Abstract
Introduction
The genus
Several studies have reported the role of climate as a major abiotic factor in shaping the bacterial communities present in the soil by controlling the rate of soil formation and its chemical composition.24–27 India possesses a large variety of climates ranging from extremely hot desert regions to high altitude locations with severely cold conditions similar to northern Europe. Climate in India can be divided into six zones according to Köppen classification system as tropical wet climatic zone (TWCZ), tropical wet and dry climatic zone, arid climatic zone (ACZ), semi-arid climatic zone ACZ, humid subtropical climatic zone (HSCZ), and mountainous climatic zone (MCZ).28,29 This climatic diversity experienced by Indian soils makes it an interesting investigation site for understanding the community profile of genus
Most of the recent work in soil microbial ecology which focuses on cataloging the diversity of soil bacteria and documenting how soil bacterial communities are affected by specific environmental changes or disturbances is gaining more importance30,31 and similar trend of studies could very well be targeted on soil
Materials and Methods
Study sites and sample collection
The sampling was performed at different locations from India falling under four climatic zones according to Köppen classification system (Supplementary Figure 1). Soil was collected in the pre-monsoon season from February 2017 to March 2017 from three different spots at each climatic zone starting from the southern part of India and moving along the northern part of India, the study sites cover four states of India each representing a different climatic zone namely, Kerala (TWCZ), Rajasthan (ACZ), Chandigarh (HSCZ), and Himachal Pradesh (MCZ) (Figure 1). All samples were collected from areas having no anthropogenic activity or animal influence and low density of vegetation. At each sampling spot, quadrates of 3.5 × 3.5 m were selected and the surface soil was removed at a depth of 15-20 cm. Thereafter, soil was collected using a sterile shovel and transferred into sterile sampling bags (Nasco: Hi-Media, India) and transported to laboratory in ice. Later, the samples from each climatic location were sieved through sterile 2 mm mesh, pooled together to make a composite sample and processed immediately.
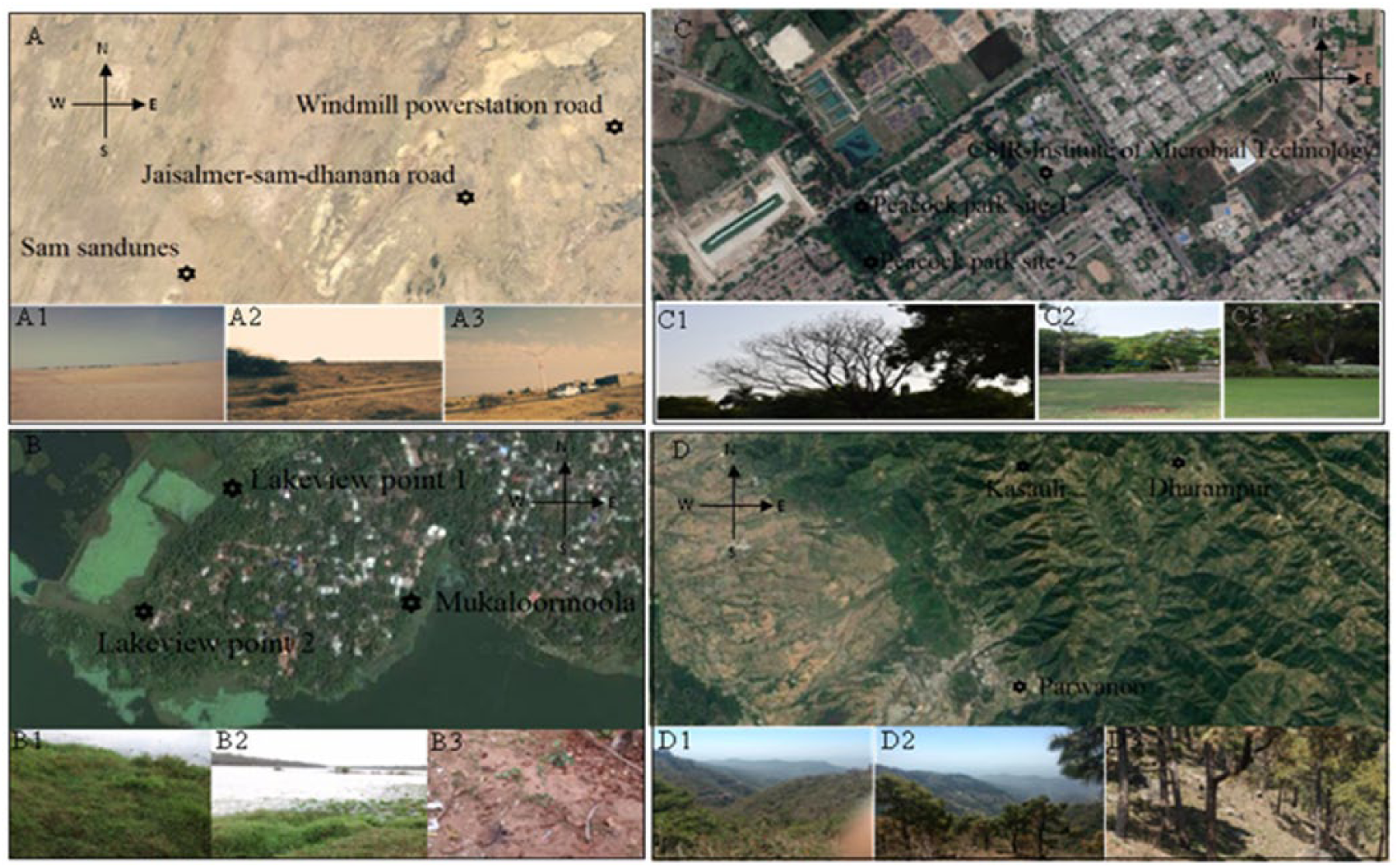
