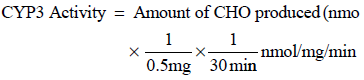Abstract
It has been well reported that complementary medicines can significantly alter the way the body handles conventional drugs, leading to potential fatal herb-drug interactions. The aim of the present study was to investigate the molecular mechanism of drug interactions involving St John's wort (SJW) (Hypericum perforatum L), a popular herbal medicine widely used for depression, particularly examining changes in the expression of cytochrome P450 CYP3A, the most abundant drug metabolising CYP enzymes in man.
Eighteen Sprague-Dawley (SD) rats were assigned randomly into 3 groups (n = 6/group): control, low dose and high dose (500 and 1000 mg/kg/day of SJW, equal to 1500 and 3000 µg/kg/day of Hypericin). Each group was treated with SJW or control preparation, by gastric gavage, for 14 consecutive days. Liver and intestinal CYP3A activity and protein and mRNA levels, from five segments of the intestine, were examined using CYP3A-dependent erythromycin N-demethylation activity assay, quantitative immuno-blotting and real-time RT-PCR.
Increase in CYP3A activity and protein level by SJW was observed in some intestinal regions, with a 3.0 fold increase in liver CYP3A activity and a 10.6 fold increase in liver CYP3A1 mRNA (p < 0.05) in a dose dependent manner. The results suggested that up regulation of liver CYP3A mRNA and differential induction of intestinal CYP3A play an important role in the molecular mechanism of herb-drug interactions.
Keywords
Introduction
Complementary medicines are widely available in the community and their uses have grown dramatically in the Western world. It has been well reported that several complementary medicines significantly alter the way the body handles numerous commonly used conventional drugs, leading to potential fatal herb-drug interactions. Knowledge of the molecular mechanisms by which complementary medicines cause such changes is therefore fundamental. It has been suggested that alteration in the expression of cytochrome P450 CYP3A, the main phase I drug metabolising enzymes in man, is one of main mechanisms of concern (Izzo, 2004; Pal and Mitra, 2006; Schulz, 2006).
St John's wort (SJW) is an increasingly popular herbal product, which is often used as an alternative to conventional antidepressants for mild to moderate depression (Josey and Tackett, 1999). Since many different preparations of SJW are available over-the-counter and may be taken without medical consultation, there are significant concerns regarding interactions linked to the use of SJW products in combination with conventional medicines (Henderson et al. 2002; Martin-Facklam et al. 2004; Mai et al. 2004). Commercial SJW preparations have been reported to contain at least nine groups of compounds that contribute to its pharmacological effect. The constituent hypericin has been reported to be the most likely candidate to be responsible for the antidepressant activity and is commonly used to standardise the various SJW products (Henderson et al. 2002). While hypericin is the component of SJW primarily believed to be responsible for its antidepressant activity, hyperforin is the most likely to cause of CYP3A inhibition and induction (Obach, 2000).
It has been extensively reported that SJW alters metabolic disposition and efficacy of many conventional drugs, in some cases resulting in potentially fatal adverse reactions (Ioannides, 2002; Izzo, 2004). These drugs include digoxin, warfarin, cyclosporine, theophylline, alprazolam, simvastatin, tacrolimus, phenprocoumon, irinotecan, methadone, HIV protease inhibitors, amitriptyline and oral contraceptives (Martin-Facklam et al. 2004; Izzo, 2004). Intake of SJW has also been found to decrease oral absorption and/or enhance metabolism of the above listed drugs, leading to a decrease in drug efficacy (Izzo, 2004). The mechanism by which SJW causes such potential deleterious drug interactions is currently not fully understood. It has been suggested that these interactions were partly due to induction of intestinal and hepatic cytochrome P450 CYP3A enzymes, resulting in diminished bioavailability of co-administered drugs (Durr et al. 2000; Bray et al. 2002; Izzo, 2004; Komoroski et al. 2004; Zhou et al. 2005).
