Abstract
The Atlas Knee System was designed to fill the gap between no longer effective conservative treatments and more invasive surgery for young patients with medial knee osteoarthritis (OA). This article reports on the 2-year results of a single-arm study of 26 subjects who previously reported favorable clinical outcomes 1 year post implantation. Western Ontario and McMaster Universities Osteoarthritis Index pain and function scores improved by a clinically meaningful amount relative to baseline, and subjects had a return to normal range of motion. This study confirmed that the benefit of a joint unloading device in the management of young patients with medial knee OA is maintained over 2 years. This trial was registered with ClinicalTrials.gov (NCT02711254).
Background
A recent article by the Centers for Disease Control and Prevention stated that more than half of adults with arthritis in the United States, an estimated 32 million, are of working age (ie, <65 years old). 1 Treating this younger, high-demand, patient demographic is a challenging situation due to their work obligations and desire to remain physically active. 2 Many of these patients with knee osteoarthritis (OA) have unicompartmental disease of the medial compartment.3-6
Biologically, the loss of hyaline cartilage increases compartmental loading, which, in turn, leads to the presence of bone marrow lesions, inflamed synovium, development of osteophytes, and further structural damage.2,7,8 This may also cause a redistribution of mechanical loads to other joint regions, including an overloaded medial compartment. 7 Conservative treatments for medial knee OA include lateral wedge insoles, unloader knee braces, and intra-articular injections (hyaluronic acid or corticosteroid).9-11 When patients are no longer responsive to such approaches, they may consider surgical intervention, namely, arthroplasty or high tibial osteotomy (HTO). In younger patients, this decision may be influenced by their desire to avoid procedures that require bone cutting or bone removal, restrict their return to high activity levels, or require lengthy rehabilitation.6,12-14 There exists a large gap between conservative treatments and bone-altering surgeries, and a clinical need exists for additional treatment options that can bridge this gap.
Unicompartmental load absorbing implants were developed to fill the gap between no longer effective conservative treatments and more invasive surgical options.4,5,14 The Atlas System (Moximed, Inc., USA) is a second-generation load absorber that is implanted during an extra-articular procedure that avoids the trade-offs associated with arthroplasty or HTO, such as bone cutting and lengthy periods of non-weight bearing. 13 The Atlas System unloads the medial knee compartment by means of an absorbing element that is compressed during weight bearing. The load absorber does not transfer excess load to the lateral compartment, rather it maintains the natural kinematics of the joint. Biomechanical studies show that this extra-articular absorber system significantly reduces medial compartment area contact pressure and peak contact pressure, comparable with that achieved with HTO, and the 1-year clinical study results of the implant were promising.15,16 The device has also shown some evidence of cartilage regeneration on magnetic resonance imaging (MRI). 13 The purpose of this study (the PHANTOM High Flex trial) was to assess the efficacy and safety of this device after 2 years in patients who may benefit from unloading of the medial compartment of the knee.
Methods
Objectives
The primary objectives of this study were to evaluate changes in pain and function at 2 years compared with baseline, using subjects as their own controls, and safety outcomes. Safety was determined using the incidence of treatment-emergent adverse events (AEs). Secondary objectives were procedural success, both patient- and physician-reported changes in outcomes scores, and radiographic assessments of the knee and implant.
Design
The study was designed, conducted, recorded, and reported in compliance with the principles of Good Clinical Practice guidelines and the International Organization for Standardization (ISO) 14155, 17 as well as in accordance with all national, state, and local laws of the appropriate regulatory authorities and the Declaration of Helsinki. The trial was registered with ClinicalTrials.gov (NCT02711254).
The study was conducted across 3 sites in Poland and 1 site in South Africa. Enrollment was completed between August and September 2015. All eligible subjects who received the implant were followed for 2 years following the procedure, with additional study visits at baseline, immediately postprocedure, 6 weeks, 3 and 6 months, and 1 year.
Eligibility criteria
The eligibility criteria were previously reported. 16 In short, included subjects were male or female, between the ages of 25 and 65 years, and had a documented pathology of the medial compartment of the knee that required unloading. The pathology qualified with either an International Cartilage Repair Society score >0 as assessed by MRI or arthroscopy older than 3 months or a Kellgren-Lawrence grade ⩽3 as assessed by X-ray. They also had continued target knee pain despite 6 months of conservative treatment prior to surgery, pain in the target knee as demonstrated by a minimum score of 40 (scale 0-100) on the Knee Injury and Osteoarthritis Outcome Score (KOOS) pain domain questions, knee flexion ⩾90º to ⩽140º, a body mass index (BMI) between 24 and 35 kg/m², and weight ⩽110 kg. A more detailed description of the inclusion and exclusion criteria is available in the supplementary data.