Location of selected soil sampling sites from different climatic zones of India. A. Map of western Rajasthan (arid climatic zone) showing sampling sites marked with 6-point star (★); A1, A2, A3 photographic images of sampling area in western Rajasthan. B. Map of southern Kerala (tropical wet climatic zone) showing sampling sites marked with 6-point star (★); B1, B2, B3 photographic images of sampling area in southern Kerala. C Map of Chandigarh city (humid subtropical climatic zone) showing sampling sites marked with 6-point star (★); C1, C2, C3 photographic images of sampling area in Chandigarh. D Map of Himachal Pradesh (mountainous climatic zone) showing sampling sites marked with 6-point star (★); D1, D2, D3 photographic images of sampling area in Himachal Pradesh.
Geochemical parameters of soil
A total of 10 geochemical parameters of soil were assessed, including pH, moisture content, organic carbon, nitrogen, potassium, calcium, magnesium, phosphorous, iron, and boron. The pH was measured using pH electrode (Shimadzu, Japan) in a saturated colloidal solution of deionized water and moisture content was calculated using oven dry method. Total phosphorus was measured colorimetrically and total nitrogen was analyzed by the micro Kjeldahl method. 32 Determination of iron, boron, calcium, potassium, and magnesium was carried out using Spectrometer (Zeenit 700; Analytik, Jena, Germany). 33 Total organic carbon was determined using partial oxidation method. 34
Plating and isolation of bacterial strains
The samples were immediately processed by serial dilution and plating technique using normal saline (0.45%) by plating on to general purpose media like TSA (Tryptic Soy Agar; Hi-Media), NA (Nutrient Agar; Hi-Media), minimal media involving TSA 1:10 and 1:100 dilutions (TSA; Hi-Media) and selective media involving AIA (Actinomycetes Isolation Agar; Hi-Media), SMA (Streptomyces Agar; Hi-Media) followed by incubation at 30°C for a week subsequently monitoring bacterial growth at 24 h, 48 h, 72 h, and so on. Quantitative analysis for bacterial growth in all the samples was examined using colony counting and determination of colony forming units (CFU) per milliliter. Qualitative analysis of colonies involved identification of bacterial species and their enumeration from all the samples. The colony morphotypes were selected using four parameters: colony size, form, color, and texture. A phenotypic variant was considered when it differed in at least one of the referred morphological parameters and further selected for identification. 35
