Abstract
Infective endocarditis is associated with significant clinical mortality and morbidity. With the increasing age of the general population and use of intra-cardiac devices it is occurring mainly in people over the age of 60 with male to female ratios ranging from 3:2 to 9. The predominant causative pathogens are staphylococci, and with the increase in the number of pathogens that are either resistant or exhibit reduced susceptibility the management of infective endocarditis is increasingly challenging. Recent guidelines have included the lipopeptide, daptomycin, in their recommendations. The body of evidence supporting the use of daptomycin in the treatment of infective endocarditis, including at doses above the licensed recommended dose of 6 mg/kg is increasing. The current review summarises the current recommendations for infective endocarditis and the place of daptomycin in its management.
Background
Infective endocarditis (IE), as defined by the European Society of Cardiology, is an ‘endovascular microbial infection of cardiovascular structures'. 1 IE is an uncommon infection of the heart that usually affects native heart valves but can also involves intra-cardiac devices. Infective endocarditis is a serious infection that was universally fatal prior to the development of antibiotics. Now it is managed with antibiotics, either alone or in combination with cardiac surgery.
More than half of the cases of IE in Europe and the United States now occur in patients over the age of 60. 2 In the majority of cases series, men predominate with male female ratios ranging from 3:2 to 9:1.2,3 Although a rare condition, it is associated with significant mortality of approximately 15% to 20% and one year mortality of 40%.1,2 Among survivors, morbidity can be significant with stroke, renal failure and secondary foci of infection commonly complicating infection. 1
Definition of infective endocarditis according to the proposed modified Duke criteria. a

Adapted from Li 72 ;
excludes single positive cultures for coagulase-negative staphylococci and organisms that do not cause endocarditis. With permission from Oxford University Press.
The predominant causative organisms worldwide are now staphylococci (Fig. 1), with Staphylococcus aureus accounting for 23% to 68% of all infections,1,2,7 although the aetiology of IE varies with clinical form with staphylococci ranging from 44% to 70% in nosocomial IE.1,8 “Viridans group” streptococci are now the second most common pathogen accounting for 17% of all infections,
1
and 17% to 26% of intracardiac device IE.
7
The aetiology of IE also varies by region. In Europe the incidence of S. aureus is lower than the United States, accounting for 28% and 43% of episodes, respectively.1,2
Organisms causing major clinical forms of endocarditis.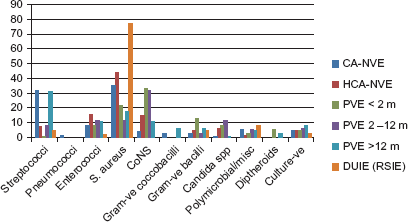
Global spread of antibiotic resistance has impacted on antibiotic therapy for IE. The 2004 recommendations for the treatment of staphylococcal endocarditis from the British Society for Antimicrobial Chemotherapy (BSAC) recommended flucloxacillin for meticillin sensitive S. aureus (MSSA) and vancomycin plus rifampicin or gentamicin or sodium fusidate for meticillin resistant S. aureus (MRSA) or patients with penicillin allergy. 9 In the presence of intracardiac prosthesis flucloxacillin plus rifampicin and/or gentamicin and/or sodium fusidate were recommended. Recently published guidelines from BSAC now recommend the following antibiotics for empiric treatment prior to obtaining blood culture results: amoxicillin or vancomycin for native valve endocarditis (NVE), vancomycin and gentamicin for NVE with severe sepsis or vancomycin plus meropenem for NVE, severe sepsis and risk factors for multiresistant Enterobacteriaceae or Pseudomonas, and Vancomycin plus gentamicin and rifampicin for prosthetic valve endocarditis (PVE) pending blood cultures or with negative blood cultures. 10 Treatment recommendations for staphylococcal endocarditis no longer include sodium fusidate. 10 Daptomycin (6 mg/kg q24 h iv) has been included for the treatment of NVE Meticillin-resistant, vancomycin-resistant (Table 2) summarises the BSAC 2011 recommendations for the treatment of staphylococcal endocarditis.
