Abstract
Background and Aims
Approximately half of persons living with HIV (PLWH) in the US smoke cigarettes. Large surveys show that 16.9%–37.3% of PLWH are never smokers compared to 57.5% of US adults. Similar proportions of PLWH and general population adults describe themselves as ex-smokers (20.3% vs 21.9% respectively). Little research has been done to characterize PLWH non-smokers. In this study, we compared a group of well characterized PLWH ex-smokers (i.e., no cigarettes for at least 5 years) to PLWH never smokers with the aim of developing a clearer understanding of the characteristics of these groups and the differences between them.
Design
Cross-sectional interview study employing audio computer-assisted self-interview (ACASI).
Setting
Comprehensive HIV care center in New York City.
Participants
In 2018–2019, we recruited a sample of PLWH never smokers (N = 54) and long-term ex-smokers (no cigarettes for at least 5 years, N = 36). Non-smoking status of participants was verified by exhaled carbon monoxide.
Measurements
We collected a range of sociodemographic, historical, clinical, and psychobehavioral data pertaining to tobacco use.
Results
Compared to never smokers, ex-smokers were older, more likely to have heterosexually acquired HIV and less likely to have same-sex-acquired infection, more likely to have parents and/or siblings who smoked, more likely to have current smoker/s in their households, and more likely to have ever used marijuana, cocaine, and/or heroin.
Conclusions
We describe important demographic and sociobehavioral differences between PLWH never smokers and ex-smokers that may be useful in crafting an effective response to the cigarette smoking epidemic in US PLWH.
Introduction
The tobacco use epidemic embedded within the HIV epidemic has received increased attention in recent years.1-6 The ultimate goal of all public health efforts directed toward this problem is to shrink the population of PLWH smokers and expand the population of PLWH ex-smokers and never smokers. Many prior studies have characterized PLWH smokers in the US, including 2 large studies of national samples within the past decade.3,6 Much less is known about PLWH who do not smoke cigarettes, that is, ex-smokers and never smokers. There are significant quantitative differences in the relative proportions of current smokers:ex-smokers:never smokers in PLWH compared to the general population in the US, and a better understanding of factors that drive these differences may be useful in addressing tobacco use in PLWH. In sharp contrast to the general population, current smokers outnumber never smokers in US PLWH.3,6 In nationally representative samples, 18.3%–20.3% of PLWH are ex-smokers and 37.3%–38.4% are never smokers.3,6 These figures contrast with an ex-smoker prevalence of 14.6%–21.9% and a never smoker prevalence of 56.2%–57.5% in all US adults.3,6 Other large surveys of PLWH have reported even lower proportions of never smokers including a New York (NY) statewide study reporting that 23.9% of PLWH were ex-smokers, and only 16.9% were never smokers. 5 PLWH smoke more and quit less than the general population.3,6
Most smokers initiate their cigarette use as teenagers, and the US government has prioritized tobacco prevention in youth and young adults over the past decade. 7 Understanding the characteristics of PLWH who have never smoked could be helpful in directing future tobacco prevention efforts in the highly vulnerable segment of the young population at risk for HIV infection.
Whereas the tobacco treatment literature focuses on PLWH who achieve abstinence in the short term, generally at time points ranging from 3 to 12 months, we are only aware of 1 clinical trial that has reported on quitters at a time point beyond 12 months. 8
There are important social and behavioral differences between ex-smokers, never smokers, and current smokers in the general population. 9 Several studies have explored the behavioral characteristics of ex-smokers in non-PLWH populations.9-11 The literature related to never smokers is dominated by the topic of lung cancer in those who have never smoked cigarettes, but a small number of studies have aimed to describe never smokers in various populations.9,12,13
Whereas multiple trials have summarized the sociodemographic and behavioral characteristics of PLWH smokers and short-term quitters, there is a paucity of published information on those PLWH who have successfully quit smoking for the long term and the additional subset of PLWH who have never smoked in their lifetimes. Such information could be useful to those seeking to improve the quit rates achieved by cessation interventions and by those involved in preventing the initiation of tobacco use in persons, particularly youth, at risk for HIV infection. In this paper, we describe a group of PLWH who have quit smoking for more than 5 years and a group of PLWH never smokers.
Materials and methods
Montefiore Medical Center’s Center for Positive Living (CPL) is a comprehensive HIV care program serving the needs of over 3000 PLWH mostly drawn from the surrounding Bronx communities.
