Abstract
Background:
Antibiotic pollution in aquatic environments is a growing global concern, posing risks to ecosystem health and human well-being. The African continent faces increasing challenges in addressing this issue, necessitating a comprehensive understanding of the current state of research and regional trends.
Aim:
This review synthesizes evidence on the occurrence and distribution of antibiotics in aquatic environments in the African continent, assessing concentration levels and evaluating the potential environmental and microbial risks using predicted no-effect concentrations (PNECs).
Methods:
A systematic search of PubMed, Scopus, Web of Science, and Google Scholar was conducted to identify relevant studies published between January 1, 2015, and November 13, 2024, reporting antibiotic concentrations in surface water, groundwater, drinking water, and wastewater from countries within the African continent.
Results:
The review encompassed 19 studies reporting antibiotic concentrations in aquatic environments from 8 African countries, with the majority being conducted in South Africa. Surface water was the most frequently sampled environment. Twenty-six antibiotics from various classes were identified, with sulfamethoxazole and trimethoprim being the most frequently detected. Surface water and wastewater exhibited the highest numbers of antibiotics. Antibiotic concentrations fluctuated over time, peaking in 2020 and 2021. Wastewater had the highest concentrations, with norfloxacin levels reaching up to 433.0000 µg/L, while drinking water had the lowest concentrations. In many instances, detected concentrations surpassed PNEC-ENV and PNEC-MIC values, indicating potential environmental and microbial risks.
Conclusion:
This review underscores the threatening high concentrations levels of antibiotic in African aquatic environments, particularly in wastewater and surface waters. The presence and levels of some antibiotics exceeding the PNEC thresholds raises concerns because they can foster the development and spread of antibiotic resistance. Antibiotic-polluted groundwater and drinking water highlight the urgent need for robust water management and treatment optimization to safeguard human health.
Introduction
Among pollutants that exert detrimental effects on aquatic ecosystem are pharmaceuticals and personal care products (PPCPs), as they have potential negative effect on humans, animals, and other aquatic life. Pharmaceuticals include antibiotics, beta-blockers, lipid regulators, anti-inflammatory drugs, antiepileptics, and contrast media.
Evidence indicates that among low-and-middle income countries (LMICs), there are varying antibiotic use among nations and regions, with Sub-Saharan Africa having the lowest estimated levels and eastern Europe and central Asia having the highest. 1 Furthermore, the global rate of antibiotic consumption in 2018 was 14·3 (95% uncertainty interval 13.2-15.6) defined daily doses (DDD) per 1000 people per day (40.2 [37.2-43.7] billion DDD), which is 46% higher than the 9.8(9.2-10.5) DDD per 1000 per day in 2000. 1 It is estimated that the projected rate of antibiotic consumption will be 52.3% amounting to 75.1 billion DDDs by 2030 compared to 49.3 billion DDDs as of 2023. 2 The cost of treating antibiotic-resistant (AR) will amount to an additional US$1 trillion in healthcare costs by 2050 and between US$1 trillion and US$3.4 trillion in annual gross domestic product (GDP) losses by 2030. 3 According to Abubakar and Salman, 4 the antibiotic consumption rate in hospitals in Africa ranges from 27.6% to 83.5% from 2010 to 2022. This comprises of West Africa (51.4%-83.5%), North Africa (79.1%), East Africa (27.6%-73.7%) and South Africa (33.6%-49.7%). Additionally, cost of treating AR in Africa could cost about US$ 2-6 billion needed annually. 5
Inappropriate usage, over-prescription, and lack of antibiotic-resistant stewardships are among the crucial causes of antibiotic resistance development and proliferation. More so, the presence of these residues in the environments provide selective pressure of microbial communities.6 -8 Water environments are one of the reservoirs for antibiotic residues responsible for AR.9 -11 Antibiotic residues from a few of these water settings, such as septic tanks, 12 municipal wastewater, 13 hospital wastewater, 14 wastewater treatment plants, and agricultural reservoirs, may end up in other aquatic environments whether or not they have been treated, consequently altering the balance of microbial communities. The mechanisms by which antibiotic residues enhance or induce resistance include selective pressure on microbial communities, 15 horizontal gene transfer, 16 co-selection, and cross-selection, 17 induction of mutagenesis and adaptive response, 18 formation of biofilms, 19 persistence and resuscitation of dormant cells, 16 and alteration of microbial communities. 20
Through selection pressure, antibiotic residues in aquatic settings promote antimicrobial resistance (AMR) by eradicating vulnerable strains of bacteria and fostering the growth of resistant ones.21 -23 When pollutants like heavy metals encourage multi-drug resistance, co-selection and cross-resistance take place.24,25 By causing genetic alterations linked to stress, mutagenesis and adaptive responses quicken resistance. Additionally, antibiotics promote the creation of biofilms, which are protective bacterial colonies that promote gene exchange.19,26 Certain bacteria go into a latent state, survive exposure to antibiotics, and then come back to life. 27 From these literatures. it is noteworthy that the presence of antibiotic residues in water environments can have detrimental effects on animals, humans, and the environments as it encourages the formation and proliferation of AR.