Cytochrome P450s are microsomal enzymes that exist mainly in the liver, intestine, lungs and kidneys (Krishna and Klotz, 1994). Recently, the intestine has been recognised as a potential site for drug metabolism due to early exposure to orally administered drugs (Emoto et al. 2000). CYP3A is the most abundant CYP enzyme in man that is responsible for the metabolism of over 70 widely used drugs. CYP3A accounts for the majority of CYP in the intestine and for 30% of total CYP content in the liver (Watkins et al. 1987; Shimada et al. 1994). Changes in CYP3A expression can lead to significant effects on the absorption and clearance of many therapeutic drugs that are commonly used.
Much research interest has focussed on altered expression of liver and intestinal CYP3A protein by SJW. However, no studies have looked at changes in liver CYP3A by SJW at the mRNA level or along the entire length of the intestine. The aim of the current study was to establish whether the induction of CYP3A by SJW is apparent along the entire length of the rat intestine or whether there is differential induction in various segments of the intestine. The study also examined whether there is up regulation of liver CYP3A mRNA by SJW. The information has significant implications for drugs that are metabolized by the liver and drugs that may be absorbed in different regions of the intestine, perhaps due to differences in solubility or release from the dosage form. The information is also important for making responsible decisions regarding the use of SJW in combination with other conventional medicines.
Materials and Methods
Chemicals
Saint John's Wort (Kira® LI 160® special extract, each tablet containing 300 mg Hypericum perforatum L., standardized to contain 900 µg Hypericin, Lot # 79090300), currently over the counter, was obtained from Lichtwer Pharma AG, Berlin, Germany. Goat anti-rabbit CYP3A antibody was supplied by Flinders University (Bedford Park, SA, Australia) and donkey anti-goat HRP conjugated secondary antibody was from Santa Cruz Biotechnology (Clayton, VIC, Australia). Biotrace NT pure nitrocellulose membrane was purchased from Pall Corporation (Lane Cove, NSW, Australia) and Bench mark prestained protein ladder was from Invitrogen (Mount Waverley, VIC, Australia). Erythromycins, NADP, isocitric acid, isocitric dehydrogenase and magnesium chloride were obtained from Sigma-Aldrich (Castle Hill, NSW, Australia). Milli-Q water was generated using a Milli-Q water purifier system, Millipore/Waters, USA. Other chemical and reagents used were at least of analytical grade. SuperScript™ III Platinum® One-Step Quantitative RT-PCR systems were obtained from Invitrogen Life Technologies (Melbourne, VIC, Australia). RNeasy Micro kit was obtained from Qiagen Pty. Ltd. (Clifton Hill, VIC, Australia), 100-bp DNA ladder from Gene Work Pty. Ltd. (Adelaide, SA, Australia), and other molecular reagents were from Progen Industries Ltd. (Brisbane, QLD, Australia).
Animal Study and Sjw Oral dosing
This study was approved by the Animal Ethics Committee of the Institute of Medical and Veterinary Science (IMVS), South Australia. Male Sprague Dawley rats (weighing 300–380 g) were obtained from the IMVS, Animal Resource Centre, Gilles Plains, South Australia. The rats were maintained in the Animal Facility of the University of South Australia under controlled temperature and humidity conditions, with a 12 hour light/dark cycle. Animals had free access to food and water at all times.
A total of 18 rats were randomly assigned into one of two treatment groups or the control group (n = 6/group). The low dose group received 500 mg/kg SJW (equal to 1500 µg/kg/day of Hypericin), the high dose group received 1000 mg/kg SJW (equal to 3000 µg/kg/day of Hypericin) and the control group received the same volume of control preparation. The doses were reported previously to induce intestinal and hepatic CYP3A activity (Durr et al. 2000). A crude suspension of SJW was made by finely grinding the SJW tablets, using a glass pestle and mortar, and then dampening the resultant powder with ethanol before diluting to the required volume with Milli-Q water. The ethanol content in each suspension was 16 µl/ml. The control preparation was made using equivalent volume of ethanol in Milli-Q water. All preparations were freshly made, protected from light and used within 48 hours. Prior to dosing, the rats were weighed and SJW doses were administered by gastric gavage once daily for 14 consecutive days, with 1 ml of preparation per 100 g body weights. On day 15 of the study, the rats were sacrificed and the liver, duodenum, jejunum, ileum, caecum and rectum were removed. Each tissue was frozen in liquid nitrogen and stored at -80 °C until required.
