Abstract
Mechanisms explaining the propensity of a primary tumor to metastasize to a specific site still need to be unveiled, and clinical studies support a link between chronic inflammation and cancer dissemination to specific tissues. Using different mouse models, we demonstrate the role of inflammation-generated extracellular matrix fragments ac-PGP (
Introduction
About 90% of cancer-related deaths are attributable to the metastatic spread of the primary tumor. 1 The importance of the relationship between microenvironment and tumor cells has become evident. However, precise mechanisms for the propensity of primary tumors to metastasize to a specific site are still to be unveiled. Tissues are in many circumstances (eg, exposure to cigarette smoke [CS], pollution, or infectious agents) infiltrated by immune cells and contain cytokines, chemokines, and growth factors that might significantly contribute to the seeding of metastatic cancer cells.2,3 Epidemiologic studies repetitively reported that many human cancers in adults are associated with chronic inflammation in different organs.4,5 Moreover, it is now clearly established that neutrophils can modulate immune responses within tumor microenvironment and interfere with tumor development in different manners (eg, establish a permissive microenvironment that favors tumor growth). 6 In humans, chronic obstructive pulmonary disease (COPD)-related inflammatory processes in smokers are associated with an increased risk of lung cancer as compared with smokers who do not have COPD. 7 In these patients, tobacco smoke exposure induces a rapid recruitment of neutrophils to lung tissues through a gradient of ELR+ (“glu-leu-arg” motif) CXC chemokine such as CXCL8.8,9
The mechanisms that might explain the favoring effect of neutrophils on cancer progression are still debated and it is established that these cells display immunomodulatory properties.
10
Hence, neutrophil-derived mediators released in lung microenvironment such as leukotrienes strongly support lung metastasis dissemination from breast cancer cells.
11
Neutrophils are able to interact with cells and extracellular matrix and the release of proteases as matrix metalloproteinase 9 (MMP-9) or other cytotoxic mediators can cause tissue damage in many diseases. MMP-9 plays important roles in tissue destruction and inflammation through proteolytic cleavages resulting in degradation of matrix proteins and activation of cytokines/chemokines. It was previously shown that collagen breakdown by MMP-8 and MMP-9 followed by a prolyl endopeptidase cleavage generates
The ac-PGP tripeptides have been reported to bind CXCR2 and trigger chemotaxis of inflammatory cells.17–19 In non–small-cell lung cancer samples, CXCR2 expression has been correlated with tumor inflammation and angiogenesis 20 and has been associated with a poorer prognosis. 21 Furthermore, in malignant breast cancer tissues, CXCR2 signaling plays a key role in tumor cell invasion and metastatic dissemination to lung tissues. 22 Interestingly, ac-PGP was also detected in the bronchoalveolar lavage (BAL), sputum, and serum from patients with COPD. 23 The molecular basis for ac-PGP chemoattractant effects on neutrophils relies on ac-PGP structural homology with the “GP” motif shared by all ELR+ CXC chemokines, including CXCL8. 24 Moreover, CXCR2 is expressed not only by neutrophils but also by a large variety of tumor cells originating from lung, melanoma, pancreatic, ovarian, and breast tissues, suggesting a possible role for ac-PGP tripeptides in tumor cell chemoattraction. 21
In this study, we explored the impact of extracellular matrix fragments (ac-PGP) generated by a pulmonary inflammatory burden induced by CS or lipopolysaccharide (LPS) instillation on cancer cell dissemination to lung parenchyma. MMP-9 originating from neutrophils generated collagen breakdown and the production of chemotactic ac-PGP that attracts tumor cells to lung parenchyma. The role of ac-PGP in cancer cells dissemination to lung was confirmed by synthetic ac-PGP instillation into the lungs. By silencing CXCR2 on tumor cells, we also demonstrate that the chemotactic effect of ac-PGP on tumor cells is dependent on CXCR2.
Materials and Methods
Cell culture
Murine melanoma B16F10 and murine mammary cancer 4T1 cell lines were purchased from Caliper Life Sciences (Hopkinton, MA, USA). Murine melanoma B16F10 cells were grown in RPMI-1640 (Roswell Park Memorial Institute; Lonza, Basel, Switzerland) supplemented with 10% fetal bovine serum (FBS) and 2 mmol/L
Mice
Male C57Bl/6J and BALB/c mice, 6 to 8 weeks old, weight ±20 g, were purchased from Charles River (Cologne, Germany). MMP-9 knockout (MMP-9−/−) mice and the corresponding wild-type (MMP-9+/+) mice were originally obtained from the Department of Anatomy, University of California, San Francisco (Z. Werb, UCSF) 25 and bred in our facility. All animal experimental procedures were approved by the ethical committee of the University of Liège.
Metastasis assays
Metastatic dissemination of murine melanoma B16F10 cells, murine mammary cancer 4T1 cells, and 4T1-shCXCR2 cells to lung tissues was investigated using 2 distinct metastasis models in syngeneic mice after inducing a pulmonary inflammation by CS or LPS exposure or ac-PGP/
Intravenous model—Mainstream CS exposure
C57Bl/6 mice (n = 8/group) were exposed to whole-body mainstream CS during 5 days per week and up to 5 weeks. For this, mice were placed in an inhalation chamber and daily exposed to 5 cigarettes with filters (Kentucky reference 3R4F; University of Kentucky Tobacco and Health Research Institute, Lexington, KY, USA). The negative-control group was exposed to ambient air. After 2 weeks of the start of CS exposure, murine melanoma B16F10 cells (1 × 105 cells/50 μL in serum-free medium) were injected into the lateral tail vein of mice.
