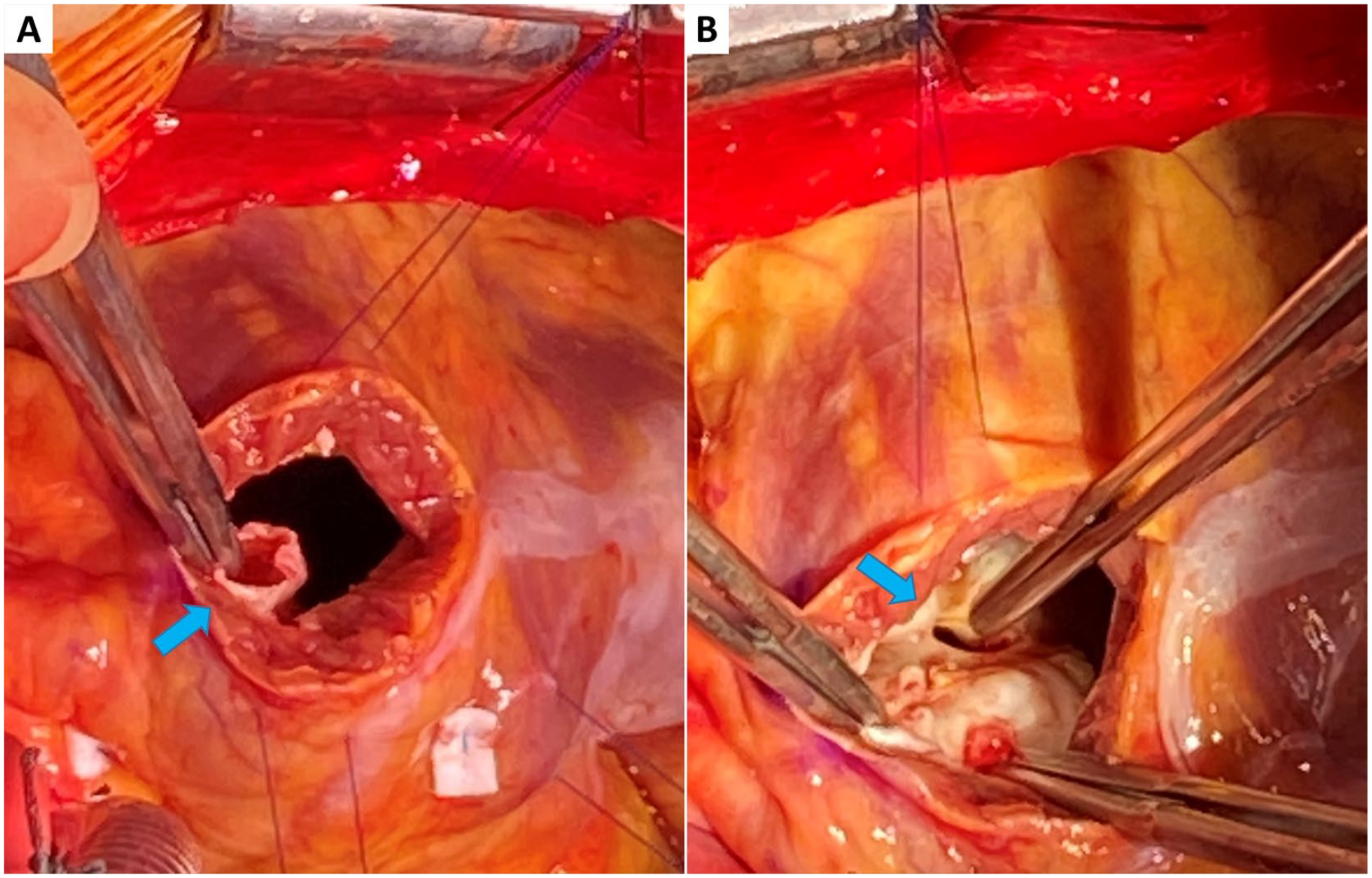
Intraoperative findings demonstrating ruptured sinus of Valsalva defect (A) and underlying supracristal ventricular septal defect (B).
