Abstract
Background:
Malaria is a public health problem, particularly in low- and middle-income countries. In Angola, it is the leading cause of death, morbidity, and absenteeism from work and school.
Objective:
To evaluate the social and clinical factors associated with resistance to in-hospital treatment.
Methodology:
A prospective analytical cross-sectional study with a quantitative approach was conducted including 220 patients with malaria.
Results:
Of the 220 patients enrolled, the majority were between 21 and 40 years old (72.7%), male (53.6%), of peri-urban areas (47.7%), employees (46.4%), and with high parasitemia levels (57.7%). Of the remaining hospitalized patients (61.4%), 20.9% were resistant to treatment. The resistance risk was higher in patients over 40 years [OR: 5.91 (95% CI: 0.76-45.7), P = .088], from rural regions [OR: 2.48 (95% CI: 0.95-6.48), P = .064], that were unemployed [OR: 1.06 (95% CI: 0.52-2.15), P = .859], presenting high parasitemia [OR: 1.95 (95% CI: 1.02-3.75), P = .043] and who remained hospitalized [OR: 5.28 (95% CI: 0.63-43.1), P = .121]. The risk to develop resistance was lower in patients that were students [OR: 0.04 (95% CI: 0.01-0.37), P = .004], patients who were treated with dipyrone [OR: 0.06 (95% CI: 0.01-0.24), P < .001], metoclopramide [OR: 0.25 (95% CI: 0.09-0.67), P = .006] and ciprofloxacin [OR: 0.22 (95% CI: 0.11-0.44), P < .001].
Conclusion:
Treatment with antimalarial drugs as well as the use of adjuvants such as dipyrone, metoclopramide, ciprofloxacin, and diazepam can reduce the chances of developing resistance to malaria treatment, however, it is necessary to carry out further in-depth studies.
Background
Malaria is among the most devastating and disseminated tropical parasitic diseases in the world, being prevalent in many developing countries, and resistance to antimalarial drugs has been a public health concern as many strains of the parasite can survive and multiply even with the administration and absorption of drugs at doses equal to or greater than those generally recommended. 1 These facts may be closely associated with different factors such as the rate of parasite mutation, the overall parasite load, drug capacity, adherence to treatment, treatment guidelines, inadequate dosage, deficient pharmacokinetic properties, use of counterfeit medications, or other factors that can lead to inadequate exposure of the parasite to antimalarials and other low-quality medications that can stimulate resistance.1,2
Resistance to antimalarial drugs has emerged as a threat to global malaria control efforts, particularly in the sub-Saharan region. Plasmodium falciparum resistance to institutionalized treatments challenges to combat and control, especially in endemic countries. Data collected in more than 1000 studies on therapeutic efficacy and on P. falciparum drug resistance molecular markers bring a response to the use of antimalarials in 10 years of surveillance (2010-2019), bringing concerns to the world community and encouraging the monitoring and protection of populations in the coming decades.3,4
With the emergence of parasites resistant to antimalarial drugs and other conditions that also allow resistance to malarial treatment, all efforts that have been achieved so far for its control are compromised, making the medical and scientific community seek new forms of treatment for the control of this disease. 5
The use of quinolones (drugs that affect the polymerization of hemozoin), antifolates (drugs that block the enzymes dihydrofolate reductase and dihydropteroate synthetase of the parasite), and others such as artemisinin (which have several mechanisms), usually administered alone or in combination to treat malaria, have been a cornerstone for its control in sub-Saharan Africa, where the use of artemether/lumefantrine and artesunate/amodiaquine has been frequent due to and by the notorious abilities of Plasmodium falciparum to develop drug resistance. Many recently developed antimalarial programs include the use of dihydroartemisinin/piperaquine as a second-generation antimalarial.6,7
Resistance to antimalarial drugs often used in emerging countries, including combination therapies, have forced scientists to search and develop drugs with novel mechanisms of action, especially with regards to resistance to P. falciparum, which are highly prevalent in Southeast Asia, Africa, and South America. 8
In the first 5 months of 2021 (January-May), Angola has recorded 3 799 458 cases of malaria and 5573 deaths.9,10 The actual figures for disease, morbidity, and mortality may differ from the figures reported above due to the weak capacity of surveillance, diagnosis, and underreporting of cases. 10
Success for the prevention, control, and cure of malaria in endemic countries such as Angola, depends on the sustained clinical efficacy of first-line therapeutic combinations (ACTs) for its treatment, however, the emergence and spread of drug resistance strains represent a constant threat to the challenges of fighting malaria, as scenario models of widespread resistance to ACTs in malaria-endemic countries, predict an impact of more than 100 000 additional deaths per year. 11
With an increase of cases and deaths from malaria in Angola, particularly with the COVID-19 pandemic, where restrictions and other measures to control the pandemic hinder medical and drug assistance for other diseases, issues related to resistance to treatment begin to rise, leading the medical community and other health professionals to investigate the factors that may be associated with resistance to the treatment of malaria. There is very little scientific evidence to show the factors associated with resistance to treatment of malaria at the hospital level in Angola.