Device description
The Atlas System and its implantation have been previously described in detail. 16 The implant is fixed subcutaneously in the medial extra-capsular space, and it can be removed without disruption to the joint and surrounding tissues. Primary joint replacement surgery can be performed in the future, if necessary. No concomitant procedures, including arthroscopy, were permitted in this study.
Outcomes
The Western Ontario and McMaster Universities Osteoarthritis Index (WOMAC) score, a disease-specific measure, was calculated based on 24 questions from the KOOS and is used to evaluate the areas of pain, stiffness, and physical function. 18 Range of motion (ROM) was also measured.
All operative and postoperative complications, whether treatment-related or not, were recorded and reported. Any event requiring surgical intervention and/or hospitalization was classified as a serious adverse event (SAE).
A responder endpoint was calculated by combining 3 elements: decrease in pain, increase in function, and safety. The final responder endpoint was achieved if a participant met all 3 of the criteria as defined in Table 1.
Responder endpoint.

Abbreviations: KOOS, Knee Injury and Osteoarthritis Outcome Score; WOMAC, Western Ontario and McMaster Universities Osteoarthritis Index.
Statistical analysis
Descriptive statistics were compiled using Microsoft Excel (Microsoft Corp.,USA). Categorical variables were presented as counts and proportions. Continuous data were presented as means with their standard deviations. We also determined the proportion of subjects who experienced a clinically meaning ful ⩾20% and ⩾10-point absolute improvement in their WOMAC pain and function scores at 2 years.19-21
Adverse events were summarized and categorized by their severity and relation to the study device or procedures. An AE that was considered recurrent was updated at the subsequent visit. Such cases were counted as one event, and the highest severity was assigned. The SAEs and AEs leading to discontinuation of the study were also recorded.
Study safety success was defined as an absence of unanticipated serious adverse device effects (USADEs).
Results
Demographics
Twenty-six subjects were enrolled and treated in the study. Baseline characteristics are available in Table 2. The mean age of the subjects at the time of surgery was 51 ± 8.8 years (range: 31-63). There were 16 men (61.5%) and 10 women (38.5%). Their average weight and BMI were 84.4 ± 9.4 kg and 28.4 ± 3.2 kg/m², respectively. Twenty-three subjects (88.5%) were employed or self-employed at the time of enrollment. Six subjects (23.1%) had a history of smoking, and mean symptom duration was 28.3 months. Half of the subjects had prior surgery in the study knee. All devices were successfully implanted, and no concomitant procedures, including arthroscopy, were performed. At 2 years, 25 of 26 subjects (96.2%) returned for follow-up visits with the device in situ.
Baseline characteristics (N = 26).
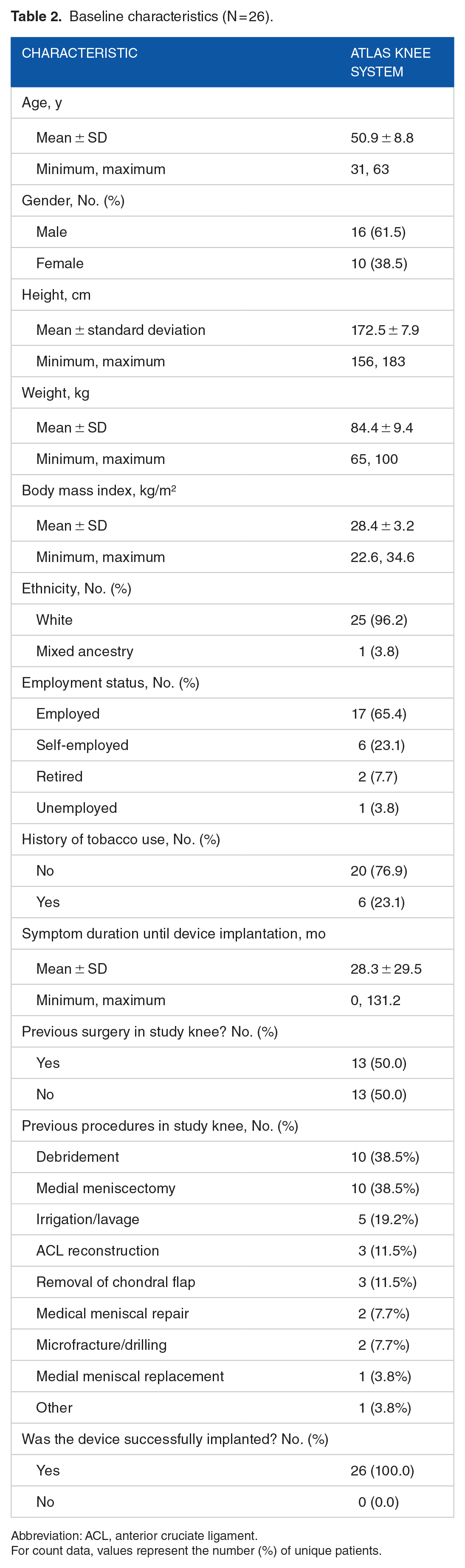
Abbreviation: ACL, anterior cruciate ligament.
