Abstract
Background:
When used for hematopoietic stem cell mobilization, plerixafor was originally recommended to be administered 11 hours prior to apheresis based on the peak effect of 10 to 14 hours translating into an administration time of 10 to 11
Objective:
The objective of this study is to compare the stem cell collection efficiency before and after the practice change at our institution.
Methods:
A retrospective chart review for patients with multiple myeloma, Hodgkin lymphoma, and non-Hodgkin lymphoma who received a plerixafor-containing mobilization regimen was conducted. The primary end point was the percentage of patients achieving the minimal CD34+ cell goal in ⩽2 apheresis days. The secondary end points included the percentage of patients achieving the preferred CD34+ cell goal in ⩽2 apheresis days, days of apheresis, total CD34+ cells Collected, and engraftment time.
Results:
A total of 208 patients (4
Conclusions:
Late afternoon administration of plerixafor provides efficient stem cell mobilization.
Background
High-dose chemotherapy followed by autologous hematopoietic stem cell transplantation (ASCT) is a standard treatment for patients with multiple myeloma (MM), Hodgkin lymphoma (HL), and non-HL (NHL).1–3 Cytokine mobilization with granulocyte colony-stimulating factor (G-CSF—filgrastim, pegfilgrastim) or granulocyte macrophage-CSF (GM-CSF—sargramostim) with or without chemotherapy has been used to mobilize hematopoietic progenitor cells (HPCs).3–5 Heavily pretreated patients or those receiving external beam radiation run approximately 20% to 25% risk of failing to collect sufficient stem cells. 4 Interaction of the stromal cell–derived factor 1 (SDF-1) expressed on bone marrow stromal cells and the chemokine receptor 4 (CXCR4) expressed on HPCs has been shown to play a major role in retaining CD34+ cells in the bone marrow regulating HPCs migration to the peripheral circulation.6,7 Plerixafor, a CXCR4 inhibitor, has improved mobilization by inhibiting the binding of SDF-1 and CXCR4 which results in HPCs’ migration from the bone marrow to the peripheral blood (PB).3,8 Plerixafor is indicated in combination with G-CSF for HPCs’ mobilization prior to ASCT. 8
In pharmacodynamic studies of plerixafor in conjunction with G-CSF in healthy volunteers, a sustained elevation in the PB CD34+ count was observed from 4 to 18 hours after plerixafor administration with a peak CD34+ count between 10 and 14 hours.
6
In response to this, the manufacturer initially recommended self-administration of plerixafor approximately 11 hours prior to initiation of apheresis. This translated into a late night administration time of 10 to 11
Materials and Methods
Our standard operating procedure (SOP) allows plerixafor administration when day 4 PB CD-34+ cell count with G-CSF alone is less than 10 to 20 cells/µL when G-CSFs are used as the mobilization strategy. In addition, plerixafor can be used at physician’s discretion in patients deemed high risk of poor mobilization. 4 To further elaborate, patients with HL, NHL, and MM, planned to undergo a autologous ASCT, are typically started on filgrastim at a dose of 10 μg/kg daily on a Friday with a (PB) CD-34+ level to be checked on Monday morning. Based on the (PB) CD-34+ and patients’ risk factors for poor mobilization, decision is made by the treating physician whether to add plerixafor or not prior to proceeding to collection of CD-34+ cells. Collection of CD-34+ is typically started when (PB) CD-34+ reaches more than 10 to 20 cells/µL. Chemotherapy plus G-CSF can be used for mobilization especially in salvage mobilization. Depending on the chemotherapy regimen used for mobilization, G-CSFs are typically started on days 5 to 7 after chemotherapy with plerixafor added after white blood cells’ recovery (typically starting day 11) if (PB) CD-34+ cell count with chemotherapy and G-CSF alone is less than 10 to 20 cells/µL. The dose of plerixafor used was initially 0.24 mg/kg when we first started using plerixafor at our institution; however, more recently, we have transitioned to using a fixed dose of 24 mg (vial size) adjusted for kidney function as appropriate. This could have created some dosing variation between patients who received plerixafor soon after it was first marketed when a weight-based approach was more commonly used compared with a fixed 24 mg dose strategy recently. Following approval from the hospital Institutional Review Board, patients with HL, NHL, and MM, who received a plerixafor-containing mobilization regimen, were identified using patients’ medical records and cell processing databases.
