Abstract
Fishes are equipped to sense stressful stimuli and are able to respond to environmental stressor such as hypoxia with varying pattern of stress response. The functional attributes of brain to hypoxia stress in relation to ion transport and its interaction during immune challenge have not yet delineated in fish. We, therefore, explored the pattern of ion transporter functions and messenger RNA (mRNA) expression of α1-subunit isoforms of Na+/K+-ATPase (NKA) in the brain segments, namely, prosencephalon (PC), mesencephalon (MC), and metencephalon (MeC) in an obligate air-breathing fish exposed either to hypoxia stress (30 minutes forced immersion in water) or challenged with zymosan treatment (25-200 ng g−1 for 24 hours) or both. Zymosan that produced nonspecific immune responses evoked differential regulation of NKA, H+/K+-ATPase (HKA), and
Keywords
Introduction
Fishes possess both innate and adaptive immune responses and are able to respond to environmental perturbations. 1 The nonspecific immune response in fish acts as a powerful attribute with versatility due to their opposing and slower specific immune response compared with mammals.2,3 Furthermore, nonspecific immunity that produces inflammatory mediators and acute phase proteins shows upregulation during an active response. 4 For example, zymosan treatment that evokes nonspecific immune response can activate phagocytic and lysozomal functions in air-breathing fish, providing evidence for a direct association of ion transporter functions with phagocytic and lysozomal activities in this fish. 5
As a consequence of physiological reaction to stressor, stress response alters the capacity of fish to maintain their homeostasis.6,7 They respond to stressors with complex and adaptive mechanisms that constitute a network of neuroendocrine and physiological interactions.8-10 Activation of neuroendocrine axis that releases catecholamines and corticosteroids from the brain-sympathetic nerves-chromaffin cell and hypothalamo-pituitary-interrenal (HPI) axes occurs during stress response.6-8 For example, a rise in cortisol after exposure to nontoxic stressor such as net confinement has been observed in a number of fishes,6,11 including the air-breathing fish Anabas testudineus. 9 In contrast, a lowered plasma cortisol level after zymosan treatment has been found in this fish and that indicates suppression of a cortisol-driven stress response during immune challenge. 5
A bidirectional communication that operates between the brain and immune components has been established in fish. 12 Here, a network of ligands and their receptors together with cytokines, chemokines, and neural pathways interact at all levels of neuroendocrine and immune components. 12 As a key event during this bidirectional communication, sharing of receptors, production of hormonal ligands, and cytokines occur in these cellular systems.13,14 Traditionally, as a sensor, immune system component informs the central nervous system (CNS) to trigger a neuroendocrine response. 15 On the contrary, stress that demands neuroendocrine interference communicates with central and autonomic nervous system and modifies HPI axis6,9 and the components of immune system.12,16 Consequently, a change in the activity of discrete populations of brain neurons may occur during immune system activation. 17 It is likely that the sensitivity of fish brain to both immune and neuroendocrine challenges may thus demand integrative functions of ion transporters that coordinate neuronal functions.
Teleost fish brain comprises 3 major segments, namely, prosencephalon (PC) (forebrain), mesencephalon (MC) (midbrain), and metencephalon (MeC) (hindbrain). Prosencephalon has telencephalon that constitutes olfactory lobes located rostroventrally to pair cerebral hemispheres18,19 and diencephalon that constitutes thalamus, epithalamus, and hypothalamus with the preoptic, postoptic, and the tuberal areas. 19 Mesencephalon that constitutes the optic tectum, torus longitudinalis, and tegmentum continues caudally into the MeC. 20 The MeC represents the most caudal part of the brain with the cerebellum and the brain stem.19,21 The 3 major serotonergic neural groups that exist in the teleost brain as pretectal population, posterior tuberculum/hypothalamic populations, and raphe populations make a network of neuronal circuitries that are sensitive to physiological challenges.22,23 Serotonergic neurons are also present in the pineal gland, area postrema, medulla oblongata, and spinal cord in the brain of teleosts. 23 In teleosts, serotonergic fibers from the brain directly project to the pituitary, 22 whereas in some teleost species, serotonin-immunoreactive cells are also present in the pituitary indicating its local production.22,24 Serotonergic neurons that are distributed in the superior raphe project to the forebrain and midbrain, extend to inferior raphe, and project to hindbrain-spinal cord region in the brain of the teleosts. 23
Dopaminergic neurons are typically located in defined groups of forebrain and midbrain 25 and possess catecholamine-containing neurons in the diencephalon. 26 Dopaminergic and noradrenergic neurons that constitute the major hindbrain of the teleost fish have been shown activated during stressful challenges.27-29 The glutamatergic neurons while distributing in the granular layers of the cerebellum, and several other diencephalic, mesencephalic, and rhombencephalic regions, dominate also in the nuclei of the dorsal telencephalon. 30 γ-aminobutyric acid (GABA)-mediated (GABAergic) neurons, however, dominate the nuclei of the ventral telencephalon. 18
Na+/K+-ATPase (NKA) that establishes electrochemical gradient of Na+ and K+ across the plasma membrane maintains resting membrane potential and electrical activity of neurons for neurotransmitter uptake and its osmotic balance. 31 This transporter that regulates Na+ and K+ transport acts as a measurable indicator of neuronal activity and excitability32,33 and its high concentrations in brain cellular membranes demands 40% to 50% of the adenosine triphosphate (ATP). 34 As a ubiquitous protein, NKA is abundant in brain regions and plays a fundamental role in maintaining cellular ionic gradients that are required for neural activity, transport of amino acids and glucose, and regulation of cell pH and volume.34-36 In addition, NKA acts as a key contributor to management of extracellular [K+] following activity-induced K+ release.37-39 Given its important role in regulating neural excitability, impairment of NKA function might lead to neural dysfunction. The activity of neuronal NKA that concentrates in the surrounding nerve ending membranes interacts with the CNS through specific receptors for classical neurotransmitter and neuropeptides. 40 For example, many neuromodulators including dopamine have been shown to regulate NKA activity, 41 probably by regulating the levels of cyclic adenosine monophosphate 42 and free radicals.36,43