Intravenous model—LPS stimulation protocol
Mice (n = 8/group) were anesthetized with 2.5% isoflurane/oxygen mixture and intratracheally injected with phosphate-buffered saline (PBS) or LPS (3 µg/100 µL, ultrapure LPS from
Subcutaneous cancer dissemination model—LPS stimulation protocol
Mice were instilled 3 times per week with PBS or LPS (3 µg/100 µL) (
Intravenous cancer dissemination model—Ac-PGP and ac-VAV stimulation protocol
Balb/c mice (n = 5/group) were anesthetized with 2.5% isoflurane/oxygen and intratracheally injected with PBS or ac-PGP (250 µg/200 µL; Eurogentec, Seraing, Belgium) or ac-VAV (250 µg/200 µL; Eurogentec), 3 times per week. After 4 ac-PGP or ac-VAV instillations, murine mammary cancer 4T1 cells or 4T1-shCXCR2 cells (1 × 105 cells/50 μL in serum-free medium) were injected into the lateral tail vein of Balb/c mice. Lung metastasis was assessed by measuring bioluminescence in vivo using the IVIS 200 Imaging System.
At the end of each experimental protocol, animals were sacrificed and a BAL was collected via intratracheal instillation of 4 mL × 1 mL PBS-EDTA 0.05 mM solution (Calbiochem, Darmstadt, Germany). Bronchoalveolar lavage fluid was collected for protein assessment, whereas cells were used for differential cell counts. Differential cell counts based on morphologic criteria were performed on cytocentrifuged preparations after staining with hematoxylin-eosin (Diff-Quick, Dade, Belgium).
Pulmonary histology and immunohistochemistry
Metastasis to lung tissues was evaluated by measuring tumor area in lungs and reporting it to the total area of lung tissues analyzed, on 8 hematoxylin-eosin–stained sections per animal using Cytomine software. 26 Proliferating and apoptotic cells were visualized on tumor sections using antibodies raised against Ki-67 and double-stranded DNA as described in supplemental experimental procedures.
Tissue processing and labeling of total cell lung suspension for flow cytometry
To identify mouse granulocyte subpopulations, digested lungs were stained with following antibodies: CD45, CCR3, CD11b, 7/4 antigen (Ly6B.2), GR1, CD11c, and CD3. More details about preparation of mouse lungs for inflammatory cells analysis by flow cytometry are described in the supplemental materials and methods section.
Tumor cell chemotaxis assay
Neutrophil supernatant (SN) preparation and tumor cell chemotaxis processing are detailed in the supplemental materials and methods section. After 48 hours of incubation in Transwell plates, migration of cells was evaluated by reporting area of cells present on the filter to the total filter area.
Lung extravasation assay
To evaluate short-term extravasation of tumor cells toward lungs of pretreated mice, Red CMTPX–stained 4T1 cells were injected intravenously. After 48 hours of cell injection, a lung perfusion was realized and extravasated tumor cells were counted by confocal microscopy. More details about this procedure are described in the supplemental materials and methods section.
