Abstract
Straw burning remains a significant environmental and agricultural challenge, releasing harmful pollutants and depleting soil fertility. This study evaluates the effectiveness of 2 novel microbial formulations, NTT-02 and NTT-03, in promoting straw decomposition and enhancing soil quality. Field experiments demonstrated that NTT-02 application resulted in a 15% increase in total organic carbon (TOC), a 12% increase in total nitrogen (TN), and a 10% increase in total phosphorus (TP) compared to untreated controls. Additionally, total aerobic microbial counts increased by 1.5 times, and cellulose-decomposing microorganisms doubled in population, indicating enhanced soil microbial activity. The microbial treatments accelerated straw decomposition, reducing the breakdown period from 28 days to 14 to 18 days. Furthermore, rice yield improved by 8% to 10% relative to conventional practices. These results highlight the potential of microbial-based straw treatments as a sustainable alternative to open-field burning, promoting improved soil health, enhanced crop productivity, and environmentally responsible agricultural practices.
Keywords
Introduction
Straw is an abundant organic material readily available to rice farmers.1,2 After harvest, about 40% of nitrogen, 35% of phosphorus, 80% to 85% of potassium, and 40% to 50% of sulfur absorbed by rice plants remain in their vegetative parts.3,4 Traditionally, straw has been used as fuel, 5 cattle fodder, 6 livestock bedding, 7 and mulch. 8 More recently, it has been utilized for bio-organic fertilizer production, straw mushroom cultivation, 9 and as a raw material in industries like paper manufacturing, biofuels, and biochar. 10 Despite these applications, a large portion of straw remains underutilized due to labor shortages and seasonal constraints, leading to widespread open-field burning. This practice contributes significantly to environmental pollution, greenhouse gas emissions, and the loss of valuable soil nutrients.
Straw burning persists across many rice-producing countries, including China, India, Vietnam, and Thailand, and even in parts of Europe and North America, despite regulatory efforts.2,11,12 Driven by the need for rapid field clearance and limited waste management options, burning is favored for its simplicity. However, it releases large quantities of pollutants such as carbon dioxide (CO₂), volatile organic compounds (VOCs), nitrogen oxides (NOₓ), hydrocarbons (HCs), fine particulate matter (PM₂.₅), and soot.13 -17 These pollutants are major contributors to respiratory diseases, cardiovascular illnesses, and climate change.18,19 Exposure to emissions from straw burning is linked to serious health effects, including cancer, developmental disorders in infants, and reproductive health risks. 20 Additionally, black carbon deposition inhibits photosynthesis and lowers crop yields, while nitrogen oxides and sulfur dioxide contribute to acid rain, degrading soil and water quality.
Global initiatives have sought to mitigate the impacts of straw burning. China provides subsidies for biochar and composting; India promotes technologies like the “Happy Seeder” for direct seeding without residue removal; Vietnam supports microbial decomposition and bio-organic fertilizers through research and education programs.2,9,14 In the European Union and the United States, stricter environmental regulations encourage biomass utilization for bioenergy and biogas production rather than open burning. 15 These combined initiatives highlight a significant shift toward sustainable agricultural waste management, focusing on eco-friendly alternatives to straw burning. In particular, integrated approaches to soil fertility improvement—such as the co-application of organic amendments and biochar—are gaining attention for their potential to mitigate environmental stress while enhancing soil health and crop productivity. 21
Various chemical methods, including pyrolysis, gasification, and liquefaction, have been investigated to convert straw into bio-oil, syngas, and biochar.22 -24 However, these methods often require pure chemicals, complex processes, and are associated with secondary pollution. In contrast, biological methods such as fermentation, anaerobic digestion, and aerobic composting offer advantages of lower energy requirements, minimal environmental impact, and simpler implementation.25 -29 Nevertheless, the slow biodegradation rate of straw, attributed to its cellulose, hemicellulose, and lignin content, remains a barrier for large-scale applications. 22 Pretreatment using microbial decomposition and enzymatic hydrolysis presents an efficient, eco-friendly solution.30 -33 By promoting soil microbial activity and improving nutrient cycling, these practices contribute to long-term soil fertility and support sustainable crop production, even under challenging conditions such as soil salinity. 21
