Abstract
Introduction:
Multidrug-resistant
Methods:
A single-center retrospective observational study between January 2017 and December 2018 for patients who had been diagnosed with carbapenem-resistant
Results:
A total of 19 patients fit the inclusion criteria, with a median age was 57 years, and 53% were female. The types of infections were nosocomial pneumonia, acute bacterial skin, and skin structure infections; complicated intra-abdominal infections; and central line–associated bloodstream infections. All of the isolates were resistant to both meropenem and imipenem. The duration of therapy was variable (average of 14 days). At day 14 of starting ceftolozane-tazobactam, 18 of 19 patients had a resolution of signs and symptoms of the infection. Only 14 of 19 patients (74%) had proven microbiological eradication observed at the end of therapy. During therapy, there was no adverse event secondary to ceftolozane-tazobactam, and no
Conclusions:
Multidrug-resistant
Introduction
Hospital-acquired infections caused by resistant gram-negative bacteria have become a frequently encountered difficult clinical challenge. Those resistant organisms account for a significant percentage of hospital-acquired infections in many countries. Extended-spectrum β-lactamase (ESBL)-producing organisms have produced multiple hospital outbreaks and become a global health concern.1,2 Moreover, increasing rates of carbapenem-resistant gram-negative bacteria are noticed in health care settings, especially with the increase of carbapenems used as essential agents to treat resistant isolates.3,4 Because of multidrug resistance in gram-negative bacteria, the use of old abandoned antibiotics (ie, polymyxins) has been recalled by the World Health Organization
4
and reclassified as critically important for human medicine. Multidrug-resistant (MDR)
Materials and Methods
This is a single-center retrospective observational study of patients treated between January 2017 and December 2018 at King Abdul Aziz Medical City-Riyadh (KAMC-RD) in Saudi Arabia. Eligible patients were those who had been diagnosed with MDR
