Abstract
Antimicrobial use in animal husbandry is a common practice in developing countries as a response to the growing demand for animal-based foods. Veterinary antibiotics account for most of the antibiotics used worldwide. This review aims to provide an overview of the impact of antibiotic usage in livestock production on the environment and its consequences in the emergence and spread of antibiotic resistance. The global estimate of antibiotic consumption in animals ranges from 63 000 to 106 000 tons annually. The most commonly used antibiotic classes include tetracyclines, sulfonamides, β-lactams, macrolides, and trimethoprim. Most of the antibiotics used in animal treatments are not fully metabolized within the animal’s body; instead, they are excreted and can enter the environment. This review also provides a brief overview of the fate of antibiotic residues and their behavior in the environment. In developing countries, in addition to the lack of data on antibiotic use trends, there are limitations in implementing scientific and regulatory frameworks for antibiotic use in livestock. To reduce the projected loss of lives associated with antimicrobial resistant infections, it is recommended to lower antibiotic contamination in the environment by implementing affordable and critical actions related to antibiotic usage and residual waste management. Actions such as rational use of antibiotics in all circumstances, reducing environmental exposure to antibiotics, research, and environmental surveillance are important to tackle the problems of antibiotic resistance, especially in low-income countries.
Plain Language Summary
Using antibiotics in farm animals is more complex than in humans because they are often given to promote growth. This widespread use, especially in developing countries with less regulation, increases environmental pollution and promotes antibiotic-resistant bacteria, making infections harder to treat. In developing countries, the problem is even worse due to the increased use of antibiotics in animals, poor monitoring, misuse, and low-quality medicines. To stop this growing threat, countries should follow WHO guidelines and learn from successful strategies used in developed nations. Simple, affordable actions—such as responsible antibiotic use, waste reduction from farms, and enhanced research and monitoring—are essential. Furthermore, investing in new treatment alternatives and improved animal feeds can help reduce the reliance on antibiotics. These steps are vital to protect both human and animal health and keep medicines effective.
Introduction
The discovery of antibiotics in the 20th century marked a pivotal milestone in human and animal healthcare. Antibiotics are extensively used in animal production for various purposes, including disease treatment, disease prevention (prophylaxis), and growth promotion. 1 Veterinary pharmaceuticals encompass of a broad range of substances, such as antimicrobials, antiparasitics, hormones, antifungals, anti-inflammatory drugs, anesthetics, tranquilizers, and sedatives. 2 Approximately 70% of veterinary antimicrobials are antibiotics, primarily derived from natural products of bacteria (40%) and fungi (60%). 3 The remaining antimicrobials are semi-synthetic or synthetic compounds.4,5 Among the first antibiotics used in food animals were penicillin and sulfonamides introduced in the 1940s. Following this breakthrough, a variety of antibiotics have been introduced not only for treating and preventing diseases but also as feed additives to enhance animal growth. 6 The widespread use of antibiotics in animals leads to the release of antibiotic residues into the environment through manure, agricultural wastes, and wastewater.7,8 A significant concern associated with antibiotic use in animals is the emergence and spread of antibiotic-resistant bacteria (ARB) that harbor antibiotic traits. 9 The release and accumulation of antibiotic residues in the environment gradually increase antibiotic concentrations, leading persistent ecological issues that threaten human and animal health. 10 Beyond the environmental and health concerns, growing public apprehension about antibiotic-resistant pathogens threatens consumer confidence in the safety of animal-derived foods.
Antimicrobial Resistance and Global Trends
One of the primary drivers of antimicrobial-resistance (AMR) in the environment is the extensive and often inappropriate use of antibiotics in agriculture. Misuse and overuse of antibiotics contribute to the development and transmission of resistance among microbes, facilitating the transfer between environmental reservoirs, humans, and animals. Global projections estimate that veterinary antibiotic consumption will reach to 104 079 tonnes by 2030, representing an 11.5% increase from the 2017 baseline. 11 During the same period, antibiotic use in humans is expected to increase by approximately 15%, suggesting that antimicrobial use (AMU) in food animals is roughly proportional to human consumption. This trend underscores the significant contribution of livestock antibiotic use to the escalation of environmental AMR. However, establishing a direct link between antibiotic use and the spread of antibiotic resistance in human pathogens remains complex, due to discrepancies in antibiotic application practices between human medicine and veterinary use. 12 Globally, more antibiotics are used in animals, particularly in poultry, swine, and cattle, than in humans. Of approximately 27 antimicrobial classes used in animals, 9 are exclusively used in veterinary applications. The most widely used antibiotic classes in animals are macrolides, penicillin, and tetracycline; however, all are considered critically important in human medicine. 13 The overlapping use of these critically important antibiotics in both humans and animals exacerbates the problem of AMR, posing significant challenges to human health and raising concerns about environmental sustainability. Developing countries such as China and India are among the largest consumers of antibiotics in animal agriculture. In China, antibiotics are extensively used as growth promoters through feed, as well as for other veterinary uses, especially in broiler and pig farming. 13 In India, several antibiotics such as tetracyclines, erythromycins, gentamicin, chloramphenicol, ciprofloxacin, cotrimoxazole, trimethoprim, amikacin, ceftriaxone, and cefotaxime continue to be used by farmers for veterinary applications. 14 Similar trends are observed in African countries; however, the extent and specifics of antibiotic use for growth promotion, prophylaxis, and disease treatment are not yet fully documented. Therefore, this review aims to highlight trends in veterinary antibiotic use in livestock, their environmental behavior, and their fate, with particular emphasis on practices in developing countries. Understanding these patterns is essential for developing strategies to mitigate the risks associated with antibiotic residues and antibiotic resistance development in the environmental contexts of developing countries.