This study is aimed to evaluate the social and clinical factors associated with resistance to in-hospital treatment of malaria since with the in-hospital assessment it is possible to identify factors of resistance to treatment and propose alternatives to reduce the impact of resistance to malaria treatment. The difficulties of treating malaria in Angola have been a constant and frequent challenge, due to several factors, especially those related to the lack of resources to obtain medicines, a large number of counterfeit drugs in the Angolan market, self-medication by patients, and many other problems that can arise from access to medical and drug services, thus, the assessment of in-hospital resistance allows to reduce biases related to the use of counterfeit drugs, lack of treatment adherence, use of insufficient doses or overdose, non-compliance with drug administration schedules and treatment at inappropriate times, since intra-hospital treatment and hospital care is performed and monitored mostly by trained health professionals and administration is mostly by the intramuscular or intravenous route, which favors pharmacokinetic and pharmacodynamics results.
Materials and Methods
Study design
A prospective analytical cross-sectional study with a quantitative approach was conducted.
Sample size determination
The study population consisted of 220 out of 410 patients admitted and hospitalized for malaria complications at the Hospital Josina Machel—Maria Pia between December 2018 and January 2020. All patients admitted to the hospital unit were included in the study, among the 410 patients treated for malaria in the study, only 220 patients presented sociodemographic and pharmacological data during 5 days of hospitalization, thus, the sample was opportunistic due to the inclusion and exclusion criteria. A 95% confidence index was maintained with a margin of error of 5%. Only patients who had moderate (above 1000 p/mm3) or high (above 10 000 p/mm3) parasitemia were included in the study, in addition, patients who did not meet the other selection criteria or did not agree to participate in the study were excluded.
Inclusion of study participants
Additional information was collected through an open and closed questionnaire for patients aged 12 to 66 years old and only patients who were hospitalized for more than 5 days were included. All patients with a history of hypertensive disease, diabetes, chronic kidney disease, cerebral malaria, other chronic diseases, low immunity, and other pathologies contributing to treatment resistance were excluded to avoid bias in data analysis. Patients who had low parasitemia (below 1000 p/mm3) were not included in the study because most of them had self-medicated with antimalarials and other drugs before being admitted to the hospital unit under study.
Laboratory procedure
Malaria diagnoses were performed using rapid malaria antigen tests (SD-Bioline Malaria AG Pf/PAN) and confirmed with microscopy technique of direct visualization of the parasite by Giemsa-stained peripheral blood thickening. 12 Patients who presented parasitemia less than or equal to 1000 p/mm3 were classified as moderate parasitemia whilst patients who presented parasitemia above 1000 p/mm3 were classified as high parasitemia.13,14 The diagnosis of resistance to treatment of malaria was made by evaluating the parasitemia of patients 5 days after finishing the treatment with antimalarial drugs (Arthemeter or Artesunate), which have already been shown in other studies that under normal conditions can reduce parasitemia from 100 000 to 0 (p/mm3) within a maximum of 5 days of in-hospital treatment.12,13,15 Patients who did not have a complete reduction in parasitemia after 5 days of treatment were classified as resistant and patients who had a complete reduction in parasitemia were considered non-resistant, meaning that delayed elimination of the parasite for more than 5 days was considered treatment resistance As rapid tests were carried out to detect P. falciparum and P. vivax and microscopic evaluations that only confirmed the presence of P. falciparum, we were able to confirm that there were no cases of infection by another parasite or even co-infection.