Microsomal Preparation
Each tissue (0.5 g) from the intestinal segments was rinsed with 0.9% sodium chloride, cut into small pieces and placed into 10 volumes (ml/g) of ice-cold phosphate KCl buffer, pH 7.4 (NaH2PO4.2H2O 0.1 M, KCl 1.15%, phenylmethanesulfonyl fluoride (PMSF) 3 mM, EDTA 5 mM). For the liver, the tissue portion was placed directly into 10 volumes (ml/g) of the ice-cold phosphate KCl buffer. The tissues were homogenised, centrifuged at 9,000 xg for 20 min at 4 °C, and the resulting supernatant was ultracentrifuged at 108,000 xg for 1 hr at 4 °C. The pellet was resuspended in phosphate KCl buffer, homogenised and ultracentrifuged as described above. The resultant pellet was then resuspended in glycerol phosphate buffer (NaH2PO4.2H2O 0.1 M, glycerol 20%, PMSF 3 mM, EDTA 5 mM) and stored at -80 °C until required. Microsomal protein concentration was determined by the method of Lowry et al. (1951), using bovine serum albumin (BSA) as the standard.
CYP3A-Dependent Erythromycin n-Demethylation Activity assay
CYP3A-dependent erythromycin N-demethylation activity assay was performed using a modified method described by Kitada et al. (1988). Other conventional methods for studying CYP3A activity include midazolam 1“-hydroxylation or testosterone 6-β hydroxylation activities. In brief, incubation samples were prepared on ice and contained 0.5 mg of microsomal protein, 200 µl of NADPH generating system (MgCl2 84 mM (initial concentration), isocitric acid 24 mM, isocitric dehydrogenase 5 I.U. and NADP 6.7 mM), erythromycin 0.4 mM and 0.1 M phosphate buffer, pH 7.4 in a final volume of 1 ml. After incubation at 37 °C for 30 min in a shaking water bath, 0.5 ml of trichloroacetic acid 1.5 M was added to the samples. The samples were then centrifuged at 3000 xg for 15 min. To 1 ml of the collected supernatant 0.5 ml of NASH reagent (ammonium acetate 5.8 M, glacial acetic acid 0.9% and acetyl acetone 0.6%) was added and the sample contents were then incubated at 60 °C for 10 min in a water bath. The absorbance of each sample was then read at 412 nm using the Shimazu UV-1601 spectrophotometer. CYP3A activity was calculated using the following formula.
Formaldehyde (CHO) concentration range from 0 to 100 nmol/ml of was used as the standard curve.