Veterinary Antibiotic Usage in Developing Countries
Livestock constitute an integral part of the livelihoods of many communities worldwide, providing essential resources such as food, income, draft power, and other environmental functions. To safeguard health and enhance productivity, Livestock production often employs the use of antimicrobial agents. Globally, the use of antibiotics in animal agriculture is substantial, and it is estimated that approximately 73% of all antibiotic consumption is attributable to the meat industry. 15 Projections indicate that antibiotic use in livestock will continue to increase, driven by rising demand for animal protein. 11 The pattern of antibiotic use in livestock varies considerably across countries, largely influenced by national regulatory frameworks, enforcement levels, and economic factors. In Africa, antibiotic utilization in livestock ranges from 77.6% in Nigeria to 100% in countries such as Tanzania, Cameroon, Zambia, Ghana, and Egypt. 16 Approximately 14 classes of veterinary antibiotics are used in Africa, with tetracyclines, aminoglycosides, and penicillin being among the most prevalent. Ethiopia, which has the largest livestock population in Africa, uses a substantial amount of antibiotics in livestock. 17 The most commonly used antibiotics include oxytetracycline, aminoglycosides, and trimethoprim-sulfonamides (Supplemental Material Table 1). Besides antibiotics, antiparasitic agents, antifungals, and acaricides are essential components of livestock health management. 18
AMR in developing countries presents a significant challenge, primarily due to inadequacies in regulating and monitoring antibiotic use in livestock. These gaps often contribute to the indiscriminate applications of veterinary antibiotics, fueling the emergence of antibiotic-resistant strains. In countries like Nigeria, Bangladesh and India veterinary antibiotics are reportedly accessible over the counter, 19 creating conditions where unregulated use is a possibility. This ease of access to veterinary antibiotics promotes the likelihood of using broad-spectrum antibiotics and sub-therapeutic doses, exacerbating resistance development among bacteria. Studies have reported an increasing prevalence of extended spectrum beta-lactamase (ESBL) producing E. coli in livestock. 20 Apart from its role in the development of AMR, veterinary antibiotics can influence microbial ecology accumulating in aquatic microbiomes leading to ecological imbalance. 21 Monitoring of AMR in the manure is also useful to trace the potential presence of antibiotics and avoid environmental risks associated with land applications.
In many developing countries, including Ethiopia, antibiotics are extensively used not only for treating infectious diseases but also as growth promoters at sub-therapeutic levels (<0.2 g/kg). 22 For example, Gemeda et al 23 reported that 84% of farmers used antibiotics to treat sick animals and promote growth, highlighting widespread reliance on these drugs. The dosage used in food animals varies significantly depending on the purpose. Relatively lower doses are used for growth promotion, intermediate doses for prophylaxis, and higher doses for therapeutic purposes. 24 However, in rural areas farmers’ limited understanding of proper dosage often leads to misuses, such as incorrect administration and inadequate withdrawal periods, exacerbating the risk of AMR development. The use of antibiotics at sub-therapeutic levels for growth promotion is particularly concerning, as it exerts selective pressure favoring resistant strains in animal productivity or health. 15 These practices can facilitate the emergence and dissemination of ARB, which may transfer to humans through direct contact or via the food chain.