Na+/K+-ATPase contains 2 major subunits, α and β, which function as a αβ heterodimer. The α-subunit is a large (110-120 kDa) protein that contains all the functional sites and is responsible for the catalytic functioning of the enzyme. The different subunit composition and cellular/subcellular distribution of NKA may confer cell type–specific properties. 44 Four isoforms of the NKA α-subunit, nkaα1, nkaα2, nkaα3, and nkaα4, have been identified in mammals, 45 and 3 isoforms, nkaα1, nkaα2, and nkaα3, have been identified in mammalian brain.45,46 The presence of distinct cellular NKA isoforms (α1, α3, β1 in neurons and α1, α2, β1, β2 in astrocytes) provides clues on distinct physiological roles of these isoforms in the CNS, 37 which could be attributed to the distinct apparent ion affinities (Km) of the different α and β isoform combinations and their subsequent neurophysical functions. 47
Na+/K+-ATPase that expresses in all cell types
45
can substitute
H+/K+-ATPase (HKA), a member of P-type transport adenosine triphosphatase (ATPase), is responsible for ATP-dependent exchange of H+ for K+ across plasma membranes. 54 H+/K+-ATPase is expressed at a very high level in mammalian gastric parietal cells, where it functions primarily as a proton pump transporting H+ in exchange for K+ into secretory canaliculi and, from there, into the gastric lumen.55,56 In addition to parietal cells, HKA has been shown to present in a variety of other cells including colonic enterocytes and renal tubular cells. 54 Presence of nongastric HKA has been found in elasmobranch gills of Atlantic stingray 57 and in the osmoregulatory epithelia of air-breathing fish A testudineus. 5 However, its role in the brain ion transport has not been looked into except a study that reported the presence of HKA in microglia of brain. 58 A critical role for HKA in the regulation of K+ and H+ gradients in the brain especially in the regulation of K+ homeostasis in microglia shows its higher magnitude for K+ transport compared with epithelial HKA. 58 It is thus plausible that the dynamics of HKA in brain would reflect the functionality of glia cells as it can contribute to glial cell volume and pH regulation which would modulate the neuronal functions in general. 58 In this context, it is reasonable to presume that HKA may act as glial marker that appears to be sensitive to both hypoxia and immune challenges at least in fish brain.
It is not certain that how these ion transporter functions operate during hypoxia stress-immune interaction in the varied neuronal clusters in fish brain. We, therefore, examined the response of ion transporters in the brain segments of an obligate air-breathing fish A testudineus Bloch to either zymosan-induced immune challenge or water immersion–induced hypoxia stress or both. The response pattern of Na+/K+, H+/K+, and
Materials and Methods
Fish holding conditions
The climbing perch (A testudineus Bloch), a tropical freshwater air-breathing fish, was used as the test species. This native teleost fish, belonging to order Perciformes and family Anabantidae, inhabits the backwaters of Kerala in Southern India. They are equipped to live in demanding environmental conditions with their well-defined physiological and biochemical mechanisms.59,60 Due to its sensitivity to various biotic and abiotic stressors, this obligate air-breathing fish serves as an excellent model to study the advanced physiological mechanisms including gas transport, acid/base, ionic, and metabolic regulations.7,60-62 These fish, approximately 40 ± 5 g, were collected from the wild conditions in their postspawning phase and held in the laboratory conditions for 3 weeks under natural photoperiod (12 hours L:12 hours D) at water temperature ranging from 28°C to 29°C with a mean water pH of 6.2. Fish were fed with dry commercial fish feed at 1.5% of body mass and were transferred to 50-L glass tanks for 2 independent static experimentations. Fish ate their normal food and there was no mortality during experimentation. The regulations of Animal Ethical Committee of the University were followed.
Experimental design
Dose-responsive effects of zymosan on brain segments
The dose-responsive in vivo effect of zymosan-induced experimental peritonitis (25-200 ng g−1) within 24 hours 63 was tested for the activity of Na+/K+-ATPase (NKA), HKA, NNA, and nkaα1-subunit isoform transcripts in PC, MC, and MeC of the fish. In total, 32 laboratory-acclimated fish kept in cement tanks were transferred into glass tanks (30 cm × 30 cm × 60 cm) and kept as groups with 8 fish in each group for another 2 weeks. The first fish group was received intraperitoneal injection of 0.65% NaCl as vehicle, which served as sham control. The remaining 3 groups of fish were given varied doses of zymosan (25, 50, and 200 ng g−1) for 24 hours.
Effect of hypoxia stress in immune-challenged fish
A selected zymosan dose was employed in another set of experiment that tested the impact of hypoxia stress on brain ion transport functions in immune-challenged fish. For that purpose, 2 subsets of fish groups, namely, nonstressed fish (I) and hypoxia-stressed fish (II), were held with 8 fish in each group. The first fish group in nonstressed subset I that served as sham controls received saline (0.65% NaCl) as vehicle. The second group fish were given intraperitoneal injection of a selected dose of zymosan (200 ng g−1) that showed substantial acid/base responses after 24 hours (unpublished data, 2017). Each fish group in the subset II that comprised 2 groups was given saline injection (control) and zymosan, respectively, for 24 hours. Before sampling, these fish groups were given hypoxia stress for 30 minutes. Water immersion was practiced to induce hypoxia stress in these fish by keeping them under water for 30 minutes without allowing gulping or breathing air. All fish groups were sampled together after the stipulated time interval.