Between November 2018 and March 2019, we recruited participants from the CPL clinic by provider referral, self-referral in response to flyers, and direct recruitment in the clinic. Smoking status and behaviors were assessed utilizing questions derived from the Centers for Disease Control Question Inventory on Tobacco. 14 For the sake of this study, we applied a rigorous definition of “ex-smoker,” requiring at least 5 years smoke-free, since earlier research in other populations has demonstrated a significant relapse rate in the early years, especially between years 2 and 3, after a quit. 15 Ex-smokers were, therefore, defined by an affirmative response to “Have you smoked at least 100 cigarettes in your entire life?” and a negative response to “Have you smoked a cigarette, even a single puff, in the past 5 years?” Never smokers were defined by negative responses to the questions “Have you smoked at least 100 cigarettes in your entire life?” and “Have you smoked a cigarette (even a puff) in the last 7 days, including today?” Non-smokers with adequate English language skills who consented completed an audio computer-assisted self-interview (ACASI) and received a $50 incentive.
Behavioral and clinical data
The following information was collected by ACASI (QDS software): Sociodemographics, clinical history, other tobacco and/or e-cigarette use, family and domestic partner histories of tobacco use, alcohol use (Alcohol Use Disorders Identification Test (AUDIT), 16 past and current illicit substance use, smoking abstinence self-efficacy (Self-efficacy/Temptations Scale), 17 loneliness (modified UCLA Loneliness Scale), 18 depression (Center for Epidemiologic Studies Depression Scale, CES-D 20), 19 anxiety (General Anxiety Disorder 7-Item Scale, GAD-7), 20 perceived stress (Perceived Stress Scale-4, PSS-4), 21 and social support (Interpersonal Support Evaluation List–12, ISEL-12). 22 The latter 7 scales are standard measures that have been previously validated in tobacco use research as noted in the reference list. In addition, we abstracted from the medical record the most recent CD4+ lymphocyte count and HIV-1 viral load. Finally, non-smoking status was verified biochemically with an exhaled carbon monoxide (ECO) level of <10 parts per million (ppm, Bedfont piCO+ Smokerlyzer).
Categorical variables were analyzed using chi-square or Fisher exact test, where appropriate. Comparisons of means were accomplished using Student t-test or Mann–Whitney U test, where appropriate. Statistical analyses were completed using SPSS V25.0 software.