Quantitative Western Blot analyses
To detect CYP3A proteins from each intestinal segment, sodium dodecylsulfate-polyacrylamide gel electrophoresis (SDS-PAGE) was performed as described by Laemmli (1970), using an 8% running gel and 5% stacking gel. Prior to electrophoresis, protein samples (100 µg) were solubilised in solubilisation buffer and heated at 100 °C for 7.5 min and then loaded onto the wells. The gel was run at 200V for 40 min, the separated proteins were then transferred onto a nitrocellulose membrane (Gelman Laboratory, NSW, Australia) at 250V overnight. The membrane was rinsed twice in tris buffered saline (TBS), twice in tween tris buffered saline (TTBS–-0.05% v/v Tween-20 in TBS) and then blocked using a 12% blocking solution at 37 °C for 40 min. After rinsing in TBS and TTBS, the membrane was incubated with goat anti-rabbit CYP3A primary antibody (in 1:1000 dilution) at 37 °C overnight. This primary antibody has been used previously in our laboratories and found to recognise most major CYP3A isoforms in most species, including CYP3A1 and CYP3A2 in rat (McKinnon et al. 1995). The membrane was then incubated in 1:25000 dilution of horseradish peroxidase (HRP) conjugated donkey anti-goat CYP3A secondary antibody at 37 °C for 40 min, after rinsed twice in TTBS the next day. Immuno-reactive CYP3A proteins were detected using a HRP colour development reagent (0.05% w/v 3,3-diaminobenzidine (DAB) and 0.05% v/v H2O2 in 20 mM imidazole buffer, pH 7.4). The relative intensity of CYP3A immunoreactive bands was quantified by densitometry scanning using the Quantity One (version 4.5.1, Bio-Rad, CA, USA) software.
Real-Time RT-PCR Quantification of CYP3A1 and CYP3A2 mRNA
Total RNA was purified using the RNeasy Micro kit in accordance with the manufacturer's instructions. Ribosome 18S RNA, CYP3A1 and CYP3A2 forward and reverse primers, CYP3A1 and CYP3A2 probes were custom made by Sigma Genosys Pty. Ltd (Castle Hill, NSW, Australia). 18S probe was purchased from Biosearch Technologies Inc. (Novato, CA, USA). The CYP3A1 and CYP3A2 probes were dual labelled at the 5'–-end with reporter molecular 6-carboxyfluorescein (FAM) and at the 3'-end with Back Hole Quencher 1 (BH1). Details of primers and probes are described in Table 1. Probes for CYP3A1, CYP3A2 and 18S ribosomal RNA were validated by amplifier liver mRNA with the following PCR conditions: 50 °C for 15 min, at 95 °C for 2 min, followed by 40 cycles of 95 °C for 15 min, and 60 °C for 1 hr. Multiplex amplification of amplicons CYP3A and 18S RNA were performed in a 25 µl reaction mixture, containing 10 µl purified water, 12.5 µl 2 x reaction mix, 0.25 µl SupperScript III RT Platinum® Tag mix, 0.5 µl forward and reverse primers 500 (nM), 0.2 µl probe for the respective target gene (100 nM), 0.25 µl primers (500 nM) and 0.1 µl probe (100 nM) for the 18S RNA as an endogenous reference.
Sequences of CYP3A1/CYP3A2 and 18S primers and probes.

Experiments were performed using a SuperScript™ III Reverse Transcriptase plus Platinum Tag DNA Polymerase kit in a 50 µl tubes on a Rotor-Gene 300™ and analysed using a Rotor-Gene 300™ operating software version 6 (Corbett Research, NSW, Australia) and Microsoft excel. The real-time PCR conditions are specified in Table 2.
Cycling parameters for real-time RT-PCR reactions.

Data were quantified by the 2-ΔΔCt method. The derivation of quantification by real-time PCR and the 2-ΔΔCt method has previously been described (Heid et al. 1996; Livak and Schmittgen, 2001). The Ct value is defined as the number of PCR cycles required for the fluorescence signal to exceed the detection threshold value (Hamalainen et al. 2001). The lower the Ct value, the more the fluorescence emitted, the higher the amount of starting mRNA in the sample tested (Mei et al. 2004). The following equation was employed to quantify the amount of message:
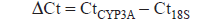
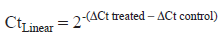
where,
CtCYP3A represents the Ct value of the target gene
Ct18S is the Ct value of housekeeping 18S ribosome RNA gene
CtLinear represents the fold change in mRNA expression between the control and SJW treated groups.
Statistical Analysis
Results from the control and each treatment group were represented as mean ± standard deviation (SD) of 6 animals per group. Statistical analysis was performed using a one way ANOVA by the Minitab (version 14) software, where p < 0.05 was considered to be significant.