BSAC treatment recommendations for the treatment of staphylococcal endocarditis. 10

Indications for infective endocarditis prophylaxis. 73

Literature Search
A literature search was conducted in PubMed on 1st November 2011. Search terms (MeSH or text word) were: daptomycin OR glycolipopeptide AND endocarditis OR endocarditis, bacterial OR endocarditis, subacute bacterial. The search was limited to human and English language publications.
Daptomycin in the treatment of IE
Daptomycin is a lipopeptide antibiotic acting largely, and unusually, through membrane depolarization. 18 It is bactericidal and active against Gram-positive bacteria. The activity of daptomycin is concentration-dependent with a prolonged post-antibiotic effect. It has linear pharmacokinetics, with a half-life of 8–9 h, the primary route of excretion is renal, it exhibits serum protein binding of approximately 92% and there is no interaction with the P450 cytochrome. The mechanism of action and in vitro activity of daptomycin have been widely reviewed in the literature.19–26
Daptomycin was initially approved by the FDA in 2003 with indications of S. aureus bacteraemia, including right-sided IE caused by MSSA and MRSA, added in 2006. It was approved by the European Medicines Agency (EMEA) in 2007 at a dose of 6 mg/kg for bacteremia including right-sided endocarditis due to S. aureus. 27 In the initial registration study by Fowler et al comparing daptomycin with standard therapy, 28 75% (90/120) of patients treated with daptomycin met the modified DUKE criteria for definite or possible endocarditis. Daptomycin was shown to be non-inferior to standard therapy patients with uncomplicated right-sided endocarditis and MSSA infections (bacteraemia and endocarditis). It has been noted that, with the increase in treatment failures in patients with MSSA treated with vancomycin, daptomycin may provide a useful empiric therapeutic option pending susceptibility information. 29
Studies and case reports of the use of daptomycin in infective endocarditis.
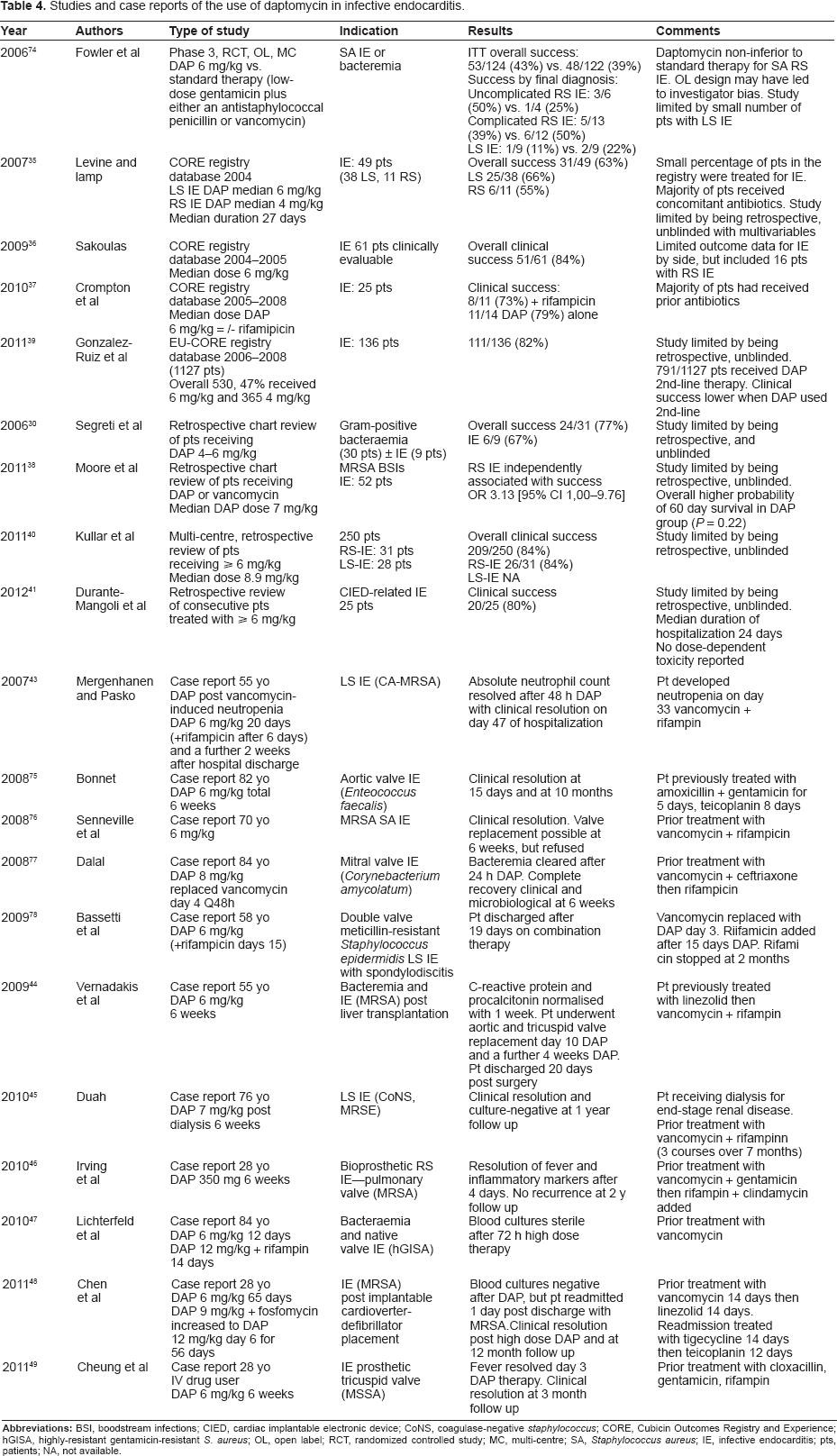