After administration, a substantial proportion of antibiotics are excreted, entering soil, water and sediments (Figure 1). This environmental dissemination promotes the proliferation of resistant bacteria and ARGs in ecosystems, posing significant public health risks. This is because farmers often use growth-promoting antibiotics to quickly reach market weight, without sufficient regard for their role enhancing AMR. 25 In addition to usage discrepancies, antibiotic distribution in developing countries is influenced by several factors that increase the risk of misuse, including the illegal trade of veterinary drugs, the prevalence of expired medications, inappropriate prescriptions, and dosage issues. 26 These issues often arise from mismanagement of antibiotics in animal husbandry. Suboptimal drug levels and low-activity drugs can lead to poor tissue penetration and incomplete eradication of pathogens in the animal. 27 Moreover, insufficient regulation and weak veterinary oversight mean antibiotics are often used without adherence to pharmacological standards, increasing the possibility of resistance development. The challenges of antibiotic use in low-income countries are also influenced by poor waste management practices, contaminated effluents, and lack of comprehensive risk assessments concerning antibiotic residues and ARB in the environment. 28 As a result, evaluating the true impact of antibiotic use in livestock remains difficult due to limited data and robust surveillance systems in these low-income settings.

Conceptual model depicting the routes of antibiotic residues in the environment.
Environmental Impacts of Antibiotic Use in Livestock
The use of antibiotics in food animals has increased markedly over the last few decades, paralleling their presence in the environment as parent compounds or partially metabolized residues. 29 This increase in antibiotic use in livestock is primarily driven by the growing global demand for animal-based food products. 30 To improve productivity, farmers often use antibiotics that promote growth in addition to their health-safeguarding roles. 6 The global trend of antibiotic usage in livestock is mainly for disease treatment, prevention, and growth promotion in decreasing order (Figure 2), but the antibiotics used for growth promotion are the most controversial application. 25 As of 2010, annual global antibiotic consumption in livestock is in the range of 63 000 to 106 000 tons, with recommended usage of 172 mg/kg for pigs, 148 mg/kg for poultry, and 45 mg/kg for cattle. 19 Nearly 70% of the antibiotics eventually enter into the environment through agricultural, medical, and animal wastes, underscoring the release of substantial amounts of antibiotics into the environment. Given that livestock are among the largest users of antibiotics, environmental contamination with antibiotics is a critical issue in Asia, Africa, and the Americas. For instance, high antimicrobial use is reported in regions such as Nile Delta, Nigeria, Tanzania, Zambia, Ghana, Egypt, and South Africa, often driven by unregulated over-the-counter sales. Ethiopia ranks mid-table in livestock antibiotic use calculated per 10 km2 pixels.15,31 In contrast, high-income countries such as the USA consume over 12,500 tons of antibiotics annually in livestock, predominantly for therapeutic purposes. This amount is comparable to the antimicrobial consumption rate in human medicine. 32

Current use trends of antibiotic in livestock and destinations of the antibiotic residues.
Major classes of antibiotics utilized in livestock include the
Over the years, there has been an alarming increase in antibiotic pollution, evidenced by the detection of residues and ARB in various environments. 36 The environment acts as an important reservoir and conduit for the horizontal transfer of ARGs. Resistance determinants often carried on mobile genetic elements like plasmids and transposons, facilitate rapid dissemination across bacterial populations. For example, the detection of extended-spectrum β-lactamases (ESBLs) and carbapenemases in environmental isolates highlights the ongoing spread of clinically relevant antibiotic resistance. The presence of antibiotics and ARB in environmental matrices poses direct and indirect risks to human health through multiple pathways, including contaminated water, food, and direct contact. Wastewater treatment plants, especially in hospitals and municipalities, act as major hotspots for AMR proliferation due to antibiotic residues and resistant bacteria.8,37 In addition, abattoirs and agricultural settings contribute to the contamination of AMR in the surface waters and soil. 38 Human and animal wastes and antibiotic residues discarded from pharmaceutical manufacturers are also important sources of antibiotic contamination.36,39 The transfer of ARGs from environmental bacteria to pathogenic strains in humans has been documented, raising concerns over treatment failures and increased morbidity. 31 In livestock, the antibiotic residues can select resistant bacteria within the gut microbiome, which can be transmitted to humans through the food chain. 36
Challenges of Antibiotic Stewardship in Livestock