All aspects of the study were reviewed and approved by the Albert Einstein College of Medicine Institutional Review Board.
Results
Sociodemographic and clinical characteristics of the study sample of HIV-infected persons who do not smoke cigarettes (n = 90).
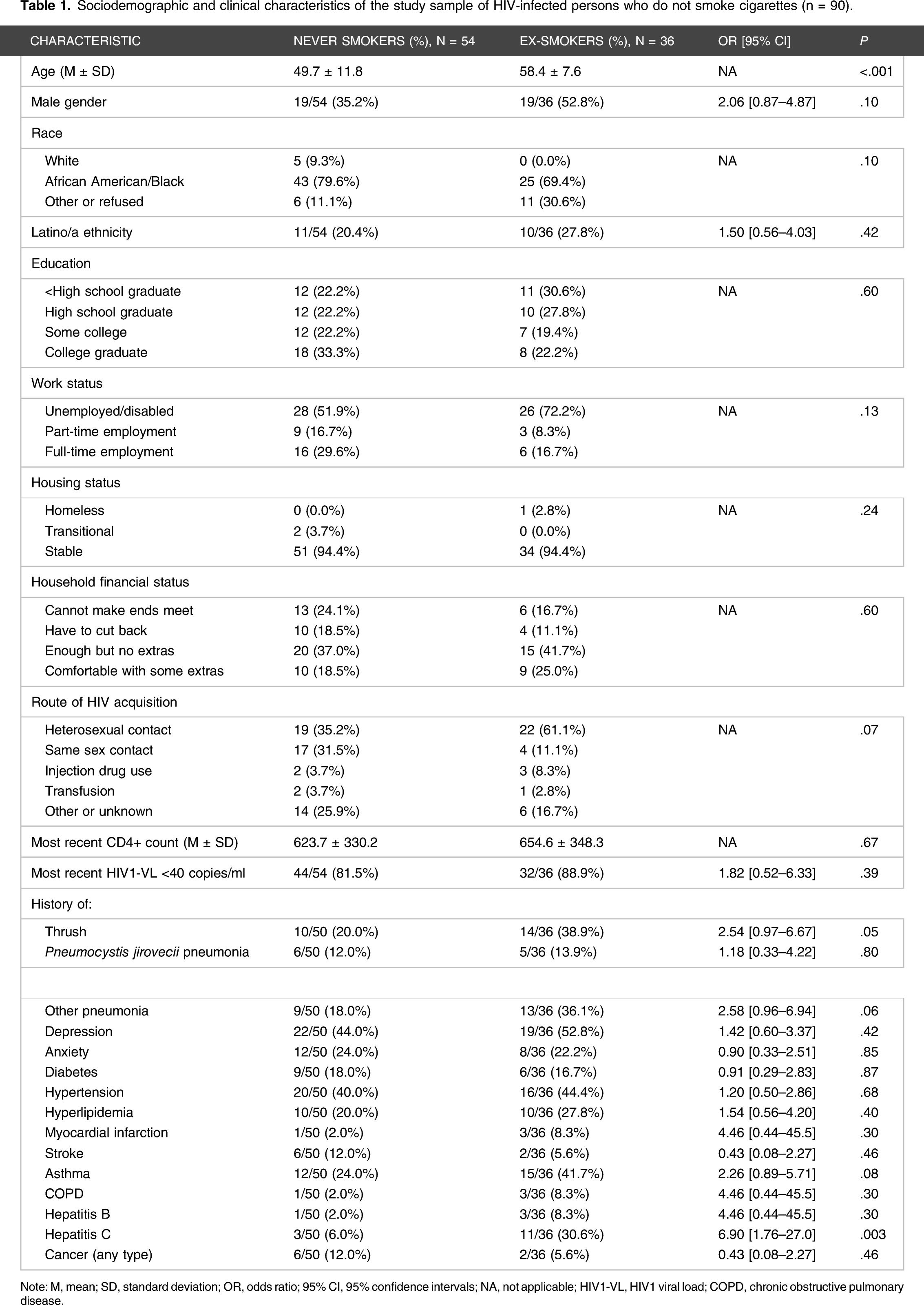
Note: M, mean; SD, standard deviation; OR, odds ratio; 95% CI, 95% confidence intervals; NA, not applicable; HIV1-VL, HIV1 viral load; COPD, chronic obstructive pulmonary disease.
Behavioral characteristics of the study sample of HIV-infected persons who do not smoke cigarettes (n = 90).

Note: M, mean; SD, standard deviation; OR, odds ratio; 95% CI, 95% confidence intervals; NA, not applicable; AUDIT, Alcohol Use Disorders Identification Test; CES-D, Center for Epidemiologic Studies Depression Scale; GAD, generalized anxiety disorder; ISEL-12, Interpersonal Support Evaluation List–12; UCLA, University of California Los Angeles.
Ex-smokers
The ex-smokers in this trial reported smoking initiation at a mean age of 15.3±4.0 years, smoking a mean of 14.2±10.0 cigarettes/day, and quitting for good a mean of 16.4±7.4 years prior to study enrollment. Almost three-fourths of the group was unemployed or disabled, and only 25.0% described their household financial situation as “comfortable.” Half of the ex-smokers were living with a cigarette smoker at the time that they quit. The great majority (86.1%) quit without any pharmacotherapy, and none of them reported use of a quitline or counseling. Some of this non-use was likely related to the enrollment requirement of at least 5 years of continuous abstinence, translating into a mean quit year of 2003 and a latest quit year of 2014.
Three participants (8.3%) had used a non-cigarette tobacco product within the past 30 days. Thirteen (36.1%) were currently living with an active smoker. Nineteen (52.8%) reported that their fathers were smokers, and 18 (50.0%) reported that their mothers were smokers. Of the 22 who were aware of siblings’ smoking behaviors, 19 (86.4%) had at least one sibling who smoked. Of the 30 who had children, 14 (46.7%) had at least one child who smoked.
On the AUDIT questionnaire, 15 (41.7%) reported complete alcohol abstinence, 32 (88.9%) scored in the low-risk category, and one (2.8%) scored in the hazardous category. Twenty-eight (77.8%) reported ever using marijuana, and 4 (11.1%) reported current marijuana use (i.e., within the past 30 days). Twenty-two (61.1%) reported ever using cocaine, and none reported current cocaine use. Eight (22.2%) reported ever using heroin, none reported current heroin use, and one (2.8%) was receiving methadone maintenance.