Addressing the issue of antibiotic resistance in aquatic environments within the African continents is crucial, as these water bodies are often used for various purposes, including drinking, bathing, irrigation, and aquaculture, thereby increasing the potential for human and animal exposure to antibiotic-resistant microorganisms. To better understand the scope and severity of this issue, it is crucial to systematically assess the current state of knowledge regarding antibiotic concentrations in the aquatic environments of the African continents. This systematic review aims to synthesize the current state of knowledge regarding antibiotic concentrations in aquatic environments (surface water, groundwater, and wastewater) of the African continent and assess the potential environmental and microbial risks using predicted no-effect concentrations (PNECs) as a benchmark.
Materials and Methods
This systematic review was conducted in accordance with the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guidelines 28 to ensure a comprehensive and transparent approach to synthesizing the available evidence.
Search Strategy
A thorough literature search was performed using 4 major electronic databases: PubMed, Scopus, Google Scholar, and Web of Science. The search strategy employed a combination of keywords related to antibiotic resistance, antibiotic use, aquatic environments, and the African continent. The specific search terms included: (“antibiotic concentration” OR “antibiotic residues” OR “antibiotic-resistant bacteria” OR “antibiotic resistance genes”) AND (“surface water” OR groundwater OR wastewater OR “aquatic environment” OR “water environment”) AND (“Africa” OR “ African continent” OR specific country names within the African continent). Full search strategy can be found in Supplemental Table S1. The search was limited to articles published in English between January 1, 2015, and November 13, 2024 to ensure the inclusion of the most recent and relevant data.
Study Selection
Two independent reviewers screened the titles and abstracts of the retrieved articles to identify studies that met the inclusion criteria.
Inclusion criteria:
The following inclusion criteria were applied to select eligible studies for this systematic review:
1. Study location: Studies conducted within the African continent, which include all 54 officially registered countries in Africa.
2. Water types: Studies reporting data on antibiotic concentrations and/or antibiotic resistance in surface water, groundwater, and wastewater environments.
3. Antibiotics and antibiotic resistance: Studies that measured the concentrations of antibiotics and/or the prevalence of antibiotic resistance in water bodies.
4. Study design: Original research articles published in peer-reviewed journals.
Exclusion criteria:
Studies that met any of the following criteria were excluded from the systematic review.
1. Geographic location: Studies conducted outside the African continent.
2. Water type: Studies that did not report data on surface water, groundwater, or wastewater.
3. Lack of antibiotic or antibiotic resistance data: Studies that did not measure antibiotic concentrations or antibiotic resistance levels in the aquatic environments.
4. Study design: Review articles, case reports, conference abstracts, and studies without original data.
Data Extraction and Quality Assessment
A standardized data extraction form was used to collect relevant information from the included studies. The extracted data included study location, water type, antibiotics measured, analytical methods, and antibiotic concentrations.
To evaluate the risk of bias in the included studies, we used the Cochrane Risk of Bias 2 (RoB 2) tool. 29 This tool provides a structured and standardized method for assessing potential bias sources in randomized controlled trials. Given that most of the included studies were observational, we adapted the RoB 2 tool to address specific concerns relevant to this study design. To visualize the results of the risk of bias assessment, we used the RobVis tool, 30 which generates a summary figure that clearly represents the risk of bias for each study across various domains. The RoB 2 assessment was conducted independently by 2 reviewers, and any discrepancies were resolved through discussion and consensus. The key domains evaluated included:
Bias arising from the randomization process (not applicable for observational studies)
Bias due to deviations from intended interventions (not applicable for observational studies)
Bias due to missing outcome data
Bias in the measurement of the outcome
Bias in selecting reported results
Each domain was assessed as having a low, high, or unclear risk of bias, and an overall risk of bias judgment was made for each study.
Statistical Analysis
Microsoft Excel 365 (Version 2503) was used for the analysis. Descriptive statistics were used to summarize the key findings on antibiotic concentrations among different water sources, including surface water, groundwater, drinking water, and wastewater. For each, the minimum, maximum, mean, and standard deviation values were calculated for the concentrations of various antibiotic classes, such as sulfonamides, fluoroquinolones, tetracyclines, and aminoglycosides. In cases where antibiotic concentrations were reported in units other than µg/L, the values were converted to µg/L to facilitate cross-comparison. The findings were synthesized thematically and conveyed through textual narratives, tables, and figures.
Assessment of Environmental and Microbial Risks
To assess the potential environmental and microbial risks associated with the presence of antibiotics in African aquatic environments, we utilized the concept of predicted no-effect concentrations (PNECs). PNECs represent the maximum concentration of a substance below which no adverse effects are expected to occur in the environment or in microbial communities.
For the environmental PNEC (PNEC-ENV), we referred to published values derived from ecotoxicological data, such as the AMR Alliance Science-Based PNEC Targets for Risk Assessments 31 (Table 1). The PNEC-ENV values typically account for safety factors to protect the most sensitive species and ensure the long-term ecological integrity of the environment.