Ac-PGP quantification in serum samples
To perform ac-PGP quantification, calibration curves were performed with ac-PGP standard solutions in the concentration range of 25 to 250 pg/mL. Internal standard, which consists of ac-PGP isotopically labeled with 13C on proline, was added in all samples at the same concentration. Details of LC-Chip nano high-performance liquid chromatography electrospray MS-MS to analyze samples are explained in the supplemental materials and methods section.
Statistical analysis
Reported values are expressed as mean ± SEM. Statistical analysis differences between experimental groups were assessed using InStat software (GraphPad, La Jolla, CA, USA). Data were analyzed by the Student
Results
Chronic exposure to CS induces a neutrophilic pulmonary inflammation and increases lung metastasis of B16F10 melanoma cells
To analyze the effects of CS on metastatic dissemination of tumor cells, C57/Bl6 mice were exposed during 5 weeks to CS or air and intravenously injected with melanoma B16F10 cells. In mice exposed to CS, a drastic increase in melanoma cell retention in lung tissue was observed (Figure 1A). Lung area occupied by tumor cells was 3-fold higher in CS-exposed mice as compared with air-exposed mice (n = 8/experimental condition; **
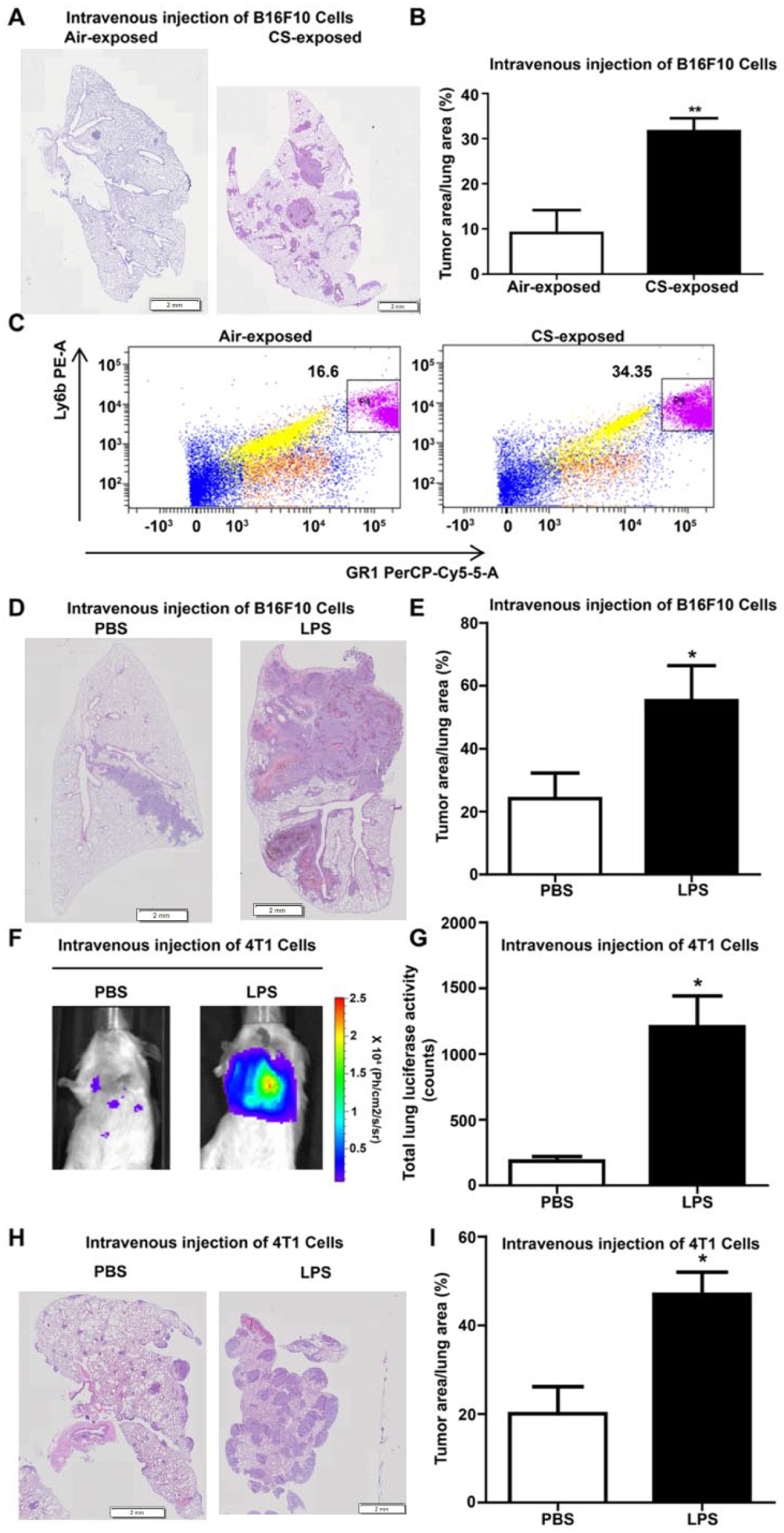
Neutrophil inflammation induces tumor cell dissemination to lung tissue. (A) Representative hematoxylin-eosin–stained sections of lung tissues (5 weeks of cigarette smoke [CS] or ambient air [air] exposure, intravenous B16F10 melanoma cell injection 2 weeks after test begins), n = 8 (hematoxylin-eosin). (C) Percentages of neutrophils in lung lysates from mice exposed to Air or CS during 2 weeks. Selected windows identify Ly6b+ GR1+ neutrophils. Results are expressed as percentage of positive cells. (D) Representative hematoxylin-eosin–stained sections of lung tissues 2 weeks after intravenous injection of B16F10 melanoma cells and pulmonary challenge with PBS or LPS, n = 8 (hematoxylin-eosin). (F) Biophotonic monitoring of lung metastasis in animals intravenously injected with luciferase-transfected 4T1 cells and treated with intratracheal injections of PBS or LPS. (G) Quantification of bioluminescence in regions of interest (ROI) determined around lungs. (H) Representative hematoxylin-eosin–stained sections of lung tissues 2 weeks after intravenous 4T1 cell injection and intratracheal stimulation with PBS or LPS, n = 8 (hematoxylin-eosin). (B, E, I) Tumor size quantification was assessed by measuring the ratio between the area of tumor foci in lungs and total lung tissue area on 8 sections per mouse in each group. Results are expressed as mean tumor area/lung area ± SEM and are representative of 2 individual experiments. **
