Abstract
Chryseobacterium indologenes is gram-negative bacteria that cause infection in humans. It is less frequently isolated in the laboratory. The development of drug-resistant and its intrinsic ability to resist a wide range of antimicrobials enables them to cause mortality in an immunocompromised patient with a longer hospital stay. Our study objectives are to investigate antimicrobial-resistant patterns, drug-resistant enzymes, and the risk factor analysis associated with multidrug-resistant (MDR), extensively drug-resistant (XDR), and Pan-drug resistant (PDR) within 2 years. Altogether 53 strains of Chryseobacterium indologens were obtained from 5000 specimens that were processed for routine bacterial culture. The bacterial identification was done using conventional techniques (colony morphology, gram staining, flexirubin test, and biochemical tests) as well as the VITEK-2 System to further confirm. The bacterial isolate were processed to observe antimicrobial susceptibility test (AST) using disk diffusion method. MDR XDR and PDR were classified following European Centre for Disease Prevention and Control guidelines. C. indologens strains with beta-lactamases such as extended-spectrum beta-lactamases (ESBL), metallo beta-lactamases (MBL), and Amp-C beta-lactamases (Amp-C) were detected phenotypically. The highest isolation of C. indologens was observed in a sputum sample. In vitro antimicrobial susceptibility test revealed susceptibility to tigecycline followed by levofloxacin, cotrimoxazole, and piperacillin-tazobactam. From 53 isolates of C. indologens, MDR accounts for 56.60% and 22.64% for XDR. Combined antimicrobial therapy and longer hospital stay were found to be the leading risk factor. All 53 C. indologenes strains were detected as MBL. Total ESBL was detected in 16.98% of MBL producer strains and Amp-C was observed in 13.20% of MBL-producing strains. All 3 enzyme co-oproducers were seen in only 5.66% of C. indologens. Although it is rarely encountered in the laboratory, it showed a remarkable effect in patients with underlying predisposing factors and prolonged hospital stays. The presence of betalactamases determined the drug-resistant activity on a wide spectrum of tested antibiotics.
Keywords
Introduction
Chryseobacterium species are nonmotile, aerobic, nonfermenters gram-negative bacteria that produces oxidase enzyme.1,2 Pigment production ranges from distinctive yellow to orange when bacteria is cultured on blood agar and nutrient agar.3,4 Chryseobacterium indologenes were previously known as Flavobacterium indologenes. The most prevalent bacterial species in Chryseobacterium obtained from clinical specimen include Chryseobacterium indologenes, Chryseobacterium meningosepticum, and Chryseobacterium gleum. The first C. indologenes-associated bacteremia was reported in Taiwan. 5 Chryseobacterium naturally inhabits soil, water, and other food products.4,6 Other literature suggests that C. indologenes are present in an aqueous system and on moist surfaces, as a potential source of infection as it can resist disinfectants such as chlorine when treated in a water supply tank. 7
The antimicrobial resistance exhibiting microorganisms are of 3 types: multidrug-resistant (MDR), extensively drug-resistant (XDR), and pan drug-resistant (PDR). If the tested antibiotics are resistant to 1 agents of atleast 3 antibiotics then the bacterium is considered as MDR whereas if the bacterium is resistant to 1 agents of all tested antimicrobials then it is known as XDR. The bacterium that are resistant to all the tested antibiotics are known to be PDR. 8 Microorganisms resistant to novel antimicrobial classes applied in clinical practices are great challenges to treat. 9 Extended-spectrum beta-lactamases (ESBLs) breaks down beta-lactam ring of beta-lactamase antibiotics that inhibit cell wall synthesis for instance penicillins, first to third generation cephalosporins (besides cephamycins), carbapenems, and aztreonam. 9 Apart from ESBLs, AmpC beta-lactamases production is the other resistant mechanism that confers resistance to broad-spectrum cephalosporins. AmpC beta-lactamases are chromosomally encoded cephalosporinase present in gram-negative bacteria, including Chryseobacterium indologens, which mediate resistance to cephalosporin, penicillin, and other groups of beta-lactamase inhibitor antibiotics. 10
Metallo-beta-lactamase (MBL) enzymes present in multidrug-resistant bacteria are widely known for their potential to hydrolyze the beta-lactamase ring of beta-lactam antibiotics (aminopenicillins, cephalosporins) including carbapenems that makes bacterium a threat in treatment compared to ESBL and Amp-C where carbapenem drugs remain susceptible. Additionally, MBL can be detected in disk diffusion tests by the application of ethylenediaminetetraacetic acid (EDTA). 11 Numerous reports have suggested that hospital-acquired infections caused by C. indologens are mainly medical device associated with respective to longer healthcare stay.3,7 Furthermore, biofilm production and protease enzyme activity in Chryseobacterium indologens play a significant role in leading to invasive infections. 3 There are various knowledge gap that our current study fills; the emerging resistant and lack of sufficient antimicrobial pattern, risks factors, and clinical correlation scarcity as well as less knowledge on intrinsic resistant to this bacterium creates havoc amongst clinicians to treat infections caused by this bacterium. Therefore our study was conducted to observe antimicrobial resistance patterns, clinical correlations, and risk factor analysis as well as to observe emerging beta-lactamases enzyme on Chryseobacterium indologens that is still responsible for evolving from MDR to XDR- Chryseobacterium. The methodology and experiments employed in our study for identification of this rare bacterium along various betalactamases (ESBL, MBL, and Amp-C) that causes rise in resistant rates can be performed in resource limited settings. Our work aids in solving misdiagnosis, underdiagnosis issues, discussion on empirical therapy on encounter of Chryseobacterium indologens which was a serious threat as CLSI Guidelines lacks to explain regarding this bacterium. In addition, the current research work on Chryseobacterium indologens was conducted for the first time in Nepal.
Materials and Methods
Sample size
Out of 5000 processed clinical specimens, 53 strains of Chryseobacterium indologens were obtained during the 2 years study period (July 2019 to July 2021).
Design of study
Prospective cross-sectional study was performed in a duration of July 2019 to 2021 at Grande International Hospital, Nepal. All age group of patient’s clinical specimens (urine, wound swab, pus, body fluids, CSF, blood, and endotracheal tube) were involved and the specimens were processed for routine culture and sensitivity testing. Chryseobacterium indologens obtained from different clinical specimens were stored in a nutrient agar vial. The Single growth of Chryseobacterium indologens in all the clinical specimen besides urine were considered significant whereas, in the urine specimen, the bacterial colony count >105 colony-forming unit/ml were accepted in our study. Clinical samples of patients lacking demographic details and clinical history as well as those clinical samples that yielded bacterium other than Chryseobacterium species were excluded from the study.