PNEC Targets for Selected Antibiotics (AMR Industry Alliance, February 2023). 31

The microbial PNEC (PNEC-MIC) was determined based on the minimum inhibitory concentrations (MICs) of antibiotic-susceptible bacterial strains, which represent the minimum concentration of an antibiotic required to inhibit the growth of these strains. The PNEC-MIC values were calculated by applying appropriate assessment factors to account for the potential development and spread of AMR in microbial communities 31 (Table 1).
For each antibiotic reported in the included studies, we identified the highest mean concentration in different water types (drinking water, groundwater, surface water, and wastewater). These values were then compared with the corresponding PNEC-ENV and PNEC-MIC thresholds to assess the potential environmental and microbial risks, respectively.
Results
Search Result
The systematic literature search identified 799 records from the electronic databases (Figure 1). After removing duplicates (n = 341), 458 records were screened based on title and abstract. During the screening process, 419 records were excluded as they did not meet the inclusion criteria, such as being conducted outside the African continent or focusing solely on sediments without reporting water-based data, reporting antibiotic resistance genes, focusing on resistance prevalence, or being reviews.

Study selection process.
The remaining 39 full-text articles were assessed for eligibility, and 20 were excluded for the following reasons: 3 studies were reviews, 5 studies focused solely on sediments without reporting water-based data, and 12 did not measure antibiotic concentrations. Ultimately, 19 studies32 -50 were included in the final qualitative synthesis of this review on antibiotic concentrations and antibiotic resistance in the aquatic environments of the African continent.
Study Settings and Regions
The review identified 19 studies that reported antibiotic concentrations in aquatic environments from 8 different countries across the African continent, South Africa (n = 6), Nigeria (n = 3), Ghana (n = 3), Kenya (n = 3), Uganda (n = 1), Zambia (n = 1), Benin (n = 1), and Tunisia (n = 1; Figure 2). The study setting encompassed various water types, including surface water, groundwater, drinking water, and wastewater, thereby contributing to a comprehensive understanding of antibiotic distribution in aquatic ecosystems (Supplemental Table S2). The majority of the studies (n = 15) were conducted in surface water environments including river, sea, rivers, canals, lake, dams, reservoirs and lagoons, while other (n = 10) focused on wastewater (including influents), groundwater (n = 2) including borewell, and well and drinking water (n = 1) including tap, sachet water, and bottled water.

Distribution of studies.
Study Period
The studies were conducted between 2015 and 2024, and most research was conducted in the last 5 years. Figure 3 illustrates the fluctuating pattern of antibiotic concentrations, covering the years from 2015 to 2024, showing peaks in 2020 and 2021, and a notable decrease in 2018. After 2021, a general decline in concentrations was observed through 2024. Antibiotic concentrations exhibit fluctuations rather than a distinct upward or downward trend over time. Notably, high levels of antibiotic contamination are observed in wastewater, especially in studies conducted in 2020 and 2021 (Supplemental Table S3).

Pattern of antibiotic concentrations across all aquatic environments over the years (2015-2024).
Detection Methods
Out of the 19 studies reviewed, high-performance liquid chromatography (HPLC) was used most (n = 9, 47.4%) for detection of antibiotics, followed by liquid chromatography-mass spectrometry (LC-MS; n = 6, 31.6%), ultra-performance liquid chromatography (UPLC; n = 3, 15.8%), and ELISA kit (n = 1, 5.3%).
Classes and Frequency/Occurrence Analysis of Detected Antibiotics
The studies examined 26 different antibiotics from classes such as penicillins (amoxicillin, ampicillin), cephalosporins (cefuroxime), sulfonamides (sulfamethoxazole, sulfadiazine, sulfathiazole, sulfamethazine), trimethoprim, tetracyclines (tetracycline, doxycycline), macrolides (erythromycin, clarithromycin, azithromycin), fluoroquinolones (ciprofloxacin, norfloxacin, ofloxacin, levofloxacin), aminoglycosides (gentamicin, amikacin, streptomycin, kanamycin B, neomycin), phenicols (chloramphenicol), lincosamides (clindamycin), nitroimidazoles (metronidazole), and nitrofurantoin.
Among the identified antibiotics, sulfamethoxazole was the most frequently detected antibiotic, with a total occurrence of 21 across various water sources: drinking water (n = 1), groundwater (n = 2), surface water (n = 11), and wastewater (n = 7). It was closely followed by trimethoprim, which was detected 17 times, including in drinking water (n = 1), groundwater (n = 2), surface water (n = 8), and wastewater (n = 6). In contrast, antibiotics such as clarithromycin, clindamycin, levofloxacin, sulfadiazine, and sulfamethazine were the least frequently detected 7 antibiotics (27%) were found exclusively in 1 type of water source, with sulfamethazine, sulfadiazine, levofloxacin, clarithromycin limited to surface water, and neomycin, ofloxacin, and clindamycin restricted to wastewater. The remaining antibiotics (73%) were detected in 2 or more water sources (Figure 4).

Occurrence of various antibiotics in aquatic environment.
Notably, surface water contained the highest number of antibiotics (43%), followed closely by wastewater (40%), whereas drinking water had the lowest number of antibiotics (8%; Figure 5).