In animal husbandry, antibiotics are among the most important inputs, used as feed additives, for disease prevention (prophylaxis), and for treatment. 40 However, irresponsible and indiscriminate use of antibiotics in livestock poses significant risks to both human and animal health, primarily through the development of antibiotic resistance. After administration to animals, a substantial proportion of antibiotics are released from the animal body either as parent compounds or active intermediate metabolites into the environment.41,42 The persistent presence of antibiotics in the environmental exerts selective pressure on bacterial populations, promoting the evolution, propagation, and dissemination of ARB and ARGs. 43 The fundamental principle guiding the therapeutic application of antibiotics is to ensure the recovery of diseased animals by administering doses sufficient to eradicate or inhibit the growth of pathogenic organisms, contingent upon the pathogens’ susceptibility to the antibiotics. Nevertheless, the clinical efficiencies of numerous antibiotics have been compromised over time, resulting in the withdrawal of several antibiotic agents from the market due to increased AMR. Beyond AMR challenges, the development and approval of new antibiotics are hampered by complex clinical trial requirements and regulatory barriers, leading in lengthy approval processes. 44
Evidence suggests that antibiotic use in animal husbandry contributes more significantly to the emergence and dissemination of ARGs and ARB than use in humans, particularly in low- and middle-income countries. 15 Studies have reported a higher prevalence of multidrug-resistant bacteria in animal sources compared to human sources. 31 For example, Cheng et al 45 demonstrated that the gut flora of chickens rapidly acquired tetracycline resistance following consumption of tetracycline-supplemented feed for a week. The continued presence of antibiotic residues in the environment remains a threat to both human and animal health. This prompted the initiation of global actions aligned with one health approach to safeguard human, animal, and environmental health. These measures include national assessment and regulatory initiatives aimed at promoting the prudent use and management of antibiotics in livestock production. 22 In developing countries, surveillance and regulatory infrastructures concerning antibiotic use remain inadequate, with limited data on antibiotic usage patterns. As of 2020, several African countries did not report national sales data of veterinary antibiotics to the global AMR monitoring networks. 12 In such contexts, antibiotics could be readily accessible over the counter, often leading to irrational drug use. 26 This issue is compounded by the presence of substandard pharmaceuticals lacking active ingredients in the market with huge economic costs. 46 These substandard products fail to deliver effective doses, and coupled with low awareness among farmers, their affordability and easy accessibility contribute to extensive antibiotic use. This, in turn, exacerbates environmental contamination and the proliferation of AMR. Despite these challenges, over 80% of farmers in Africa continued to utilize antimicrobial agents in livestock to meet the rising demand for animal-derived foods. 16
Addressing the AMR issues associated with antibiotic use in livestock within developing countries necessitates adherence to global strategies promoting the judicious applications of antimicrobials. This includes establishing robust regulatory frameworks and surveillance systems for antibiotic usage. 36 However, failure to regulate and monitor antibiotic deployment risks reversing progress in controlling AMR, potentially returning societies to the pre-antibiotic era. It is important to recognize that the current reports on AMR in developing countries are likely underestimations, as they mostly rely on phenotypic detection methods, thereby representing only a fraction of the circulating ARGs. In low-resource settings, lack of access to advanced molecular techniques limits comprehensive AMU and AMR surveillance. 15 Therefore, support is required from the global AMR network to facilitate the adoption of molecular diagnostic tools, enabling a deeper understanding of the complex dynamics governing the evolution and transmission of resistance genes at the genome level.
Environmental Behaviors of Antibiotics
Antibiotics are detected across various environments using sensitive analytical techniques, however, much remains to be understood about their ecological fates. 47 Although administered at optimal therapeutic concentrations, the majority of antibiotics are excreted by animals and subsequently enter the environment, where they can remain pharmacologically active.32,48 It is only about 30% of the antibiotics metabolized within the animal body are used effectively, and the rest are eliminated through the excreta.40,49 As a result, most antibiotics consumed by animals are released into the environment, posing undesirable ecological and health effects. 30 Their presence in the environmental exerts selective pressure on bacterial communities, fostering the development and transfer of ARGs through vertical and horizontal transmissions.9,50