Statistical analysis
Initially, the data obtained in the study were compiled into an Excel 2017 database, later transferred, categorized, and analyzed using SPSS v25. Absolute and relative frequencies were determined. The chi-square (χ2) and logistic regression tests were used to assess the relationship between categorical variables. The odds ratio (OR) and its 95% confidence intervals (CI) were calculated to assess the strength and direction of the relationship. All reported P-values are 2-tailed and deemed significant when P < .05. Parasitemia data were summarized as mean ± standard deviation for admission data (Initial) before the start of treatment and 5 days after admission (final).
Ethical considerations
It was approved by the Human Research Ethics Committee of the Higher Institute of Health Sciences (Official Letter No. 755/GD/ISCISA/UAN/2018) and by the clinical directorate of Hospital Josina Machel—Maria Pia in Luanda (Official Letter No. 260/DPC/HJM/2018). All patients who agreed to participate have signed the informed consent form after being enlightened about the nature and objectives of the study.
Results
Sociodemographic and clinical characteristics related to resistance
Among the sociodemographic and clinical characteristics related to resistance to in-hospital malaria treatment among patients admitted to hospitals in Luanda, we found that a total of 220 patients diagnosed with malaria between October 2018 and December 2019 were included (Table 1). The age of the study participants was ranged from 12 to 66 years with a mean age was 28 ± 10 years. All patients lived in Luanda province and had been infected with P. falciparum and no other types of infection by other species of Plasmodium or even co-infection by different types of Plasmodium were identified. Patients aged between 21 and 40 years old were the majority (72.7%, n = 160/220), male (53.6%, n = 118/220), from the peri-urban areas of Luanda (47.7%, n = 105/220) and employed (46.4%, n = 102/220). Patients with high parasitemia (57.7%, n = 127/220) and who remained hospitalized for more than 5 days (61.4%, n = 135/220) represented the majority of the individuals studied, while mortality was observed in 5.9% (n = 13/220) of patients. A total of 46/220 (20.9%) patients were considered resistant to treatment. A significant relationship was observed between occupation, level of parasitemia, and hospital outcomes, with resistance to in-hospital treatment of malaria. On the other hand, there was no relationship observed between age, gender, and place of residence with resistance to in-hospital treatment for malaria (P > .05).