Percentage of antibiotics detected in aquatic environment.
Distribution of Antibiotic Concentrations in Aquatic Environment
The analysis revealed significant regional variation in the overall concentration of antibiotics across Africa (Figure 6). Western Africa has the highest concentration at 737.9195 µg/L, followed by Southern Africa at 249.5808 µg/L, Eastern Africa at 150.5831 µg/L, and Northern Africa with the lowest at 22.3000 µg/L. Wastewater exhibited the highest concentrations of antibiotics, ranging from 0.0400 µg/L to a peak of 433.0000 µg/L, with an overall average concentration of 975.4147 µg/L, highlighting its significant role as a source of antibiotic pollution. Notably, norfloxacin exhibited the highest concentration among all antibiotics in wastewater (Table 2, Figure 7). Surface water concentration ranged from 0.0010 to 49.7000 µg/L, with an average of 183.3567 µg/L. Sulfamethoxazole had the highest concentration in surface water (Table 2, Figure 8).

Overall concentration of all antibiotics found in the various African continent.
Antibiotic Concentrations in All Aquatic Environments.

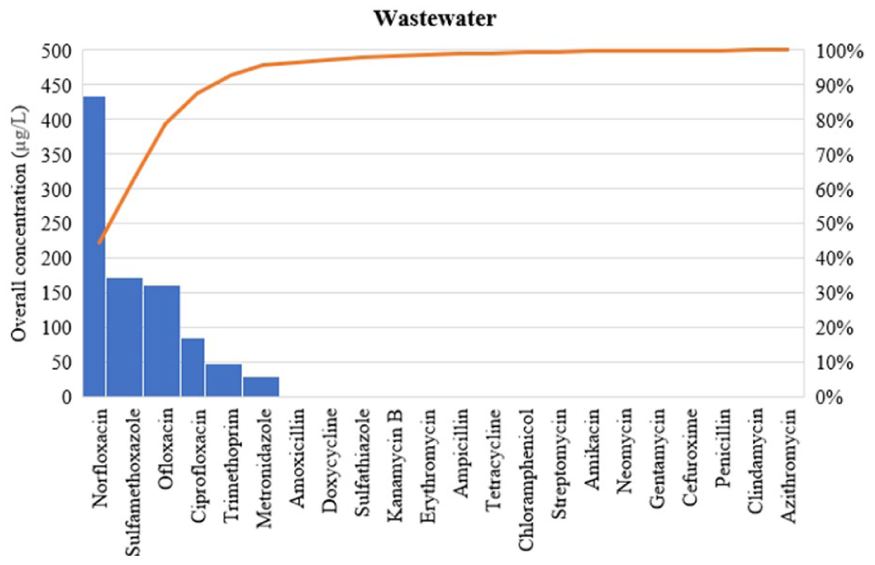
Overall concentration of all antibiotics found in wastewater.

Overall concentration of all antibiotics found in surface water.
In contrast, groundwater generally had lower concentrations, varying from 0.0010 to 0.7600 µg/L, with a total concentration of 1.2510 µg/L, indicating relatively lower levels of contamination. Amoxicillin had the highest concentration among all antibiotics detected in groundwater (Table 2, Figure 9). Drinking water, which is more protected, had the lowest antibiotic levels, ranging from 0.0010 to 0.3580 µg/L, with a total of 0.3610 µg/L, reflecting effective treatment processes. Like groundwater, amoxicillin had the highest concentration in drinking water (Table 2, Figure 10).

Overall concentration of all antibiotics found in groundwater.

Overall concentration of all antibiotics found in drinking water.
PNEC Assessment
In drinking water, the highest mean amoxicillin concentration was above the PNEC-MIC value (Table 3). In contrast, the groundwater concentrations exceeded both the PNEC-ENV and PNEC-MIC values. For surface water, the highest mean sulfamethoxazole concentration surpassed both PNEC thresholds. In addition, in wastewater, the mean concentrations of norfloxacin and ofloxacin exceeded both the PNEC-ENV and PNEC-MIC values.
Highest Mean Concentration of Dominant Antibiotics in Each Aquatic Environment Compared to Their Respective PNEC Values.

Risk of Bias
The risk of bias assessment indicated that the majority of the included studies (n = 17, 89.5%) were classified as having a low or unclear risk of bias, while only 2 studies (n = 2, 10.5%) were deemed to have a high risk of bias (Figure 11). The most prevalent sources of bias were associated with outcome measurement, particularly regarding the reliability and validity of the analytical methods used to detect and quantify antibiotic concentrations in aquatic samples.
