Abstract
The development of the nervous system requires precise outgrowth, extension, and wiring of both axons and dendrites to generate properly functioning neural circuits. The molecular mechanisms that shape neurite development, in particular dendritic development, remain incompletely understood. Dendrites are often highly branched and coated with actin-filled, thorny protrusions, called dendritic spines, that allow for increased numbers of synaptic contacts with neighboring neurons. Disruptions in dendritic spine development have been implicated in many neurological disorders such as autism, schizophrenia, and Alzheimer’s disease. Although the development of dendritic spines is vital for cognitive function, understanding the mechanisms driving their outgrowth and stabilization
Introduction
During neuronal development, a single axon branch and multiple dendritic arbors emerge from the soma and extend toward specific partners to form the complex networks of connections that comprise the nervous system. As dendrites continue their growth, thorny protrusions called dendritic spines appear and coat the highly branched dendritic arbors. These postsynaptic structures are specialized compartments where most excitatory synaptic communication occurs. Dynamic changes in the density of spines provide a mechanism for structural plasticity at synapses and are associated with learning and memory.
1
Spine plasticity is regulated by altering the rates of spine formation and disassembly, and disruptions in these processes are linked with neurological diseases such as autism spectrum disorders, schizophrenia, and Alzheimer’s disease.
2
Recent work has identified a variety of pathways involved, however, our understanding of the mechanisms that govern spine extension and the formation of postsynaptic specializations
Our work investigated molecular mechanisms shaping partner-specific synaptic connectivity using the motor circuit of the nematode
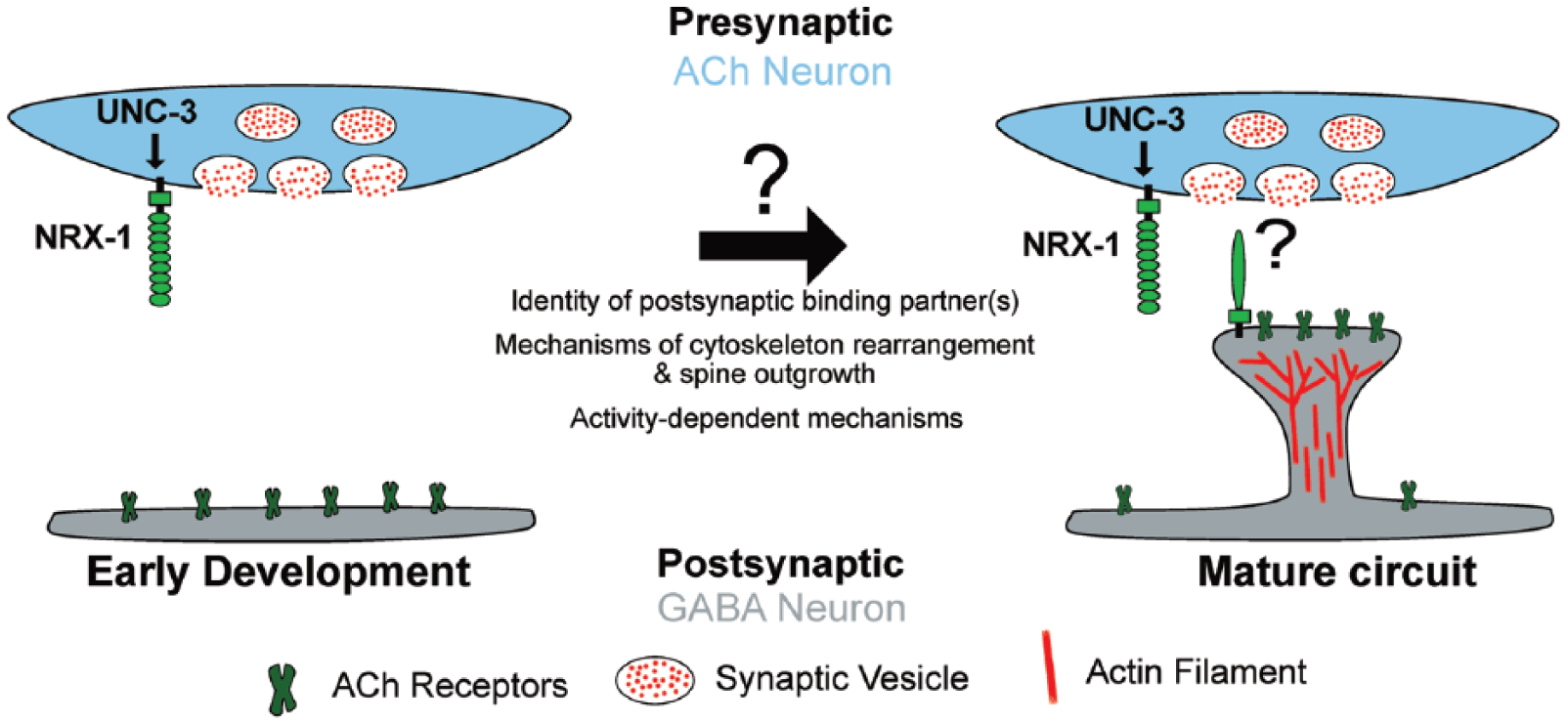
Presynaptic
Novel Spine-Like Protrusions in the C. elegans Motor Circuit
We sought to define and investigate genetic pathways important for specifying the connectivity of cholinergic motor neurons with their postsynaptic partners, GABAergic motor neurons, and muscles. To accomplish this, we used clusters of the GFP-tagged postsynaptic acetylcholine receptor subunit (ACR-12) as markers for synapses onto GABAergic neurons.4,5 Strikingly, we found that ACR-12::GFP clusters appear to protrude from the main dendritic shaft of DD neurons, where cholinergic inputs are known to be located from prior electron microscopy (EM) studies and localize to the tips of dendritic spine-like protrusions.6,7 These finger-like projections range from 0.3 to 1 µm in length and are clearly evident with selective labeling of DD neurons. Initial electron microscopy studies had reported the presence of dendritic spines on DD neurons, 6 but these structures had remained previously undetected and uncharacterized by light microscopy. We hypothesized that spine-like protrusions may perform similar roles to mammalian dendritic spines, and their location would then demarcate important sites of synaptic input. We examined a marker that labeled the presynaptic domains of the cholinergic neurons and found that DD spine-like protrusions were apposed to clusters of cholinergic synaptic vesicles in presynaptic axons, consistent with this proposed synaptic role.