Detection of antibiotic residues in the environment involves solid-phase extraction (SPE) followed by liquid chromatography-tandem mass spectrometry (LC-MS/MS), which offers sensitivity and specificity to detect trace-level antibiotics. 51 In soil and manure samples, antibiotic extraction applies the use of ultrasonic extraction coupled with cleanup procedures such as the dispersive solid phase extraction. Detecting antibiotics in the environment suffers from matrix effect, low analyte concentrations, and complexity of environmental samples, which necessitate rigorous sample cleanup and validation methods. Amounts of veterinary antibiotics detected in different environments differ based on several factors including the efficiency of wastewater treatment, and land applications of wastes emanating from agricultural sources. 38 Tetracyclines, fluoroquinolones, sulfonamides macrolides, and beta-lactams are among the most commonly detected veterinary antibiotics in aquatic and terrestrial environments. 52 In surface water, the antibiotics sulfonamides have been detected from 24 to 385 ng/L, particularly in waste disposal impacted rivers. In soil and sludge samples, antibiotic residues were detected in several magnitudes higher than the surface and groundwater environments. Mobile antibiotics such as sulfonamides can be disseminated into groundwater. 53
Antibiotics are complex organic molecules characterized by various functional groups, which confer unique physiological and physicochemical properties (Figure 3). These functional groups influence their stability under different environmental conditions (pH, moisture, temperature, and radiation). Such conditions affect their physicochemical and biological behaviors, including their sorption, photoreactivity, and antimicrobial activity.41,43,54 Hydrophobicity, an important property of antibiotics, is commonly measured by an octanol-water partition coefficient (Kow). Most antibiotics have log Kow values below 5, indicating they are relatively non-hydrophobic (see Supplemental Table 1). Additionally, many of the antibiotics exhibit water solubility exceeding 1 g/L, further supporting their hydrophilic properties. 55 The physicochemical properties of antibiotics in the soil are also influenced by factors such as molecular structure, size, shape, solubility, and hydrophobicity. For instance, polar compounds can interact with suspended organic matter, which amplifies their environmental concentrations. 56 Other factors, including pH, soil structure, organic carbon content, ion exchange capacity, and climate conditions, also significantly influence the environmental behavior of antibiotics. 57 The extent of impacts of antibiotics on the environment including their bioconcentration and toxicity within the ecosystem remain largely unknown. 9 Assessing the mechanisms and concentrations of antibiotics that impart the selection of resistant bacteria strains in natural microcosms of soil, sediment, and water bodies is also a challenging task. Since antibiotic use in agriculture exceeds the antibiotic use in human medicine, most environmental antibiotic residues originate from agricultural sources, intensifying their impact on the environment. Therefore, given their complicated behaviors, the widespread presence of antibiotics in the environment can only exacerbate the development and dissemination of AMR-determinant genes, intensifying the pressure on human and animal health.57,58

The structure and selected physicochemical properties of some antibiotic classes [adapted from (54)].
Transmission and Fates of Antibiotics in the Environment
The extensive use of antibiotics in livestock, coupled with human medicinal use, leads to the dissemination and presence of antibiotic residues in the environment. Annually, approximately 3000 to 27 000 tons of animal drugs and intermediate bioactive compounds are released into the environment through animal waste disposal, free discharge of dung by grazing animals, manure dust, pharmaceutical spills, and wastewater discharges.33,59 Soil and water environments also receive antibiotic residues from container disposal, unused medicines, and wastewater discharged from drug manufacturing processes (Figure 4). From these sources, antibiotic residues and ARGs circulate among microbes, animals, humans, and the environment.27,60 The dissemination of antibiotics depends on the antibiotics’ physicochemical properties such as sorption, solubility, and persistence, and the characteristics of the transport medium as well as the receiving environment. 33 Water plays a key role in spreading antibiotics, ARGs and ARB in the environment 61 originating from human and animal sources. Wastewater from agriculture, municipal, and industrial sources often contains residual antibiotics which are important sources of antibiotics to the aquatic environment.7,29 This is a particular problem in developing countries where wastewater treatment systems including the 3-stage treatment systems are insufficient to remove antibiotics from wastewater. Hence, residual antibiotics can escape from wastewater treatment plants and contaminate water bodies such as rivers often used for irrigation contaminating the soil. 56 Detectable antibiotic residue concentrations in the soil and water environments range from nmol/L to μmol/L, often at low or sub-therapeutic levels. 62 Despite their low concentrations, the antibiotic residues can aggregate over time to a level that potentially affects the profiles of microbial communities in the environment. Poor management of solid waste generated from treatment plants can further disseminate resistance in the environment when used in soil fertilization and landfill applications. In the environment, the fate of antibiotic residues varies depending on the nature of the antibiotics and environmental conditions. 63 Once released, antibiotic can either undergo degradation or remain adsorbed onto soil and sediment particles.