For real-time RT-PCR analysis of CYP3A levels, one tail t-test was used to determine the changes in CYP3A1/CYP3A2 expression between the control and the treated samples, where p < 0.05 was considered to be significant.
Results
cyp3a-Dependent Erythromycin n-Demethylation Activity assay
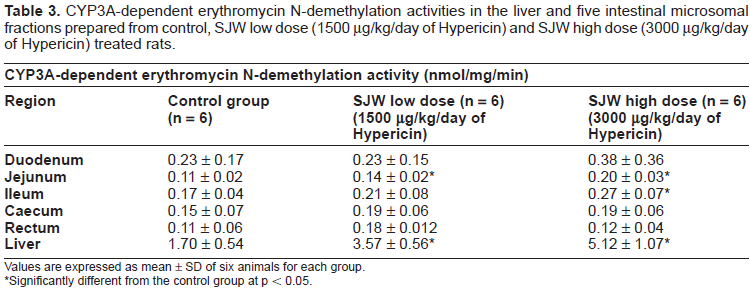
Data for CYP3A-dependent erythromycin N-demethylation activities in the liver and five intestinal microsomal fractions prepared from control and SJW treated rats are summarized in Table 3.
CYP3A-dependent erythromycin N-demethylation activities in the liver and five intestinal microsomal fractions prepared from control, SJW low dose (1500 µg/kg/day of Hypericin) and SJW high dose (3000 µg/kg/day of Hypericin) treated rats.
Values are expressed as mean ± SD of six animals for each group.
Significantly different from the control group at p < 0.05.
The results are expressed as the mean ± standard deviation (SD) of 6 animals per group. CYP3A-dependent erythromycin N-demethylation activity was higher in some segments of the intestine prepared from the SJW high dose treated group in comparison to the control group. In the jejunum from the SJW high dose treated group, erythromycin N-demethylation activity was 1.8 fold higher in comparison to the control (p < 0.05). In the ileum from SJW high dose treated group, erythromycin N-demethylation activity was 1.7 fold higher than that from the control (p < 0.05). In the duodenum, caecum and rectum from SJW high dose treated rats, erythromycin N-demethylation activities were 1.7, 1.3 and 1.1 fold higher than that from the controls however, these differences were not statistically significant (p > 0.05). No significant differences in CYP3A-dependent erythromycin N-demethylation activity were observed between the control and the SJW low dose treated groups, except for the jejunum (Table 3). To compare the induction of CYP3A by SJW in rat intestine with that in the liver and to investigate the impact of SJW exposure dose on hepatic CYP3A, we examined CYP3A-dependent erythromycin N-demethylation activity in the livers from the control and SJW low dose and high dose treated groups. Liver CYP3A-dependent erythromycin N-demethylation activity was found to be 2.1 and 3.0 fold higher in the SJW low and high dose groups in comparison with the control, respectively (p < 0.05) (Table 3).
Quantitative Western Blot analyses
Quantitative Western blot analyses were performed to detect and quantify CYP3A proteins in five intestinal regions prepared from control, SJW low dose and high dose treated rats. CYP3A immunoreactive bands of greater intensity were seen in upper regions of the intestine prepared from SJW high dose treated rats in comparison to the controls. The results for the western blots are provided in Figure 1. The results for the densitometry scanning of intestinal CYP3A immunoreactive bands are summarized in Table 4. The results are expressed as the mean ± standard deviation (SD) of 4 to 6 animals per group.

