Abstract
Objective
Human cytomegalovirus (HCMV) infection is common after bone marrow transplantation (BMT), and it increases morbidity and mortality for transplant recipients. HCMV infection may cause hepatitis and elevate the liver enzymes aspartate transferase (AST) and alanine transferase (ALT). This study aimed to analyse the associations between liver enzyme levels and infection with HCMV antigenaemia after BMT.
Methods
Data from 30 patients after BMT were collected at different time points (0.5, 1.0, 1.5, 2.0, 2.5, 3.0, 4.0, 5.0, and 6.0 months post-transplantation). The patients were divided into the HCMV antigenaemia-positive and HCMV antigenaemia-negative groups according to a peripheral blood pp65 antigen assay. Immunohistochemistry was used to identify HCMV pp65 antigen and conventional methods were used to detect liver enzyme levels.
Results
Twelve patients were pp65 antigenaemia-positive and 10 patients were positive in the first 3 months post-transplant. Liver enzyme levels were increased after positivity for HCMV antigenaemia (p = 0.034 and p = 0.018 for ALT and AST, respectively). One month before antigenaemia, AST levels were higher in the HCMV antigenaemia-positive group compared with the negative group (p = 0.006).
Conclusion
HCMV antigenaemia mostly occurs in the early stage of post-BMT and early abnormal liver enzyme levels may increase the chance of HCMV antigenaemia after BMT.
Abbreviations
AST: aspartate transferase ALT: alanine transferase HCMV, human cytomegalovirus.*P < 0.05, comparison of the number of patients in the HCMV antigenaemia-positive and HCMV antigenaemia-negative groups at the same time point (Fisher’s exact test, two-sided)
Introduction
Over the past half century, tremendous advances have been made in bone marrow transplantation (BMT). Despite these advances, infection is a severe complication in patients undergoing BMT.1–4 Human cytomegalovirus (HCMV) is a common viral infection, which is a major factor in morbidity and mortality undergoing BMT.4–6 Rapid laboratory methods for the early detection of cytomegalovirus (CMV) are needed for preventing CMV disease in transplant recipients. Some sensitive and specific laboratory tests should be used to rapidly diagnose CMV infection after BMT. Detection of the HCMV antigen pp65 in peripheral blood mononuclear cells (PBLs) is preferred for screening for HCMV antigenaemia. This is because this method is more rapid and sensitive than culture and has a good positive predictive value compared with CMV-DNA by polymerase chain reaction.7,8 Wirgart et al. 9 (1996) found that the best marker for monitoring kidney transplant patients might be the quantitative CMV pp65 antigen detection assay. In their study, positivity for antigen pp65 was used for screening for HCMV antigenaemia. HCMV infection may cause hepatitis and elevate the liver enzymes aspartate transferase (AST) and alanine transferase (ALT).
Our study aimed to investigate the incidence of HCMV antigenaemia during 6 months post-BMT. Because HCMV hepatitis is associated with solid organ recipients and has little relation to BMT,10,11 we also investigated the associations between HCMV antigenaemia and ALT and AST levels at different time points post-BMT.
Methods
Patients
Characteristics of patients in the study.

Abbreviations: M, male; F, female; ALL, acute lymphoblastic leukaemia; AML, acute myeloid leukaemia; CML, chronic myeloid leukaemia; HCMV, human cytomegalovirus. P < 0.05 (dependent-samples t test).
Immunosuppressive therapy
Chemotherapy before transplantation consisted of busulfan (4 mg/kg/day from days −7 to −4) and cyclophosphamide (60 mg/kg/day from days −3 to −2). Prophylaxis for graft-versus-host disease consisted of mycophenolate mofetil (1.0 g/day for 3 months after BMT) and cyclophosphamide (3 mg/kg/day for 24 h by intravenous infusion, to maintain blood concentrations at 300–500 ng/ml until medicine could be taken orally at 6 mg/kg/day).
Antiviral treatment
All BMT recipients received ganciclovir intravenously at a standard dosage of 5 mg/kg/day until the day of transplantation. Antiviral treatment for HCMV infection was started in each BMT recipient according to a previously reported preemptive therapy regimen.12,13 Briefly, all patients received ganciclovir intravenously at a standard dosage of 5 mg/kg every 12 h for 14 days or until negative antigenaemia results were observed.
Methods of detection
From day + 1 until day + 90 after BMT, HCMV pp65 antigen monitoring was performed twice a month. From day + 90 until 6 months after BMT, the pp65 antigen assay was performed every month. At the same time, conventional methods were used to assay liver enzyme levels. Liver enzyme levels are usually less than 40 U/L for AST and less than 50 U/L for ALT. All of the patients were monitored twice weekly during the next 2 months after BMT, and then every month during the next 4 months post-6 months after BMT. A total of 296 blood samples were taken from the 30 patients. Immunohistochemistry was used to identify CMV pp65 antigen. The CMV antigen assay was performed as described previously, with minor modifications.11,14 A standard two-step immunohistochemical method was used to assay CMV antigen expression in PBLs. In brief, leukocytes were separated from EDTA-anticoagulated blood and spread on slides. Anti-CMV-PP65-Ag monoclonal antibody (DAKO, Denmark) and the Envision™ + System peroxidase (DAB) kit (DAKO) were used. The stained samples were analysed under an optical microscope with an image recording system (BH-2, OLYMPUS, Japan). Cells that were stained yellow or brown were positive and blue cells were negative. The results are shown as the number of positive cells per 50,000 leukocytes, as previously described. 15
Diagnosis of HCMV antigenaemia
HCMV antigenaemia is defined by serological conversion to CMV in a patient who had been seronegative before transplantation. In our study, we used antigenaemia as the gold standard for detection of CMV, as previously reported. 16 The standard of antigenaemia is in accordance with a previous report. 17 HCMV infection was diagnosed when more than one positive cell was observed among 50,000 leukocytes in the blood samples. The patients were divided into the infection group and the control group according to the peripheral blood pp65 antigen assay results.