The primary efficacy end point was the percentage of patients achieving the minimal CD-34+ cell goal (defined as 2 × 106 CD-34+ cells/kg) in ⩽2 apheresis days. The secondary efficacy end points were the proportion of patients achieving the preferred CD-34+ cell goal (defined as 5 × 106 CD-34+ cells/kg) in ⩽2 apheresis days, the fold increase in PB CD-34+ cells after administration of plerixafor, days of apheresis to achieve minimal and preferred CD-34+ cell goals, total days of apheresis, number of doses of plerixafor administered, total CD-34+ cells collected, and the number of days to neutrophil and platelet engraftment. Neutrophil engraftment was defined as an absolute neutrophil count ⩾0.5 × 109/L for 3 consecutive days. Platelet engraftment was defined as platelet count ⩾20 × 109/L without a transfusion for the preceding 7 days.
Statistical Analysis
The χ2 and Fisher exact tests were used to compare the proportion of patients meeting the primary end point between the groups and for categorical data analysis. The Mann-Whitney test was used for numerical data analysis.
Results
A total of 208 patients received plerixafor-facilitated mobilization for ASCT. Of these, 140 patients received plerixafor at 10

Patients accrual (January 2009-September 2015). MM indicates multiple myeloma.
Baseline characteristics.

Abbreviations: G-CSF, granulocyte colony-stimulating factor; WBC, white blood cell.
Days of apheresis completed prior to first plerixafor dose: 1 (n = 23).
Days of apheresis completed prior to first plerixafor dose: 1 (n = 29), 2 (n = 2).
Any of the following agents: anthracycline, bortezomib, cyclophosphamide, melphalan, and vincristine.
Radiation location (spine n = 7, abdomen n = 3, jaw/neck n = 2, pelvis n = 1, groin n = 1).
Radiation location (spine n = 5, arm/shoulder n = 4, chest n = 4, abdomen n = 3, mediastinum n = 2, pelvis/sacrum n = 2, testis n = 2, brain n = 1, neck n = 1, sinus n = 1).

Primary end point.
Secondary end points.
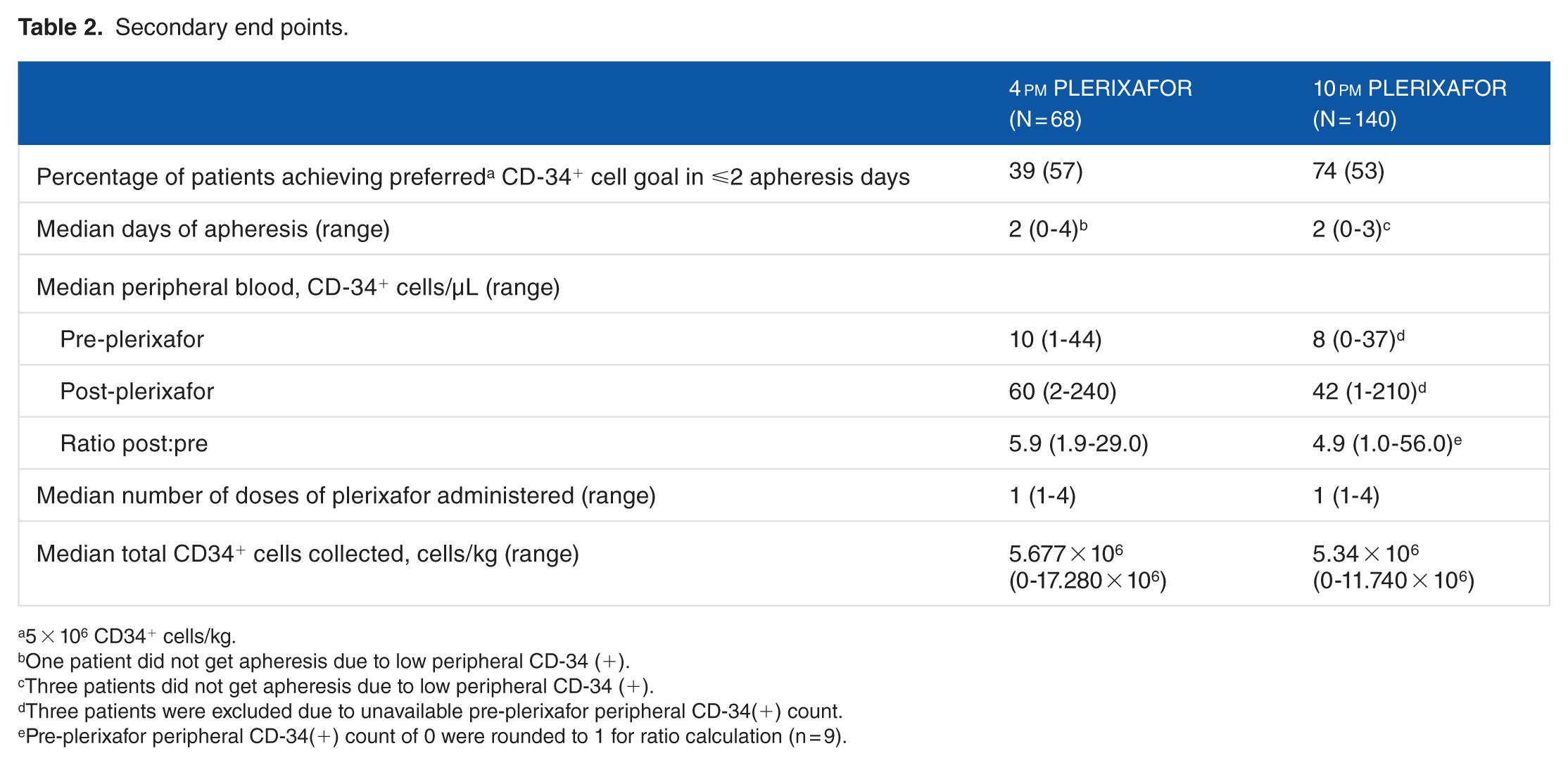
5 × 106 CD34+ cells/kg.
One patient did not get apheresis due to low peripheral CD-34 (+).
Three patients did not get apheresis due to low peripheral CD-34 (+).
Three patients were excluded due to unavailable pre-plerixafor peripheral CD-34(+) count.
Pre-plerixafor peripheral CD-34(+) count of 0 were rounded to 1 for ratio calculation (n = 9).