Sociodemographic and clinical characteristics related to antimalaric resistance among patients with malaria in Luanda, Angola.
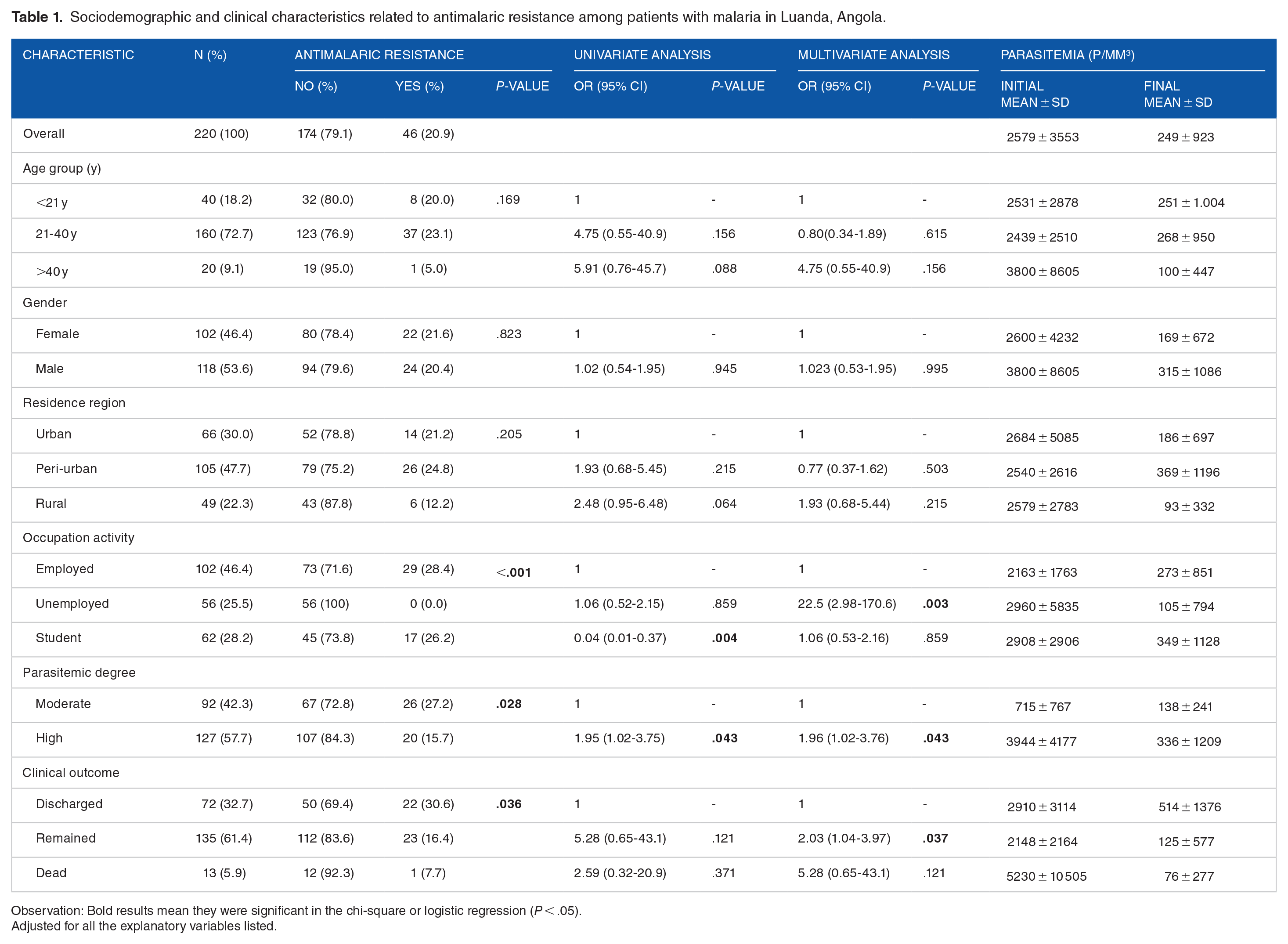
Observation: Bold results mean they were significant in the chi-square or logistic regression (P < .05).
Adjusted for all the explanatory variables listed.
In the univariate analysis, it was found that although there is no significant relationship between the risk of resistance to in-hospital treatment and age, in patients older than 40 years, the risk of resistance to in-hospital treatment seemed to be higher [OR: 5.91 (95% CI: 0.76-45.7), P = .088] and from rural regions [OR: 2.48 (95% CI: 0.95-6.48), P = .064]. On the other hand, there was a significant relationship between sociodemographic and clinical data with resistance to in-hospital treatment of malaria, patients who were unemployed [OR: 1.06 (95% CI: 0.52-2.15), P = .859], with high parasitemia levels [OR: 1.95 (95% CI: 1.02-3.75), P = .043] and who remained hospitalized for more than 5 days [OR: 5.28 (95% CI: 0.63-43.1), P = .121] with a high probability of presenting resistance to in-hospital treatment in malaria compared to the other subgroups. Only patients categorized as students were less likely to develop resistance to in-hospital treatment for malaria [OR: 0.04 (95% CI: 0.01-0.37), P = .004]. Multivariate analysis also showed high chances of 40-year-old male patients, residents of rural areas, unemployed, with high parasitemia, and patients who died in hospital, presenting resistance to in-hospital treatment for malaria.
An interesting finding was that at admission, a higher mean (with standard deviations close to or above the mean) of parasitemia was observed in patients aged over 40 years old (3800 ± 8605 p/mm3), male (3800 ± 8605 p/mm3), from the urban region (2684 ± 5085 p/mm3), unemployed (2960 ± 5835) and who died (5230 ± 10 505 p/mm3) and this fact was indifferent to the greater or lesser possibility of presenting resistance to in-hospital treatment, however, after 5 days of hospitalization, all subgroups showed a significant reduction in the mean value of parasitemia (less than 600 p/mm3) although these standard deviations remained above the means, especially in patients under 20 years old, women, from the peri-urban region, students and those who were discharged from the hospital still showed a high standard deviation (above 1000 p/mm3).