Sampling and analysis
Feeding was discontinued for 24 hours prior to experiment to ensure standardized experimental conditions. While handling for injection, care was taken to ensure minimum disturbance to fish. After the experimental treatment, the fish were quickly netted and dipped in 0.2% 2-phenoxyethanol (SRL, Mumbai, India) solution for 2 minutes. These anesthetized fish were held on a wet towel and blood was immediately drawn from the caudal artery. The fish were killed immediately by spinal trans-section, and the whole brain was then segmented into 3 portions based on the morphological and functional properties. The first segment, the PC, including the telencephalon and diencephalon; the second segment, the MC, including optic tectum, torus longitudinalis, and tegmentum; and the third segment, the MeC, comprising cerebellum, medulla, and a part of the spinal cord were separated. These 3 brain segments were blotted, weighed, and kept in ice-cold brain mitochondrial extraction (BME) buffer (pH 7.4) containing 0.25 M sucrose, 10 mM HEPES, 0.5 mM EDTA, and 0.5 mM EGTA and stored at −80°C for further analysis. Samples were extracted for mRNA expression analysis and were aseptically isolated and preserved in RNAlater solution (Ambion, Invitrogen, USA.) at −80°C for further analysis.
Quantification of ion-specific ATPases
NKA, HKA, and NNA-specific activities
Frozen PC, MC, and MeC were thawed and a homogenate (50 mg/500 μL) was prepared in BME buffer (pH 7.4) using glass homogenizer fitted with Teflon pestle giving 3 strokes as described previously (Samuel et al., 2014). 64 Homogenates were centrifuged at 2000g for 3 minutes at 4°C (Eppendorf 5430R, Germany.), and the membrane fraction prepared (Samuel et al., 2014) was used for quantifying the activity of ATPases, namely, NKA, HKA, and NNA. The protein concentrations in the tissues were determined using modified biuret assay 65 with bovine serum albumin as standard.
NKA-specific activity
The ouabain-sensitive NKA-specific activity in the PC, MC, and MeC was quantified adopting the method of Peter et al. (2000) modified for microplate assay. 60 Saponin (0.2 mg protein−1) was routinely added to optimize substrate accessibility. Samples in duplicates containing 1.0 µg protein were added to a 96-well microplate containing 100 mM NaCl, 30 mM imidazole (pH 7.4), 0.1 mM EDTA, and 5 mM MgCl2. About 0.13 mM KCl was used as the promoter and 0.14 mM ouabain was used as the inhibitor. After vortexing, the assay mixture was incubated at 37°C for 15 minutes. The reaction was initiated by the addition of 0.3 mM ATP and was terminated with addition of 8.6% trichloroacetic acid (TCA). The liberated inorganic phosphate was measured against phosphate standard at 700 nm in Synergy HT Biotek Microplate Reader, USA. The change in absorbance between promoter and inhibitor assays was calculated and regression analysis was employed to derive the rate of activity of NKA and expressed in micromoles of Pi liberated per hour per milligram of protein.
HKA-specific activity
SCH28080-sensitive HKA assay for brain homogenates was developed after extensive standardization including kinetics (data not shown) based on NKA microplate assay, 60 but SCH28080 was used as inhibitor instead of ouabain. As inhibitor, 0.1 mM SCH28080 (2-methyl-8-(phenylmethoxy)-imidazo[1,2-a]pyridine-3-acetonitrile) was used, whereas 0.13 mM KCl was used as promoter to measure HKA activity. Saponin-treated samples in duplicates (1.0 µg protein) were added to a 96-well microplate containing above reaction mixture, and the reaction was initiated by the addition of 0.3 mM ATP and incubated at 37°C for 15 minutes. The reaction was terminated by adding 8.6% TCA, and the inorganic phosphate content was measured in Synergy HT Biotek Microplate Reader. The change in absorbance at 700 nm between promoter and inhibitor assays was calculated using regression analysis, and the rate of transport activity of H+/K+-ATPase was expressed in micromoles of Pi liberated per hour per milligram of protein.
NNA-specific activity
An ouabain-sensitive NNA assay system has been developed for brain based on the previously described NKA microplate assay 60 but used 0.02 mM NH4Cl instead of 0.13 mM KCl. Extensive standardization of this assay system including kinetics was done (data not shown). Samples were treated with saponin (0.2 mg protein−1) to optimize substrate accessibility. Samples in duplicates containing 1.0 µg protein were added to a 96-well microplate containing 100 mM NaCl, 30 mM imidazole, 0.1 mM EDTA, and 5 mM MgCl2. As promoter, 0.02 mM NH4Cl and 0.14 mM ouabain were used in the assay as the inhibitor. After vortexing, the assay mixture was incubated at 37°C for 15 minutes. The reaction was initiated by the addition of 0.3 mM ATP and was terminated with addition of 8.6% TCA. The liberated inorganic phosphate was measured against phosphate standard at 700 nm in Synergy HT Biotek Microplate Reader. The change in absorbance between promoter and inhibitor assays was calculated, and regression analysis was employed to derive the rate of activity of NNA and expressed in micromoles of Pi liberated per hour per milligram of protein.
mRNA expression of NKA isoforms by quantitative reverse transcription-polymerase chain reaction
Fresh PC, MC, and MeC were excised immediately after sampling and kept in RNAlater solution (Ambion, Invitrogen) and stored at −80°C. Total RNA was isolated from the PC, MC, and MeC tissues and purified using the PureLink RNA Mini Kit (Ambion, Invitrogen, Life technologies). RNA purity was validated by optical density (OD) absorption ratio (OD 260/280 nm) using BioPhotometer (BioPhotometer Plus; Eppendorf), and the samples that showed above 2.0 were taken for complementary DNA (cDNA) synthesis. Complementary DNA was synthesized from 1 µg of total RNA in StepOne Real-Time qPCR System using the High Capacity cDNA Reverse Transcription Kit (Applied Biosystems, Life Technologies, Massachusetts, USA.) following the manufacturer’s protocol. The thermal cycling conditions were as follows: 25°C for 1 minute, 37°C for 120 minutes, 85°C for 5 minutes, and finally 4°C holding stage. The cDNA sequences of NKA α-isoforms; nkaα1a (JN180940), nkaα1b (JN180941), and nkaα1c (JN180942) were obtained from GenBank (Table 1). The assay mix contained the custom-made primers with FAM-labeled MGB probe (Applied Biosystems, Life Technologies). Primers for all genes were designed (Table 1) using Primer Express Software (version 2.0.0; Applied Biosystems).