For count data, values represent the number (%) of unique patients.
Procedure and return to activity information is summarized in Table 3. The average procedure time was 92.8 minutes, and the average time to discharge was 28.8 hours. Subjects, on average, returned to their preoperative activity level within 23 days (range: 5-63).
Procedure and return to activity information.

Responder endpoint
Of the 25 subjects who had their implant in situ, all (25 of 25, 100%) had a clinically meaningful ⩾20% improvement in their WOMAC pain score with a change ⩾10 points at 2 years; 24 of 25 (96%) had a clinically meaningful improvement in their WOMAC function score at 2 years (Table 1). Pain and function scores are described in the following section. There were no USADEs reported during the study.
Clinical outcomes
WOMAC pain and function subscales all improved over the 2 years relative to baseline (Table 4; Figures 1 and 2). Pain scores improved from 53.5 ± 8.6 at baseline to 15.0 ± 10.8, and function scores improved from 48.4 ± 17.2 at baseline to 18.8 ± 14.8.
Results of clinical outcome measures.
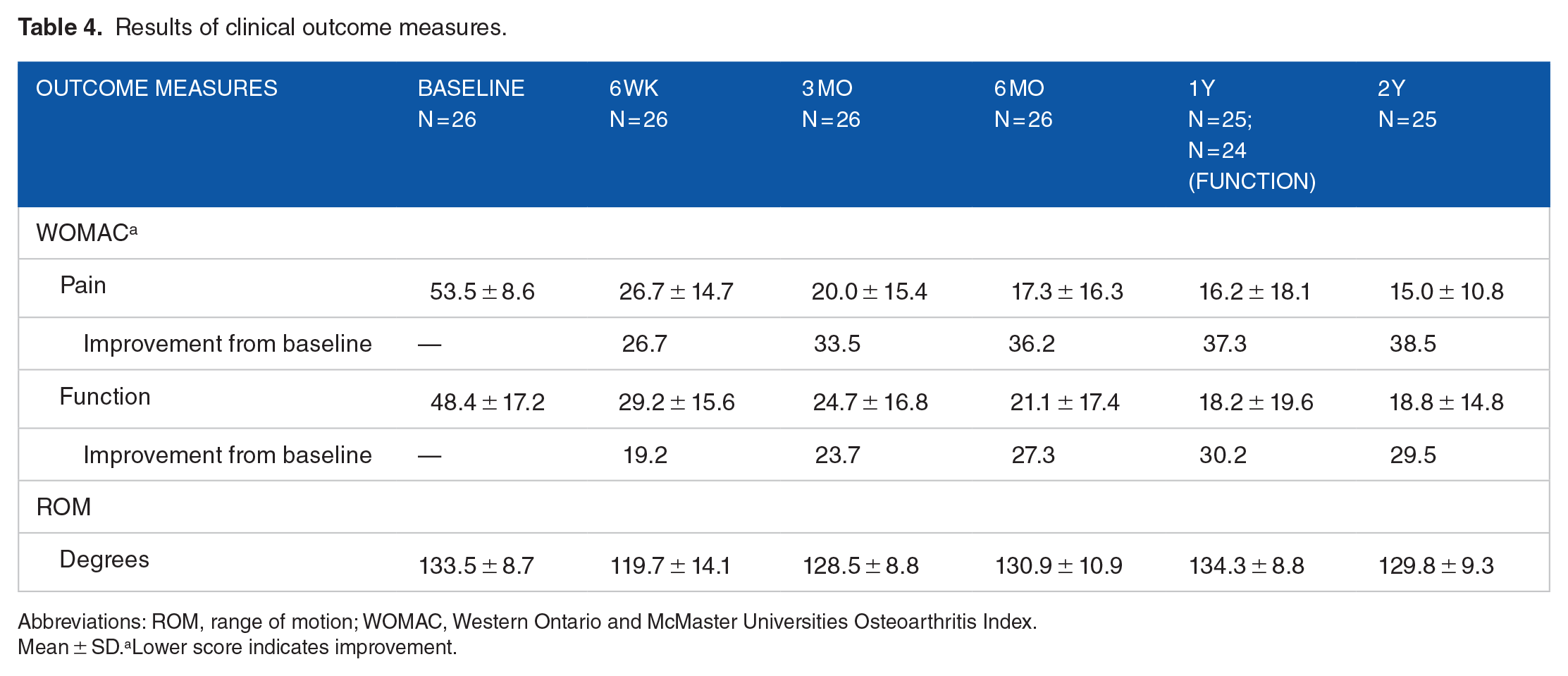
Abbreviations: ROM, range of motion; WOMAC, Western Ontario and McMaster Universities Osteoarthritis Index.