Results
A total of 19 patients with MDR
Demographics and clinical characteristics of 19 patients.
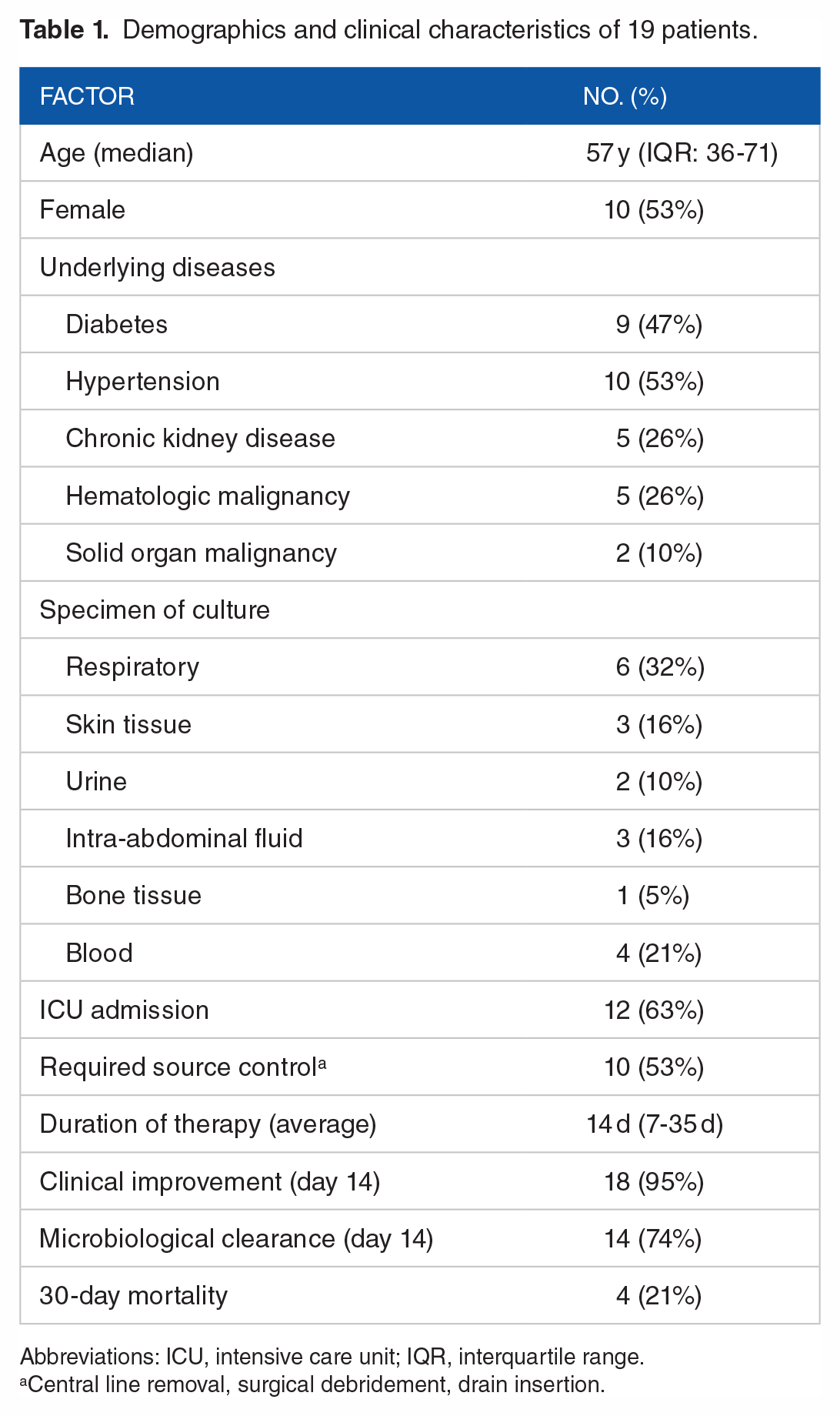
Abbreviations: ICU, intensive care unit; IQR, interquartile range.
Central line removal, surgical debridement, drain insertion.
Types of infection were nosocomial pneumonia (6/19, 32%), where half of them had ventilator-associated pneumonia, acute bacterial skin, and skin structure infections (3/19, 16%), and one of those had complicated bacteremia, pyelonephritis (2/19, 10%), complicated intra-abdominal infection (3/19, 16%), including cholangitis and intra-abdominal collection and bone infection (1, 5%). Central line–associated bloodstream infection was documented in 4 cases, and 1 had a complication with endocarditis.
Of 19 patients (63%), 12 were in intensive care unit (ICU) at the time of starting ceftolozane-tazobactam (Table 1). The rest of the patients did not require ICU admission during their hospitalization.
All the isolated
Ceftolozane-tazobactam was used as a single therapy in 11 patients, whereas the combination with other antibiotics was done in 8 patients with no apparent impact on the patient’s outcome. Colistin, aztreonam, and amikacin were the antibiotics used as accompanying antipseudomonas agents. The average time between obtaining the culture and starting ceftolozane-tazobactam was 5 days. The antibiotic duration of therapy was widely variable between patients, ranging from 7 days to more than a month, with an average of 14 days. Only 2 patients required more than 3 weeks of therapy, and it was secondary to poor source control for osteomyelitis and persistent liver abscesses.
Among subjects with a creatinine clearance >50 mL/min, the approved dose of 1.5 g every 8 hours was used in 8 cases, and 2 patients received 3 g every 8 hours as decided by the treating physician. Ceftolozane-tazobactam dosing among the remaining patients was calculated according to the changes in creatinine clearance and requirement of hemodialysis, ranged from 1.5 to 0.375 g every 8 hours, following the recommended protocol.