Prior to establishment of the mature circuit organization, DD neurons undergo a specialized form of developmental synaptic remodeling, where the axonal and dendritic compartments are exchanged. 7 In newly born larvae, DD neurons receive cholinergic synaptic input along their dorsal side and innervate ventral muscles. After the first larval stage, DD neurons relocate their synaptic connections to innervate dorsal muscle and receive ventral synaptic inputs from newly born VA/VB cholinergic motor neurons. When do spines first appear? We noted that spine outgrowth coincides with the completion of DD synaptic remodeling, suggesting that these structures may serve specialized functions in the mature circuit.
Postsynaptic Development Requires NRX-1
Using a candidate approach to identify regulators of AChR clustering in GABAergic neurons, we found that the synaptic organizer neurexin/
To assess the functional requirement for
The requirement for presynaptic NRX-1 raises important questions about how
Future Directions
The discovery of spiny postsynaptic processes presented in this work raises many new and exciting questions. Are these GABAergic spine-like protrusions evolutionary precursors to mammalian dendritic spines? Spine-like structures have been observed in flies,
10
bees,
11
and at squid giant synapses,
12
suggesting that these specializations may have emerged early in the evolution of the nervous system. In the mammalian brain, most excitatory synapses occur on dendritic spines.
1
Recent technological advances in microscopy and molecular techniques have enhanced our understanding of dendritic spines. Spines can vary in size and number, through development and across neuron type but can be broadly classified into morphological categories that often correlate with their function and maturity.
1
Cytoskeletal actin and actin regulators appear particularly important in determining spine morphology and plasticity.
1
Further analysis of dendritic spines in
Our studies revealed that
Mammalian neurexins encode three isoforms, α, β, γ-neurexins, each with various neuronal functions.
8
In
Deletions in
Are spines a general feature of worm neurons? Diverse studies of neuronal morphology from both our lab and others provide evidence that this is not the case. The presence of these specialized structures on GABAergic neurons therefore suggests that they may perform specialized roles in these neurons. One possibility is that the requirement for spines arises as a consequence of synaptic remodeling that occurs in DD neurons. Our analysis suggests that spines are formed either coincident with, or shortly after the completion of remodeling. Adult synaptic inputs to DD neurons are formed post-embryonically, and spine outgrowth may be critical for forming proper synaptic contacts with newly born cholinergic neurons. Indeed, outgrowth of spines may represent a second phase of the remodeling program. Alternatively, the development of spines may provide a spatial mechanism permitting increased numbers of synaptic inputs to GABAergic neurons. Future examination of spine outgrowth in the context of DD neuron remodeling, as well as additional efforts to search for spines in other neuronal classes, will address whether spines perform specialized functions in GABAergic neurons or are a more generalized feature of
Our recent work has uncovered novel postsynaptic specializations that appear morphologically similar to mammalian dendritic spines and identified presynaptic NRX-1 as an important player in directing spine outgrowth and postsynaptic assembly. Future studies will exploit the genetic and experimental strengths of
Footnotes
Funding:
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests:
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Author Contributions
DO and MMF both conceptualized, prepared, and finalized the commentary. KA reviewed commentary draft.