To analyze the effects of CS on the recruitment of inflammatory cells, a BAL was performed after mice were exposed to CS during 5 weeks and showed increased neutrophil percentages in CS-exposed mice injected with B16F10 cells (2.82% ± 0.94% in control group versus 5.90% ± 1.18% in CS group) or not injected with tumor cells (1.33% ± 0.33% in control group versus 5% ± 2.21% in CS group). This CS-induced neutrophil influx was also measured by flow cytometry performed on digested lungs after 2 weeks of CS exposure. An increased proportion of GR1+ Ly6b+ population identified as neutrophils (P4) was detected in lung lysates of CS-exposed mice (34.35%) as compared with control mice (16.6%) (Figure 1C).
Neutrophils promote cancer cell migration and extravasation
To investigate whether the environment-induced neutrophilic inflammation triggers organ colonization by metastasis, C57Bl/6 mice were treated with a pulmonary, acute neutrophil-inducing stimulus (LPS). Mice were pretreated by intratracheal instillations of PBS or LPS and intravenously injected with B16F10 cells. Mice stimulated with LPS displayed increased BAL neutrophil percentages (n = 8 per group, 1.37% ± 1.05% and 1.93% ± 0.67% in control (PBS) and PBS + B16F10 group versus 70.15% ± 6.79% and 77.6% ± 1.02% in LPS and LPS + B16F10 group, respectively). Interestingly, lung tissue area occupied by B16F10 cells was significantly higher in LPS-challenged animals as compared with PBS-treated animals (n = 8; *
Neutrophils recruited in lung microenvironment promote metastasis from distant tumors
To confirm the effects of neutrophils on spontaneous tumor cell dissemination from a primary tumor, a neutrophilic inflammation was induced by LPS instillations in vivo and metastatic spread of subcutaneously injected tumor cells to lung tissues was analyzed. As expected, LPS-exposed mice showed higher BAL neutrophils as compared with control mice (PBS) (n = 8 per group, 1.92% ± 0.67% and 34.52% ± 4.01% in PBS and PBS + 4T1 group versus 60.95% ± 11.65% and 52.46% ± 2.71% in LPS and LPS + 4T1 group, respectively). Although no difference of primary tumor volume was seen between groups (data not shown), tumor cell dissemination to the lung parenchyma was significantly increased in mice exposed to LPS as compared with PBS-treated mice (n = 8; *
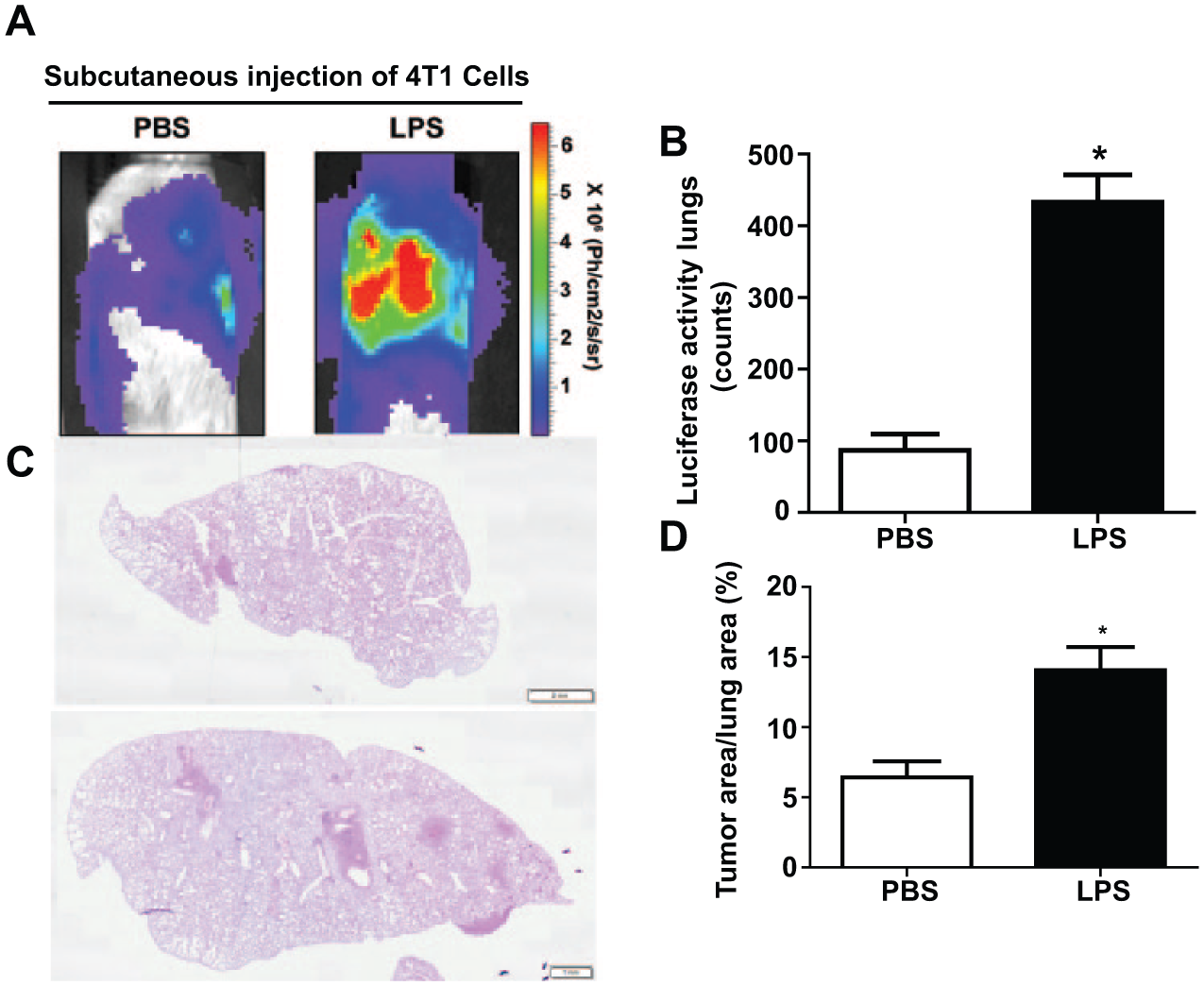
LPS-induced chronic neutrophilic inflammation correlates with lung metastasis. (A) Representative biophotonic monitoring images of lung metastasis in animals subcutaneously injected with 4T1 cells and treated with intratracheal injections of either PBS or LPS. (B) Bioluminescence quantification of tumor foci in lungs by determining regions of interest (ROI) around lungs. (C) Representative hematoxylin-eosin–stained sections of lung tissues of animals intratracheally instilled with PBS or LPS. Scale bar: 1 mm (hematoxylin-eosin). (D) Tumor size was quantified by measuring the ratio between tumor area and total lung tissue area 28 days after subcutaneous cell injection (n = 8). Results are expressed as mean ± SEM. *