Strain identification and phylogenetic analysis
All the strains were identified using through genomic DNA isolation and 16S rRNA gene sequencing. Genomic DNA was extracted using Nucleospin nucleic acid kit followed by PCR amplification using eubacterial universal primers 27F (5′-AGA GTT TGA TCM TGG CTC AG-3′) and 1492 R (5′-CGG TTA CCT TGT TAC GAC TT-3′). 36 Furthermore, the purified PCR product was amplified using 1100R (5′-GGG TTG CGC TCG TTG-3′) primer subjected to Sanger sequencing.37,38 The 16S rRNA gene sequences obtained after sequencing were searched for similar sequences from 16S rRNA gene database of EzTaxon server 39 (Supplementary Table 1). To determine the phylogenetic relationship of the identified isolates, their 16S rRNA gene sequences were aligned using Mega version 7.0 40 and phylogenetic trees were constructed using the neighbor joining as well as maximum likelihood and maximum parsimony algorithms. 41 Bootstrap analysis of 1000 bootstrap replications was performed to assess the confidence limits of the branching. 42
Nucleotide sequence accession numbers
The 16S rRNA gene sequences of the
Results
Chemical analysis of soil samples
The soil samples collected at each climatic zone were pooled together to make a composite sample and the highest values of moisture were recorded in soils collected from TWCZ followed by MCZ, HSCZ, and lowest in ACZ. The pH (3.94) was lowest in TWCZ soil which reflects its acidic nature whereas the pH in soils collected from ACZ (8.05) and HSCZ (8.13) was alkaline. The soil from MCZ was mildly acidic having a pH value of 5.44. There was a much less difference in the values of organic carbon, phosphorous and iron in all soil samples. The highest values of organic carbon and phosphorous were recorded in soils collected at MCZ, whereas highest values of iron were recorded in ACZ. Greater differences were observed in values of calcium and magnesium, the value of calcium was highest in MCZ and lowest in HSCZ. The value of magnesium showed higher levels in ACZ and lower levels in HSCZ. The value of boron was highest in ACZ and lowest in TWCZ. The percentage of potassium had similar range in ACZ (1.86%) and MCZ (1.88%) together. Similarly, it was observed as 2.9% for both TWCZ and HSCZ (Table 1).
Physiochemical properties of soil samples collected at different climatic zones.