Primer sequences of Na+/K+-ATPase (NKA) α1-subunit isoforms of Anabas testudineus Bloch.
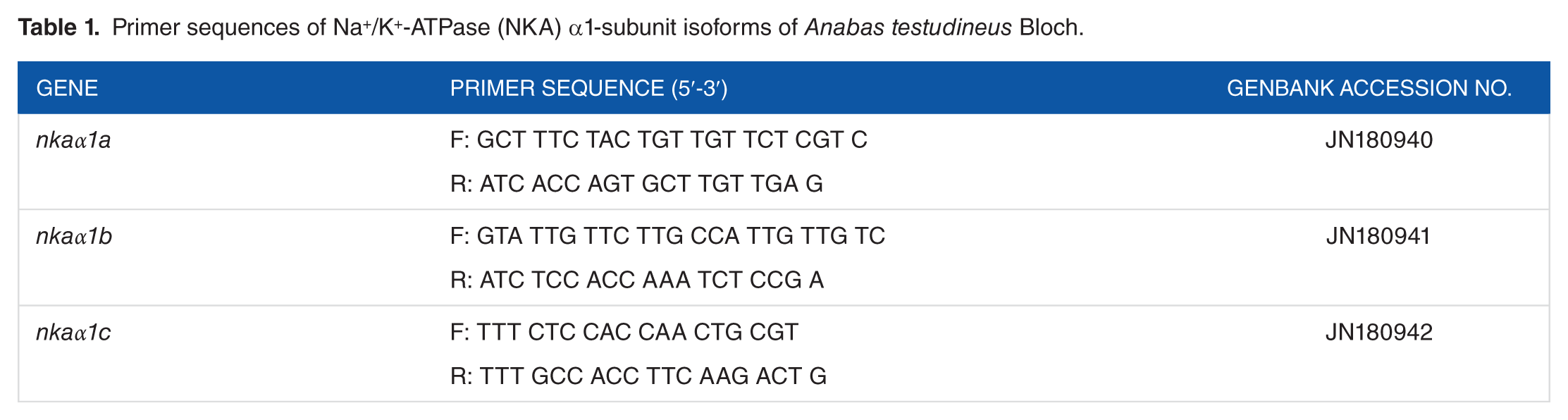
Quantitative reverse transcription-polymerase chain reaction was performed in duplicates using the StepOne Real-Time qPCR system (Applied Biosystems) using TaqMan assays (Applied Biosystems, Life Technologies). The reactions contained 5 µL of 2× TaqMan Universal Master Mix, 0.5 µL 20× custom-made primers (Table 1), and cDNA (1 ng) or standard (1 µL) in a total volume of 10 µL. Cycling conditions were kept as 50°C for 2 minutes and 95°C for 10 minutes for 1 cycle followed by 40 cycles of 95°C for 15 seconds and 60°C for 1 minute. 18S ribosomal RNA was used as the endogenous control (Applied Biosystems). Data (threshold cycle [Ct] values) were collected at real time. The Ct, slope, PCR efficiency, y-intercept, and correlation coefficient (R2) were calculated using the default setting of StepOne Software (Applied Biosystems). The amplification efficiency for nkaα isoforms was 95% to 100%, and the mRNA expression was calculated by adopting 2−ΔΔCt comparative Ct method (where ΔCt = ΔCt sample − ΔCt control. 66 The results were statistically analyzed using Relative Expression Software Tool 2009 (REST), and the values were then expressed relative to the control sample.
Statistics
Data were collected from 8 animals in each group. Before statistical analyses, data were checked for normal distribution and variance homogeneity. Data were subjected to 1-way analysis of variance followed by Student-Newman-Keuls test (GraphPad InStat-3; GraphPad Software, Inc., San Diego, CA, USA). Stastistical changes between the means were accepted as significant if P < .05. In dose-responsive zymosan treatments, the significance levels were represented as “
The relative mRNA expression of NKA α1-subunit isoforms nkaα1a, nkaα1b, and nkaα1c in brain regions after varied doses of zymosan treatment was statistically analyzed using REST, and the significant upregulation (x) and downregulation (y) between fish groups were recorded after depicting significance at P < .05.