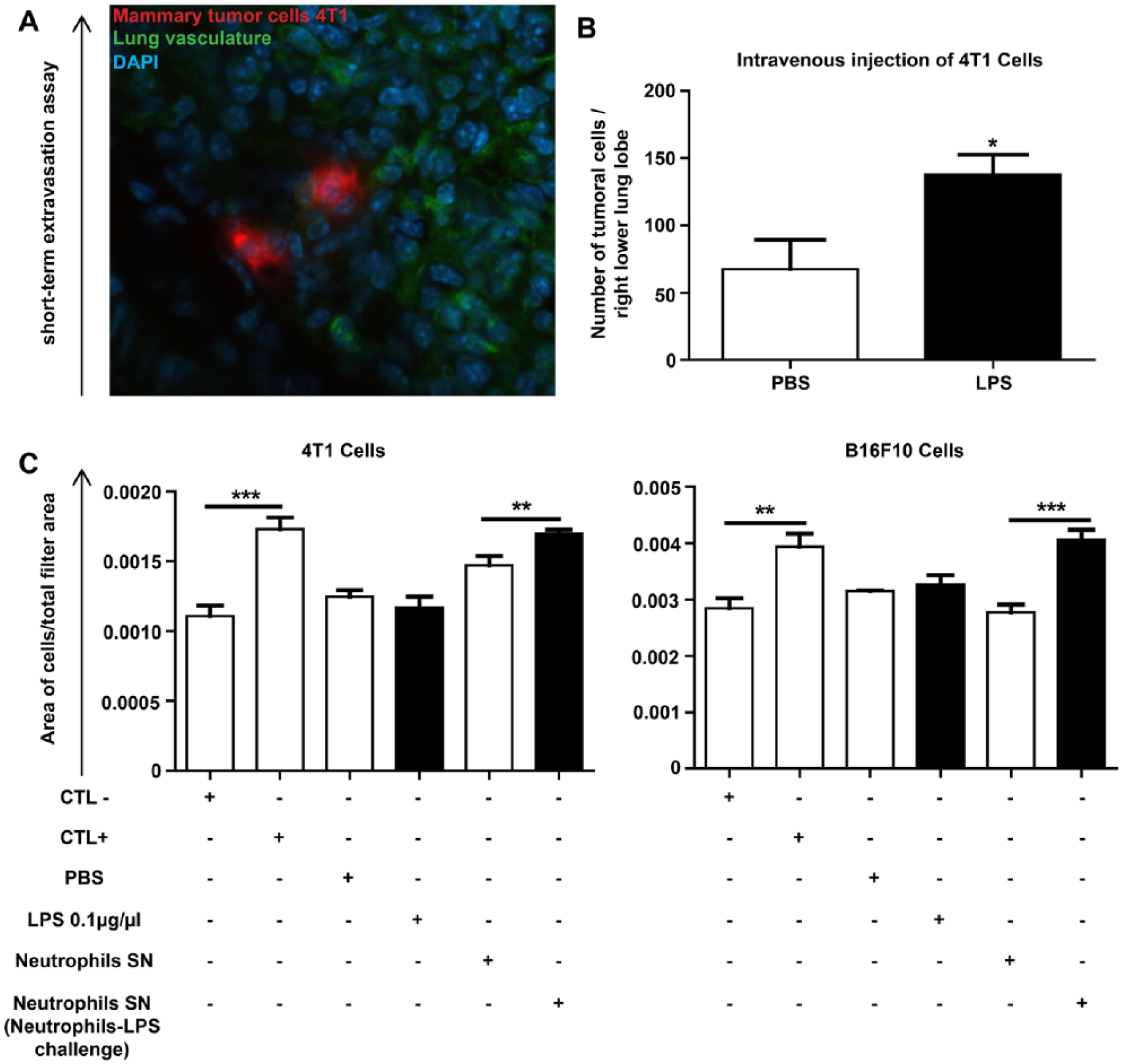
Soluble factors released on neutrophil activation favor tumor cell migration and extravasation. (A) Intravenously injected red-fluorescent 4T1 cells, which extravasated from the FITC-lectin–labeled lung blood vessels (green). Blue staining represents cell nuclei (DAPI) (scale bar: 50 µm). Images were taken 48 hours after tumor cell injection. (B) Quantification of extravasated tumor cells to lungs of mice pretreated with PBS or LPS instillations (n = 6). (C) Quantification of tumor cell chemotaxis (Boyden chamber assay). Tumor cell (4T1 or B16F10 cells) migration was estimated by calculating the ratio between the areas occupied by tumor cells on total filter area from 6 random fields (original magnification 200×). Experiments have been performed in triplicates. Results are expressed as mean ± SEM. *
Neutrophils produce increased levels of MMP-9 that generate ac-PGP peptide
Cytokine Antibody Array performed on lung protein extracts from mice bearing subcutaneous tumors identified Pro-MMP-9 as being overexpressed in LPS-treated samples (Supplementary Figure 1). MMP-9, a potential trigger for tumor cell metastasis, was reported to be active on native collagen resulting in the formation of ac-PGP. Interestingly, in this model, serum samples of mice treated with LPS and subcutaneously injected with mammary 4T1 tumor cells displayed detectable levels of ac-PGP (40.5 pg/mL), whereas serum samples derived from PBS-treated animals did not (Figure 4A). To evaluate whether ac-PGP peptides contribute to tumor cell colonization in lungs, mice were treated with intratracheal instillations of synthetic ac-PGP (250 µg/200 µL PBS) or PBS and intravenously injected with 4T1 cells. Interestingly, tumor cell colonization in lungs significantly increased when mice were instilled with ac-PGP (n = 5,*
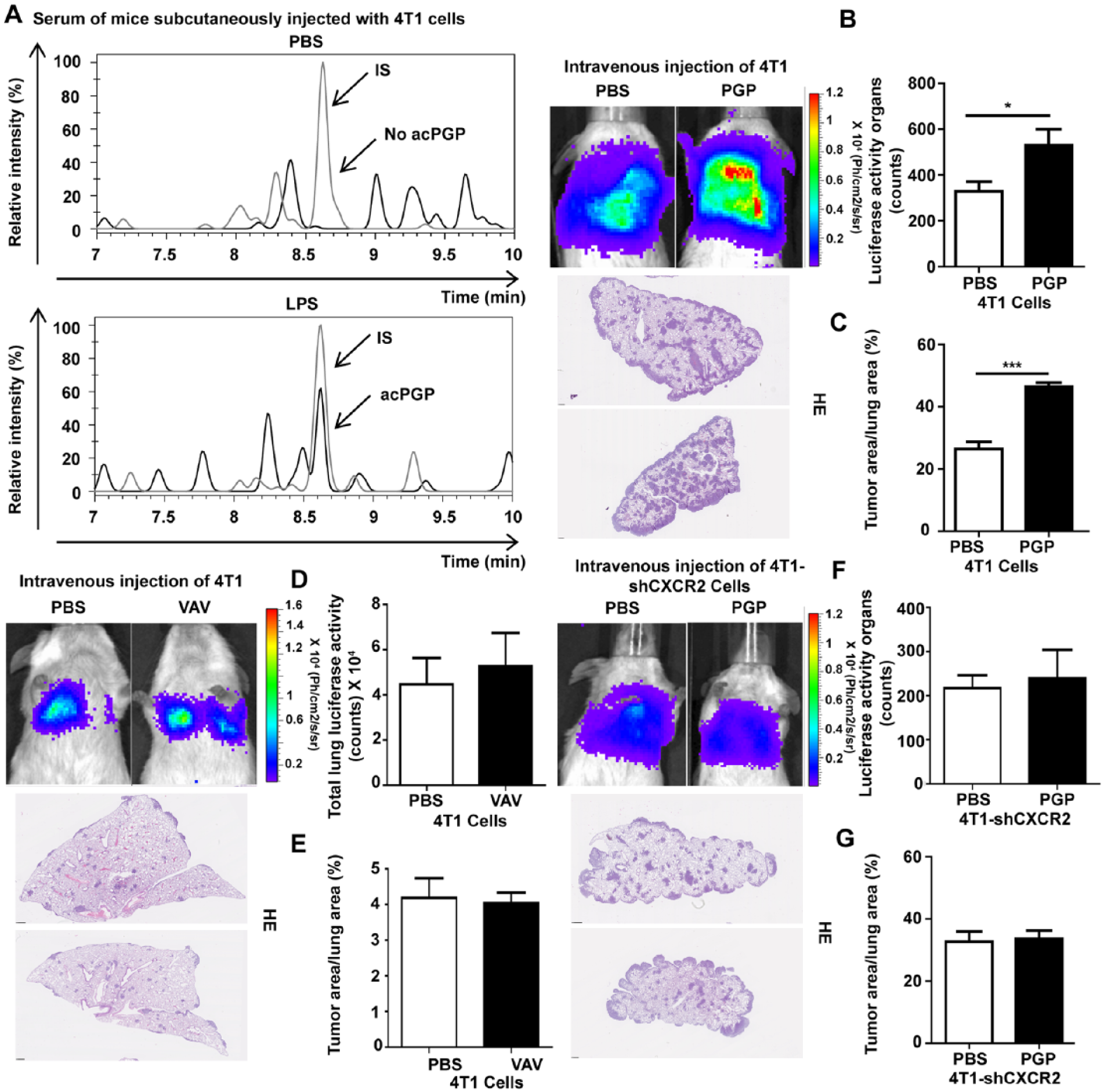
Ac-PGP originating from MMP-9–mediated collagen matrix breakdown induces the recruitment of tumor cells to lung tissues by producing ac-PGP peptides that recruit tumor cells via CXCR2 receptors. (A) Measurement of ac-PGP in serum of mice subcutaneously injected with 4T1 cells and treated with PBS or LPS using LC-MS/MS (representative run). (B, D, F) Bioluminescent monitoring of animals intravenously injected with 4T1 cells or 4T1—shCXCR2 cells and treated with intratracheal instillation of PBS, ac-PGP or ac-VAV (n = 5/group). Bioluminescence quantification of tumors by determining regions of interest (ROI) around lungs. (C, E, G) Representative hematoxylin-eosin–stained sections of lung tissues (hematoxylin-eosin). Scale bar: 1 mm. Tumor size was quantified by measuring the ratio between tumor area and total lung tissue area 14 days after intravenous cell injection. Results are expressed as mean ± SEM. *