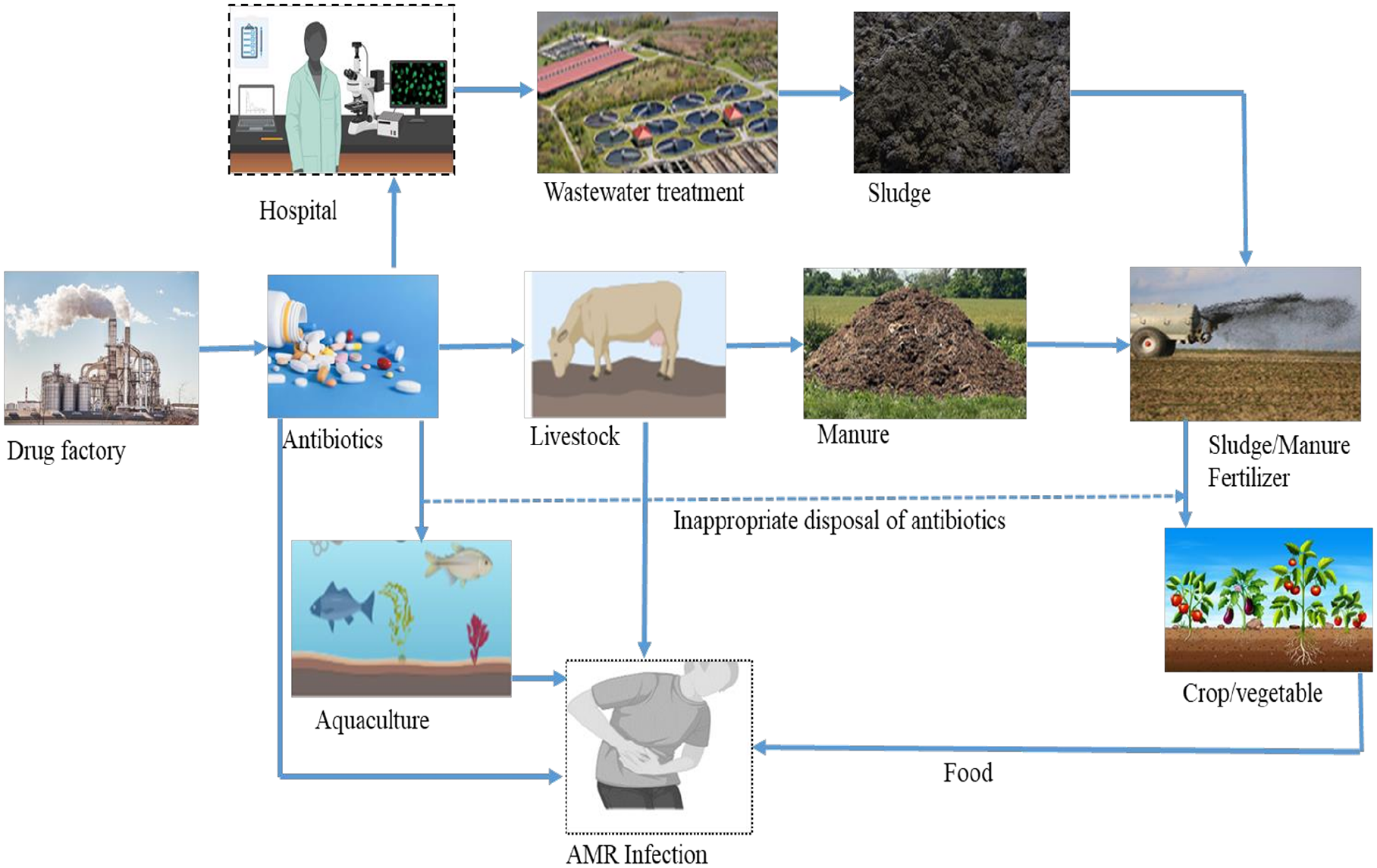
Release of antibiotic residues into the environment and human exposure routes to antimicrobials and antibiotic-resistant bacteria [modified from (24)].
Degradation
Antibiotics present in the environment are subjected to biological and non-biological factors that can lead to their breakdown. In the soil environment, the primary pathways for antibiotic degradation involve microbial activity such as decomposition, organic transformation, oxidation, and physical and chemical processes like photolysis and hydrolysis. 64 For instance, microbial degradation of antibiotics in soil amended with manure and sludge involves hydroxylation and oxidative decarboxylation reactions catalyzed by microbial enzymes. Under normal conditions, the antibiotics present in manure are degradable in the soil with a half-life of less than 30 days. 4 Some antibiotics, such as sulfachloropyridazine, degrade rapidly, whereas others like ivermectin and chlortetracycline persist for several months. Although degradation can reduce the activity of parent antibiotics, some degradation products may retain toxicity levels comparable to the original antibiotics. 65 Manure composting is another means of degrading antibiotics through microbial agents. However, the rate of degradation depends on factors such as the specific antibiotic, manure type, and composting conditions. 66 At concentrations near the minimum inhibitory concentration (MIC), biological degradation of antibiotics may be inhibited due to their antimicrobial effects. 33
Photodegradation is another mechanism by which antibiotics are broken down through photolytic processes in soil and water environments. The process of photodegradation occurs mainly at the interface between soil and atmosphere as well as on water surfaces. The rate of photolytic transformation varies significantly between soil and water due to differences in their physical conditions as soil matrices tend to be more complex and variable compared to water. Antibiotics can also undergo degradation by hydrolytic pathways in aquatic environments. Some antibiotics such as
Adsorption
Some antibiotics have a strong affinity for binding to environmental materials like soil, sediment, and sludge particles. This tendency arises from the chemical properties of antibiotic compounds, which allow them to interact with and adhere to solid surfaces in the environment. These materials often act as reservoirs or hiding spots of antibiotics, effectively sequestering them away from degradation processes. 58 As a result, the presence of antibiotics in the environment is prolonged due to their attachment to the solid particles that shield them from chemical (hydrolysis and photolysis) and biological breakdown by microorganisms that would otherwise mineralize or metabolize them. The degree of sorption can vary among different antibiotics. Even within the same class of antibiotics, sorption behaviors of antibiotics are not necessarily similar, as some antibiotics can bind tightly to sediments and soil, while others are loosely associated. This affinity is quantified by the sorption coefficient, often expressed as the distribution coefficient (Kd). The sorption coefficient of antimicrobials varies widely, from less than 1 L/kg—indicating weak sorption—to 600 L/kg, indicating strong binding affinity. 67 For instance, chloramphenicol in marine sediment exhibits a low adsorption coefficient of around 0.2 L/kg, implying limited binding and higher mobility in the environment. Conversely, enrofloxacin, a fluoroquinolone antibiotic, shows a much higher adsorption coefficient of 561 L/kg when present in soil matrices, suggesting its strong binding affinity to soil particles. 65 The mechanisms underlying these adsorption processes are multifaceted and include interactions at the molecular level. The interaction involves hydrophobic partitioning, ion interactions, and charge transfer which facilitate the binding of antibiotics to sediment and soil. 33 When antibiotic residues are present in the environmental compartments at levels exceeding the natural degradation rate, they accumulate over time in soil layers. This accumulation process often referred to as terraccumulation, involves the progressive buildup of antibiotics in the soil, especially in agricultural settings where manure and sludge are used as fertilizer. 57