Despite advances, practical challenges like straw collection, transportation, and storage after harvest limit widespread adoption of biological treatments.34,35 Consequently, many farmers resort to field burning. On-field microbial treatments present a promising alternative by accelerating decomposition, enriching soil organic matter, and improving soil conditions under controlled irrigation systems.36,37 Several microbial products containing cellulolytic bacteria, actinomycetes, and Trichoderma fungi have demonstrated potential in enhancing straw degradation and converting it into humus. 38
While rice straw is the primary focus due to its abundance in Vietnam, other biomass sources such as corn stalks, sugarcane bagasse, wheat straw, legume residues, coconut coir, forestry waste, and food processing by-products also offer viable substrates for microbial decomposition. Their application can improve soil health, enhance water retention, and reduce agricultural waste, contributing further to sustainable farming systems. 39
Despite available alternatives, open-field straw burning remains widespread, exacerbating air pollution, greenhouse gas emissions, and soil degradation. Conventional alternatives such as composting and biochar production require significant labor and investment, limiting their scalability. In this context, microbial decomposition offers a sustainable, cost-effective solution that can be integrated directly into existing farming practices. By enhancing soil microbial activity and nutrient cycling, it supports long-term soil fertility and sustainable crop production. 40
Despite advancements in microbial decomposition techniques, most previous studies have relied on single-strain inoculants and controlled composting processes, limiting their practical application in large-scale agricultural systems. 41 Moreover, the need for external facilities and labor-intensive straw collection further constrains the adoption of these methods. To address these challenges, this study introduces 2 novel multistrain microbial formulations, NTT-02 and NTT-03, composed of fungi (Trichoderma spp., Chaetomium spp.), bacteria (Bacillus subtilis), actinomycetes, yeast (Saccharomyces cerevisiae), and cellulase enzyme complexes. These formulations are designed to accelerate lignocellulose breakdown through synergistic microbial activities and are directly applied to rice fields without prior straw collection. By developing and validating these in-field microbial treatments, this study aims to bridge the gap between laboratory-based microbial technologies and practical, scalable solutions for sustainable straw management in agriculture.
This study evaluates the effectiveness of 2 novel microbial formulations, NTT-02 and NTT-03, developed specifically to accelerate straw decomposition and improve soil health. Unlike previous approaches focusing on single-strain inoculants or off-field composting, this research applies multistrain microbial products directly in the field, eliminating the need for straw collection and enabling scalable adoption by farmers. Soil quality indicators, including pH, electrical conductivity (EC), total organic carbon (TOC), organic matter (OM), total nitrogen (TN), total phosphorus (TP), aerobic microbial populations, and cellulose-degrading organisms, were comprehensively analyzed.
The novelty of this study lies in the development and direct field application of multistrain microbial formulations designed to enhance both cellulose and lignin degradation, enabling rapid in-situ straw decomposition without labor-intensive processes. Its importance is underscored by its contribution to sustainable agriculture through reducing chemical fertilizer dependency, improving soil health, and offering a viable alternative to environmentally harmful straw burning practices, particularly in rice-producing regions vulnerable to climate change.
Experiment
Model of Biological Products
In this study, 3 microbial preparations were used, representing 1 commercial product on the market and 2 preparations researched at Nguyen Tat Thanh University. Specifically, these include:
- Commercial straw decomposer (Bio decomposer) with the main component being Bacillus Subtilis: 1 × 106CFU/mL. It includes 11 decomposing microbial strains: Aspergillus oryzae, Aspergillus terreus, Emericella nidulans, Pseudo eurotium zonatum, Mucor plumbeus, Penicillium variabile, Trichoderma hamatum, Trichoderma harzianum, Bacillus sp., Streptomyces sp.
- Straw decomposer NTT02 with the main components being beneficial microbial strains such as Trichoderma hamatum (1 × 108 CFU/mL), Trichoderma harzianum (1 × 108 CFU/mL), Trichoderma asperellum (1 × 108 CFU/mL), Trichoderma viride (1 × 108 CFU/mL).