Identification of pathogens by conventional and Vitek system
Isolates were identified based on Bergey’s microbiology book guidelines. That includes the study of colony morphology, colony pigment, hanging drop motility test, and other conventional biochemical tests including catalase, oxidase, triple sugar iron agar test, sulfite indole motility test, citrate utilization test, urea hydrolysis test as well as flexirubin test. 12 The pigment production test, that is, flexirubin test was done by the addition of 10% potassium hydroxide as shown below in Figure 1.
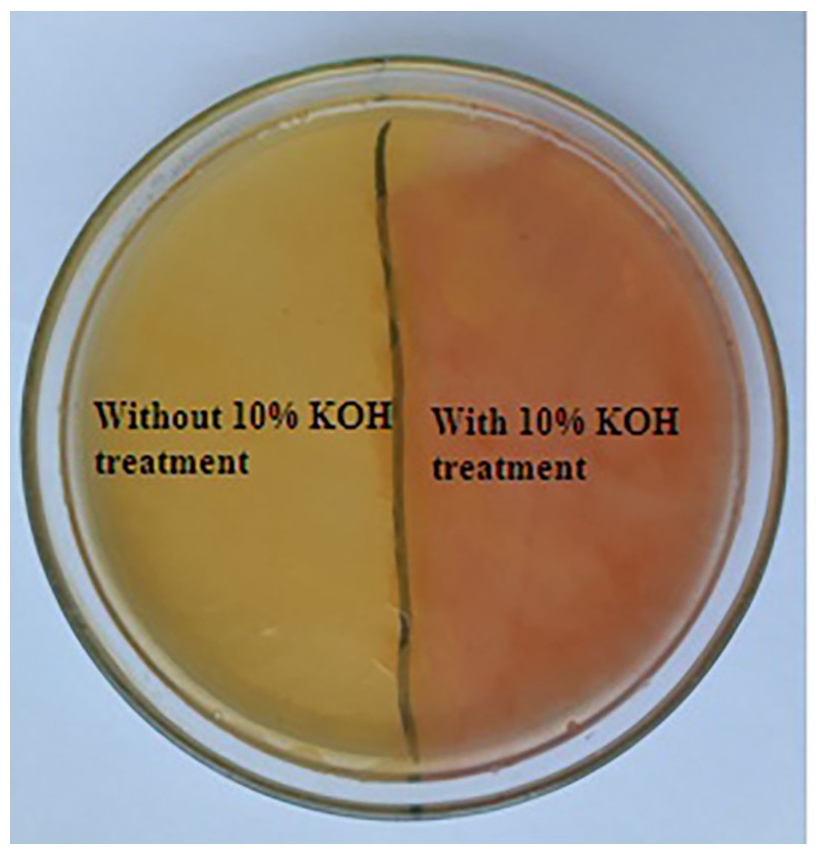
Flexirubin production test shown by Chryseobacterium indologens using 10% KOH (right) and without KOH treatment original bacterial pigment (left).
Identification of pathogens by Vitek-2 system
The characteristics of Chryseobacterium indologens were confirmed using the VITEK-2 system with GNB identification cards following the instructions recommended by manufacturer. The VITEK-2 system comprises 64-wells that conduct 40 biochemical tests. Eighteen biochemical tests includes sugar tests, that is, fermentation and assimilation tests, 3 amino acid decarboxylation tests, and the 2 remaining miscellaneous tests (for urea hydrolysis and malonate utilization test). Inoculation was prepared by matching turbidity with 0.5 M Mcfarland carried out in a card prepared from isolates that were incubated in blood agar for 24 hours and inserted inside the VITEK 2 module (reader and inoculator module). The fluorescence emission was measured at interval of 15 minutes, and the identification process were completed after 3 hours. 13
Antimicrobial susceptibility testing
Antimicrobial Susceptibility Testing of the Chryseobacterium indologens was carried out by using Kirby-Bauer disk diffusion assay on Mueller-Hinton agar (MHA) medium that was tested against 9 antibiotic disks following the SENTRY antimicrobial surveillance program during 1997 to 2001 14 as there were no specific guidelines for Chryseobacterium indologens in Clinical and Laboratory Standard Institute (CLSI). The following antibiotics applied are as follows: Cefepime (30 µg), amoxicillin/clavulanate (20/10 µg), ceftazidime (30 µg), imipenem/meropenem (10 µg), tigecycline (15 µg), levofloxacin (5 µg), azithromycin (15 µg), piperacillin/tazobactam (100/10 µg), and cotrimoxazole (23.75/1.25 µg). The turbidity of bacterial suspension were compared with 0.5 Mcfarland and subjected to lawn culture in MHA plates. The following illustrated antibiotic disks were applied in each plate and the plates were incubated at 37°C for 24 hours. For the quality control of antibiotics disk ATCC 25922 strain of Escherichia coli was tested when subjected to AST.
Definition and classification of multidrug-resistant (MDR), extensively drug-resistant (XDR), and Pan drug-resistant (PDR).
MDR is defined as non-susceptibility to any 1 agent in 3 or more antimicrobial classes. XDR is defined as non-susceptibility to 1 agent in all antimicrobial categories but only 1 or 2 antimicrobial class remains susceptible. Microorganisms nonsusceptible to all agents in every antimicrobial category are referred to as PDR.
MDR, XDR, and PDR Chryseobacterium indologens are determined in our study following European Centre for Disease Prevention and Control (ECDC) guidelines. 8
Screening for extended-spectrum beta-lactamase (ESBL), AmpC beta-lactamase, and metallo beta-lactamase
Chryseobacterium indologens isolates were screened for Ceftazidime (30 μg) for ESBL, Cefoxitin (30 μl) for AmpC, and Imipenem (10 μg) for Metallo beta-Lactamase. Chryseobacterium isolates that were resistant to the above-mentioned antibiotics were suspected of beta-Lactamase enzyme producers. Hence further phenotypical confirmation was done.