Mean ± SD.aLower score indicates improvement.
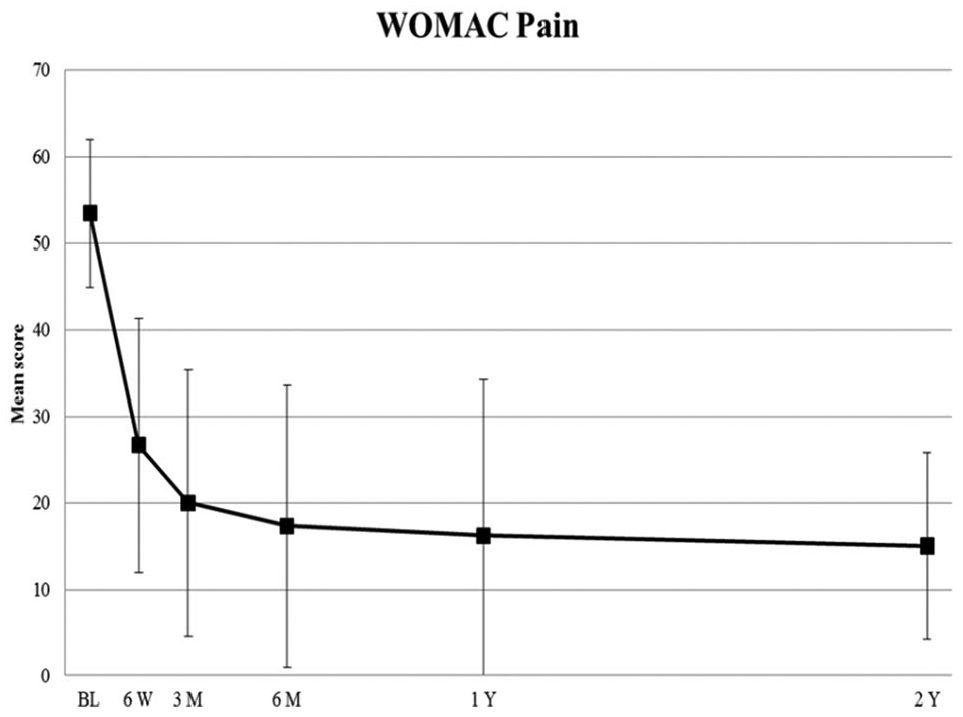
Mean (±SD) WOMAC pain scores from baseline to 2 years. Lower scores indicate improvement. BL indicates baseline; M, months; W, weeks; Y, years. WOMAC indicates Western Ontario and McMaster Universities Osteoarthritis Index.

Mean (±SD) WOMAC function scores from baseline to 2 years. Lower scores indicate improvement. BL indicates baseline; M, months; W, weeks; Y, years. WOMAC indicates Western Ontario and McMaster Universities Osteoarthritis Index.
Range of motion values initially decreased from baseline (133.5 ± 8.7) to 6 weeks (119.7 ± 14.1) but returned to normal at the 6-month visit, and this ROM was maintained over the 2 years (Table 2; Figure 3).