- Straw decomposer NTT03 with the main components organic matter: 15%; Trichoderma spp: 1 × 106 CFU/g; moisture content: 30%; a fermented product of billions of beneficial microorganisms such as Actinomycetes sp., Aspergillus sp., Saccharomyces cerevisiae, Chaetomium spp., Mucor sp., Bacillus subtilis, along with many beneficial microorganisms combined with a cellulase enzyme complex. The dosage differences between NTT-02 and NTT-03 were based on prior optimization trials and microbial density assessments. NTT-02 contained a higher viable microbial density (approximately 1 × 109 CFU/mL), while NTT-03 had a density of approximately 5 × 108 CFU/mL. To achieve comparable total microbial application loads across treatments, 350 mL of NTT-02 and 700 mL of NTT-03 were applied per hectare. These dosages were also aligned with preliminary efficacy tests and manufacturer recommendations to balance microbial efficacy with practical application feasibility in field conditions.
This study presents a novel approach to microbial-based straw decomposition by integrating NTT-02 and NTT-03, two innovative microbial formulations designed to improve lignocellulosic breakdown and improve soil fertility. Unlike conventional decomposers that rely on a limited number of microbial strains, NTT-02 contains high concentrations of Trichoderma species, which are highly effective in the degradation of cellulose and hemicellulose while promoting beneficial soil microbial activity. NTT-03 expands on this by incorporating actinomycetes, Bacillus subtilis, Aspergillus spp., Saccharomyces cerevisiae, Chaetomium spp., and a cellulase enzyme complex, ensuring a more efficient breakdown of straw residues and improving nutrient cycling.
The microbial formulation NTT-03 was designed to leverage the complementary mechanisms of multiple microbial groups. Trichoderma spp. are filamentous fungi known for producing a wide range of cellulolytic and ligninolytic enzymes, including cellulase, hemicellulase, and laccase, which facilitate the rapid breakdown of straw lignocellulosic structures. Meanwhile, Actinomycetes secrete extracellular enzymes capable of degrading complex polysaccharides and contribute to lignin decomposition while producing bioactive compounds that suppress opportunistic soil pathogens. The inclusion of both Trichoderma spp. and Actinomycetes is intended to harness a synergistic effect: fungal hyphae mechanically disrupt straw tissues, increasing the surface area for bacterial enzyme access, while bacterial metabolism further degrades released compounds. Preliminary compatibility tests showed no significant antagonism between the strains, and the functional complementarity between fungal and bacterial enzymatic systems is anticipated to enhance the overall decomposition rate and nutrient release in treated soils.
Another key distinction of this study is its direct in-field application of microbial products. Many previous studies have focused on composting or biogas production in controlled environments, whereas this research applies microbial treatments directly to rice fields. This eliminates the need for labor intensive residue collection and transportation, accelerating the decomposition process while simultaneously enriching soil fertility in situ. By integrating microbial decomposition into existing agricultural practices, this approach offers a practical and scalable solution for farmers, reducing dependence on chemical fertilizers.
The study also distinguishes itself through a comprehensive assessment of the environmental and soil impacts. Instead of focusing on single-parameter evaluations, the research examines a broad set of soil health indicators, including pH, electrical conductivity, total organic carbon, total nitrogen, total phosphorus, total aerobic microbes, and cellulose-decomposing microorganisms. This multidimensional analysis provides a deeper understanding of how microbial treatments influence both soil quality and crop productivity over time.
Beyond its agronomic benefits, the study also evaluates the economic feasibility and environmental impact. The findings demonstrate a significant reduction in fertilizer and pesticide costs, making microbial treatments not only environmentally sustainable but also economically viable for large-scale adoption. Additionally, by offering a practical alternative to straw burning, this approach contributes to reducing air pollution and greenhouse gas emissions, addressing a critical environmental issue in rice farming regions. By integrating innovative microbial formulations, real-world field applications, multiparameter soil assessments, and economic evaluations, this study provides a holistic and scalable solution for sustainable straw management. This combination of factors sets it apart from previous research and highlights its contribution to advancing microbial-based agricultural sustainability.