Phenotypic confirmation of ESBL- and AmpC beta-lactamase and metallo beta-lactamase
Combined disk method for ESBL
In the beginning step, the bacterium turbidity was compared with 0.5 Mc Farland solution and bacterial suspensions were swabbed in MHA plates and ceftazidime (30 µg) and ceftazidime-clavulanic acid (30/10 µg) disk were applied in Mueller Hinton agar (MHA). The zone of inhibition exceeding >5 mm in ceftazidime-clavulanic acid (30/10 µg) compared to ceftazidime (30 µg) disk determined the formation of the extended-spectrum beta-lactamase enzyme. 9
Inhibitor-based method for AmpC beta-lactamase
The test isolate’s turbidity was matched with 0.5 Mc Farland suspensions and uniformly inoculated on a MHA plates. Two disk cefoxitin (30 µg) and Cefoxitin + phenylboronic acid (400 µg) were placed 30 mm apart on the agar plate’s surface. Following 24 hours of aerobic incubation at 37°C, if a zone of inhibition appears exceeding 5 mm or more in the disk of cefoxitin impregnated with phenylboronic acid than the cefoxitin disk alone then the tests isolates were denoted as an AmpC beta-lactamase producer. 10
Combined disk diffusion method for metallo beta-lactamase
Metallo beta-lactamases (MBL) were identified using imipenem/meropenem in combination with EDTA. The test isolates’ lawn cultures (0.5 Mc Farland suspensions) were prepared. After allowing MHA plates to dry, the imipenem/meropenem disks, 1 impregnated with 0.5 M EDTA and the other imipenem disk alone, were placed on the surface of MHA agar plates approximately 30 mm apart. Following the 24 hours of incubation at 37°C, a 7 mm increase in zone diameter around the imipenem + EDTA disk compared to the imipenem disk alone indicated MBL production. 15
Phenotypical methods for the identification of ESBL, Amp-C, and MBL with its co-producers, that is (MBL + ESBL) or (MBL + ESBL + AmpC) were shown in Figure 2 under 2a to 2d.
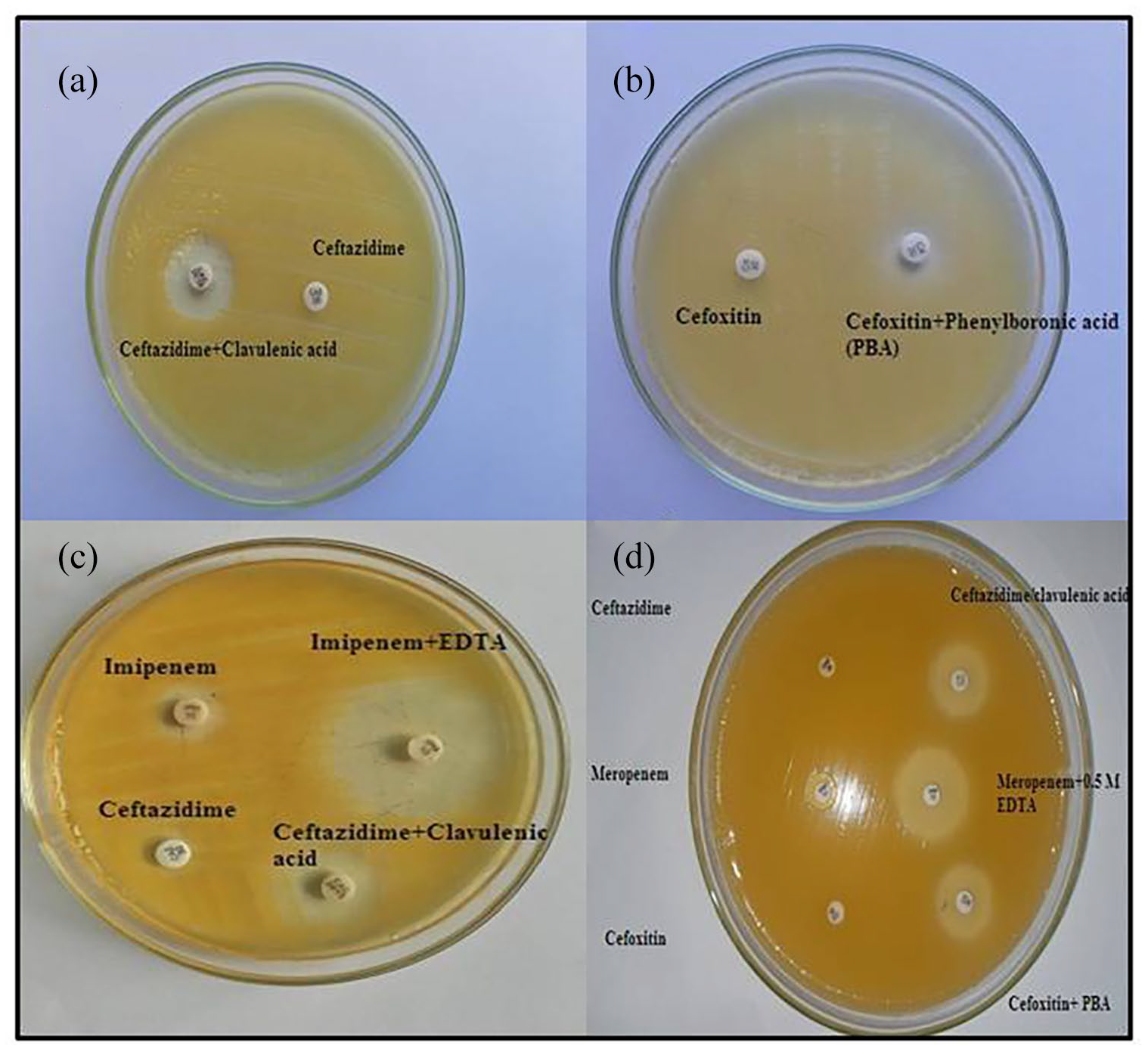
Phenotypical detection of various beta-lactamases (a: ESBL, b: Amp-C, c: MBL + ESBL coproducer, d: ESBL + MBL + Amp-C co-producers) in Chryseobacterium indologens.
Clinical outcome
The primary outcomes were the survival and mortality of patients. The obtained data on patients’ mortality was obtained from the medical records. The secondary outcome was the longer hospital stay of patients admitted to the ICU from the culture date.
Statistical analysis
The Statistical Package for Social Sciences (SPSS 16.0) was used to analyze the data. The outcome data was analyzed using simple descriptive statistics. The Chi-square and Fisher exact tests were used to compare differences in demographics, clinical conditions, and predisposing factors. The MDR and XDR strains of Chryseobacterium indologens were correlated with various risk factors of patients. For each variable, the data was subjected to multivariate analysis and logistic regression analysis. The level of significance differences between the tests was observed on the basis of P value (P < .05) at 95% confidence interval.