Persistence in the Environment
Antibiotics can persist in both aquatic and terrestrial environments for extended periods.
63
The persistence of antibiotics in the environment raises concerns about selective pressure on bacterial communities, often promoting the development of resistant strains. Some antibiotics tend to remain longer in water due to their hydrophilic properties, which are conferred by the presence of polar functional groups.
64
Hydrophilic antibiotics may occur freely in water or interact with suspended particles. Over time, their concentrations can gradually increase in aquatic environments, potentially surpassing environmental threshold levels.
56
Antibiotics like chlortetracycline and sulfamethoxazole (SMZ) can bind to soil particles, resulting in accumulation and extended persistence.
66
Similarly, antibiotics such as erythromycin and tetracycline can persist in the environment, fostering conditions favorable for resistant bacteria proliferation. The environmental half-lives of macrolides, tetracyclines, sulfonamides, and
Effect of Antibiotic Residues on Environmental Resistome
The environment, particularly the soil and sediment, acts as a reservoir for the transmission of antibiotics, ARB, and ARGs. 68 Antibiotic pollution coupled with poor sanitation, limited surveillance, and expanding reservoirs of ARGs, collectively contributes to the proliferation of resistome—the entire collection of ARGs within a specific environment. Wastewater is also recognized as a critical reservoir of residual antibiotic compounds and ARB, thereby facilitating the spread of AMR. 38 Moreover, many antibiotics prescribed for animals are similar in chemical structure and mechanism of action to those used in human medicine. For example, amoxicillin and erythromycin, commonly used in treating human infections, are also administered to animals to promote feed efficiency. 69 Additionally, any inappropriate use of antibiotics in animals, particularly in countries with weak regulatory systems, contributes to the emergence and dissemination of AMR in the environment.
Antibiotics typically exert their effects by either killing bacteria (bactericidal action) or inhibiting their growth (bacteriostatic action). They target essential bacterial cell functions such as cell wall synthesis, DNA replication, and protein synthesis by targeting the ribosome.1,2 However, these compounds often have complex, pleiotropic effects on bacteria, affecting multiple molecular targets simultaneously. 70 This complex interaction can promote the development of resistance mechanisms in bacteria within the environment. Historically, the development of antibiotic resistance in both pathogenic and non-pathogenic bacteria was primarily attributed to clinical use of antibiotics in human medicine. However, recent evidences suggest that this perspective is too narrow to consider human medical practices as the major drivers for the emergence of antibiotic resistance. The inappropriate use of antibiotics in animals combined with poor management of animal waste significantly contributes to the expansion of AMR and environmental resistome. 35 These practices promote the selection and spread of ARB diverse ecosystems creating a broad ecological network in which ARGs can circulate among bacteria in soil, water sediment, animals, and humans.
The amounts of antibiotics detected in the environment are often at sub-therapeutic levels ranging from ng/L to

Concentration-dependent effect of antibiotics in transcription [adapted from (71)].