Experimental Model
The experiment was carried out during the summer-autumn crop season in Song Ray Commune, Cam My District, Dong Nai Province, Vietnam, as shown in Figure 1, over a period of 3 weeks. A completely randomized block design was applied, with 4 treatment groups, each replicated 3 times on a total experimental area of 2600 m², divided into 4 equal plots of 650 m². The study used plowed rice straw left after harvesting, with an estimated coverage of approximately 3 to 5 tons per hectare.

Experimental model of this study.
The microbial treatments were applied in a 2-step process. In the first application, the microbial preparations were dissolved in water and sprayed evenly over the straw-covered fields according to the recommended dilution ratios. The field was then flooded to facilitate microbial decomposition. After 7 days, a second microbial application was performed, followed by plowing and leveling of the field. The treated straw was allowed to decompose for an additional 6 to 8 days prior to the final field preparation for rice cultivation.
The microbial treatments included:
• Biodecomposer (commercial product): 350 mL per 650 m2, dissolved in a 16 to 18 L spray bottle and applied evenly.
• NTT-02 (Microbial formulation with Trichoderma strains): 350 mL per 650 m2, applied similarly.
• NTT-03 (Microbial formulation with mixed beneficial microbes and cellulase enzymes): 700 mL per 650 m2.
• Control plot: No microbial application for comparison.
Soil and microbial activity assessments were conducted at 3 key intervals: before treatment, during treatment (7 days after application), and after treatment (14 days after application). The experimental design ensured an accurate evaluation of the effectiveness of microbial decomposition, improvement of soil quality, and nutrient enhancement. By incorporating these details, the study provides a clearer and more reproducible methodology for sustainable straw management.
The experiment was arranged in a completely randomized block design with 3 replications, which involved the plowing of straw into the soil with and without the use of 3 different microbial preparations.
- Sample A: Straw was plowed into the soil and sprayed with Bio Decomposer microbial preparation.
- Sample B: Straw plowed into the soil and sprayed with the NTT02 microbial preparation.
- Sample C: Straw plowed into the soil and sprayed with the NTT03 microbial preparation.
- Sample D: Straw plowed into the soil, no microbial preparation sprayed (control).
This study provides several new insights into straw treatments, contributing to both scientific understanding and practical applications in sustainable agriculture. The results demonstrate that NTT-02 and NTT-03 outperform conventional microbial decomposers due to their unique multistrain compositions. NTT-02, enriched with Trichoderma species, accelerates cellulose and hemicellulose, while NTT-03, which includes actinomycetes, Bacillus subtilis, Aspergillus spp., Saccharomyces cerevisiae, Chaetomium spp., and a cellulase enzyme complex, enhances lignin degradation, resulting in faster and more efficient decomposition compared to traditional single strain treatments.
Singh et al 7 highlighted the economic and environmental advantages of sustainable rice straw management practices over open field burning, demonstrating how alternative methods can reduce air pollution and improve soil health. Their study emphasized the importance of integrating microbial treatments as a feasible solution to improve nutrient recycling and crop productivity. 38 Similarly, Singh and Arya 13 reviewed various rice straw management strategies, concluding that microbial decomposition is among the most cost-effective and environmentally friendly options for large-scale agricultural adoption. 42
These findings align with the results of this study, in which the application of microbial treatments NTT-02 and NTT-03 was shown to accelerate straw decomposition, enrich soil nutrients and reduce the need for chemical fertilizers. Building on previous research, this study provides quantitative evidence supporting the large-scale feasibility of microbial-based straw decomposition as an economically viable and sustainable alternative to conventional disposal methods.
Beyond improving straw decomposition efficiency, the study confirms that direct in-field microbial application is a practical and scalable alternative to straw burning. By integrating microbial decomposition into existing farming practices, this method reduces labor costs, eliminates the need for transportation, and simultaneously improves soil fertility, making it a feasible solution for farmers. Additionally, microbial treatments positively influence multiple soil health indicators, including pH balance, electrical conductivity, total organic carbon, nitrogen retention, and microbial diversity, reinforcing their role in long-term soil improvement beyond the immediate benefits of residue decomposition.