Results
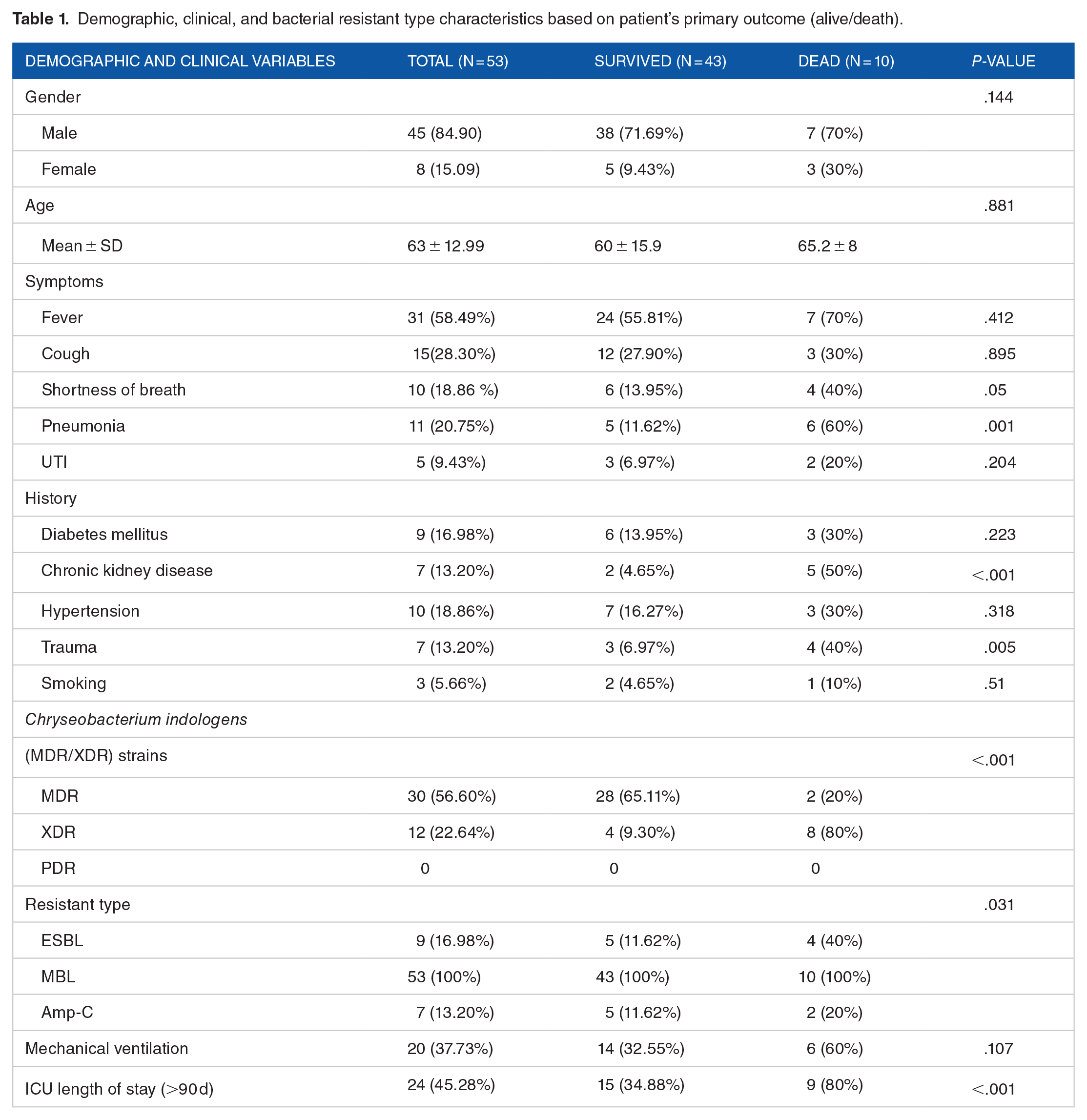
Altogether 5000 clinical specimens were processed, 53 strains of Chryseobacterium indologens were identified during the 2 years of period, 30 (56.60%) were MDR and 12 (22.64%) were XDR in culture. PDR-Chryseobacterium indologenes were absent. Mortality of individuals following longer healthcare stay was reported in 18.86% (10/53) of the patients. The majority of individuals infected by Chryseobacterium indologens was male 45 (84.90%) and the mean age group was expressed in form of mean ± standard deviation was 63 ± 12.9 years. Majority (>50%) of the patients had pre-existing co-morbidities. Altogether 9 (16.98%) were ESBL producers from the clinical isolates in our study and 7 (13.20%) were Amp-C beta-lactamases. Twenty patients were ventilated (37.73%) before enrollment in the study. When classification of results was done based on survival status, there was absence of significant differences in the demographic and clinical features of patients except for pneumonia, longer health care stay, chronic kidney disease, MDR/XDR strains of Chryseobacterium indologens, and drug-resistant type enzymes (MBL, ESBL, and Amp-C). The majority of surviving patients were male. These results were classified in Table 1:
Demographic, clinical, and bacterial resistant type characteristics based on patient’s primary outcome (alive/death).
A total of 53 Chryseobacterium indologenes strains were processed for series of conventional biochemical tests and confirmed by the Vitek-2 system. All the grown isolates were further processed for AST to observe susceptibility and resistant pattern on various tested antibiotics. From various sources of the clinical specimen, a maximum number of Chryseobacterim indologens isolates was observed in the sputum (47.1% ) followed by blood (22.64%), endotracheal aspirate (16.9% ), body fluid (7.5%), urine (3.7%), and pus (1.8%) based on in-patient department (IPD) and out-patient department (OPD) as shown inside table of Supplemental File 1.
The AST pattern of Chryseobacterium indologens in our study determined the most susceptible antibiotics were found to be tigecycline followed by co-trimoxazole, levofloxacin, and piperacillin-tazobactam. Ceftazidime and Imipenem were found to be the most resistant antimicrobials against isolated strains of Chryseobacterium indologens. The results were summarized in a bar diagram given below in Figure 3.