Pharmacological procedures and resistance to malaria treatment
When evaluating if pharmacological procedures could be related to resistance to malaria in-hospital treatment (Table 2), it was found that most patients were treated with Artemether (90.9%, n = 200/220), saline solution was performed in 52.3% (n = 115/220) of the patients, the use of dipyrone as an antipyretic occurred in 64.1% (n = 141/220) of the patients, metoclopramide (19.5%, n = 43/220) and complex B (16.4%, n = 36/220) were used to reduce vomiting, some patients were treated with metronidazole (10.0%, n = 22/220) to reduce gastrointestinal infection. The use of ciprofloxacin as antibiotics occurred in 22.7% (n = 50/220) of the patients and 9.1% (n = 20/220) of them received diazepam to be able to sleep. A statistically significant relationship was shown between the prevalence of resistance to in-hospital treatment of malaria and the use of antipyretics, antiemetics, and antibiotics (P < .05), while the use of antimalarials and infusions, treatment with antiparasitic and hypnotic drugs were not related to the in-hospital treatment of malaria (P > .05).
Pharmacological procedures and resistance to malaria treatment.
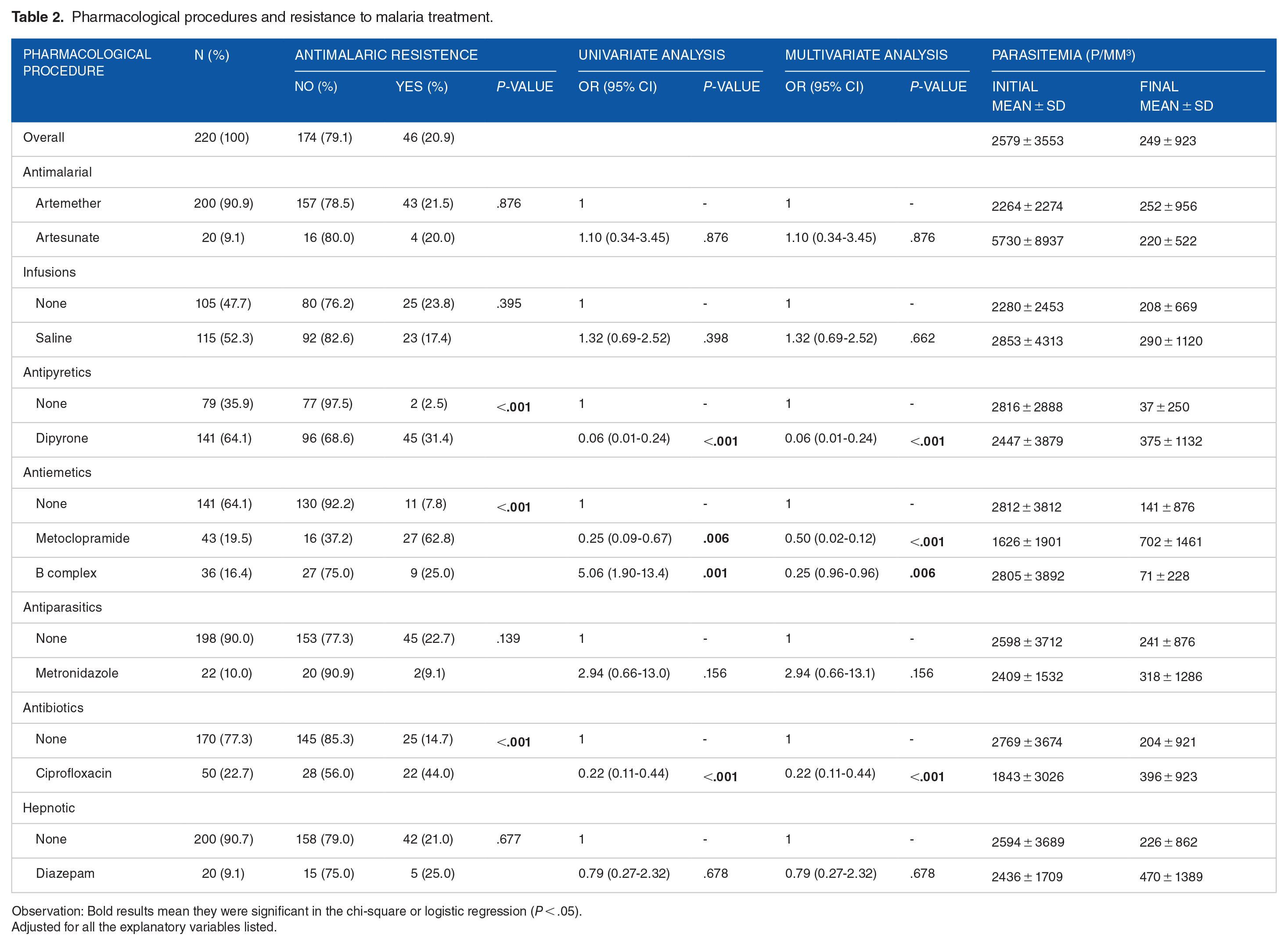
Observation: Bold results mean they were significant in the chi-square or logistic regression (P < .05).