Statistical analysis
All statistical analyses were performed using SPSS 16.0 (Chicago, IL, USA). The chi-square test was performed for dichotomous variables. The dependent samples t test was used to compare differences in the median values of continuous variables. A P value of less than 0.05 was considered statistically significant.
Results
Comparison of ALT and AST levels at different time points in patients who were positive for HCMV antigenaemia.

AST: aspartate transferase; ALT: alanine transferase; HCMV, human cytomegalovirus. *P < 0.05, comparison of the number of patients in the HCMV antigenaemia-positive group among different time points (Fisher’s exact test, two-sided).
Comparison of ALT and AST levels between patients in the HCMV antigenaemia-positive and negative groups at the same time point.
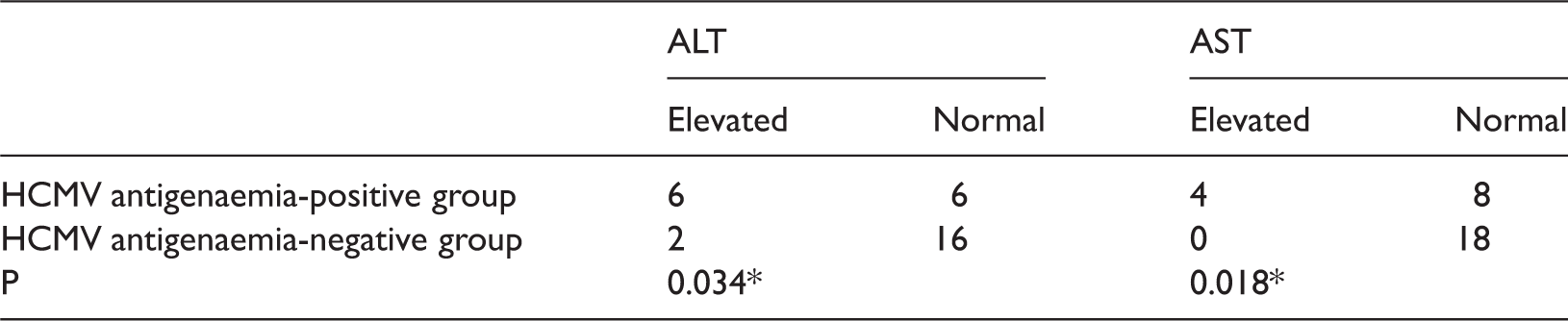
Abbreviations: AST: aspartate transferase; ALT: alanine transferase; HCMV, human cytomegalovirus. *P < , comparison of the number of patients in the HCMV antigenaemia-positive group and the HCMV antigenaemia-negative group (Fisher's exact test, two-sided).
Discussion
HCMV infects 70–100% of adults in populations worldwide. 18 For BMT recipients, infection with HCMV is still a major problem regarding morbidity and mortality. Clear evidence shows that HCMV donor seropositive/recipient seronegative (D+/R−) BMT may increase the incidence of HCMV infection post-transplantation for recipients.19,20 There were no HCMV-seropositive donors in our series to reduce the incidence of transmission of HCMV infection by leukoreduced blood product.2,20
In the present study, 83.33% (10/12) of patients were positive for HCMV antigenaemia during the first 3 months post-transplant. HCMV replication of most BMT recipients typically occurs after engraftment until approximately 100 days post-transplantation. 1 HCMV is a ubiquitous herpes virus that belongs to the β-herpes virus family. Similar to other herpes viruses, the cellular immune response plays a critical role in viral clearance after HCMV infection. Cell-mediated immunity for BMT recipients during the first 100 days post-transplantation is impaired because some infection may occur, particularly HCMV.1,20
We found that HCMV infection was significantly associated with liver enzyme levels. A previous study showed that ALT and AST values started to increase at the time of distinct CMV infection (day 12), suggesting an association between CMV infection and elevated liver enzyme levels. 21 In our study, the number of patients whose ALT/AST levels were elevated at different time points in the HCMV antigenaemia-positive group and the HCMV antigenaemia-negative group was significantly different (P < 0.05). Specifically, ALT and AST levels at 1 month before HCMV antigenaemia were significantly elevated. Some studies have shown no significant differences between the infection and control groups regarding other factors that produce an increase in liver enzyme levels, such as age and the use of immunosuppressive agents after transplantation.22,23 Therefore, we suggest that there is a significant association between HCMV infection and an increase in liver enzyme levels (ALT and AST) before HCMV antigenaemia. HCMV induces activation of nuclear factor κB (NF-κB) after infecting fibroblasts and macrophage cells. NF-κB response elements are present in the enhancer region of the CMV major immediate early promoter, and activity of this promotor is strongly upregulated by NF-κB in transient transfection assays. 24 This activation of NF-κB may activate cytokines, which may lead to liver injury and increased ALT and AST levels.
We conclude that HCMV antigenaemia often occurs in the early stage of post-BMT, and that HCMV antigenaemia may be detected early by measures of liver function post-BMT. A limitation of our study was the relatively small number of patients, which might be associated with large interpersonal variation. Future larger-scale studies are required to verify the relationship between HCMV antigenaemia and early abnormal liver enzyme levels in patients who have BMT.
Footnotes
Acknowledgements
We would like to thank all of the participants who were recruited in this study.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