On the CESD-20 depression scale, 18 (50.0%) scored in the none–mild range, 5 (13.9%) in the moderate range, and 10 (27.8%) in the depressed range. On the GAD-7 anxiety scale, 22 (61.1%) scored in the none–minimal range, 8 (22.2%) scored in the mild range, 4 (11.1%) scored in the moderate range, and 2 (5.6%) scored in the severe range. On the Self-Efficacy/Temptations Scale, which describes a variety of positive affect, negative affect, and craving situations related to cigarette smoking and queries how tempted the participant would be to smoke cigarettes in those situations, answers were almost uniformly “Not at all tempted” or “Not very tempted.” However, 5 participants indicated higher levels of temptations “at a bar and having a drink.”
Never smokers
Four participants who reported never smoking cigarettes (7.4%) had used a non-cigarette tobacco product, and 3 (5.6%) had used an e-cigarette within the past 30 days. More than half of the group was unemployed or disabled, and only 18.5% described their household financial situation as “comfortable.” Eight (14.8%) participants were currently living with an active smoker. Nineteen (35.2%) reported that their fathers were smokers, and 10 (18.5%) reported that their mothers were smokers. Of the 28 who were aware of siblings’ smoking behaviors, 12 (42.9%) had at least one sibling who smoked. Of the 31 who had children, 11 (35.4%) had at least one child who smoked.
On the AUDIT questionnaire, 22 (40.7%) reported complete alcohol abstinence, 47 (87.0%) scored in the low-risk category, and one (1.9%) scored in the hazardous category. Twenty-one (38.9%) reported ever using marijuana, and 5 (9.3%) reported current marijuana use (i.e., within the past 30 days). Five (9.3%) reported ever using cocaine, and none reported current cocaine use. Three (5.6%) reported ever using heroin, none reported current heroin use, and none were receiving methadone maintenance.
On the CESD-20 depression scale, 31 (57.4%) scored in the none–mild range, 9 (16.7%) in the moderate range, and 11 (20.4%) in the depressed range. On the GAD-7 anxiety scale, 28 (51.9%) scored in the none–minimal range, 13 (24.1%) scored in the mild range, 5 (9.3%) scored in the moderate range, and 6 (11.1%) scored in the severe range. On the Self-Efficacy/Temptations Scale, answers were almost uniformly “Not at all tempted” or “Not very tempted.” However, 5 participants indicated higher levels of temptations “at a bar and having a drink.”
Comparison of ex-smokers to never smokers
As detailed in Tables 1 and 2, a number of statistically significant differences were noted on univariate analyses comparing PLWH ex- and never smokers. Compared to never smokers, ex-smokers were older, more likely to report acquiring HIV through heterosexual contact and less likely to report it from same-sex contact, more likely to report histories of thrush and hepatitis C, more likely to have parents and/or siblings who smoked, more likely to have current smoker/s in their households, and more likely to have ever used marijuana, cocaine, and/or heroin.
Discussion
Tobacco use has emerged as a major problem in the PLWH population, and efforts to confront it have met with significant challenges. Understandably, the literature and research in this area have focused on PLWH who actively smoke cigarettes. Building a profile of PLWH ex-smokers, especially long-term ex-smokers, has importance because a primary goal of tobacco control measures targeting PLWH is to convert as many current smokers as possible into long-term ex-smokers. We established a very rigorous definition of “long-term ex-smokers” in this study since in the general population there is a significant relapse rate in the early years, especially between the second and third year, that drops gradually over time. 15 Although we required at least 5 years smoke-free to enroll, our ex-smoker sample had quit a mean of 16.4 years prior to the study and, thus, had most likely quit for life. A clearer understanding of what a successful PLWH ex-smoker “looks like” may help to guide intervention development and better assist PLWH trying to change their smoking behaviors.
There is a marked disparity in the proportion of never smokers in the PLWH community as compared to the US adult population as a whole. The vast majority of PLWH smokers began smoking cigarettes prior to their HIV diagnoses. There is little doubt that multiple factors act individually or in concert to promote smoking initiation as well as HIV acquisition. However, many individuals subjected to these influences manage to completely avoid tobacco use. A better understanding of the factors that induce young adults at risk for HIV infection to avoid tobacco use could prove useful in promoting smoke-free living at the young ages during which smoking prevention is so critical.