Data for daptomycin use in left-sided endocarditis, pacemaker lead infection and prosthetic valve endocarditis are limited. The study by Fowler et al included a small number of patients with left-sided endocarditis treated with daptomycin after a protocol amendment. 28 The success rate in evaluable patients was 11% (1 of 9) for daptomycin and 22% (2 of 9) for the dual therapy comparator using the strict definitions required for success 6 weeks after completed therapy. Data on 49 patients with endocarditis treated with daptomycin from the Cubicin Outcomes Registry (CORE®) database represents the largest published series to date and 38 patients with left-sided or bilateral endocarditis with a documented cure of 37% (18/49). 35 2004–2005 from the CORE database included 87 patients with endocarditis. 36 The median initial dose was 6 mg/kg and clinical outcomes were available for 61 patients. Overall clinical success in this series was 84% (51/61), and included patients with left-, right- or both sided IE. Data from CORE published in 2010 included 25 IE patients, of these 6 had vancomycin MICs ≥ 2 mg/L. 37 The clinical success rates with or without rifampicin in this later report were 73% (8/11) and 79% (11/14), respectively. More recently, a case control study of daptomycin versus vancomycin for bloodstream infections with high vancomycin MICs has been published. 38 A total of 118 vancomycin-treated patients were compared with 59 daptomycin treated subjects, of whom 11 had righ-sided IE and 6 left-sided. Right-sided endocarditis was independently associated with clinical success (OR, 0.07 [95% CI, 0.01–0.83].
Data from the European CORE (EU-CORE) registry (2006–2008) includes 136 patients with endocarditis, of whom 65% received doses of 6 mg/kg. 39 Of these, 111 patients were recorded as having a successful clinical outcome and 8 as clinical failures. Of note in this series is that the majority of patients (70%) had received prior antibiotic therapy, with treatment failure being recorded and the most common reason for initiating daptomycin.