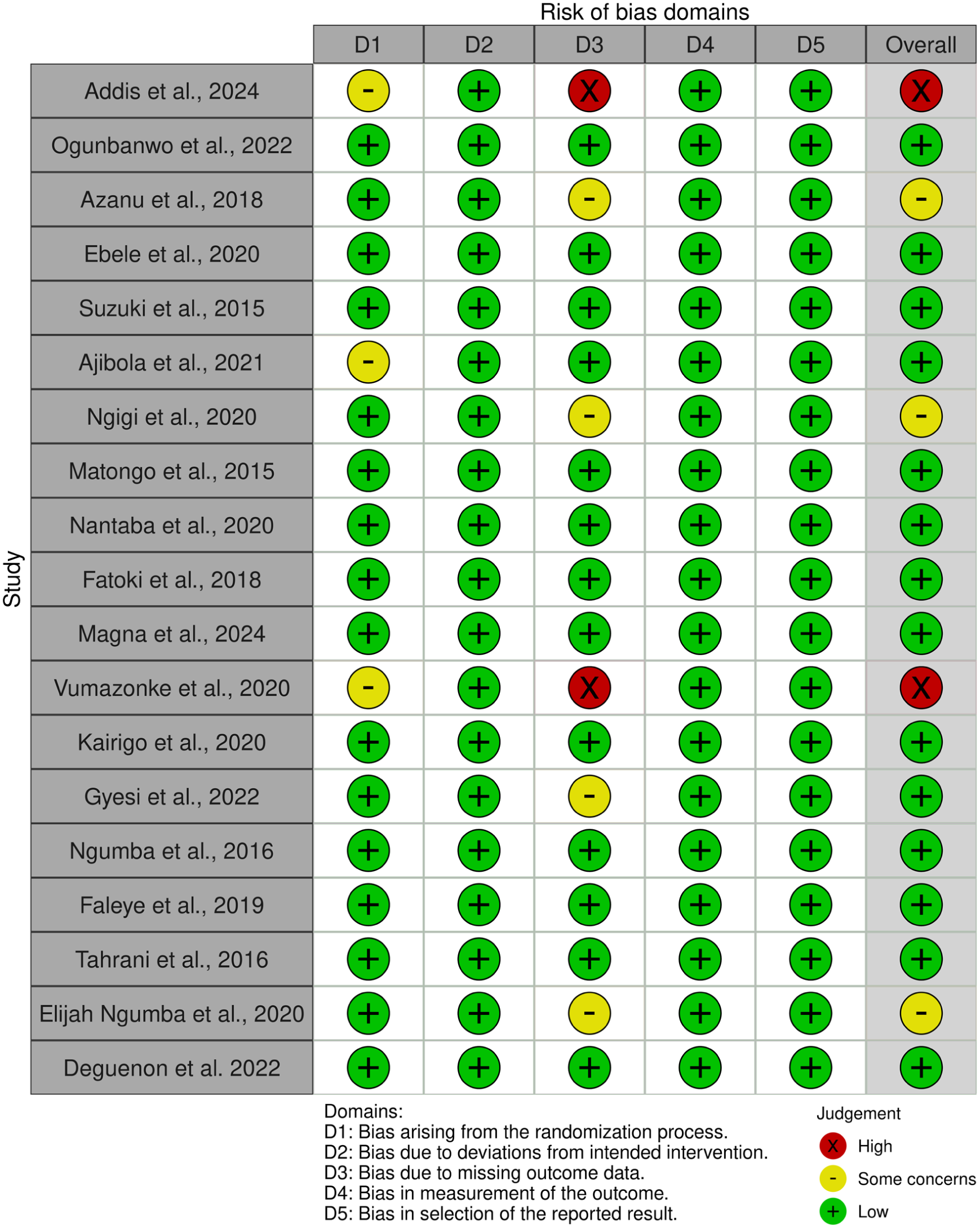
Furthermore, some studies lacked sufficient details on sampling procedures, sample handling, and laboratory protocols, complicating the assessment of potential bias in outcome measurement. Additionally, issues such as missing data and inadequate reporting of flow and exclusions contributed to an unclear risk of bias in the domain of missing outcome data.
Discussion
This comprehensive review of the literature on antibiotic contamination in the aquatic environments of the African continent provides a wealth of insights into the scope, severity, and implications of this critical environmental issue. While previous reviews may have covered broader global or regional perspectives, this review specifically focuses on the African continent, providing a more in-depth analysis of the situation in this particular geographical region. A unique feature of this review is the inclusion of a PNEC assessment for the detected antibiotic compounds. Previous reviews may have lacked this type of systematic environmental risk assessment.51,52
The geographical diversity of the studies included in this review provides a valuable regional perspective on the prevalence and distribution of antibiotics in the aquatic environments of the African continent. Most of the studies (31.6%) were conducted in South Africa reflecting the country’s proactive environmental monitoring and research effort on emerging contaminants, including antibiotics. As one of the more developed nations in the region, South Africa has likely benefited from greater research building capacity, funding, and access to advanced analytical instruments, enabling extensive investigations into the extent of antibiotic contamination in its aquatic systems. However, other countries, such as Nigeria, Ghana, and Kenya, are also making significant efforts, underscoring the growing research interest and recognition of the importance of addressing antibiotic pollution in the region.