Results
Dose-responsive effect of zymosan in intact fish brain
A dose-dependent rise (P < .001) in the NKA activity was found after zymosan challenge in PC, where serotonergic neurons were abundant (Figure 1A). The NKA activity that showed a rise (P < .05) in MC after a medium dose of zymosan treatment elevated (P < .001) its activity in MeC after a low dose of zymosan treatment (Figure 1A). Dose-dependent sensitivity of serotonergic neurons in the MC and dopaminergic neurons in the MeC to zymosan was observed in these fish brain segments. Likewise, a dose-dependent rise (P < .001) in HKA activity after zymosan treatment was observed in PC (Figure 2A). A low dose of zymosan treatment was necessary to produce a rise (P < .001) in HKA activity in MeC, where it showed a dose-dependent decline (P < .001) after medium and high dose of zymosan treatment (Figure 2A). On the contrary, the HKA activity of the MC remained unaffected after zymosan treatment (Figure 2A). A high dose of zymosan treatment was necessary to produce a rise (P < .01) in NNA activity in PC, where it declined (P < .01, P < .001) after low and medium doses of zymosan treatment (Figure 3A). A dose-dependent rise (P < .01, P < .001) in NNA activity after zymosan challenge was found in MC (Figure 3A). Likewise, NNA activity increased (P < .01, P < .001) after low and high doses of zymosan treatment in MeC (Figure 3A). Spatial and differential participation of both serotonergic and dopaminergic neurons could be found in these segments that transport

Dose-responsive effects of (A) zymosan (ZYM; 25, 50, 200 ng g−1) treatment for 24 hours on Na+/K+-ATPase activity in the prosencephalon (PC), mesencephalon (MC), and metencephalon (MeC) segments of Anabas testudineus. The activity pattern of Na+/K+-ATPase in the (B) prosencephalon, (C) mesencephalon, and (D) metencephalon brain segments after 30 minutes hypoxic stress in immune-challenged (zymosan; ZYM; 200 ng g−1) fish. Each point is mean ± SE for 8 fish. In Figure 1A, the significance levels are represented as “

Dose-responsive effects of (A) zymosan (ZYM; 25, 50, 200 ng g−1) treatment for 24 hours on H+/K+-ATPase activity in the prosencephalon (PC), mesencephalon (MC), and metencephalon (MeC) segments of Anabas testudineus. The activity pattern of H+/K+-ATPase in the (B) prosencephalon, (C) mesencephalon, and (D) metencephalon brain segments after 30 minutes hypoxic stress in immune-challenged (zymosan; ZYM; 200 ng g−1) fish. Each point is mean ± SE for 8 fish. In Figure 2A, the significance levels are represented as “

Dose-responsive effects of (A) zymosan (ZYM; 25, 50, 200 ng g−1) treatment for 24 hours on
Glowchart showing the differential activity pattern of Na+/K+-ATPase (NKA), H+/K+-ATPase (HKA), and

 indicates activation,
indicates activation,  indicates inactivation, and
indicates inactivation, and  indicates unaffected.
indicates unaffected.
Chart showing the functional response of NKA activity during hypoxia-stress-immune interaction in the neuronal clusters of Prosencephalon (PC), mesencephalon (MC) and metencephalon (MeC) of air-breathing fish.

Abbreviations: MC, mesencephalon; MeC, metencephalon; NKA, Na+/K+-ATPase; PC, prosencephalon.
Effect of hypoxia in immune-challenged fish brain
A rise (P < .05) in NKA activity after hypoxia stress was observed in the PC and MeC of intact brain, whereas its activity decreased (P < .05) after hypoxia stress in MeC of immune-challenged brain (Figure 1B and D). A compensatory role of dopaminergic neurons in these brain segments could maintain Na+ and K+ homeostasis after hypoxia stress and immune challenge. On the contrary, the NKA activity in the MC remained unaffected after hypoxia stress in the intact and immune-challenged brain (Figure 1C). The analysis of activity pattern of NKA revealed a shift in its activity pattern after hypoxia stress in the PC and MeC of the immune-challenged fish (Table 4).
Glowchart showing the differential activity pattern of Na+/K+-ATPase (NKA), H+/K+-ATPase (HKA), and

 indicates activation,
indicates activation,  indicates inactivation, and
indicates inactivation, and  indicates unaffected.
indicates unaffected.
A rise (P < .05) in HKA activity occurred after hypoxia stress in the MeC of intact fish brain (Figure 2D). An increased sensitivity of the dopaminergic neurons to hypoxia stress could be found in these neuronal cells. On the contrary, the HKA activity in PC and MC remained unaffected after hypoxia stress in intact and immune-challenged fish brain (Figure 2B and C). The analysis of activity pattern of HKA revealed that alterations in the MeC of intact fish brain showed a reversed pattern after hypoxia stress in immune-challenged fish, although its activity remained unaffected in the PC and MC (Table 4).
A rise in NNA activity occurred after hypoxia stress in PC and MC of the intact fish brain (Figure 3B and C). Activated serotonergic neurons that showed a demand for transporting
Dose-responsive effect of zymosan challenge on nkaα1 mRNA expression in intact fish brain
The mRNA expression of nkaα1a showed an upregulation after low dose of zymosan treatment in PC, whereas medium and high doses of zymosan downregulated its expression (Figure 4A). On the contrary, low and high doses of zymosan treatment upregulated the mRNA expression of nkaα1a in MC, whereas a medium dose of zymosan produced a downregulation (Figure 4A). An upregulation in the mRNA expression of nkaα1a after medium and high doses of zymosan treatment was found in the MeC (Figure 4A).

Dose-responsive effects of zymosan (ZYM; 25, 50, 200 ng g−1) treatment for 24 hours (Figure A) on the relative messenger RNA (mRNA) expression of NKA α-subunit isoform nkaα1a in the prosencephalon (PC), mesencephalon (MC), and metencephalon (MeC) segments of Anabas testudineus. The relative mRNA expression pattern of NKA α-subunit isoform nkaα1a in the (B) prosencephalon, (C) mesencephalon, and (D) metencephalon brain segments after 30 minutes hypoxic stress in immune-challenged (zymosan; ZYM; 200 ng g−1) fish. The data were statistically analyzed using Relative Expression Software Tool 2009 (REST), and the significant upregulation (x) and downregulation (y) between fish groups were recorded after depicting significance at P < .05.
Likewise, an upregulation of nkaα1b mRNA expression in PC was observed only at low dose of zymosan treatment, whereas medium and high doses of zymosan downregulated its expression (Figure 5A). On the contrary, low and high doses of zymosan treatment upregulated the mRNA expression of nkaα1b in MC, whereas medium dose produced a downregulation (Figure 5A). A downregulation of nkaα1b mRNA expression was found in MeC after zymosan challenge (Figure 5A). Likewise, a downregulation in the mRNA expression of nkaα1c after medium and high doses of zymosan treatment was found in PC (Figure 6A). Low and high doses of zymosan treatment upregulated the mRNA expression of nkaα1c in MC, whereas medium dose of zymosan downregulated its expression (Figure 6A). An upregulation in the mRNA expression of nkaα1c after low dose of zymosan treatment was observed in MeC, whereas its medium and high doses downregulated its expression (Figure 6A). The analysis of the expression pattern of nkaα1 subunit isoforms after zymosan treatment showed a dose-responsive pattern in the varied brain segments. A substantial sensitivity of MC that possesses more dopaminergic neurons to immune challenge was also observed (Table 5).