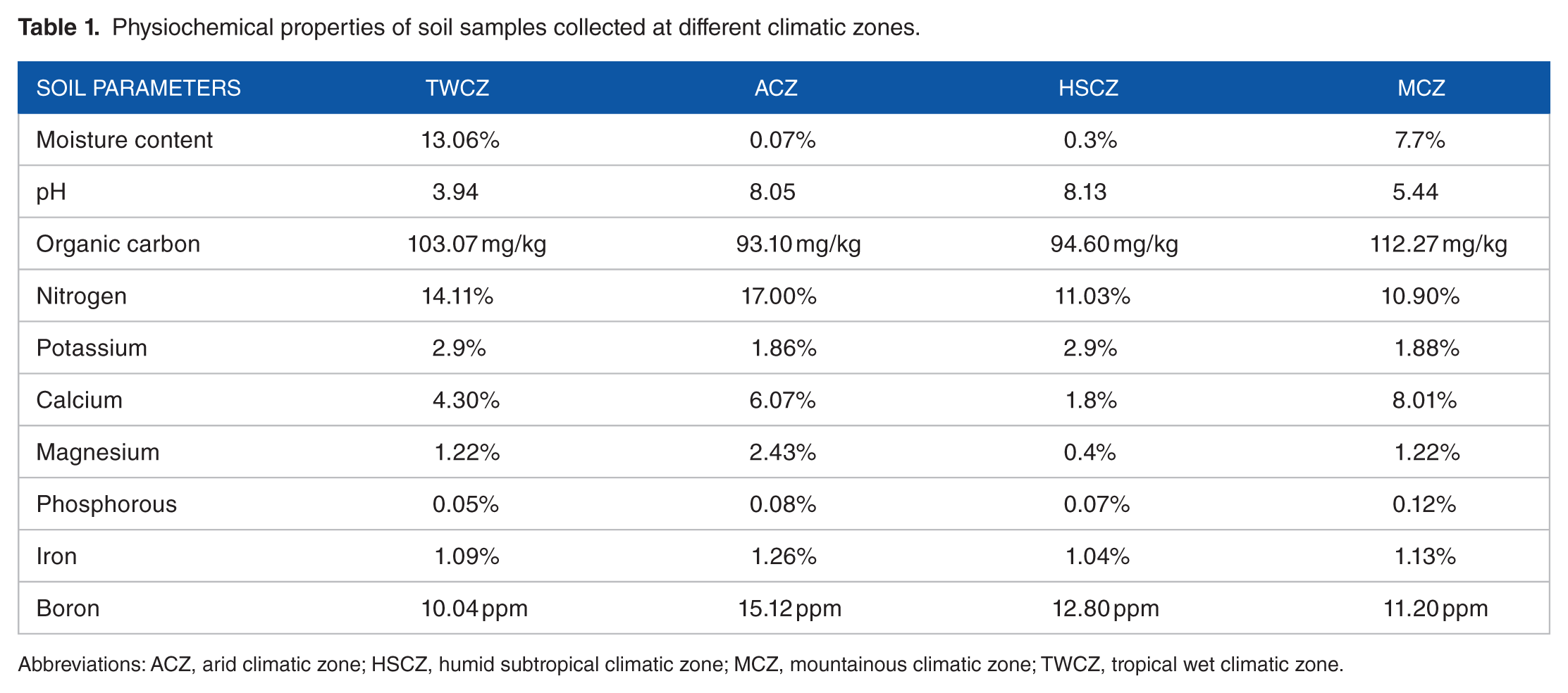
Abbreviations: ACZ, arid climatic zone; HSCZ, humid subtropical climatic zone; MCZ, mountainous climatic zone; TWCZ, tropical wet climatic zone.
Culturable diversity of Bacillus species
The viable bacterial population was measured using plating and CFU determination on five different media namely, TSA, NA, SMA, and AIA. Of all these media, the highest CFU (2.58 × 104 CFU/mL) was recorded in TSA in sample TWCZ. The phylum Firmicutes dominated among other identified phyla Actinobacteria and Proteobacteria which could be correlated with the depth of soil collected and presence of rhizospheric soils in the sampling sites. In the phylum Firmicutes, 20 different
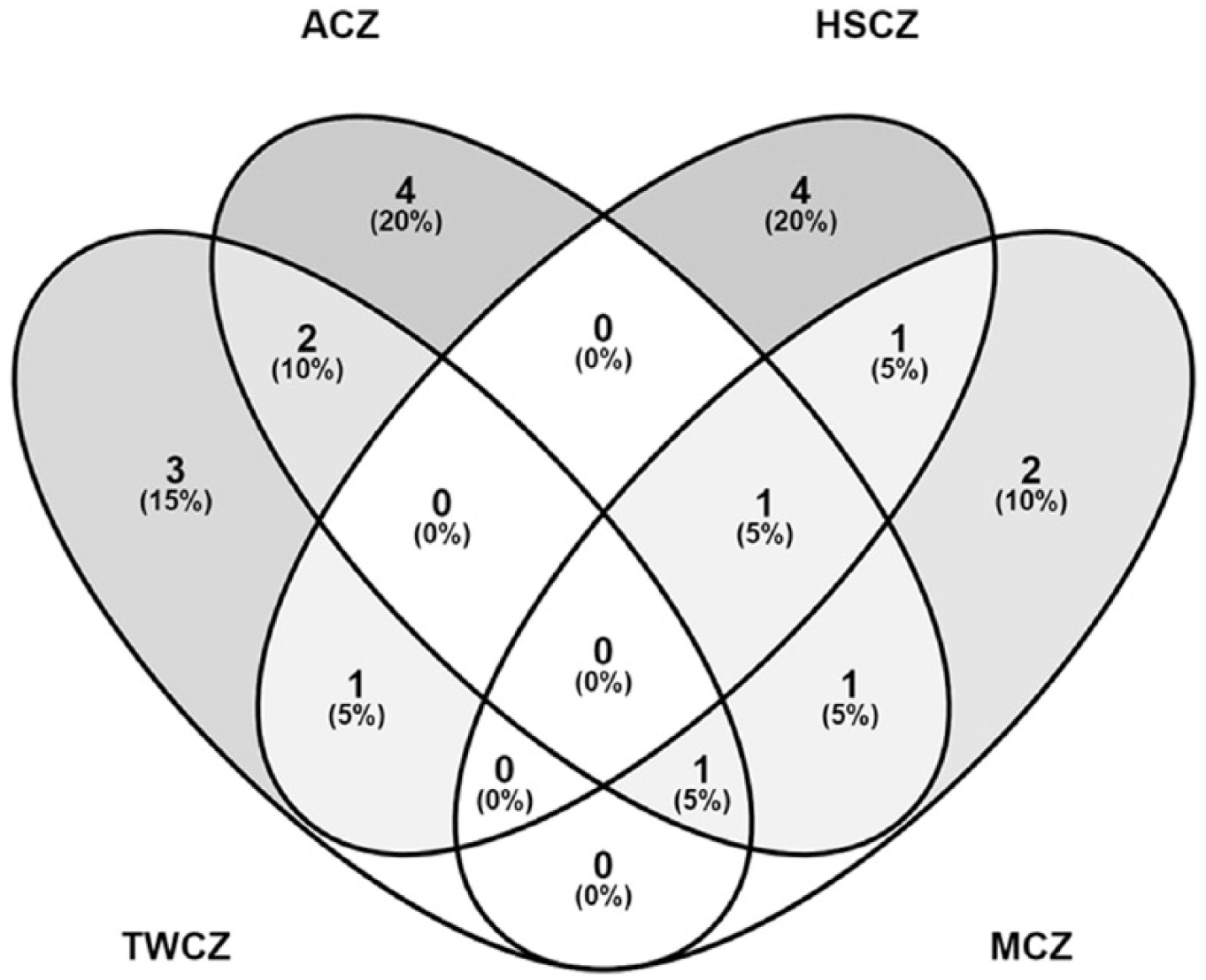
Venn diagram showing the different
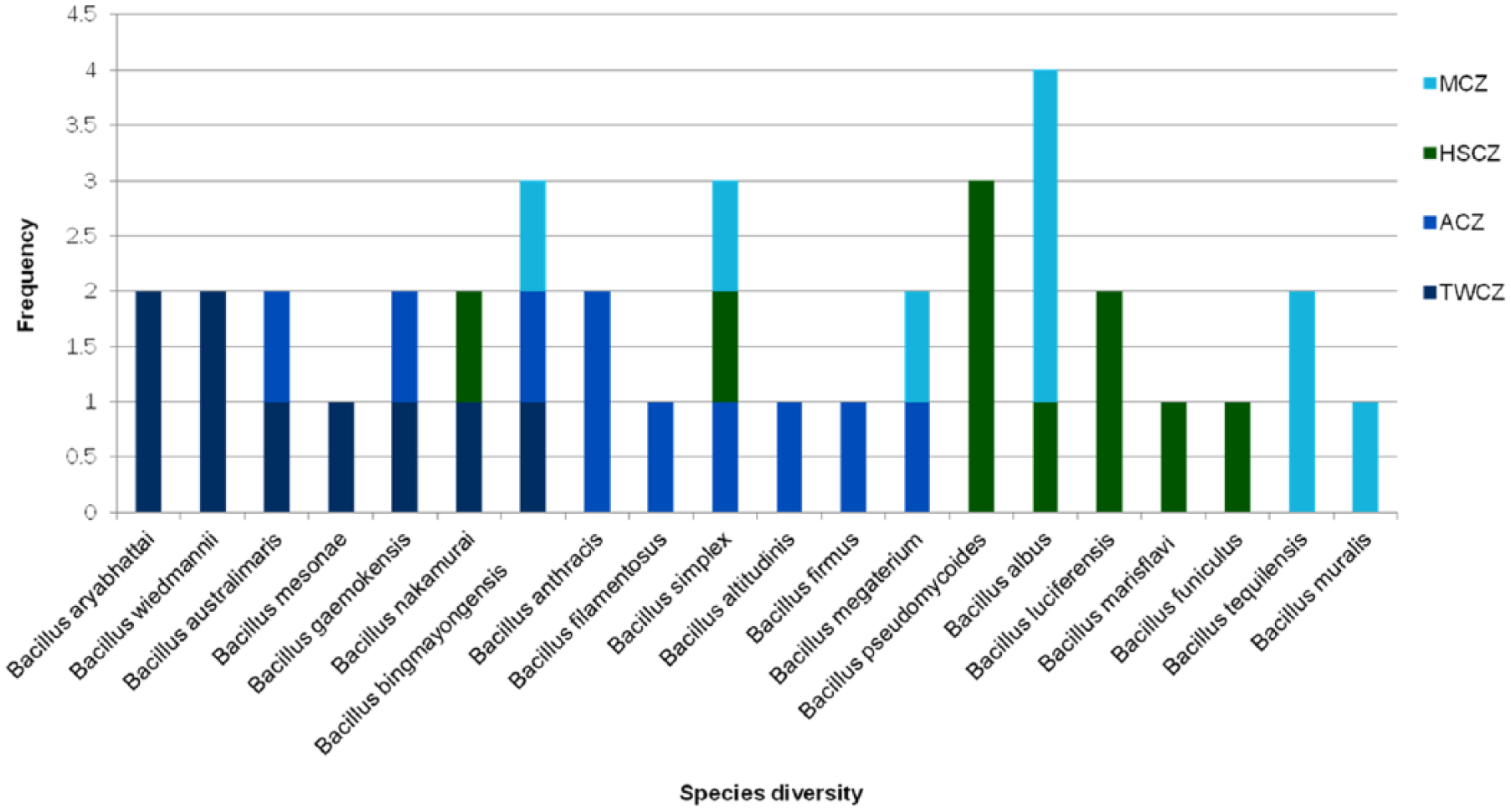
Distribution of