A large retrospective review of high dose daptomycin (>8 mg/kg) from 5 American institutions included 59 patients (23.6%) with endocarditis. 40 In this series the overall clinical and microbiological success were reported as 83.6% (209/250) and 80.3% (175/218), respectively. High-dose daptomycin (>6 mg/kg and 10 mg/kg q24) has also been reported in a series of 25 patients with cardiac implantable electronic device-related endocarditis (CIED) with an overall clinical success rate of 80% and microbiological success of 92%. 41 18 of the 25 patients had been treated with prior antibiotics. These data highlight the need for comparative studies of different doses and schedules.
Bassetti et al reported the successful outcome in a patient with meticillin-resistant S. epidermidis (MRSE) double-valve left-sided endocarditis and spondylodiscitis treated for 12 weeks with daptomycin 6 mg/kg (in combination with rifampicin for 2 months) after initial treatment for 3 days with vancomycin. 42 The patient was managed for 19 days in hospital and then discharged with continued treatment in an outpatient regimen. After 4 weeks of therapy TEE showed no vegetations. Rifampicin was stopped after 2 months therapy because of gastric and liver toxicity, but no adverse events or elevations in creatinine phosphokinase (CPK) were associated with this prolonged course of daptomycin. Mergenhagen and Pasko reported a positive outcome in a patient treated with daptomycin 6 mg/kg with left-sided endocarditis associated with MRSA subsequent to vancomycin-induced neutropaenia. 43
Other case reports of the use of daptomycin include, successful salvage therapy in a patient with persistent MRSA bacteraemia and endocarditis following orthoptic liver transplantation, following treatment failure with linezolid followed by vancomycin/rifampicin combination therapy 44 and successful treatment of meticillin-resistant Staphylococcus epidermidis native valve endocarditis with 7 mg/kg of daptomycin 3 times/week for 6 weeks post treatment relapse following vancomycin therapy (despite the MIC remaining stable at 2 mg/L). 45 Clinical success without surgical intervention in the management of S. aureus endocarditis on a bovine pericardial pulmonary valve with no recurrence at 2-year follow up was also recently reported. 46 Successful treatment with high-dose daptomycin (6 mg/kg increased on day 12 to12 mg/kg with rifampicin 300 mg q12 added) of IE caused by S. aureus with reduced susceptibility to glycopeptides and loss of susceptibility to daptomycin during therapy was also recently reported. 47 This case report also highlights the need to consider alternative therapeutic options in serious infections caused by non- or reduced susceptibility to conventional therapy. The addition of fosfomycin (6 mg q4) to high dose daptomycin (9 mg/kg) in a patient with implantable cardioverter-defibrillator (ICD) device-related endocarditis complicated with osteomyelitis caused by daptomycin-nonsusceptible (DNS) MRSA 48 and vegetations refractory to standard therapy 49 have also been reported, 48 suggesting that daptomycin may be an alternative agent in these difficult to manage patients where surgical intervention is not an option. The addition of daptomycin to rifampicin has also been shown to be effective at reducing the subpopulation of bacteria that develop rifamipicin resistance.50,51
In vitro pharmacodynamic modelling with simulated endocarditis vegetations has demonstrated rapid bactericidal activity with daptomycin-susceptible strains, with more rapid killing at a dose of 10 mg/kg. 52 These authors reported an improvement in activity with combination regimens against some, but not all, isolates, and suggest that in the presence of strains with reduced susceptibility high-dose daptomycin or combination therapy may be a reasonable treatment option. Cunha et al have reported a successful clinical outcome using high-dose (12 mg/kg) daptomycin after continuing bacteraemia and mitral valve IE following 10 days of daptomycin (6 mg/kg) in combination with linezolid (600 mg 12 h). Use of the higher dose resulted in a rapid clinical improvement and negative blood cultures within 24 hours of administration. High-dose daptomycin was continued on a CoPAT basis for a total of 8 weeks and no adverse events were reported. 53 However, the same group have more recently reported a treatment failure using high-dose daptomycin in a patient IE who had been previously treated with vancomycin, 54 highlighting the need for caution interpreting the limited data pertaining to refractory IE.