Western blot analyses of CYP3A immunoreactive proteins in the duodenum, jejunum, ileum, and caecum of control and St John's Wort treated animals, using a goat anti-rabbit CYP3A primary antibody. C1&3, C2&4: intestinal samples from control animals (n = 4), upper channel: animal 1 & animal 2, lower channel: animal 3 & animal 4. L1&4, L2&5, L3&6: intestinal samples from low dose (1500 µg/kg/day of Hypericin) SJW treated animals (n = 6), upper channel: animal 1, animal 2 & animal 3, lower channel: animal 4, animal 5 & animal 6. H1&4, H2&5, H3&6: intestinal samples from high dose (3000 µg/kg/day of Hypericin)) SJW treated animals (n = 6), upper channel: animal 1, animal 2 & animal 3, lower channel: animal 4, animal 5 & animal 6. Rc: untreated-liver samples, previously collected by other studies were included for the purpose of control experiment. A dense CYP3A immunoreactive band was detected for the liver of control experiment in all blots.
Densitometry scanning of western blot analysis of CYP3A immunoreactive proteins in upper intestinal segments prepared from the control and SJW low dose (1500 µg/kg/day of Hypericin) and high dose (3000 µg/kg/day of Hypericin) treated rats.
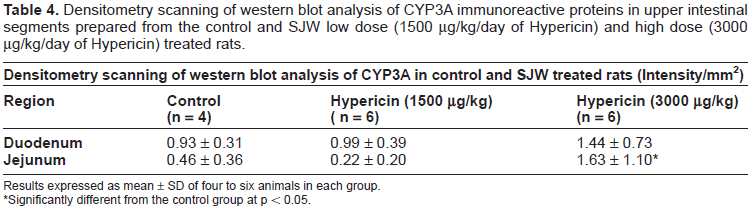
Results expressed as mean ± SD of four to six animals in each group.
Significantly different from the control group at p < 0.05.
In the jejunum from the SJW high dose treated group, the average relative density of CYP3A immunoreactive bands was 3.5 fold higher than that from the control group (p < 0.05). In the duodenum from the SJW high dose treated group, the average relative density of CYP3A immunoreactive bands was 1.5 fold higher in comparison to the control, but the difference was not statistically significant (p > 0.05). No significant differences were seen in most segments from the SJW low dose in comparison to that from the controls. In the ileum, caecum, and rectum, immunoreactive bands detected were not clearly visible and defined for all samples. Therefore, densitometry scanning was not performed for these regions since it is unlikely that the scanning would give accurate data.
Real-Time rt-Pcr Analysis of Liver and Intestinal cyp3a1 and cyp3a2 Mrna levels
Changes in intestinal CYP3A1 and CYP3A2 mRNA levels in SJW high dose and control groups are summarised in Table 5. Efficiency was calculated using the method in Liu and Saint (2002) and an equal efficiency was calculated. Ct Linear was calculated for the group and then meaned. A statistically significant 10.60 fold increase in liver CYP3A1 mRNA was observed in the SJW high dose treated group in comparison with the control group (p < 0.05), with a 1.27 fold increase in CYP3A2 which was found to be not statistically significant. Intestinal CYP3A1 and CYP3A2 mRNA levels appeared to be higher in some regions, such as jejunum for CYP3A1 and ileum for both CYP3A1 and CYP3A2. As the data was from only 2 animals per group, further work will need to be performed in order to draw any firm conclusion about regulation of intestinal CYP3A mRNA by SJW. Lower level of CYP3A1 and CYP3A2 was also noticed in other intestinal regions with a relatively small magnitude.
Changes in CYP3A1 and CYP3A2 mRNA levels by SJW high dose (3000 µg/kg/day of Hypericin) in liver and five intestinal regions in comparison with the control group a .