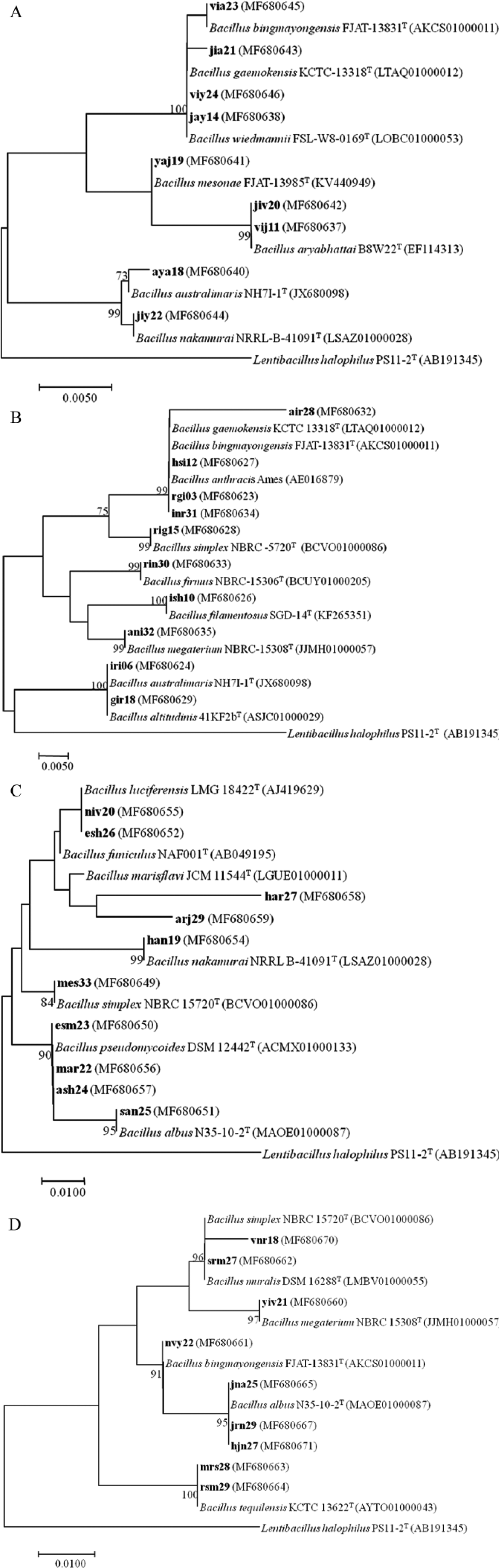
A. Phylogenetic tree based on 16S rDNA sequences showing relationship between different members of phylum Firmicutes isolated from TWCZ. Boot-Strap values (1000 replicates) of >70% are given at the nodes.
Discussion
There are number factors that mold the microbiota surviving in the soil, one of which is the climate and considered as a major abiotic factor controlling the other sub-factors that shape the
The Indian subcontinent has a varied climatic exposure having a tremendous effect on the terrestrial environments that could modulate the soil physiochemical properties in-turn affecting the bacterial community profile of a particular habitat. This makes lot avenues for exploring the microbial community diversity in Indian soils on a climatic and geographical scale which are given much less attention. Several