Dose-responsive effects of zymosan (ZYM; 25, 50, 200 ng g−1) treatment for 24 hours (Figure A) on the relative messenger RNA (mRNA) expression of NKA α-subunit isoform nkaα1b in the prosencephalon (PC), mesencephalon (MC), and metencephalon (MeC) segments of Anabas testudineus. The relative mRNA expression pattern of NKA α-subunit isoform nkaα1b in the (B) prosencephalon, (C) mesencephalon, and (D) metencephalon brain segments after 30 minutes hypoxic stress in immune-challenged (zymosan; ZYM; 200 ng g−1) fish. The data were statistically analyzed using Relative Expression Software Tool 2009 (REST), and the significant upregulation (x) and downregulation (y) between fish groups were recorded after depicting significance at P < .05.

Dose-responsive effects of zymosan (ZYM; 25, 50, 200 ng g−1) treatment for 24 hours (Figure A) on the relative messenger RNA (mRNA) expression of NKA α-subunit isoform nkaα1c in the prosencephalon (PC), mesencephalon (MC), and metencephalon (MeC) segments of Anabas testudineus. The relative mRNA expression pattern of NKA α-subunit isoform nkaα1c in the (B) prosencephalon, (C) mesencephalon, and (D) metencephalon brain segments after 30 minutes hypoxic stress in immune-challenged (zymosan; ZYM; 200 ng g−1) fish. The data were statistically analyzed using Relative Expression Software Tool 2009 (REST), and the significant upregulation (x) and downregulation (y) between fish groups were recorded after depicting significance at P < .05.
Glowchart showing the differential pattern of messenger RNA expression of Na+/K+-ATPase α1-subunit isoforms in the prosencephalon (PC), mesencephalon (MC), and metencephalon (MeC) after varied doses of zymosan treatment (25, 50, 200 ng g−1) for 24 hours.

 indicates activation,
indicates activation,  indicates inactivation, and
indicates inactivation, and  indicates unaffected.
indicates unaffected.
mRNA expression of nkaα1 subunit isoforms in fish brain after hypoxia in zymosan-challenged fish
An upregulated nkaα1a mRNA expression after hypoxia stress was found in PC of immune-challenged fish (Figure 4B). But, hypoxia stress downregulated its mRNA expression in MC of intact fish brain, whereas upregulating its expression after hypoxia stress in immune-challenged fish brain (Figure 4C). On the contrary, hypoxia stress produced an upregulation in nkaα1a expression in the MeC of both intact and immune-challenged fish brain (Figure 4D). A downregulation of nkaα1b mRNA expression after hypoxia stress was found in PC and MC of intact fish brain, whereas an upregulation of nkaα1b after hypoxia stress was found in immune-challenged fish (Figure 5B and C). On the contrary, an upregulation in the mRNA expression of nkaα1b was found after hypoxia stress in the MeC in both intact and immune-challenged fish brain (Figure 5D). Likewise, a downregulation of nkaα1c mRNA expression was observed after hypoxia stress in PC of intact fish brain, whereas it upregulated after hypoxia stress in immune-challenged fish brain (Figure 6B). On the contrary, hypoxia stress in intact and immune-challenged fish produced a downregulation of nkaα1c mRNA expression in MC and MeC (Figure 6C and D).
The mRNA expression analysis of nkaα1 isoforms showed a shift in the pattern of nkaα1a and nkaα1b after hypoxia stress in the PC and MC of immune-challenged fish (Tables 6 and 7). Likewise, the analysis of nkaα1c pattern showed a shift in its expression after hypoxia stress in PC of immune-challenged fish brain (Tables 6 and 7). The mRNA expression analysis of nkaα1 isoforms showed differential pattern during hypoxia-immune challenges in the varied neuronal clusters, indicating a spatio-temporal regulation of NKA function in the neuronal clusters.
Glowchart showing the differential pattern of messenger RNA expression of Na+/K+-ATPase α1-subunit isoforms in the prosencephalon (PC), mesencephalon (MC), and metencephalon (MeC) after hypoxia stress in intact and immune-challenged fish.

 indicates activation,
indicates activation,  indicates inactivation, and
indicates inactivation, and  indicates unaffected.
indicates unaffected.
Chart showing the functional response of nkaα1a, nkaα1b and nkaα1c isoforms during hypoxia-stress-immune interaction in the neuronal clusters of Prosencephalon (PC), Mesencephalon (MC) and Metencephalon (MeC) of air-breathing fish.

Abbreviation: GABAergic, GABA-mediated.
Discussion
Similar to vertebrate brain, fish brain also coordinates the spatial and temporal physiological activities through the activation/inactivation of their specific neuronal groups. The membrane-bound NKA that maintains the asymmetric distribution of Na+ and K+ in neuronal cells shows synaptic and neuronal activity and is connected to neuronal functions. 67 In our study, we found a differential pattern of NKA regulation in both immune-challenged and hypoxia-stressed fish brain which suggests a differential sensitivity of brain NKA to these challenges. This further points to the differential regulation of neuronal groups including serotonergic and dopaminergic neurons to immune challenge as these neuronal clusters are more abundant in the PC and MeC of fish brain.22,23 The increased NKA activity after hypoxia stress in the PC and MeC of the intact fish brain showed a decrease in MeC of immune-challenged brain, indicating a spatial sensitivity of NKA to hypoxia stress and immune challenge. A reversal of this activation was found in the MeC during hypoxia stress-immune interaction. This deactivation of dopaminergic neurons during hypoxia stress-immune interaction probably points to a compromised dopamine release, which appears to be a protective action against hypoxia-stress and immune challenges.