At day 14 of starting ceftolozane-tazobactam, 18 of 19 patients had a resolution of signs and symptoms of the infection (as reported by treating physicians). Only 14 patients (74%) had proven microbiological eradication observed at the end of therapy. Of the 12 patients, 7 patients who were in ICU at the initiation of treatment were discharged to the medical/surgical ward, whereas the remaining 5 patients stayed for other reasons. During therapy, there was no adverse event secondary to ceftolozane-tazobactam in our cohort, and no cases with
The 30-day mortality rate was 21% (4/19). Two of those deaths were related to the primary infection. Although 1 of those 2 had shown a clinical improvement initially, the cultures remained positive due to lack of source control (Table 2). The other 2 patients deteriorated later and died due to complications of catastrophic antiphospholipid syndrome and severe aspiration.
Clinical and microbiological descriptions of cases associated with treatment failure.
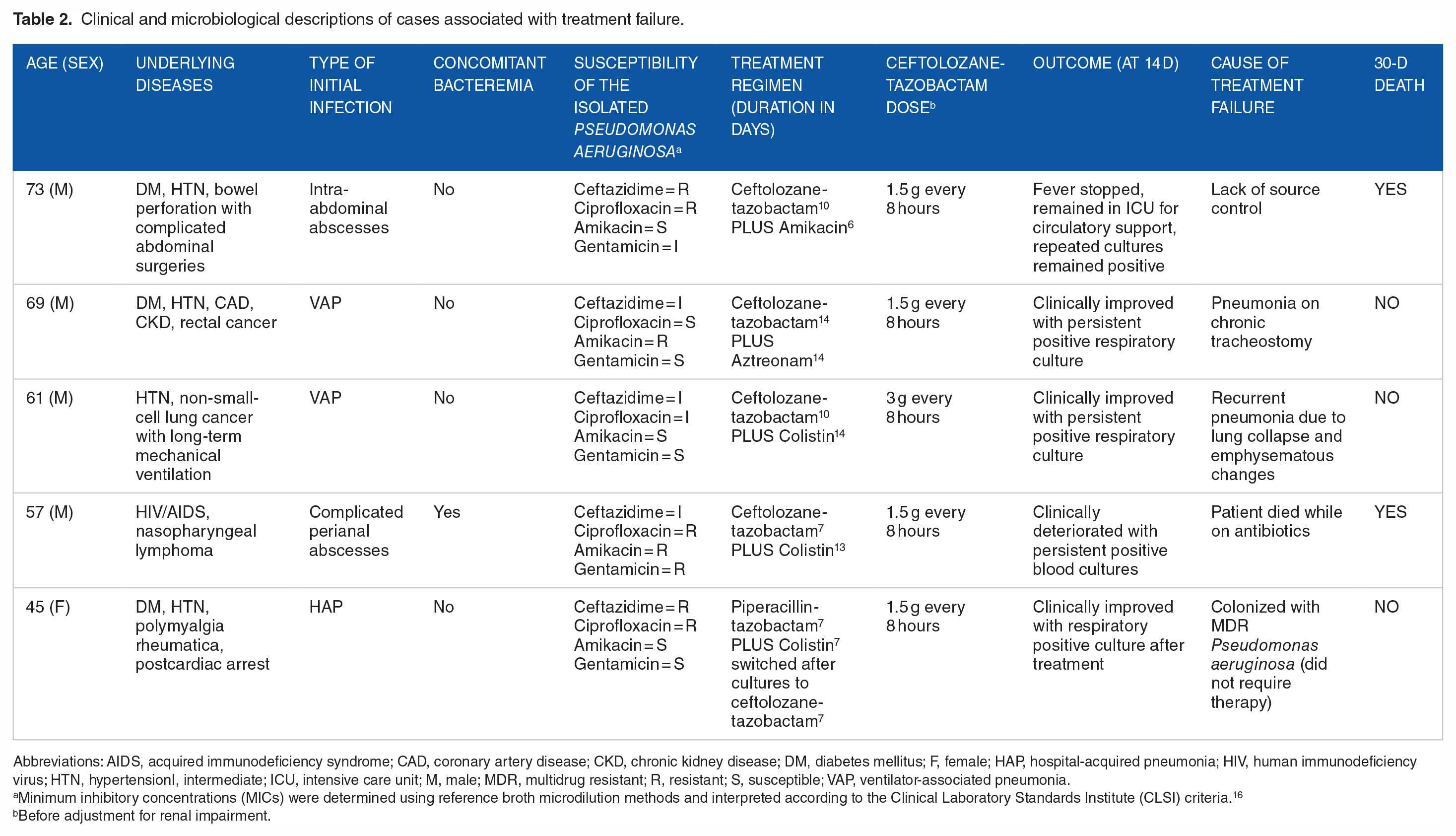
Abbreviations: AIDS, acquired immunodeficiency syndrome; CAD, coronary artery disease; CKD, chronic kidney disease; DM, diabetes mellitus; F, female; HAP, hospital-acquired pneumonia; HIV, human immunodeficiency virus; HTN, hypertensionI, intermediate; ICU, intensive care unit; M, male; MDR, multidrug resistant; R, resistant; S, susceptible; VAP, ventilator-associated pneumonia.