Adjusted for all the explanatory variables listed.
Although no statistical significance was found in the univariate analysis, high chances of resistance to in-hospital malaria treatment were observed in patients treated with the antimalarial artesunate [OR: 1.10 (95% CI: 0.34-3.45), P = .876], patients treated with saline solution [OR: 1.32 (95% CI: 0.69-2.52), P = .398] and patients treated with metronidazole [OR: 2.94 (95% CI: 0.66-13.0), P = .735], whereas low odds were observed in patients treated with dipyrone [OR: 0.06 (95% CI: 0.01-0.24), P < .001 ], with metoclopramide [OR: 0.25 (95% CI: 0.09-0.67), P = .006] and with ciprofloxacin [OR: 0.22 (95% CI: 0.11-0.44), P < .001] when compared to patients in their subgroups. Multivariate analysis also showed a high chance of resistance to in-hospital treatment of malaria in patients treated with artesunate, saline solution, and metronidazole, while a low chance of resistance to in-hospital treatment was observed in patients treated with dipyrone, metoclopramide, complex B, and patients who were treated with ciprofloxacin.
When evaluating the pharmacological procedures about the mean (standard deviations) of parasitemia, it was found that on admission to the hospital all groups of patients had a mean parasitemia above 2000 p/mm3, except patients that were treated with metoclopramide and ciprofloxacin, means of infection were higher in patients treated with artesunate (5730 ± 8937 p/mm3) being this group the one that was more likely to present resistance to in-hospital treatment for malaria. However, after 5 days of hospitalization, all subgroups showed a reduction in the mean value of parasitemia (less than 800 p/mm3), although the standard deviation remained above the mean in all subgroups, especially in patients treated with saline solution, dipyrone, metoclopramide, omeprazole, and diazepam, who presented very high standard deviations (over 1000 p/mm3).
Discussion
Results related to age, gender, region of residence, occupation, and length of stay (Table 1), are similar to a previous study carried out by our research team, in the same hospital unit in 2016, to assess whether the degree of parasitemia increased the chances of kidney damage, also showed that the majority of the studied population (71%) were male, with high parasitemia (27%). 14
In the present study, it was found that about 20.9% of patients were resistant to hospital treatment for malaria, there was a statistically significant relationship between resistance to occupation, degree of parasitemia, and clinical outcomes, but age, sex, and place of residence were not related. Resistance to hospital treatment for malaria (Table 1). At certain points, our data resembles some data found in the African multicentric study that stated resistance to P. falciparum at medium level increased in Tanzania (64.0%), Sudan(55.4%), Mozambique (45.7%), Kenya (29.7%), and Malawi (8.7%) between 2000 to 2010 and suffered a slight decline in Sudan (76.0%), Kenya (65.7%) and Tanzania (17.4%), from 2010 to 2020. In the same study, in Central Africa, levels increased in Equatorial Guinea (28.9%) and Congo (85.3%) from 2000 to 2020, while in other countries it remained practically unchanged, and in West Africa, the levels remained low from 2000 to 2020, except in Nigeria where there was a reduction of 14.4%, an increase of 7.0% in Mali and 5.5% in Malawi. 16
In the present study, univariate analysis showed that some sociodemographic and clinical data, although not having a significant relationship with the risk of resistance (P > .05), increased or decreased the probability of presenting resistance to the treatment, on the other hand, the multivariate analysis also confirmed that these data (Table 1). Our data is similar to data from the study carried out in Cape Verde, which showed that of the 131 patients and 137 close contacts followed, malaria was predominantly alone (73.3%), in male (65.6%), and younger than cases not associated with malaria (P < .001), students and tertiary sector activities accounted for 80.2% of malaria cases and multiple logistic regression models suggested an increased risk of malaria because of gender male (OR = 4.99; 95% CI 2.90-8.58) and single (OR = 1.92, 95% CI 1.02-3.58), with potential risk in older age (OR = 0.98; 95%CI 0.97-1.00). 17