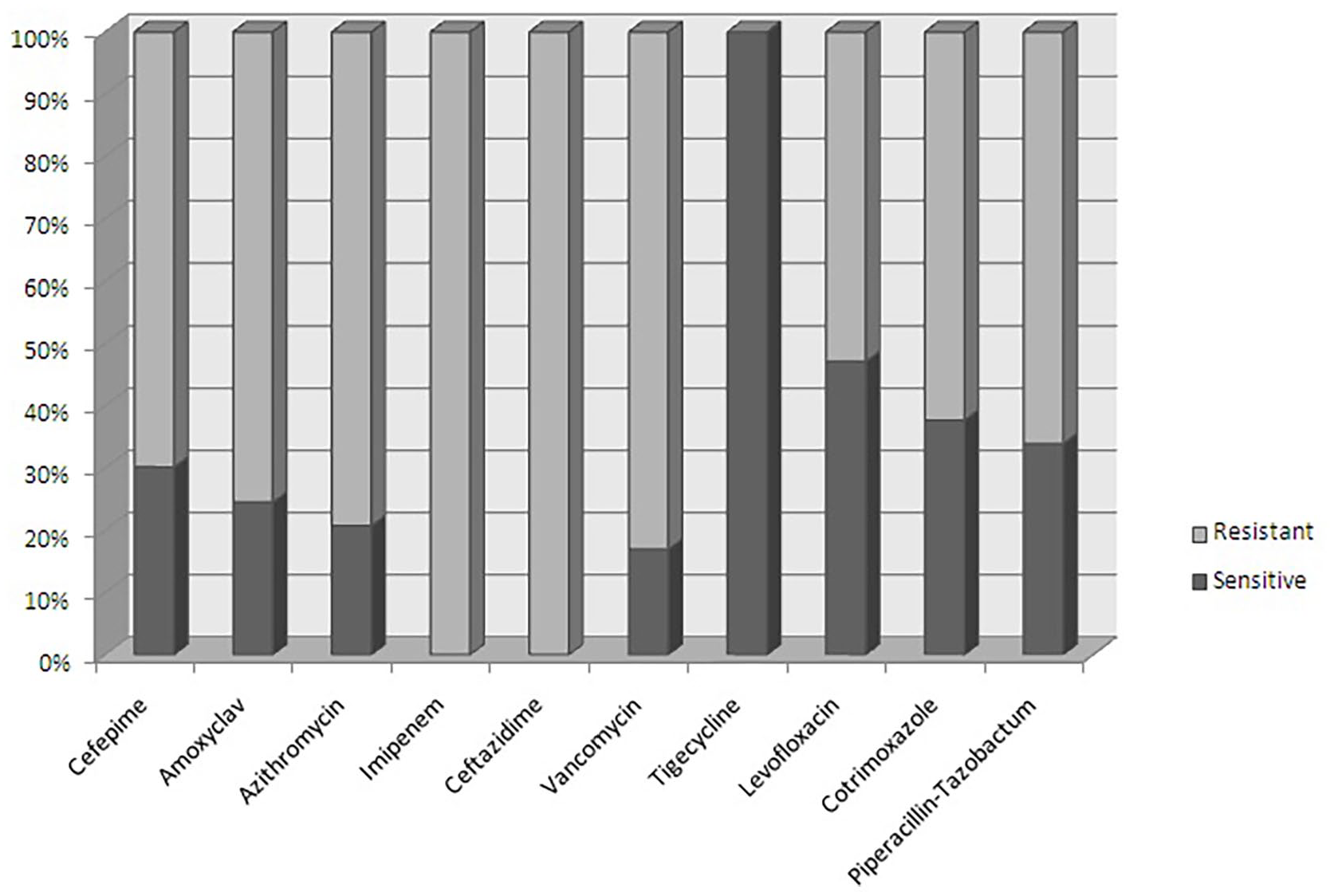
Antimicrobial suceptibility test of Chryseobacterium indologens.
The average duration of healthcare stay of the patient in our study was 60 days. Eighteen multidrug-resistant and 10 extensively drug-resistant strains of Chryseobacterium indologens were observed among those patients subjected to a long hospital stay. About a 10% mortality rate was observed due to MDR and XDR strains. Both the outcome concerning MDR and XDR strains were statistically significant (P < .05) as shown below in Table 2.
Comparison of clinical outcomes with MDR and XDR strains of Chryseobacterium indologens.

The risk factors were analyzed in 53 cases from the significant growth of Chryseobacterium indologens. All variables were incorporated into the logistic regression analysis for both MDR and XDR strains. After applying binary logistic regression analysis, Three factors remained statistically significant for MDR Chryseobacterium indologens and 2 factors remained statistically significant for XDR Chryseobacterium indologens as displayed in Table 3.
Multivariate analysis of risk factors for developing MDR and XDR strains of Chryseobacterium indologens.
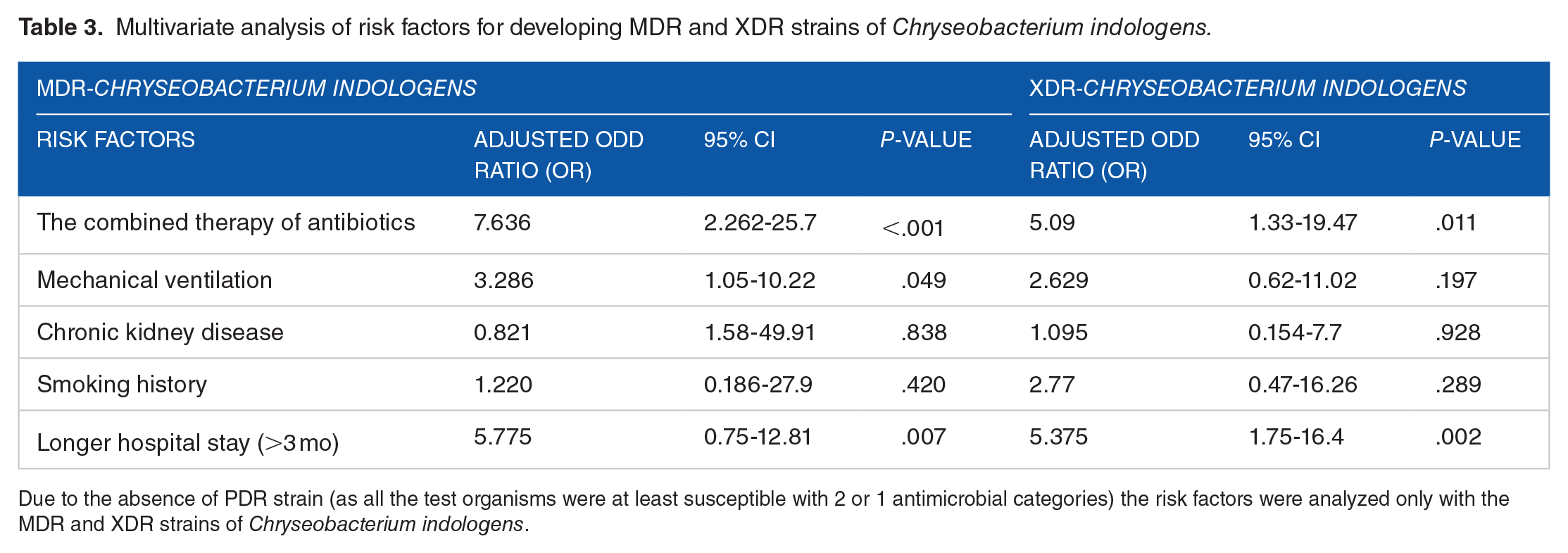
Due to the absence of PDR strain (as all the test organisms were at least susceptible with 2 or 1 antimicrobial categories) the risk factors were analyzed only with the MDR and XDR strains of Chryseobacterium indologens.
Throughout the various risk factors, combined therapy of antibiotics were leading factor determining the risk for colonization of multidrug-resistant Chryseobacterium indologens (OR = 7.63, 95% CI 2.262-25.7) followed by prolonged hospital stay (OR = 5.775, 95% CI 0.75-12.81). For XDR, a longer hospital stay (OR = 5.375, 95% CI 1.75-16.4) was found to be the leading risk factor for Chryseobacterium indologens followed by combined antibiotics therapy (OR = 5.09, 95% CI 1.33-19.47).