Minimum inhibitory concentrations (MICs) were determined using reference broth microdilution methods and interpreted according to the Clinical Laboratory Standards Institute (CLSI) criteria. 16
Before adjustment for renal impairment.
Discussion
In this report, we are presenting a real-world experience with ceftolozane-tazobactam treatment of MDR
It is difficult to evaluate the actual effects of the use of concomitant intravenous antibiotics or high-dose ceftolozane-tazobactam due to the small number of patients. Recent studies are now recommending using high-dose ceftolozane-tazobactam for the treatment of hospital-acquired bacterial pneumonia and ventilator-associated bacterial pneumonia in patients ⩾18 years old. Ceftolozane-tazobactam monotherapy may be sufficient for the treatment of
The median time from culture collection to the start of ceftolozane-tazobactam dosing in our study was 5 days. This indicates that a de-escalation approach was used in these cases, which is commonly recommended by clinical guidelines for severe infections. The emergence of resistance after courses of ceftolozane-tazobactam has been described in some reports highlighting the importance of setting strict standards for the drug’s use and the continued urgency for new antibiotics. 18
The limitations of this study include the retrospective nature of the data and dependence on documentation for assessing clinical outcomes via medical records review. Moreover, the decision to use ceftolozane-tazobactam as well as different doses and antibiotic combinations was at the consideration of the treating clinicians rather than prespecified in a protocol. Finally, the presence of different types of infections also limited the analysis. However, considering the severity and high mortality rates of such infections, it is necessary to have clinical data on the efficiency of new antibiotics with potent activity against resistant
In conclusion, our results suggest that ceftolozane-tazobactam shows an anticipated great advantage for treating severe infections caused by carbapenem-resistant
Footnotes
Funding:
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests:
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Author Contributions
MB contributed to conception or design of the work, data analysis and interpretation, drafting the article, and final approval of the version to be published. AAh contributed to conception or design of the work, data collection, drafting the article, and critical revision of the article. AAla contributed to conception or design of the work, data collection, and critical revision of the article. EM contributed to conception or design of the work, data collection, data analysis and interpretation, drafting the article, and critical revision of the article. LA contributed to data collection, data analysis and interpretation, drafting the article, and critical revision of the article. AbA contributed to data collection, data analysis and interpretation, and critical revision of the article. SA and MA contributed to data analysis and interpretation, drafting the article, and critical revision of the article. BA contributed to data collection, drafting the article, and critical revision of the article. AAlo contributed to conception or design of the work, data analysis and interpretation, critical revision of the article, and final approval of the version to be published.
Ethical Approval
This study was conducted in compliance with all the applicable institutional ethical guidelines in King Abdullah International Medical Research Center (KAIMRC), and consent was not required as there was no direct contact with patients.