A startling finding of our study was the significant rate of non-cigarette tobacco use in both never-(cigarette) smokers and long-term ex-(cigarette) smokers. These individuals would likely be classified as non-tobacco users on cursory screening in clinic, but 7.8% reported the current use of cigars, cigarillos, pipes, hookahs, blunts, and/or chewing tobacco. We repeated all analyses excluding participants who admitted to other tobacco product use, and there were no appreciable changes to the findings that we observed. These statistics highlight the need for comprehensive screening for non-cigarette tobacco and nicotine product use in all PLWH.
Given the reality that every study participant was either a never smoker or a long-term ex-smoker, it is not surprising that almost all of them expressed high levels of confidence in their ability to resist smoking temptations in a variety of situations. It is interesting that within both categories of non-smokers, the setting that was most tempting was in a bar while having a drink.
We are not aware of prior research investigating family and household smoking histories in PLWH. As noted in Table 2, never smokers were significantly less likely than ex-smokers to grow up with nuclear family members who smoked cigarettes. These differences were significant for mothers, both parents in the aggregate, and siblings. Our finding of transgenerational smoking patterns is consistent with observations made in the general population. 23 At the time of the study interview, never smokers were significantly more likely than ex-smokers to live in non-smoking households. Half of ex-smokers were living with another smoker at the time that they quit for good, and 36.1% of them were living with a smoker at the time of the study interview. These findings suggest that never smokers may preferentially assort with non-smokers as domestic/housing partners.
In addition to contributing to the corpus of knowledge relating to tobacco use behaviors in PLWH, our findings may have practical value to those caring for PLWH non-smokers and smokers. It is clear that screening for tobacco use should not be limited to questions about cigarettes since a surprisingly large minority of PLWH non-smokers use other tobacco products. We are not aware of any prior research delineating the multigenerational nature of tobacco use in PLWH and their families. This should be acknowledged in behavioral interventions and is worthy of future scientific inquiry. The temptation of long-term ex-smokers and even never smokers to smoke in social situations involving alcohol is interesting and suggests that abstinence counseling should advise PLWH to anticipate and perhaps avoid such high-risk situations. Finally, the frequency that long-term ex-smokers and never smokers share living space with current smokers is important for several reasons: (1) these “low-risk” individuals may still incur the significant health risks associated with second-hand smoke, (2) counseling should provide practical advice about maintaining abstinence in a home where cigarette smoking occurs, and (3) the myth that “it is impossible to quit” if there are other smokers in the home needs to be dispelled.
Our study had limitations that require mention. All participants were recruited from a single center in the Bronx, and they may not be representative of PLWH in other geographic areas. Although a number of prior studies have characterized PLWH smokers, our sample does not include a contemporaneous group of current smokers for comparison, and this may limit the ability to contextualize some of the findings. Our sample size was small, creating the possibility that some important associations were undetected and precluding us from completing more sophisticated, multivariate analyses. We employed an ECO definition for non-smoker (<10 ppm) that accorded with expert guidance at the time that the study was conceived, 24 but this cut point has been lowered in the most recent guidelines. 25 However, the authors of those guidelines do note that non-smokers commonly have ECO values up to 8 ppm in industrialized urban centers, and 94% of our sample had ECO levels below this lower cut point.
This study is the first to carefully characterize the clinical, sociodemographic, and behavioral profiles of PLWH who do not smoke cigarettes. As tobacco use continues to be the leading killer of PLWH in the US, and as effective measures to control the rampant tobacco use in this population remain elusive, 26 a more thorough understanding of the factors that drive tobacco use and non-use in the PLWH community may be helpful in guiding practice and policy in the future.
Footnotes
Author contributions
The content is solely the responsibility of the authors and does not necessarily represent the official views of the National Institute on Drug Abuse or the National Institutes of Health.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by award 1R01DA036445 (JS) from the National Institutes of Health (NIH)/National Institutes on Drug Abuse (NIDA) and by the Einstein-Rockefeller-CUNY Center for AIDS Research (P30-AI124414) which is supported by the following NIH co-funding and participating institutes and centers: NIAID, NCI, NICHD, NHBL, NIDA, NIMH, NIA, FIC, and OAR.