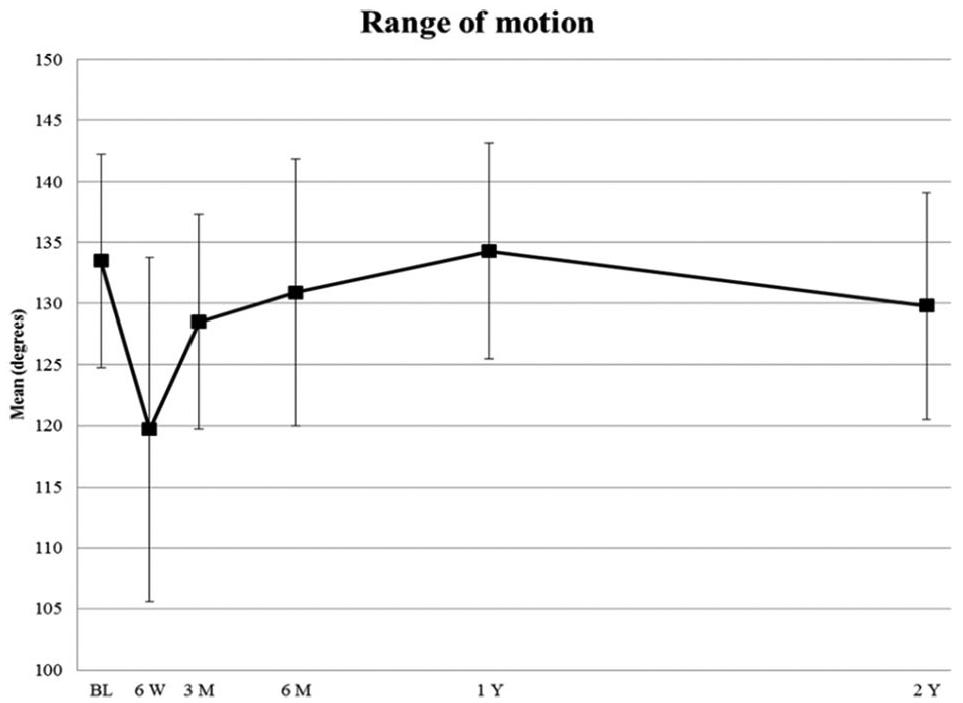
Mean (±SD) range of motion values (in degrees) from baseline to 2 years. BL indicates baseline; M, months; W, weeks; Y, years.
Safety
There were no unanticipated SAEs in the study. All operative and postoperative complications, whether treatment-related or not, were recorded (Table 5). None of them resulted in study discontinuation. The most commonly reported event was knee pain (15.4% of subjects). No infection or deep vein thrombosis (DVT) occurred.
Adverse events.

Abbreviation: ROM, range of motion.
On review of X-rays, radiolucency around screws were observed, which are typical and anticipated for metal implants.
Subject with thin subcutaneous tissue and prominent hardware.
Of 5 SAEs, 2 were related to the device or procedure (2 counts of knee pain; Table 6). The unrelated SAEs were for uterine bleeding, pneumonia, and a painful trochlear lesion.
Serious adverse events.

At the end of the 2-year period, 4 subjects had a total of 5 secondary operations. One subject had the implant removed after the 1-year visit (due to knee pain and stiffness). One subject had uterus surgery, one subject had a bronchoscopy, one subject had arthroscopic debridement and microfracture of a trochlear lesion, and one subject had arthroscopic meniscectomy and medial osteophyte removal. None of the subjects required device revisions.
Discussion
The current study evaluated the effectiveness and safety of the Atlas System for individuals with medial compartment knee OA. Of the 25 subjects who had their implant in situ at 2 years, all had a clinically meaningful improvement in pain and 96% had a clinically meaningful improvement function. In addition, the subjects reported a rapid return to preoperative activity levels within a mean of 23 days, which compares favorably to a prior report that patients were unable to work for a median of 87 days after an HTO procedure. 22 These results indicate that the treatment is well-targeted toward the difficult-to-treat young arthritic population. All concomitant procedures, including arthroscopy, were prohibited in this study of the Atlas System, so the outcomes can be attributed to the joint unloading provided by the implant.
This form of joint unloading therapy is not intended to cure the underlying OA disease, and pain with activity is possible. As the population comprised working-age subjects with mild to moderate knee OA, it is not surprising that knee pain was the most common AE. As such, appropriate expectations should be set preoperatively with each candidate patient and, in this active population, a previous patient preference study suggested that knee OA sufferers will often tolerate some pain if they can avoid bone-cutting treatments and maintain desired activities. 23 Therefore, the potential of an implantable unloader as a bridge treatment to help patients delay an eventual arthroplasty can be a valuable option.