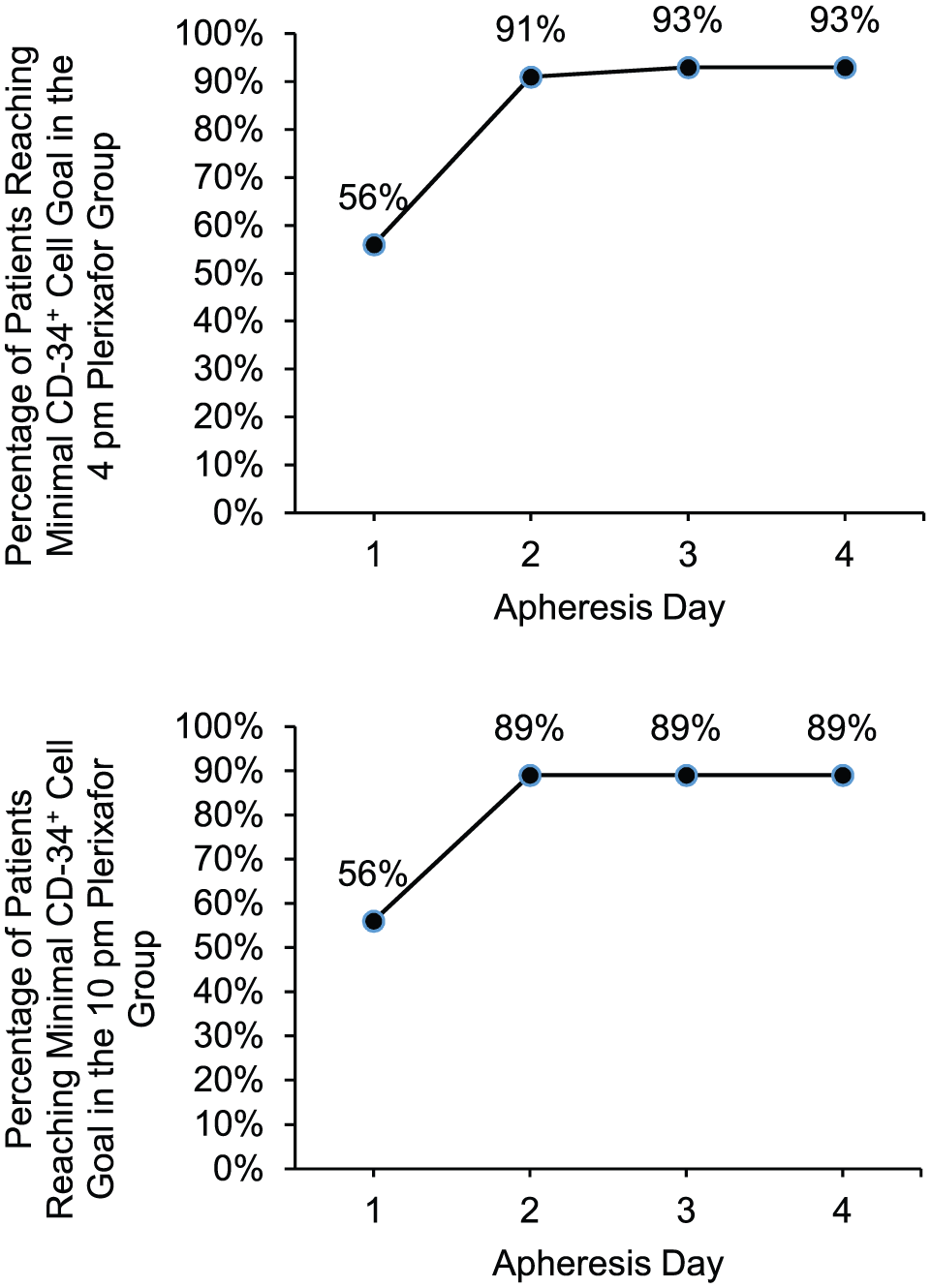
Minimal cell dose outcomes.

Preferred cell dose outcomes.
Discussion
In this study, 4
The results of our analysis should be interpreted with the following limitations in mind. First, our study is a retrospective review and the decision to add plerixafor to a mobilization regimen was in some instances based on physician’s discretion. Deviation from our SOP was noted in 14 patients (20%) in the 4
Conclusions
Late afternoon administration of plerixafor allows efficient stem cell mobilization and provides practical solution when used as part of the mobilization strategy in ASCT.
Footnotes
Acknowledgements
The authors thank David Putney, PharmD, MPH, BCPS (AQ-Cardiology), Department of Pharmacy, Houston Methodist Hospital. Poster presented at the hematology oncology pharmacy association (HOPA) meeting 2016. Abstract presented at the American Society for Blood and Marrow Transplantation (ASBMT) 2017.
Declaration of conflicting interests:
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding:
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Author Contributions
CER, JEC, RM, GC, and RK: Project conception or design and collection, analysis, and/or interpretation of data. CER, JEC, GC, and RK: Drafting the article or revising it critically for important intellectual content. CER, JEC, GC, GOA, AS and RK: Revising the article critically for important intellectual content approval of the version to be published.