The sample-wise prevalence of MDR, XDR strains of Chryseobacterium indologens producing beta-lactamases was observed in the sputum sample. The lowest sample-wise prevalence of MDR and XDR was found in pus. The beta-lactamases and their co-producers such as MBL, MBL + ESBL, AmpC + MBL, and AmpC + MBL + ESBL were observed maximum in the sputum sample. The sputum sample accounts for the isolation of 18 (58%) MDR and 6 (50%) XDR strains of Chryseobacterium indologens followed by endotracheal aspirate (MDR = 9.6%, XDR = 50%). Blood, body fluids, and pus samples revealed low isolation of MDR and XDR strains with drug-resistant beta-lactamases as compared to respiratory samples, that is, sputum and endotracheal aspirate. The equal highest frequency of XDR was observed in both sputum and endotracheal aspirate (50%). Our results estimated all the bacterial isolates n = 53 were MBL producers, n = 9 (16.98%) were both ESBL and MBL producers, and n = 7 (5.66%) were found to be both MBL and AmpC producers. Three (5.66%) isolates were co-producer for all MBL, ESBL, and AmpC beta-lactamases. All 3 co-producers of drug-resistant enzymes were detected in the sputum sample (66.6%) followed by endotracheal aspirate (33.3%). Moreover, the increase in the frequency of ESBL and Amp-C beta-lactamase was observed in respiratory samples which were summarized below in Table 4.
Sample-wise distribution of MDR, XDR, and beta-lactamase producer Chryseobacterium indologens..
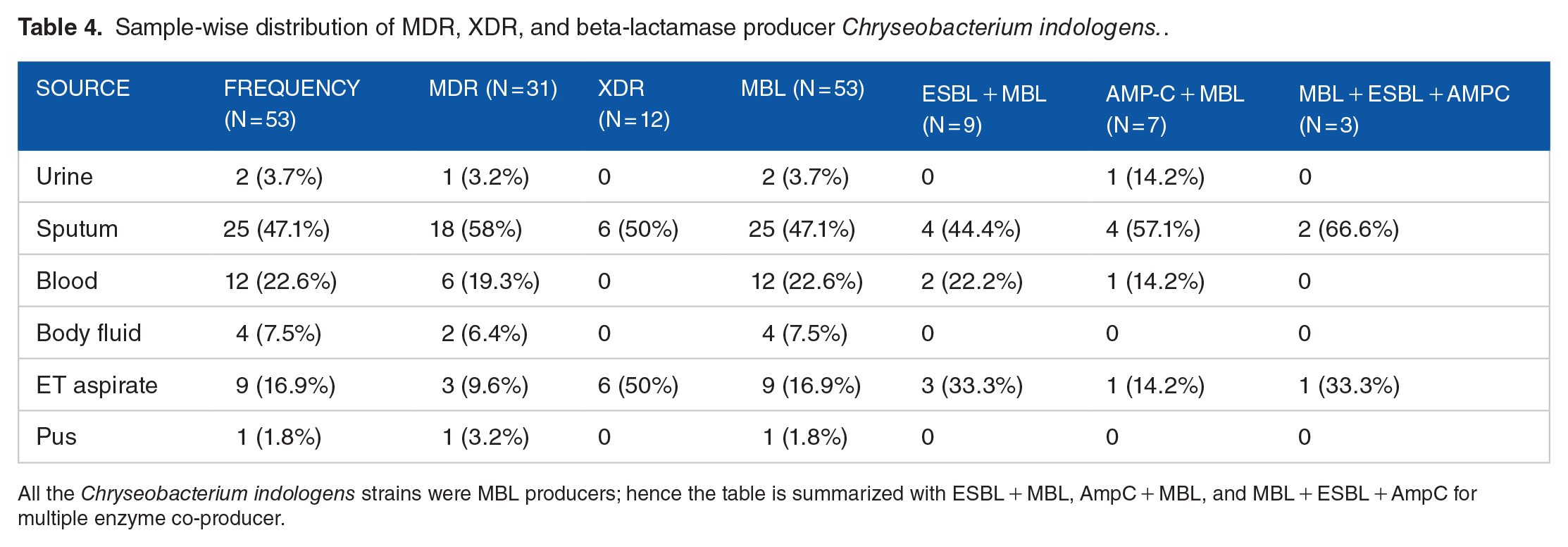
All the Chryseobacterium indologens strains were MBL producers; hence the table is summarized with ESBL + MBL, AmpC + MBL, and MBL + ESBL + AmpC for multiple enzyme co-producer.
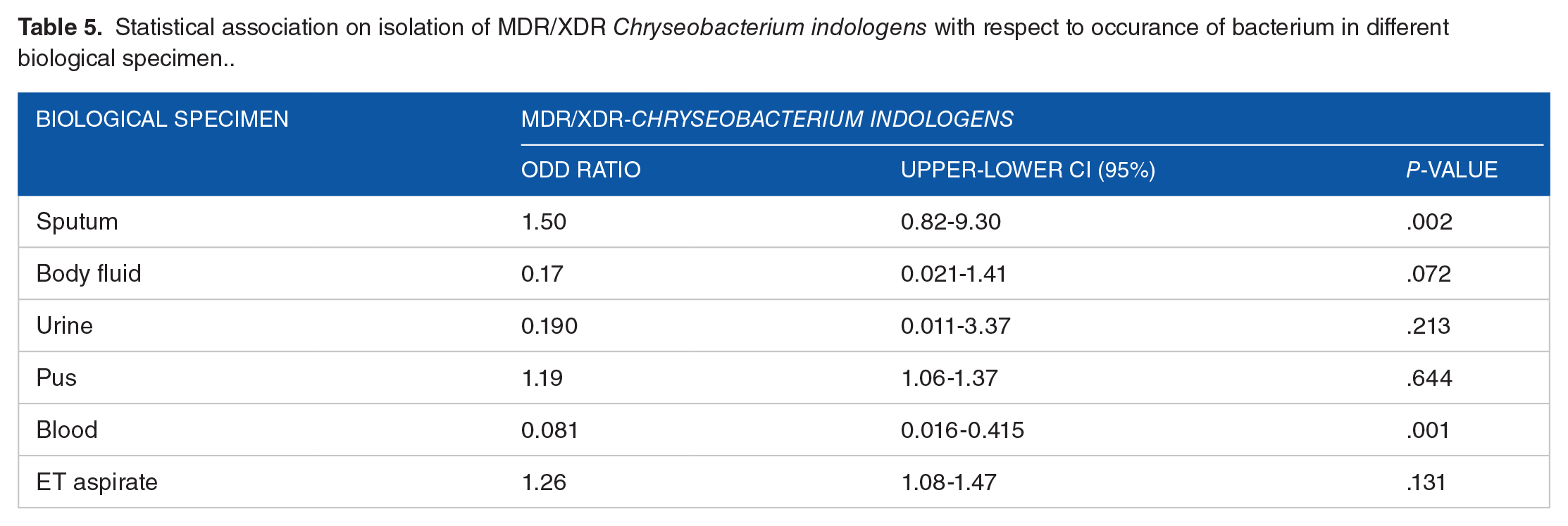
Various clinical specimens of infected patients resembles the biological source for the isolation of Chryseobacterium indologens. From the results depicted in Table 5, the positive cases in various biological specimen were statistically analyzed to explain the occurrence of MDR and XDR- Chryseobacterium indologens between different clinical sources. Our results showed the maximum yield of drug resistant Chryseobacterium indologens, particularly on respiratory specimen (sputum and endotracheal aspirate). Sputum specimen represents highest odds [OR = 1.5, 95% (0.82-9.30)], that is, 1.5 times greater compared to other biological specimens for the recovering the occurrence of MDR and XDR-Chryseobacterium indologens which was found to be statistically significant (P < .05) followed by endotracheal aspirate [OR = 1.26, 95% (1.08-1.4)]. Despite total Chryseobacterium indologens recovered from the blood samples had lower odds (OR = 0.081) for drug resistant potential compared to other biological specimens when analyzed with it is total number, the blood samples showed a useful source for isolation of Chryseobacterium indologens (P < .01). Rest of the clinical specimens (body fluid, urine, and pus) were found to be statistically insignificant (P > .05).