Advancing Strategies and Future Perspectives on Antibiotic Use in Livestock
After the golden age of antibiotics in the 1950s, several antibiotics have been withdrawn due to the emergence of resistance by pathogens. Currently, more than 70% of pathogenic bacteria are resistant to at least one of the antibiotics in use. 74 The annual global mortality rate attributable to AMR is estimated to exceed 700 000 deaths. 75 If antibiotic use is not carefully managed, projections suggest that this number could rise to 10 million deaths per year by 2050, with nearly 9 million deaths occurring in developing countries. Overall, deaths related to AMR are expected to account for approximately 44% of total global mortality in this period.76,77 Furthermore, the economic burden of AMR is substantial, impacting both human and animal healthcare systems. It is estimated to reduce gross domestic product (GDP) by 2% to 3% and incur health security costs of up to $100 trillion worldwide. 78
The spread of AMR is especially pronounced in developing countries, driven by factors such as poor surveillance, weak regulatory frameworks, inadequate monitoring of antibiotic use, insufficient diagnostic capacity, poor infection prevention, and control, and inappropriate antibiotic use practices. 79 These challenges highlight the growing concerns about environmental resistome, especially considering incidences of continued misuse of antibiotics in agriculture and over-prescription in clinical settings. 80 Additionally, the circulation of substandard antimicrobials—antibiotics that fail to deliver the correct pharmaceutical dosage or quality—further exacerbates the problem in developing countries. 81 Exposure of animals to sub-therapeutic doses of low-quality antibiotics can accelerate the development of AMR, posing significant risks to both human and animal health worldwide.
The future of antibiotic use in livestock hinges on a comprehensive, multidisciplinary approach that balances animal health and productivity with the imperative to curb antimicrobial resistance and protect public health. Advancements in alternative therapies—such as vaccines, probiotics, and improved biosecurity measures—offer promising avenues to reduce reliance on traditional antibiotics. Integrating innovative technologies like precision farming, data analytics, and IoT devices enables targeted medication practices, optimizing interventions and minimizing unnecessary antibiotic administration. Enhanced surveillance systems and stricter regulatory frameworks are essential for monitoring antibiotic usage and resistance patterns across farms and regions, facilitating timely and effective responses. Policy initiatives that incentivize responsible stewardship, coupled with education and awareness campaigns targeting farmers and stakeholders, foster a culture of sustainable and judicious antibiotic use. Additionally, ongoing research into novel therapeutics, rapid detection systems for resistant pathogens, and diagnostic tools will further strengthen efforts to combat antimicrobial resistance. Adopting a comprehensive One Health approach—integrating human, animal, and environmental health—is crucial for sustainable and responsible antibiotic use across these interconnected sectors. 9 Ultimately, collaborative efforts that combine technological innovation, policy reform, and stakeholder engagement are crucial for promoting responsible antibiotic use, reducing environmental dissemination of resistance, and safeguarding both animal welfare and human health against the global threat of AMR.
Conclusions and Recommendations
The widespread and often unregulated use of antibiotics in animal agriculture, especially for growth promotion, poses complex challenges that threaten environmental health and contribute to the global rise of AMR. This problem is particularly acute in developing countries, where limited surveillance, misuse, and weak regulations exacerbate resistance development, threatening public health and ecological stability. The use of antibiotics in livestock not only increases environmental residues and promotes resistant pathogens but also complicates efforts to trace and manage their behavior and impact. To effectively address this multifaceted crisis, comprehensive strategies are needed, including implementing rational antibiotic policies that restrict use to therapeutic purposes, strengthening surveillance systems, improving waste management, and promoting alternatives like vaccines and probiotics. Additionally, fostering international collaboration, raising awareness among stakeholders, supporting affordable interventions tailored to resource-limited settings, and investing in research for new therapeutics are vital components. Tailoring these efforts to the capacities of low-income countries and prioritizing sustainable, innovative approaches will be crucial in reducing antibiotic misuse, breaking transmission pathways of AMR, and safeguarding both human and animal health for future generations.
Supplemental Material
sj-docx-1-ehi-10.1177_11786302251357775 – Supplemental material for Antibiotic Use in Livestock and Environmental Antibiotic Resistance: A Narrative Review
Supplemental material, sj-docx-1-ehi-10.1177_11786302251357775 for Antibiotic Use in Livestock and Environmental Antibiotic Resistance: A Narrative Review by Tesfaye Admassu Abate and Alemayehu Godana Birhanu in Environmental Health Insights
Footnotes
Author Contributions
TAA and AGB conceptualized and wrote the review. Both authors approved the final manuscript.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