studies have evaluated the culturable microbial diversity of Trans-Himalayas of Himachal Pradesh Similarly, the diversity of culturable
The change in the pattern of diversity in the soil
Supplemental Material
Supplementary_Fig_xyz1078749fd14d3 – Supplemental material for Climate and Soil Properties Influence Species Diversity of Soil Bacillus Community in India
Supplemental material, Supplementary_Fig_xyz1078749fd14d3 for Climate and Soil Properties Influence Species Diversity of Soil Bacillus Community in India by Girish R Nair and Suresh SS Raja in Microbiology Insights
Supplemental Material
Supplementary_table_1_xyz10787872bd34a – Supplemental material for Climate and Soil Properties Influence Species Diversity of Soil Bacillus Community in India
Supplemental material, Supplementary_table_1_xyz10787872bd34a for Climate and Soil Properties Influence Species Diversity of Soil Bacillus Community in India by Girish R Nair and Suresh SS Raja in Microbiology Insights
Footnotes
Funding:
The author(s) disclosed receipt of the following financial support for the research, authorship and/or publication of this article: This work was supported by Council of Scientific and Industrial Research, India (to Girish R Nair), CSIR-SRF direct fellowship. The funder had no role in study design, data collection and analysis, decision to publish, or preparation of the manuscript.
Declaration of conflicting interests:
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Author Contributions
Conceptualization and Study design: Suresh S S Raja (SSR), Manuscript preparation, data analysis, review and editing: Girish R Nair (RGN).
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