Statistical association on isolation of MDR/XDR Chryseobacterium indologens with respect to occurance of bacterium in different biological specimen..
Discussion
The Chryseobacterium genus falls under family of Flavo-bacteriaceae and is naturally found in soil and water.1,2 The presence of this bacteria in medical devices containing liquid such as respirators, intubation tubes, humidifiers, etc that are used in a hospital lead to complications in critical cases. The mean age group of patients most commonly affected by Chryseobacterium indologens in our study was 63. Based on SENTRY antimicrobial surveillance program (1997-2001), the highest prevalence of Chryseobacterium indologens was observed in the old age patients implying similar trends to our study. 14 The majority of Chryseobacterium indologens were isolated from sputum and blood specimens. The first existence of C. indologenes in a respiratory specimen (tracheal aspirate) in a ventilated patient with pneumonia was recognized by Bonten et al. 16 Similarly, the study conducted in Taiwan on 215 cases of Chryseobacterium species revealed its maximum isolate obtained from sputum (138 isolates) and a blood sample (39 isolates) in which the sources of clinical specimens are similar to our findings. 17 The isolation of drug resistant Chryseobacterium indologens (MDR/XDR) were isolated in higher proportion from the respiratory specimens such as sputum (OR = 1.5, 95% (0.82-9.30) in our study. The drug resistance observation corresponds to respiratory specimen from the similar studies conducted in Asia8-17 that coincides with our study that was also statistically significant P < .05.
The antimicrobial surveillance program also revealed that the most active antimicrobials against Chryseobacterium indologenes were the quinolones group and trimethoprim-sulfamethoxazole (>95% susceptibility) 14 which supported the AST pattern in which tigecycline, cotrimoxazole, levofloxacin, and piperacillin-tazobactam remained to be sensitive compared to other groups of antibiotics in our study as shown above in Figure 3. However, despite of large in-vitro susceptibility of tigecycline, several studies suggests to follow other drugs instead of tigecycline for the treatment of patient in critical cases.18,19,20,21 In addition, Lin et al 18 suggested that cotrimoxazole was the most appropriate antibiotic against Chryseobacterium indologens in the study.
The proper choice of drugs used for treating Chryseobacterium indologenes is complex due to unavailable data in clinical laboratory standard institute guidelines, and its possibility to acquire antimicrobial resistance that involves resistance to many selected antibiotics that are used in empirical therapy for various gram-negative bacterial infections. Chryseobacterium indologenes are intrinsically resistant to aminoglycosides, cephalosporins (first generation), penicillins, aztreonam, carbapenem, and other cell membrane synthesis drugs like colistin and polymixin B.17,18,19,20 The information was evident enough to determine the least susceptibility to ceftazidime and imipenem. Hsueh et al 5 denoted the incidence of hospital-acquired infection associated with Chryseobacterium indologens that correlates with our findings in patients with a prolonged hospital stay.
Regarding on discussion of empirical therapy, the treatment of infection caused by Chryseobacterium indologens remains complicated due to problems in selection of antibiotics: One of the reason is due to its intrinsic resistant ability to resists to commonly used antibiotics such as aminoglycosides, carbapenems, and polymixins in intensive care unit whereas the other issue was resistance acquired by bacterium on that particular selected antimicrobial. In contrast, the carbapenem antibiotics which has been used as a “Magic bullet” for empirical therapy of wide range of gram negative bacteria were found to be useless for Chryseobacterium indologens infection. Interestingly, the Chryseobacterium indologens isolates from outside of Asia were relatively sensitive to beta-lactams and fluoroquinolones, whereas isolates from the Asia-Pacific region were non suceptible. This might be the reasons of emerging beta-lactamases highlighted in our study. According to past antibiogram survey, the majority of antibiogram data on SENTRY projects on Chryseobacterium indologenes from worldwide of local hospital determined the highest susceptibility of this organisms to trimethoprim-sulfamethoxazole/Cotrimoxazole followed by fluroquinolones group that had showed reasonable activities.14,17-20 Therefore, based on the available published reports and SENTRY project data, several reports determines cotrimoxazole as well as fluroquinolones antimicrobial could be better options in terms of empirical therapy.14,17-23
Our study showed that combined antibiotics therapy and prolonged hospital stays are more prone to colonizing and developing drug-resistant Chryseobacterium indologens. Despite limited studies being conducted in Chryseobacterium indologens, Alkofide et al 24 explained the affiliation between the existence of drug-resistant pathogens with the mutual antibiotics therapy as well as longer hospital stays in ICU. Another study suggests that Chryseobacterium infections in humans are frequently associated with patients undergoing treatment by broad-spectrum antibiotics for prolong periods as well as patients with low immune status.25,26 Similar trends were obtained in our study that showed the highest risk factor (OR = 7.63, 95% Cl 2.262-25.7) of developing MDR- Chryseobacterium indologens infection.