Patients with IE often require protracted courses of antibiotics, and the once daily dosage schedule and a 2 minute bolus injection make daptomycin an attractive option for use in community-based outpatient antimicrobial therapy (CoPAT) programmes. In a retrospective analysis of the study by Fowler et al, which included patients with right- and left-sided endocarditis, 28 51.5% (103/200) patients received part of their treatment in the community setting. 55 These patients received daptomycin for a mean of 25.9 days (range 11–74), of which 15.2 days were administered in the community setting, and demonstrated that the use of daptomycin in the community was an appropriate treatment option.
Cost Effectiveness
Bhavnani et al
56
demonstrated median cost effectiveness ratios of daptomycin compared with vancomycin-gentamicin in terms of:
drug acquisition costs plus cost of therapy for treatment failures and adverse events (a) plus (b) plus hospital bed days costs.
Although the drug acquisition costs of a typical vancomycin-gentamicin course of therapy were much lower than the costs for daptomycin, $560 vs. $4082, respectively (Table 5), the vancomycin-gentamicin regimen was not more cost-effective when the total cost of therapy (including therapeutic drug monitoring), treatment failures and adverse events ($11635 vs. $4582, respectively) and inpatient care was considered ($26073 vs. $23639, respectively). The two main reasons for this finding were differences in response rates and costs associated with treatment failures, which favoured daptomycin over the vancomycin-gentamicin regimen. Clinical response rates favoured daptomycin 44% vs. 32%). The costs associated with vancomycin plus gentamicin treatment failure showed a trend toward a higher cost for follow-up and a longer duration of follow-up therapy. Other contributing factors were the fact that vancomycin is typically administered twice daily vs. daptomyicin once per day and the lack of the need for therapeutic drug monitoring. In addition, there were a higher number of renal adverse events requiring management in the vancomycin-gentamicin group (4 of 9) than the daptomycin group (0 of 6).
Costs of daptomycin versus standard therapy for patients with MRSA bacteremia and/or infective endocarditis.
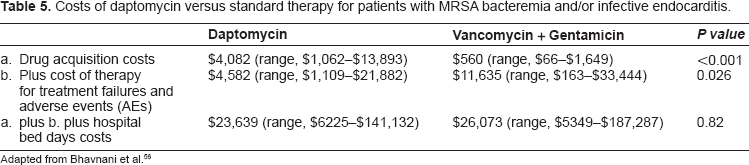
Adapted from Bhavnani et al. 56
Treatment failures with daptomycin
There have been a limited number of reports of treatment failure with daptomycin in IE. The clinical significance reduced susceptibility to daptomycin is, as yet, unknown, although a recent report of daptomycin resistance and treatment failure in a patient with MRSA mitral valve IE, 54 treatment failure in a patient with left-sided IE, 57 relapse after treatment with daptomycin 58 and the report of an MRSA IE caused by the USA300 strain where the MIC of daptomycin increased from 1 to 8 μg/mL 59 highlight the need for vigilance in these cases. Daptomycin demonstrates rapid concentration-dependent activity against Gram-positive bacteria and has also been shown to demonstrate a reduction in biofilm production in vitro for S. aureus and S. epidermidis. 18 However, where resistance has developed during treatment, 60 there have been reports of linked increases in vancomycin and daptomycin MICs.54,58,61,62 Hidron et al reported a clinical and bacteriological failure in a patient with E. faecalis mitral valve IE, which had developed resistance during vancomycin therapy. 63 Possible explanations for the loss of daptomycin activity are an increase in cell wall thickness induced by vancomycin, the effect of daptomycin on peptidoglycan synthesis 18 or decreased daptomycin binding. 64 Currently there are no studies on the mechanisms of daptomycin resistance in enterococcal species. 63
Safety
To date, clinical trials, retrospective reviews, case reports and in vitro simulation models have documented the safety and tolerability of daptomycin even at doses in excess of those licensed by the authorities and for prolonged durations.39,65–69 Retrospective analyses of data from the phase 3 studies of daptomycin in patients with or without IE reported that a daptomycin C(min) of 24.3 mg/L was associated with an increased probability (0 to 0.01) of elevated creatine phosphokinase (CPK) levels after 7 days treatment and recommend monitoring CPK levels 3 to 5 days after initiating therapy. 70 However, these authors note that a proportion of these CPK elevations maybe related to other factors such as the use of statins, diabetes mellitus or surgical procedures.