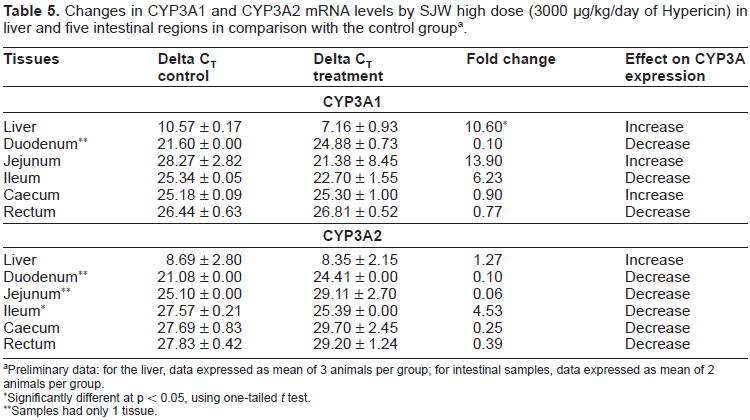
Preliminary data: for the liver, data expressed as mean of 3 animals per group; for intestinal samples, data expressed as mean of 2 animals per group.
Significantly different at p < 0.05, using one-tailed t test.
Samples had only 1 tissue.
Discussion
Ingestion of SJW over a period of time has been reported to induce hepatic and intestinal CYP3A4 enzymes (Durr et al. 2000). However, no in vivo studies have examined the inducing effect of SJW on CYP3A along the entire length of the intestine or at the mRNA level.
In the present study, after 14 days of consecutive daily administration of a standardised SJW extract, it was found that up regulation of CYP3A protein by SJW only occurred in upper segments of the rat intestine, the duodenum and jejunum, especially at high dose. A direct correlation between immunoreactive CYP3A protein and enzyme activity was shown in the duodenum and jejunum, but not in other regions. The later regions are known to have lower capacities in drug metabolism (Kolars et al. 1992; Paine et al. 1997; Zhang and Benet, 2001). Although both CYP3A activity and protein were higher in the duodenum prepared from the SJW high dose treated group in comparison with the control, the differences were not statistically significant, probably due to the great inter-individual variations. In man, a 11-fold variation in CYP3A protein content in intestinal biopsies from 20 patients has been reported (Lown et al. 1994). In rat, the current study is the first study that examined CYP3A in five segments of the intestine.
Administration of SJW for 14 days in man was reported to cause a 1.5 fold increase in intestinal CYP3A activity (Durr et al. 2000). In the present study, the increases in CYP3A-dependent erythromycin N-demethylation activity in the rat duodenum, jejunum and ileum were consistent which that observed in man intestine (1.7-1.8 fold). It is worth to note that practically, the dose of SJW used in man is less than that used in rat model (Durr et al. 2000). While no studies have reported the fold increase in CYP3A activity by SJW in rat intestine, it has been reported that when rats were dosed with SJW extracts for 14 days, an approximately 2.5 fold increase in hepatic CYP3A was seen (Durr et al. 2000, Bray et al. 2002). Erythromycin N-demethylation activity was 3.0 and 2.1 fold higher in the livers from the SJW high dose and low treated groups in comparison with the controls respectively (p < 0.05). Our results here were not only supported by other studies (Durr et al. 2000; Bray et al. 2001; Perloff et al. 2001; Komoroski et al. 2004) but also indicated the impact of SJW exposure dose on induction of hepatic CYP3A. In addition, significant up regulation of CYP3A1 mRNA by SJW was observed in the rat liver, with a small change in CYP3A2 mRNA. This reflects variation in drug metabolism and the distribution of drug metabolising enzyme observed in rat. The preliminary experiment to investigate the effect of SJW on intestinal CYP 3A1 and CYP3A2 mRNA suggested a differential increase in CYP3A across different regions of the rat intestine. Further RNA work will need to be performed in order to establish a firm conclusion about changes in intestinal CYP3A mRNA expression by SJW.
In conclusion, our preliminary results demonstrated up regulation of liver CYP3A1 mRNA, and to a smaller extent CYP3A2, by SJW, and suggested up regulation of intestinal CYP3A protein mainly in the upper segments, in a dose-dependent manner. This has significant implications for drugs that target a specific region of the intestine. The finding provides better understanding of the molecular mechanisms of drug interactions involving complementary medicines, promoting more appropriate use of complementary medicines in combination with conventional drugs.