Ac-PGP peptide promotes tumor cell recruitment via CXCR2 receptors
CXCR2, a receptor that binds ac-PGP peptides, is present on 4T1 tumor cell surface (data not shown). To evaluate the involvement of ac-PGP-CXCR2 axis in neutrophil-stimulated tumor cell colonization, 4T1 cells transfected with shRNA targeting CXCR2 (4T1-shCXCR2) were intravenously injected in mice previously instilled with either ac-PGP (250 µg/200 µL) or PBS. Animals injected with 4T1-shCXCR2 cells displayed no differences in lung tumor area after ac-PGP treatment (n = 5, Figure 4F and G) as compared with mice injected with 4T1 cells which showed an increase in tumor area/lung area ratio (Figure 4B and C).
MMP-9 depletion reduces ac-PGP production and lung metastasis
To demonstrate the importance of MMP-9 in ac-PGP release in our model, C57Bl/6 MMP-9+/+ and MMP-9−/− mice were intravenously injected with B16F10 and treated with LPS. As expected, lower levels of ac-PGP were observed in serum samples of MMP-9−/− mice treated with LPS (121 pg/mL) as compared with MMP-9+/+ mice (194 pg/mL) (Figure 5A). Histologic quantification of lung metastasis showed a significant increase in lung tumor area in LPS-treated MMP-9+/+ and MMP-9−/− mice. However, area occupied by lung metastasis in LPS-treated MMP-9−/− was significantly lower than in MMP-9+/+, suggesting an implication of MMP-9 in the neutrophil-driven metastatic dissemination of tumor cells via ac-PGP peptides (n = 6, *
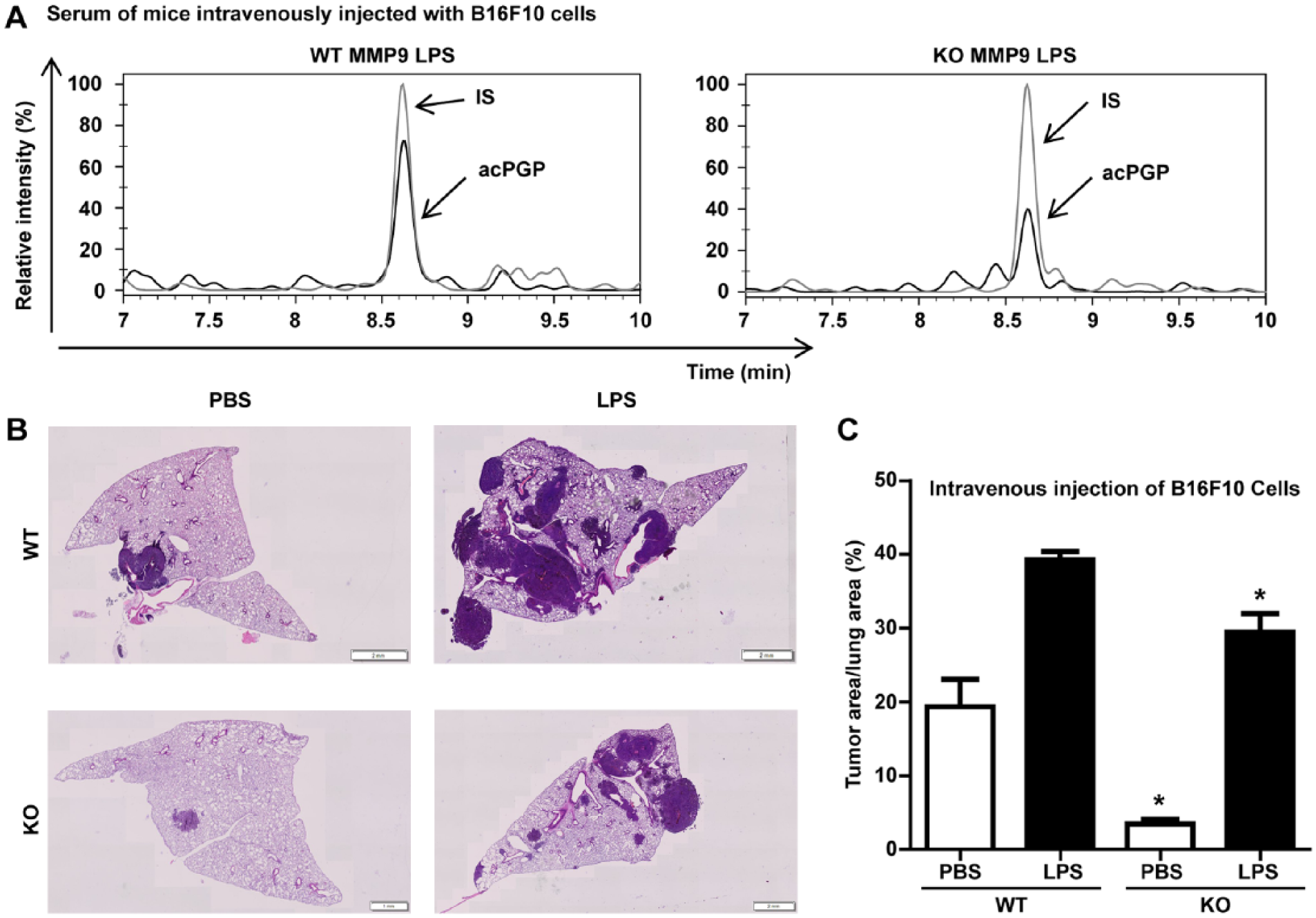
MMP-9 participates in the ac-PGP release in lungs. (A) Measurement of ac-PGP in serum of MMP-9+/+ and MMP-9−/− mice intravenously injected with B16F10 cells and treated with LPS using LC-MS/MS (representative run). (B) Representative hematoxylin-eosin–stained lung sections of MMP-9+/+ and MMP-9−/− mice 2 weeks after intravenous B16F10 melanoma injection (hematoxylin-eosin). Scale bar: 1 mm. (C) Tumor size was quantified by measuring the ratio between area occupied by tumor foci and total lung tissue area on hematoxylin-eosin–stained tissue sections (n = 6/group; hematoxylin-eosin). Results are expressed as mean ± SEM. *
Discussion