Induction of hypoxia stress in immune-challenged fish brain, on the contrary, shows a shift in the pattern of NKA activity, and this further points that hypoxia stress has a direct control on brain Na+/K+-pump function. This would probably be due to a differential activation of functional neuronal clusters in the brain segments to the challenges in oxygen delivery and Na+ availability. This view is consistent with the fact that NKA shows more sensitivity to intracellular Na+ concentration besides its K+-dependent activation. 68 The dose-responsive inhibition of NKA activity to zymosan challenge in the PC indicates inactivation of neuronal clusters such as serotonergic, dopaminergic, glutamergic, and GABAergic neurons. On the contrary, hypoxia stress demands an activated NKA pattern in the PC that reveals activation of these neuronal clusters. These differential sensitivities of neuronal clusters further support the view that immune challenge could disturb the neuronal activity of neuronal clusters in the brain that shows spatial and differential responses. This further provides evidence for the critical role of NKA in the regulation of Na+ and K+ transport as a major target Na+ transporter. Interestingly, the pattern of NKA activity that showed little response to hypoxia stress in immune-challenged fish brain further points that hypoxia stress demands a modified NKA function during hypoxia stress-immune interaction. This classic NKA response pattern in fish brain presents clues on the specific target response of NKA to both hypoxia stress and immune-induced disturbance in ion transport. Consistent observation has also been found in the osmoregulatory epithelia of air-breathing fish where NKA responded differentially to both immune challenge and hypoxia stress. 5
Neuronal Na+ accumulation occurs gradually via ligand-gated and voltage-gated Na+ channels and in parallel to the release of K+ to the extracellular space. 67 It is evident in our study that both immune and hypoxia stress target intracellular Na+ signals that prompt an enhanced activation of neuronal NKA as reported earlier.69-72 It is likely that the immune challenge or hypoxia stress can extrude Na+ and uptake of K+ and that permits the neurons to reestablish the concentration gradients of these ions. This activation of the neuronal clusters in the tested segments of brain further indicates its differential sensitivity of varied types of neuronal clusters including serotonergic and dopaminergic neurons to hypoxia stress or immune challenges. Moreover, it is likely that the differential and spatial responses of these neurons appear to be due to the altered neuronal membrane potential. It is known that the release of dopamine from the dopaminergic neuronal synaptosomes is sensitive to pH changes.73,74 Similar sensitivity of neuronal clusters to acid/base variables after zymosan challenge has been found in our test species (Peter et al, unpublished data, 2017).
The abundant dopaminergic neurons in the MeC that respond to immune challenge as well as to hypoxic stress further provide clues that NKA in this brain segment could function as a neuronal marker that could fine-tune its neuronal activity during hypoxia stress-immune interaction. It has been further shown that the failure of NKA that leads to dissipated transmembrane H+ and Na+ gradients may contribute to unregulated dopamine efflux.75,76 It is further plausible to assume that the differential sensitivity of nkaα1 isoform expression may also be related to the varied demands for intracellular Na+ concentration in brain segments. Similar changes in Na+ signaling and its sensitivity have been attributed to a modified NKA function as it has been linked with neuronal activity.77-79 A lead role for NKA in the regulation of Na+ and K+ gradients has been well established in neurons where any alterations of this pump could lead to its altered physiological functions. Likewise, changes in the neuronal crosstalk with astrocytes could also lead to changes in brain function as neuronal activity requires association with astrocytes for the clearance of K+ from the extracellular space. 80
H+/K+-ATPase possesses a catalytic α-subunit and a regulatory β-subunit that use energy from ATP hydrolysis to transport H+ in exchange for K+. 81 The large, ~100-kDa, α-subunit with 10 transmembrane helices contains the catalytic site and translocates H+ and K+. A second, smaller glycosylated β-subunit, ~30 kDa, is required for proper trafficking and processing of the enzyme. 81 The HKα1 H+-K+-ATPase, also known as the gastric HKA, is sensitive to micromolar concentrations of imidazopyridine, 2-methyl-8-(phenylmethoxy)imidazole[1,2a]-pyridine-3-acetonitrile (SCH-28080), 81 whereas the HKα2 H+-K+-ATPase is sensitive to relatively high concentrations of ouabain. 82 Studies have shown that respiratory/metabolic acidosis82-84 and ammonia toxicity could stimulate SCH-28080–sensitive HKA activity in mice. 85 Recently, we found that SCH-28080–sensitive HKA transport system in the osmoregulatory epithelia of immune-challenged air-breathing fish responds to hypoxia stress. 5 The presence of this ion transporter in the varied brain segments that possess serotonergic and dopaminergic neurons provides evidence that the membrane potential of these neurons sensitizes both H+ and K+ ions. It appears that immune challenge that modifies HKA activity activates the serotonergic neurons in the PC while inactivating the dopaminergic neurons in the MeC. This spatial and differential regulation thus for the first time provides evidence for a role of H+ and K+ gradients in integrating ion signals in the brain neuronal clusters. There are reports showing that dopamine release could modify the neuronal transmembrane H+ and Na+ gradients due to its dependence on energy metabolism and Na+/H+ Exchanger (NHE) activity. 74 HKA activity pattern in the MeC during hypoxia stress and immune challenge in our test species also supports this view. The rise in HKA activity in the MeC of hypoxia-stressed fish showed a reversal after hypoxia stress in immune-challenged fish, indicating that hypoxia stress may activate the dopamine release from the MeC during these challenges. The unaffected HKA activity in the PC and MC also support this view. Overall, our HKA data indicate that this transporter is sensitive to both hypoxia stress and immune challenge as neurons are shown to be more permeable to K+ than to any other cation, and the reduction in its ion permeability (“channel arrest”) can be considered as a major strategy for saving energy during anoxia as reported earlier.86-88 Studies have found that anoxia can induce reduction in K+ permeability in the brain of the freshwater turtle Trachemys scripta. Likewise, in crucian carp, hypoxia can reduce the neural membrane “leakiness” for ions such as Na+ and Ca2+, a strategy that should lead to reduce neuronal excitability. 89
Fish can tolerate high internal ammonia compared with humans, and at high blood ammonia concentrations, NH3 probably enters the brain via diffusion.