Discussion
Ruptured SoV aneurysm may present with a sudden hemodynamic collapse but can have a more insidious onset depending on the site and size of the perforation. 5 Our patient presented with subacute heart failure symptoms and was noted to have a harsh continuous “machinery” murmur, which has been traditionally described in patients with patent ductus arteriosus, and which results from continuous systolic and diastolic flow. In our patient, the continuous nature of the shunt from the aortic root to the RVOT closely mimicked patent ductus arteriosus physiology resulting in a similar auscultatory finding. Additionally, due to the high shunt ratio, increased left-sided pulmonary venous return, and continuous flow in diastole with resultant high pulse pressure, the patient had examination findings mimicking those of chronic aortic insufficiency including “water hammer” pulses and “pistol shot” sign. The patient did not have evidence of aortic insufficiency on TTE or intraoperative TEE. Due to the continuous nature of the large left to right shunt, there is equalization of end-diastolic pressures between the right ventricle and the pulmonary artery. Importantly, the pulmonic valve does not fully close as the flow jet is directed into the subvalvular area as depicted on CCTA. The most common site of rupture of SOV aneurysm is into the right ventricle followed by right atrium, left ventricle, and the left atrium. 5
The use of multimodality imaging in SoV aneurysms has been described and recommended previously.2,6,7 The goal of imaging includes diagnosis, accurate characterization of the defect including origin, extent, and relation to the coronary arteries, defining associated cardiac anomalies, and delineating potential associated complications. In case of rupture, the site of rupture, its effect on surrounding structures, and degree of shunting should also be assessed. TTE is the modality of choice for diagnosis and assessment of the involved sinus and the presence of fistulae, as well as any associated aortic valve pathologies or other anomalies. 6 TEE can further delineate aortic root anatomy and the relationship of the sinuses to surrounding structures. CCTA has emerged as an important complementary tool in the evaluation of SoV aneurysms. It allows simultaneous evaluation of the coronary arteries prior to surgical interventions, dynamic assessment of aortic valve motion, left ventricular and right ventricular function quantification when retrospective processing is used. The dataset can be reformatted in any desired plane making it less operator dependent. 6
In our patient, an interesting imaging finding was the area of calcifications in the supracristal portion of the interventricular septum inferior to the right SoV aneurysm that we postulated corresponded to a healed VSD responsible for her childhood murmur. The embryological basis of the SoV aneurysm is a defect in the development of the distal bulbar septum base that forms the right coronary sinus and adjoining noncoronary sinus. This also explains the association with an otherwise uncommon defect, a supracristal VSD.4,8 This latter finding could present a diagnostic challenge. In an isolated VSD, the Doppler waveform shows a high-velocity systolic and a low-velocity diastolic flow, whereas a continuous high-jet velocity is observed in a ruptured SoV aneurysm, which is accentuated in diastole. 2 There was no evidence of flow across the area on either of the imaging modalities. Intraoperative findings, however, revealed a VSD in the aforementioned area. It is possible that intraoperative inspection and manipulation of the tissue between the aneurysm and the calcified supracristal region of the interventricular septum lead to the dehiscence of the healed defect. VSD is one of the most common congenital defects associated with aneurysm of SoV. Another interesting finding was that of a quadricuspid pulmonic valve, which is mostly a benign finding but has been associated with other congenital anomalies predominantly that of aortic valve. While there is no direct association between quadricuspid pulmonic valve and SoV aneurysm, quadricuspid aortic valve has been described in association with SoV aneurysm in multiple reports. 9
Importantly, long-standing increased flow across the RVOT can lead to infundibular hypertrophy creating a dynamic obstruction explaining the discrepancy between the echocardiographic pulmonary arterial pressures and the hemodynamic pressures. In the presence of such an obstruction, pulmonary arterial pressures cannot be assumed to accurately reflect right ventricular systolic pressure. An interesting intraoperative observation after closure of the defect and weaning of cardioplegia was worsening of the RVOT infundibular dynamic obstruction similarly to what has been described after tetralogy of Fallot repair. This highlights the importance of maintaining adequate cardiac preload with avoidance of anemia (with resultant tachycardia) and adequate fluid resuscitation. Regression of right ventricular hypertrophy after relieving obstruction has been demonstrated to be dependent on the duration of obstruction. 10
Conclusion
Sinus of Valsalva aneurysm rupture is a rare but potentially fatal occurrence and should be considered on the differential of a shunt in a young individual presenting with dilated cardiomyopathy. TTE is the test of choice to establish the diagnosis and characterize the defect. CCTA can provide complementary information and allow for noninvasive evaluation of coronary artery anatomy in preparation for surgical intervention. Although there are no specific guidelines, multimodality imaging is recommended for diagnosis confirmation. Definitive management usually involves surgical repair.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethics Approval
Our institution does not require ethical approval for reporting individual cases or case series.
Informed Consent
Verbal informed consent was obtained from the patient(s) for their anonymized information to be published in this article.