Our findings indicated that surface water was the most extensively studied water source, highlighting its critical role in the environmental fate and transport of antibiotics. Surface water bodies, including seas, rivers, canals, lakes, dams, and streams, often receive various anthropogenic inputs, such as discharges from wastewater treatment plants, agricultural runoff, and industrial effluents.53,54 As a result, these environments are particularly vulnerable to direct contamination by antibiotics, making them essential targets for environmental monitoring and assessment. 55 Wastewater is the primary pathway through which antibiotics are introduced into aquatic ecosystems. In the presence of a diverse mix of pharmaceutical residues and microbial contaminants, wastewater often acts as a point source for antibiotic pollution, especially in regions with inadequate wastewater treatment infrastructure. 56 The limited number of studies focusing on groundwater and drinking water highlights the low level of interest in studying those areas due to confidence in the treatment process, especially for drinking water. Although groundwater and drinking water sources are generally more protected from direct anthropogenic influences, they can still be at risk of low-level antibiotic contamination through pathways such as leaching from polluted surface water or insufficient treatment processes. 57 Nonetheless, the low number of studies in these areas, especially in drinking water, paves the way for more focus to be directed toward them.
We identified 26 antibiotics across various classes, including penicillins, cephalosporins, sulfonamides, trimethoprim, tetracyclines, macrolides, fluoroquinolones, aminoglycosides, phenicols, lincosamides, nitroimidazoles, and nitrofurantoin. This variation is not exclusive to Africa, as similar findings have been reported in studies from Europe and Asia.58,59 Antibiotics were detected using several methods, with high-performance liquid chromatography (HPLC) being the most employed technique in the reviewed studies, highlighting its widespread use in environmental monitoring. HPLC has been a reliable choice for decades, allowing for the robust separation and quantification of various antibiotics such as sulfonamides, quinolones, tetracyclines, macrolides, and penicillin. 60 In addition to HPLC, some studies utilized other analytical tools such as liquid chromatography-mass spectrometry (LC-MS) and ultra-performance liquid chromatography (UPLC). LC-MS combines the separation capabilities of liquid chromatography with the sensitivity of mass spectrometry, enabling accurate identification and quantification of a broader range of antibiotics, even at trace levels. 60 UPLC, an advanced analytical technique on the other hand, offers enhanced resolution, sensitivity, and separation efficiency compared to conventional HPLC, allowing for the detection of a wider array of antibiotics, including macrolides, sulfonamides, and aminoglycosides.61,62 The adoption of these advanced analytical techniques in Africa indicates a positive trend toward the development of environmental monitoring and research capabilities.
Sulfamethoxazole and trimethoprim were the most detected antibiotics in our review, and they were present in multiple water sources, including drinking water, groundwater, surface water, and wastewater. Our results are consistent with global trends, as sulfamethoxazole and trimethoprim are among the most widely used antibiotics worldwide and have been frequently reported in environmental monitoring studies across various regions.63,64 Sulfamethoxazole has been identified as a persistent and ubiquitous contaminant in aquatic environments, with the ability to accumulate in sediments and potentially re-enter the water column through desorption or resuspension. 65
Wastewater had the highest antibiotic concentrations, ranging from 0.0400 µg/L to an extreme value of 433.0000 µg/L. These elevated levels surpass those reported in China, where antibiotic concentrations in wastewater have been documented between 0.401 and 57.106 µg/L. 66 High antibiotic levels in wastewater highlight the critical need for effective treatment and management strategies to prevent the discharge of antibiotic-contaminated effluents into the environment. 67 Inefficient and ineffective treatment of wastewater can cause the release of either trace amounts or high concentrations of these antibiotics into surface waters, adversely affecting aquatic ecosystems. Surface water in the African continent exhibited several antibiotic concentrations. 32 Although these levels are generally lower than those found in wastewater, they can lead to exposure and bioaccumulation in aquatic organisms, disrupt ecosystem functioning, and facilitate the development and spread of antibiotic resistance.18,68 The potential for surface water contamination to impact drinking water sources and human health further emphasizes the need for comprehensive water management strategies. 69 Groundwater typically has antibiotic concentration. 51 While these levels might seem relatively low, the persistence and potential for groundwater contamination should not be overlooked, as groundwater can act as a long-term reservoir for antibiotics and gradually contaminate drinking water sources. 70 Drinking water, which is more regulated and subject to treatment processes, exhibited the lowest levels of antibiotic contamination. This finding aligns with findings from other regions, where treated drinking water generally has lower antibiotic levels than other aquatic environments.71 -73 However, trace amounts of antibiotics in drinking water are concerning, because they can contribute to the selection and spread of antibiotic-resistant bacteria, posing risks to human health. 74
Concerning specific antibiotics, our analysis revealed the maximum concentration of ciprofloxacin and norfloxacin across all aquatic environments. High levels of critically important antibiotics, often used as last-resort treatments for severe infections, can exert significant selective pressure on microbial communities, promoting the emergence, and proliferation of antibiotic-resistant pathogens. 75 Similarly, sulfamethoxazole and amoxicillin exhibited the highest concentrations in surface water and groundwater, respectively, with elevated norfloxacin and ofloxacin levels in wastewater further highlighting the need for targeted interventions to address the sources and fate of these specific antibiotics in aquatic environments. Notably, we found that amoxicillin had the highest concentration in both groundwater and drinking water; however, the concentration in groundwater was twice that in drinking water. Since the sources of drinking water in Africa mainly come from groundwater, this observation suggests that groundwater treatment reduces antibiotic concentration. Continuous monitoring, optimization of water treatment processes, and implementation of effective strategies are needed to minimize the transfer of contaminants into the final water supply. 76
The overuse or underuse of antibiotics for human and animal health and for improving growth in agriculture and aquaculture leads to the channeling of the unmetabolized low concentrations into some water environments.