Although there are not much sufficient data available for phenotypical testing of multidrug-resistant enzymes in Chryseobacterium indologens despite several literature searches, it is less frequently isolated in a clinical sample. Phenotypical examination of beta-lactamase enzymes in our study consisting ESBL, MBL, and AmpC detection was possible due to application of ceftazidime, imipenem, and cefoxitin disk. Our study showed altogether n = 12 Chryseobacterium indologens ESBL producers that were detected phenotypically. Due to a lack of sufficient literature review on Chryseobacterium indologens because of its rare encounter in the diagnostic microbiology laboratory. Our findings were supported by a French study that found in-vitro synergistic activity between third generation cephalosporins with and without clavulanic acid as well as cefoxitin in multidrug drug-resistant Chryseobacterium isolates using 2 disk assay for phenotypical identification. 17 Other studies had provided information on the presence of ESBL in Chryseobacterium species.19,25
According to the case report conducted in Kuwait designated phenotypically detection of highest Metallo-beta-lactamase producer Chryseobacterium species, that is, n = 8 out of 10 strains, with a similar methodology followed in our study which was later confirmed by MBL gene detection applying PCR. These findings show similar trends to our study in which the majority of beta-lactamases were identified as MBL (n = 53) producers in Chryseobacterium indologens. The study is further supported by a phenotypical study conducted in Argentina that depicted the presence of MBL following its drug-resistant characteristics. 26 The study conducted in Japan also revealed the existence of metallo beta-lactamase property that acquires resistance to Chryseobacterium indologens. 27 Another study conducted in the UK further determined the presence of chromosomal-mediated MBL enzymes in Chryseobacterium species. 28
The MDR bacterium evolving into XDR by acquiring resistant to antibiotics, which were used to be previously administered for the treating MDR results major threat that can lead to the crisis of antibiotics for human worldwide. Current evidence indicates poor awareness programs on health and hygiene, lack of accurate diagnostic tools, and inappropriate prescription or overprescription in clinical settings, most cases in developing nations where the antibiotics access are easy led to existing antibiotics crisis. To solve this crisis we should strongly prepare to deal with such superbugs. According to WHO, initial step is to accept the crisis and implement better strategy by analyzing the surveillance data which shows drift of antibiotics resistant to design the alternate therapies. Several mitigation approaches should be implemented for the management of MDR and XDR bacteria including Chryseobacterium indologens, that is, following effective sterilization and disinfectant methodology, proper infection control practices such as hand hygine, controlled access in antimicrobial purchases as well as conducting educational program to enhance health and hygiene practices and following guidelines to prevent overprescription for both humans and farm animals and avoiding unnecessary prescription of antibiotics (without the idea of intrinsic resistant nature of bacteria) can minimize the emerging bacteria including MDR-Chryseobacterium indologens. Hence, implementing effective strategy for antimicrobial administration can play vital role in dealing with these superbugs. 29
Conclusion
Although Chryseobacterium indologenes is an extremely rare bacterium in the diagnostic microbiology laboratory, numerous reported cases show its tendency to increase the rate of infection that determines the advancement of diagnostic tools and proper methodologies. As due to lack of specific guidelines for this bacterium it causes significant mortality in patients with immunocompromised states, comorbidities, and long hospital stays. The existence of MDR and XDR strains Chryseobacterim indologens in our study possessed several types of drug-resistant beta-lactamases that conferred developing resistance to a wide range of antibiotics that had created an enormous challenge for microbiologists and clinicians to treat the patient.
Limitations and Strength of the Study
Our studies possess certain limitations; our study was based on phenotypical methodology and we were unable to conduct molecular assay in a low resource country due to lack of funding as molecular assay detects specific betalactamases (ESBL, Amp-C, and MBL) producing genes that were responsible for causing MDR and XDR despite it is expensive. Other limitations were our study is single center based study. There would be more possibility to add sample sizes if multicenter study had been conducted with heath care associated to several other areas. Regarding on strength, study on Chryseobacterium indologens are still less because of its rare encounter in the laboratory. The drug resistant patterns and beta-lactamases enzyme are studied in a scanty manner after the SENTRY antimicrobial surveillance during 2004. Our results highlights proper methodology for detection of Chryseobacterium indologens that can be applied in a limited resource settings where the expensive molecular methods are sole financial problems in the laboratory of undeveloped and developing countries. Interestingly, the CLSI guidelines still suggests phenotypical confirmation to determine the drug resistant enzymes (ESBL, MBL, and Amp-C) in a lawn culture plates with semi-confluent growth. Other interesting approach obtained from the available literature search 30 had also claimed that phenotypical detection of betalactamases are more sensitive than molecular tests due to its ability to detect betalactamase enzymes however it is only applicable in terms of sensitivity. The study on drug resistant patterns, formation of beta-lactamase enzyme as well as statistical correlations of various clinical findings, risk factor, and discussion on empirical therapy in our study could aid in the development of the treatment outcome associated to Chryseobacterium infections.
Supplemental Material
sj-docx-1-mbi-10.1177_11786361221150755 – Supplemental material for Phenotypical Detection of β-Lactamases in a Multidrug-Resistant and Extensively Drug-Resistant Chryseobacterium indologens: A Rare Human Pathogen With Special References to Risk Factor
Supplemental material, sj-docx-1-mbi-10.1177_11786361221150755 for Phenotypical Detection of β-Lactamases in a Multidrug-Resistant and Extensively Drug-Resistant Chryseobacterium indologens: A Rare Human Pathogen With Special References to Risk Factor by Ranjana Parajuli, Trishant Limbu, Raina Chaudhary, Kundan Gautam and Pragyan Dahal in Microbiology Insights
Footnotes
Acknowledgements
The authors would like to acknowledge 2 microbiology laboratory personnel Manisha Maharjan and Ashmi Khadka for the preservation of bacteria and the medical record section of Grande International Hospital for providing detailed clinical information of patients.
Declaration of Conflicting Interests:
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding:
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Author Contributions
Data Availability
The majority of the data representing our findings were already included in our study. The raw data can be obtained by contacting the correspondence author.
Ethical Approval
Ethical clearance was obtained via institutional review board of Grande International Hospital (GIH IRC Ref no: 05).
Consent
Consent was obtained in written form from all the ages of patient’s that were enrolled in our study.
Significant Statement
As Chryseobacterium indologens are less frequently encountered in the laboratory. The studies conducted on this bacterium are very rare and noble. More importantly, this bacterium is intrinsic resistant to aminoglycosides, first generation cephalosporins, penicillins, aztreonam, carbapenem, and drugs used in last resorts of treatment in most gram-negative bacteria, that is, colistin and polymixin B. Furthermore the emerging beta-lactamases (ESBL, Amp-C, and MBL) that enables them to acquire drug-resistant to more applied antibiotics makes them difficult to treat and cause high mortality rate in immunocompromised patients as well as patients with co-morbidities.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