Understanding molecular mechanisms governing metastatic dissemination is a key issue for the development of cancer therapies. In this study, our main purpose was to decipher the impact on lung metastasis of extracellular matrix fragments generated in the context of inflammation. We report that neutrophil accumulation generated by CS and LPS facilitates the implantation of metastatic cells into lung tissues in an MMP-9-ac-PGP-CXCR2–dependent manner. This paradigm is supported by (1) the measurement of increased tumor cell dissemination to lungs following the induction of a pulmonary neutrophilic inflammation, (2) the measurement of increased levels of ac-PGP tripeptides in the serum of LPS-treated animals and subcutaneously injected with tumor cells, (3) the demonstration of an MMP-9–dependent collagen breakdown into chemotactic ac-PGP, (4) enhanced tumor cell colonization to lung tissues after pulmonary ac-PGP treatment, and (5) the inhibition of the effects of ac-PGP on tumor cells accumulation in lungs once CXCR2 is knocked down in tumor cells (Figure 6).
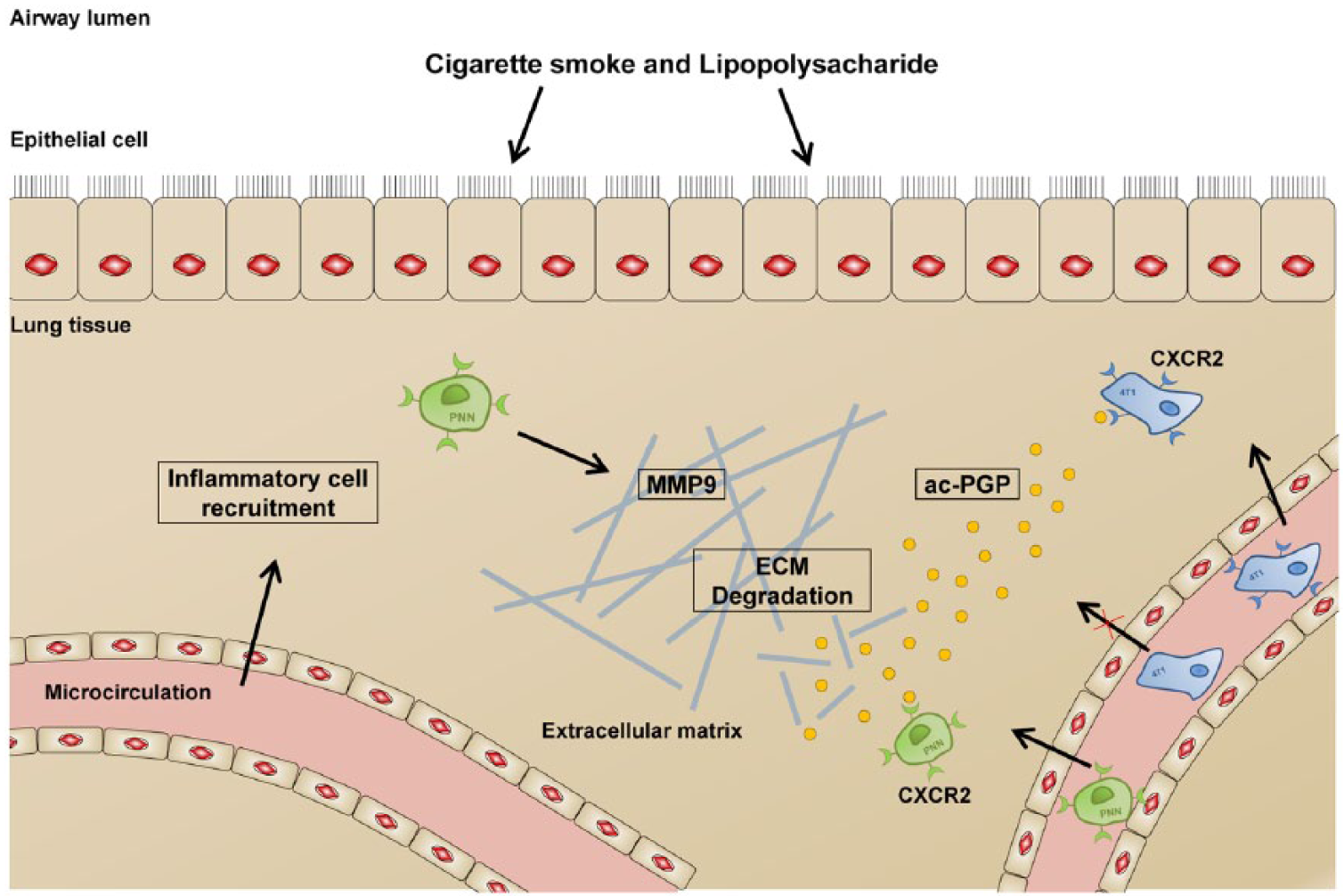
Proposed mechanisms for tumor cell progression in the context of neutrophilic inflammation in the lung. Ac-PGP indicates
Epidemiologic studies and clinical observations have established links between inflammation, carcinogenesis, and metastasis processes. However, mechanisms linking chronic inflammation to increased tumor development and metastasis remain enigmatic. Recent studies support that 25% of all human cancers result from chronic inflammation.27–31 Chronic inflammation includes microbial infections,
32
autoimmune diseases,
33
or pollution-driven inflammation. Tobacco smoke appears as a very common pollutant composed of no less than 4700 compounds, comprising many oxidants and influencing cell proliferation, angiogenesis, tumor growth, and metastasis.
34
Nicotine is one major bioactive component of CS displaying a carcinogenic potential by directly inducing cell proliferation and angiogenesis and by protecting cancer cells from apoptosis.
35
We therefore evaluated direct effects of CS on cell proliferation in vivo. We report that CS per se does not enhance tumor cell proliferation in our experimental setting. Although previous authors suggested that tobacco smoke compounds could positively interfere with tumor progression,
36
others have shown a cytotoxic effect of CS due to, eg,
Many therapeutic perspectives could derive from these data as ac-PGP or CXCR2 blockade might represent attractive therapeutic targets to decrease lung metastasis in patients with established primary tumors or in a preventive manner in high-risk patients. Some therapeutic options already exist for patients with COPD to decrease ac-PGP levels using azithromycin, 43 and valproic acid is able to reduce the production of ac-PGP by inhibiting the downstream cleavage of collagen-derived peptides by prolyl endopeptidase. 44 Another promising therapeutic perspective would be to enhance the ac-PGP clearance by a restoration of the leukotriene A4 aminopeptidase activity through a dedicated pharmacologic agent. 45 Other studies will be needed to confirm our findings and establish the validity of such a therapeutic strategy in patients with cancer.