77
Significant environmental antibiotic presence puts bacterial strains in that niche under selective pressure, forcing them to evolve a variety of resistance mechanisms that serve as antibiotic defense mechanisms.
78
This allows the bacteria to survive in an antibiotic-loaded environment and leads to the development of AMR. Studies have reported on the mechanisms and types of resistance some antibiotic residues confer being present in water environments. One of the methods by which antibiotic residues induce resistance is through mutations in the bacteria due to long exposures.
79
These mutations can persist and accumulate for years leading to transfer of mutated resistant genes. The exposure of
Our temporal analysis revealed a fluctuating pattern in antibiotic concentrations over time. We observed particularly high antibiotic levels in aquatic environment during 2020 and 2021, which is particularly concerning, because it suggests a potential surge in antibiotic consumption or improper disposal practices during this period. This finding aligns with global trends observed during the COVID-19 pandemic, in which increased antibiotic use for the treatment of secondary bacterial infections in hospitalized COVID-19 patients has been widely reported. 88 The subsequent decline in antibiotic concentrations observed after 2021 may be indicative of improved antibiotic stewardship and the implementation of more effective wastewater treatment measures. Nonetheless, continual monitoring and sustained efforts are necessary to maintain this positive trajectory. These common patterns across varied water sources underscore the dynamic and multifactorial nature of antibiotic pollution, which is influenced by global and regional shifts in pharmaceutical consumption, healthcare practices, and environmental management strategies.18,75
Widespread contamination of African surface waters, groundwater, and wastewater with antibiotics raises significant concerns due to their potential adverse effects on aquatic ecosystems. High concentrations of antibiotics, such as amoxicillin in drinking and groundwater, along with elevated levels of sulfamethoxazole, and metronidazole in surface water, exceed the established PNEC-ENV thresholds. This situation poses risks to sensitive aquatic organisms, potentially disrupting their growth, reproduction, and overall ecological balance. 11 Additionally, antibiotics can bioaccumulate and bio-magnify through the food chain, impacting higher trophic levels and jeopardize the health and functionality of aquatic ecosystems. 89 Moreover, the presence of antibiotics in irrigation water can lead to the contamination of agricultural produce, further threatening human health and the wider food system. 90
Detected concentrations of antibiotics, including norfloxacin and ofloxacin, in wastewater exceeding PNEC-MIC thresholds raise significant concerns about antibiotic resistance. This scenario increases the risk of developing and spreading antibiotic-resistance in aquatic environments. Resistance genes can propagate through various pathways: untreated or inadequately treated wastewater discharge, the use of contaminated water for irrigation and livestock, and human consumption of polluted food and water. 91 Such dissemination routes may lead to increased resistance in both the environment and human populations, potentially undermining the effectiveness of antibiotic therapies and resulting in adverse health outcomes. Groundwater pollution, a crucial drinking water source in many African continents, directly exposes threatened communities to antibiotic polluted water sources, increasing the risk of antibiotic resistance. This issue is especially critical in areas with limited access to clean water and healthcare, where the consequences of antibiotic-resistant infections can be severe. Furthermore, the global impact of antibiotic resistance originating from this region could hinder international efforts by reducing the effectiveness of antimicrobial treatments worldwide.
The elevated antibiotic concentrations reported in African aquatic environments, particularly the extreme levels found in wastewater, are not merely the result of high consumption, or poor disposal practices in isolation. Rather, they reflect interconnected systemic challenges rooted in weak healthcare infrastructure, regulatory gaps, inadequate urban planning, and prevailing socioeconomic conditions across much of the continent. In many African countries, self-medication and the over-the-counter sale of antibiotics without prescriptions are widespread, driven by limited access to formal healthcare, especially in rural and peri-urban areas. This unregulated use directly increases the volume of antibiotics entering human waste streams. Hospitals and clinics, particularly in urban centers, often lack on-site pharmaceutical waste management protocols. Studies from Nigeria, Kenya, and Benin confirm that hospital effluents serve as direct conduits for high-concentration antibiotic residues into municipal sewers or open drains. Compounding the problem, most African cities rely on hybrid or informal sanitation systems that combine septic tanks, open drains, pit latrines, and limited sewer networks. Only a small fraction of urban wastewater undergoes any form of treatment, and even where wastewater treatment plants (WWTPs) exist, such as in parts of South Africa or Tunisia, they are typically designed to remove organic matter, and nutrients, not micropollutants like antibiotics. Consequently, these compounds pass through treatment systems largely unattenuated, ultimately entering rivers and lakes that serve as critical downstream water sources. Although some African nations have established environmental quality standards, these rarely include thresholds for pharmaceuticals, let alone enforceable limits for antibiotics in water. Environmental monitoring remains sporadic, and compliance mechanisms are chronically under-resourced, further undermining efforts to control antibiotic pollution at its source.