Discussion
Since the publication of the pivotal Phase 3 registration study 28 which reported non-inferiority of daptomycin versus standard therapy in patients with bacteraemia or endocarditis there have been a number of publications from the Cubicin Outcomes Registry and Experience database.35–37,39 Although these reports are limited by the fact that they are uncontrolled and retrospective, they do present real world clinical experience for IE where it is difficult to conduct randomized controlled studies and which provide useful addition information.
A number of the case studies included in the current review illustrate the difficulty successfully treating IE, particularly in patients with multiple co-morbidities, 33 where salvage therapy is impossible or where intolerance to conventional antibiotics is a problem.31,32 The published case reports also provide support for the use of daptomycin where patients have been treated unsuccessfully (sometimes for prolonged periods and with resistant organisms 45 ) with vancomycin ± rifampin. Although the data in this difficult to treat group of patients is limited, it would appear that daptomycin may be a useful therapeutic alternative in complex cases of IE or those caused by organisms with high vancomycin MICs or resistant organisms. Unfortunately it is unlikely that controlled clinical trials will be conducted to guide therapy in the foreseeable future, making the publication of retrospective studies and case reports an important addition to the available data.
Indeed, it has been estimated that almost 25% of daptomycin use in the USA has been for the treatment of bacteraemia and endocarditis, most of which has been off label because of the dose used or the treatment of left-sided endocarditis. It is not know what level of use in the UK is currently off label. However, there may be patients with left-sided endocarditis or IE associated with resistant organisms who may benefit from treatment with daptomycin. 71 However, the apparent association between vancomycin MIC creep and reduced susceptibility to daptomycin57,58,61,62 needs to be considered in the clinical setting, particularly before initiating daptomycin therapy in patients who have previously failed on vancomycin. Further clinical studies are needed to extend experience with daptomycin for infective endocarditis and its use at high doses that are currently outside the current licensed recommendations.
Conflict of Interest
Neither AG or AJ have any conflict of interest to declare. The authors confirm that they have complied with the ICMJE guidelines and have contributed equally to the review.
Funding
AGR has received fees from Novartis, Pfizer and Gilead for staff training, being a member of Advisory Boards and being a member of speakers panels. He also has received support to attend scientific conferences including accommodation and travel payments and a Novartis grant to support a research project. AJ works as an independent scientific consultant to a number of bodies and pharmaceutical companies including the British Society for Antimicrobial Chemotherapy, International Society for Chemotherapy, Becton Dickinson (Diagnostics), Novartis (Anti-Infectives), Astellas (Anti-Infectives), Smith and Nephew (Wound Care), Wyeth and Pfizer (Anti-infectives and Vaccines) and has received consultancy fees for the services provided.
Author Contributions
Conceived and designed the experiments: AG-R, AJ. Analysed the data: AG-R, AJ. Wrote the first draft of the manuscript: AG-R, AJ. Contributed to the writing of the manuscript: AG-R, AJ. Agree with manuscript results and conclusions: AG-R, AJ. Jointly developed the structure and arguments for the paper: AG-R, AJ. Made critical revisions and approved final version: AG-R, AJ. All authors reviewed and approved of the final manuscript.
Footnotes
As a requirement of publication author(s) have provided to the publisher signed confirmation of compliance with legal and ethical obligations including but not limited to the following: authorship and contributorship, conflicts of interest, privacy and confidentiality and (where applicable) protection of human and animal research subjects. The authors have read and confirmed their agreement with the ICMJE authorship and conflict of interest criteria. The authors have also confirmed that this article is unique and not under consideration or published in any other publication, and that they have permission from rights holders to reproduce any copyrighted material. Any disclosures are made in this section. The external blind peer reviewers report no conflicts of interest. Provenance: the authors were invited to submit this paper.