In conclusion, our data demonstrate that pulmonary accumulation of neutrophils increases lung metastasis of tumor cells in an MMP-9–dependent manner by the production of collagen-derived ac-PGP chemotactic peptides that engage CXCR2 present at the surface of cancer cells.
Footnotes
Acknowledgements
The authors thank Gaël Cobraiville, Christine Fink, Pascale Heneaux, Elodie Kip, Quentin Leclercq, Fabienne Perin, and Céline Vanwinge for technical support. They also acknowledge the GIGA-Imaging and Flow Cytometry platforms.
Funding:
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was financially supported by grants from the Fonds National pour le Recherche Scientifique (FRS-FNRS Télévie, grant n°7463012F), the Centre AntiCancéreux (University of Liège), the Foundation against Cancer (foundation of public interest, Belgium), Interuniversity Attraction Poles Program-Belgian State-Belgian Science Policy-Project P7/30, and the Fonds Léon Fredericq (University of Liège).
Declaration of conflicting interests:
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: D.C. is the founder of Aquilon Pharmaceuticals, received speaker fees from AstraZeneca, Boehringer-Ingelheim, Novartis, Mundipharma, Chiesi, and GSK and received consultancy fees from AstraZeneca, Boehringer-Ingelheim, and Novartis for the participation to advisory boards. None of these activities have any connection with oncology or development of drugs in the field of oncology.
Author Contributions
SB and NR contributed to conception and design of the work, data collection, analysis interpretation, preparation of figures, and manuscript preparation. MF contributed to data collection, performed the mass spectrometry measurements, and interpreted the results. BD contributed to data collection and interpretation of results; MP helped with FACS studies, contributed to data collection, and interpretation of results; NR contributed to image analysis and supported the project by affording his expertise in quantifications of tumor sizes in histology; AN did take part to project supervision, contributed to manuscript preparation, and critically revised the manuscript. DC conceived the research program, applied to grants for funding, supervised the project, supervised and validated the results, supervised manuscript preparation, approved the final version to be published, and submitted the manuscript to the editor.
Availability of data and material
Data sharing is not applicable to this article as no data sets were generated or analyzed during this study.
Ethical Approval and Consent to Participate
Ethical Committee of the University of Liège approved the protocols used for animal studies under the references #736, #918, #1667. There were no clinical samples in this study.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