As with any treatment, the overall benefit-risk assessment is crucial. In this study, 25 of 26 subjects maintained the device in situ at the 24-month follow-up visit, with one elective removal, due to knee pain and stiffness, occurring at 1 year post implant. One of 26 subjects (3.8%) experienced a device malfunction, and further inspection of the study data confirms a clean safety profile with no infections, DVTs, or unanticipated SAEs during the study.
The results of the current study of the Atlas System support the clinical utility of unicompartmental joint unloading implants.5,24-27 More conservative joint unloading techniques have been proposed in the past (ie, unloader knee braces and lateral wedge insoles) and, although positive results have been shown with these biomechanical interventions, adherence and discomfort are primary concerns. The orthotic will need to be tailored to the individual patient; can cause problems in the foot, ankle, and hip; and does not correct the underlying pathology.9-12,14,28-30 Sustained knee joint unloading may only truly be possible via surgery. 29 Of note, it is important to be aware of the contraindications of this particular device and to consider other surgical options (ie, HTO or arthroplasty) when a patient presents with one or several of these contraindications.
Although an HTO can unload the joint, the procedure involves invasive bone alterations, a potentially prolonged recovery, and may actually accelerate OA in the lateral compartment as a result of the longer term load shifting to this compartment.3,4,13 The procedure is also contraindicated in those with malalignment based purely on degenerative changes.8,15 Joint distraction has also been investigated, but the distraction fixator can cause discomfort, pin site infection, and osteomyelitis2,15; in comparison, no infections occurred during this trial.
A strength of this study was the strict eligibility criteria and rigorous protocol, which ensured that the sample included the targeted patient population and treatments effects could only beat tributed to the investigation device. The instruments included in this investigation are validated, disease-specific patient-important outcome measures. These results also confirmed that the findings seen at 1 year were sustained to 2 years. A limitation of this trial was its limited sample size. There was also no comparator to evaluate how the Atlas Knee System performs against one of the many knee OA treatments. Randomized trials are considered the gold standard study design when estimating comparative effects between treatments; however, studies involving surgical interventions may have additional concerns to address regarding ethics and feasibility.31-33 In this particular case, we must also consider patient preferences to avoid potential randomization to a more invasive procedure, the different indications and contraindications of these various therapies, and surgeon experience. For example, arthroplasty tends to be recommended for patients who have more advanced disease, are older, and less active than those who may be offered a joint unloading device. Finally, this investigation took place at clinical sites in Poland and South Africa only, so it is unclear whether the results would also be generalizable to patients with knee OA in other geographical locations.
Conclusions
This study highlights the potential benefit of a joint unloading device in the management of younger patients with mild to moderate medial knee OA who failed conservative treatment and do not want to undergo a more invasive surgical intervention. The clinically meaningful improvements in pain and function, clean safety profile, and rapid return to preoperative activity level suggest that the implantable unloader may serve as a potential bridge therapy for patients with medial knee OA who hope to delay arthroplasty.
Supplemental Material
Supplementary_data_xyz2097119f28066 – Supplemental material for Two-Year Results of the PHANTOM High Flex Trial: A Single-Arm Study on the Atlas Unicompartmental Knee System Load Absorber in Patients With Medial Compartment Osteoarthritis of the Knee
Supplemental material, Supplementary_data_xyz2097119f28066 for Two-Year Results of the PHANTOM High Flex Trial: A Single-Arm Study on the Atlas Unicompartmental Knee System Load Absorber in Patients With Medial Compartment Osteoarthritis of the Knee by Konrad Slynarski, Jacek Walawski, Robert Smigielski and Willem van der Merwe in Clinical Medicine Insights: Arthritis and Musculoskeletal Disorders
Footnotes
Acknowledgements
The authors thank Christopher Vannabouathong for his assistance in preparing the manuscript.
Funding:
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The study was funded by Moximed, Inc.
Declaration of Conflicting Interests:
K.S., J.W., R.S., and W.v.d.M. were clinical investigators in the PHANTOM High Flex trial. The authors declare no other conflicts of interest.
Author Contributions
Conceived and designed the experiments: KS, JW, RS, WV. Analysed the data: KS, JW, RS, WV. Wrote the first draft of the manuscript: KS, JW, RS, WV. Contributed to the writing of the manuscript: KS, JW, RS, WV. Agree with manuscript results and conclusions: KS, JW, RS, WV. Jointly developed the structure and arguments for the paper: KS, JW, RS, WV. Made critical revisions and approved final version: KS, JW, RS, WV. All authors reviewed and approved of the final manuscript.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