When evaluating how pharmacological procedures were related to resistance to malaria treatment, it was observed that although almost all patients received antimalarial treatment, especially Artemether, that the use of certain drugs from other groups, with antibiotics, antipyretics, and others, when combined with antimalarial treatment, may increase or reduce the possibility of resistance (Table 2). Our data do not show associations between artemether-lumefantrine and artesunate-amodiaquine, which seems to be more effective drugs due to their mechanism of action and therefore have been proposed as alternatives in the treatment of parasites drug-resistant. 18 The data from the present study seem to corroborate the study developed by our research team in 2018, in which the majority of patients studied were treated with Artemether (83%), artesunate (15%), and quinine (2%). 14 On the other hand, the data diverge from another study carried out in 2016, where most of the patients were treated with quinine (61%). 19
For the pharmacological procedure, the chi-square test showed a statistically significant relationship between the prevalence of resistance to treatment for in-hospital malaria and the use of some drugs, while the use of other drugs did not show a statistically significant relationship with resistance to treatment of in-hospital malaria. The univariate analysis showed a high chance of resistance in patients treated with some drugs, including one of the antimalarials used, which was also confirmed by the multivariate analysis (Table 2). This data supports the idea that unfortunately, in recent decades, partial artemisinin resistance has emerged, probably due to the historical use of monotherapy with artemisinin and this may cause the use of pharmacological combinations to substantially reduce the parasite load in Africa, however, considerable levels of malaria remain high in African countries and if widespread, resistance to it will develop in Africa and there would be potential to seed malaria epidemics in African countries. 18 In any case, the pharmacological combinations found in the study were not defined as a combined treatment to combat resistance, but rather as alternatives to reduce certain symptoms presented by patients, which may suggest that non-resistant parasites have symptoms that may require the use of certain drugs when compared to resistant parasites. Should also be considered that the use of some drugs can potentiate or inhibit the actions of antimalarials, so there is a need for further studies to assess the impact of data related to pharmacological procedures.
Some potential limitations should be considered when analyzing these results. One of the limitations of this study was the fact that the study was not based on antimalarial resistance markers, in addition, we did not assess how sociodemographic data and a combination of drugs and non-pharmacological procedures can impact resistance to treatment of malaria. Despite these limitations, our findings highlight potential characteristics related to antimalarial drug resistance in patients with malaria from Angola. The antimalarial resistance, and their relationship with other drugs used during the treatment of malaria in Angola, deserve further investigation.
Conclusion
Our findings suggested that sociodemographic, clinical, and pharmacological procedures could be related to antimalarial resistance among patients with malaria in Angola. However, the use of adjuvant drugs such as dipyrone, metoclopramide, ciprofloxacin, and diazepam during malaria treatment could reduce the chances of resistance to in-hospital treatment for malaria.
Footnotes
Acknowledgements
The authors would like to thank the patients for their participation and the management and the workers from the Josina Machel Hospital for institutional support. We are grateful for the logistic and scientific support given by the CISA/INIS researchers.
Funding:
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was financially supported by the International Society of Nephrology (ISN) in the program (Research and Prevention Program and Saving Young Lives, 2018.2).
Declaration of Conflicting Interests:
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Author Contributions
ENMS, CSS, JM: 1. Conception and design or analysis and interpretation of data; ENMS, JLFA, AKZ, and CSS: 2. Writing of the article or relevant critical review of the intellectual content; DVSB, CSS, JM, and ENMS: 3. Final approval of the version to be published; ENMS: 4. Be responsible for all aspects of the work to ensure the accuracy and completeness of any part of the work.
Availability of Data and Materials
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
Ethics Approval
The study was approved by the Human Research Ethics Committee of the Higher Institute of Health Sciences (Official Letter No. 755/GD/ISCISA/UAN/2018) and by the clinical directorate of Hospital Josina Machel—Maria Pia in Luanda (Official Letter No. 260/DPC/HJM/2018).
Informed Consent
All patients who agreed to participate have signed the informed consent form after being enlightened about the nature and objectives of the study.