Limitations
First, the review was limited by the heterogeneity of the included studies in terms of their methodologies, sampling locations, and the specific antibiotics analyzed. This variability can introduce challenges when making direct comparisons and synthesizing the evidence, potentially leading to biases or inconsistencies in the reported findings. Second, the review relied on the availability and quality of the published literature. The identification of only 19 relevant studies, with the majority originating from South Africa, suggests that there are significant geographical gaps in the existing research. This uneven distribution of data across the African continent limits the generalizability of the findings and the ability to draw comprehensive conclusions about the overall antibiotic pollution status in the continent’s aquatic environments. Finally, the review was limited to the available antibiotic concentrations data, which may not always be comprehensive, or representative of the actual levels present in the environment. Factors such as temporal and spatial variability, the influence of seasonal changes, and the potential for fluctuations in antibiotic use and discharge patterns were not always accounted for in the included studies.
Conclusions
This systematic review provides a compelling and multifaceted understanding of antibiotic contamination in the aquatic environments of the African continent. The findings revealed widespread contamination, with several antibiotics detected at concentrations exceeding the established PNEC-ENV and PNEC-MIC thresholds, indicating the potential for adverse impacts on aquatic ecosystems, and the selection and proliferation of antibiotic-resistant bacteria. The contamination of surface water, groundwater, and even drinking water sources highlights the pervasive nature of this problem and the urgent need for comprehensive, region-wide action. According to this review, the mean levels of antibiotics in drinking water across the African continent are low. However, increased monitoring and regulation of drinking water sources and suppliers should be encouraged to further reduce antibiotic concentrations, as even as trace amounts can have long-term effects on humans and animal health. Addressing this issue requires a multifaceted approach, including improved wastewater treatment, stricter regulations on antibiotic use and disposal, and enhanced monitoring and surveillance programs.
Recommendations
Enforce on-site pre-treatment of hospital and pharmaceutical facility effluents: Mandate all hospitals, clinics, and pharmaceutical distributors to install and operate on-site pre-treatment systems (eg, granular activated carbon or ozonation units) before discharging wastewater into municipal sewers or open drains.
Adopt PNEC-based discharge limits for high-risk antibiotics in national water regulations: Integrate PNEC-MIC and PNEC-ENV thresholds (eg, from the AMR Industry Alliance) into national wastewater discharge standards, starting with priority antibiotics that consistently exceed safe levels: norfloxacin, ofloxacin, sulfamethoxazole, and metronidazole.
Pilot and scale low-cost, decentralized wastewater treatment in high-risk urban informal settlements: Launch government-supported pilots of modular or nature-based treatment systems (eg, constructed wetlands, biochar filters, or solar photo-Fenton reactors) in informal settlements near rivers like the Nairobi River (Kenya), Odaw River (Ghana), or Apapa Canal (Nigeria), where untreated domestic and medical waste directly enters surface waters.
Establish a regional antibiotic monitoring network with standardized protocols: Create an African Antibiotic Surveillance in Water (AASW) Network, coordinated by Africa CDC and AMR National Focal Points, to harmonize sampling, analytical methods (eg, LC-MS/MS), and data reporting across countries.
Supplemental Material
sj-docx-1-ehi-10.1177_11786302251391299 – Supplemental material for Antibiotic Concentrations in Aquatic Environments of the African Continent: A Systematic Review and Predicted No-Effect Concentration (PNEC) Assessment
Supplemental material, sj-docx-1-ehi-10.1177_11786302251391299 for Antibiotic Concentrations in Aquatic Environments of the African Continent: A Systematic Review and Predicted No-Effect Concentration (PNEC) Assessment by Eric S. Donkor, Alex Odoom and Prince Hotor in Environmental Health Insights
Footnotes
Author Contributions
Conceptualization, E.S.D., A.O. and P.H.; methodology, A.O., P.H. and E.S.D.; software, A.O. and E.S.D.; validation, E.S.D., P.H., and A.O.; formal analysis, A.O. and E.S.D.; investigation, A.O., P.H. and E.S.D.; resources, E.S.D.; data curation, A.O. and E.S.D.; writing—original draft preparation, A.O. and E.S.D.; writing—review and editing, A.O., P.H. and E.S.D.; visualization, A.O., P.H. and E.S.D.; supervision, E.S.D.; project administration, E.S.D; funding acquisition, E.S.D. All authors have read and agreed to the published version of the manuscript.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This review paper was supported by the Fogarty International Center of the National Institutes of Health through the Research, and Capacity Building in Antimicrobial Resistance in West Africa (RECABAW) Training Program hosted at the Department of Medical Microbiology, University of Ghana Medical School (Award Number: D43TW012487). The content is solely the responsibility of the authors and does not necessarily represent the official views of the National Institutes of Health.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
All supporting data is in the manuscript and supplementary materials
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