90
Diffusion of ammonia into the brain is promoted by the lower intracellular pH of brain tissue, in which NH3 is trapped as ionized
Krogh proposed that Na+ absorption by fish is linked with
Ion-dependent ATPase plays an important role in intracellular functions and is considered as a sensitive indicator. There are 3 subunits of NKA, α, β, and γ, and each subunit has its isoforms. The minimum functional unit is a heterodimer of the α-subunit and β-subunit, which may also be co-expressed with the γ-subunit. 105 The NKA α-subunit (110-120 kDa) contains the catalytic unit responsible for binding and transporting Na+ out and K+ into the cell and the binding site for ATP. 45 Four NKA α-subunit isoforms (α1, α2, α3, and α4) have been identified in mammals 45 and nkaα1, nkaα2, and nkaα3 are expressed in the brain of all mammalian species examined.45,46,106,107 Here, we present evidence for the first time that 3 α1 isoforms such as nkaα1a, nkaα1b, and nkaα1c are expressed in brain segments of air-breathing fish as reported for the osmoregulatory tissues of this fish. 5 We also found that hypoxia stress and immune challenge can modify the mRNA expression of nkaα1 isoforms, indicating the major functional role of these isoforms in brain NKA regulation during immune and hypoxia stress challenges. The shift in the mRNA expression of nkaα1a and nkaα1b in the PC and MC to hypoxia stress indicates a differential regulation of these nka1 isoforms to both hypoxia stress and immune challenge, which in turn is associated with the differential response of serotonergic neurons that are abundant in the PC and MC of fish brain. These transcriptomic data further confirm the physiological role of brain NKA as a major Na+ transporter of brain that coordinates many physiological activities and also account for its observed spatial and differential regulation during hypoxia stress-immune interaction. In brain, NKA α1-subunit isoforms are found both in neuronal cells and astrocytes contributing to the control of membrane potential and K+ re-uptake after depolarization.44,108,109 In this sense, NKA is a crucial transporter that controls neuronal excitability and its dysregulation may lead to several dysfunctions including neurological and physiological and behavioral disturbances.
The activity patterns of the NKA ion transporter seem to be opposite to the expression pattern of the nkaα1 isoforms in the brain segments after challenges. However, a general shift in the pattern of the mRNA expression was noticed during hypoxia stress-immune interaction, suggesting the transcriptomic regulation of NKA during hypoxia stress-immune interaction. This further provides evidence for the differential sensitivity of neuronal clusters that are distributed abundantly in the PC and MC, whereas dopaminergic neurons in the MeC may play a compensatory role. While analyzing the differential expression pattern of the nkaα1 subunit isoforms, it is evident that MeC is less sensitive to the hypoxia stress in immune-challenged fish. Furthermore, the switching of nkaα1a and nkaα1b isoforms in the PC and MC clearly indicates a differential modification of sodium pump in these brain segments during hypoxia stress in immune-challenged fish. This spatial and differential genomic regulations of nkaα1 isoforms in these brain segments further demand transcriptomic regulations, which would alter the NKA activity during hypoxia stress-immune interaction. The differential activity of nkaα1 isoforms to bind with K+ or
The brain serotonergic system is crucial for the development of divergent stress coping styles and plays a central role in the processes of neuronal plasticity. 110 Activation of brain serotonergic system (serotonin, 5-hydroxytryptamine, 5-HT) along with catecholamines (norepinephrine and dopamine) has been found associated with stressful conditions in fish.111,112 We have found the involvement of many neuronal clusters mainly serotonergic and dopaminergic neuronal clusters during hypoxia stress-immune interaction in the fish brain. Similarly, stressful conditions that induce hyperactivity in the metabolism of brain serotonin both in mammals113-115 and in fish116,117 have been implicated to hormonal control. 117 Likewise, dopamine, primarily synthesized in the brain and kidney, has direct action on brain functions, where it is important in the control of locomotion, cognition, and affect neuroendocrine secretion.119-121 The involvement of dopaminergic neuronal clusters in fish brain could point to a role for dopamine in Na+ transport as reported earlier. 122 Hypothalamic proopiomelanocortin neurons that release GABA as excitatory transmitter and glutamate as inhibitory transmitter 123 are involved in neuronal control of physiological mechanisms.124,125 Furthermore, in fish, the transport of Na+ and K+ has been associated with either 5-HT3 receptor channel function 126 or stress-induced immunosuppressive effects in fish via glucocorticoids or serotonin. 127 It is thus obvious that crosstalks between neural, immune, and endocrine systems occur during immune-hypoxia stress interaction in fish.
In summary, this study explored the mechanism of hypoxia stress-immune interactions during ion transporter function in the brain segments of an air-breathing fish. We found for the first time that immune challenge evoked a varied response pattern of ion transporters that include NKA, HKA, and NNA in the different regions of fish brain that showed differential regulation to hypoxia stress. The differential and spatial modifications of these ion transporters along with transcriptomic analysis of nkaα1 isoforms in fish brain provide evidence for the regulatory functions of the serotonergic and dopaminergic neuronal clusters in fish brain to hypoxia stress and immune challenge. Our data provide evidence for the first time that highlighted the critical role of functional neuronal clusters in the integrative physiology of brain activities, where signaling of Na+, K+, H+, and
Footnotes
Acknowledgements
The authors thank DST, New Delhi, for granting a project on fish. They also thank the University Grants Commission, New Delhi, for SAP-DRS II Fellowship (S.S.), iCEIB project, Government of Kerala, and SAP facility in the Department of Zoology of the University of Kerala.
Funding:
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The authors received financial support from the iCEIB project.
Declaration of conflicting interests:
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Author Contributions
MCS designed the study, interpreted results and wrote the manuscript. SS performed the dose and stress experiments including the ATPase assays and RT-PCR analysis.