The study also highlights the economic advantages of microbial treatments, showing a reduction in fertilizer and pesticide costs while increasing rice yield by approximately 15%. This quantification of financial benefits provides new practical insights into the cost effectiveness of microbial decomposition, addressing concerns about adoption on a larger scale. Additionally, the findings emphasize the importance of adaptability to different soil and environmental conditions, since the microbial decomposition efficiency varies according to soil type, moisture levels, and temperature. These observations underscore the need for adaptive application strategies to optimize performance in diverse agricultural settings.
By integrating advances in microbial formulations, direct field application, soil health benefits, economic feasibility, and environmental adaptability, this study expands the knowledge base on microbial straw treatments and provides a scientifically validated, cost-effective, and scalable approach for sustainable agricultural waste management.
NTT-02 and NTT-03 were selected for their ability to enhance straw decomposition and improve soil health by accelerating the breakdown of lignocellulosic materials. These formulations were developed to address the limitations of conventional microbial products, particularly their slow decomposition rates, by leveraging a combination of highly effective microbial strains.
NTT-02 was chosen due to its high concentration of Trichoderma strains, including Trichoderma hamatum, Trichoderma harzianum, Trichoderma asperellum, and Trichoderma viride. These fungi are known for their strong decomposition capabilities of cellulose and hemicellulose, promoting the rapid conversion of straw into organic matter. Additionally, Trichoderma species improve soil microbial activity and contribute to plant disease suppression by outcompeting soil pathogens.
NTT-03 was selected for its broader microbial composition, which incorporates actinomycetes, Bacillus subtilis, Aspergillus spp., Saccharomyces cerevisiae, Chaetomium spp. and a cellulase enzyme complex. This diverse microbial community improves the efficiency of decomposition by targeting both cellulose and lignin components, ensuring a more comprehensive breakdown of straw residues while enhancing the nutrient cycling in the soil.
Both formulations are novel microbial treatments developed at Nguyen Tat Thanh University, designed to optimize biological decomposition in rice fields. Unlike conventional commercial decomposers that typically contain a single microbial strain, these formulations integrate multiple complementary microbial groups, enhancing decomposition rates and soil fertility. Their application in this study represents a new approach to microbial-based straw management, offering an innovative and sustainable alternative to traditional residue disposal methods.
The straw treatment process using microbial preparations is carried out in 5 steps as described in Figure 2, specifically as follows:
- After harvesting rice, if the field lacks water, flood the field to soak the straw for 1 to 2 days. Then, drain the water and evenly spread phosphorus or lime (if needed) on the field surface.
- Dissolve the microbial preparation (see the product packaging for mixture preparation) in water and spray it evenly over the straw and across the entire field. - Treat wild rice (volunteer rice, wild rice, 2-layer rice): Use a wild rice management preparation by spreading or spraying (600 g/2600 m2) the product evenly on the field surface. - Treat the straw using experimental spraying of 3 different microbial preparations (Bio Decomposer, NTT-02, NTT-03) and 1 control plot. Each plot is 650 m2. Table 1 describes the formulas and methods for preparing the microbial preparations used in the experiment.
- After spraying the microbial preparation, flood the field with water to a depth of 4 to 6 cm above the field surface. Use a plow to turn the straw, ensuring that the straw is thoroughly mixed into the soil. Soak the field for 7 days, then drain the water.
- Dissolve the microbial preparation (prepare the mixture similarly to the first application as described in Table. . .) in water and spray it evenly across the entire field surface.
- Flood the field with water to a depth of 4 to 6 cm and soak for about 7 days. Then, plowing the soil, leveling the field surface, and draining the excess water.
- Finally, leave the field fallow for 6 to 8 days, then flood the field and puddle before sowing or transplanting rice according to the usual procedure. The total treatment time is less than 3 weeks.
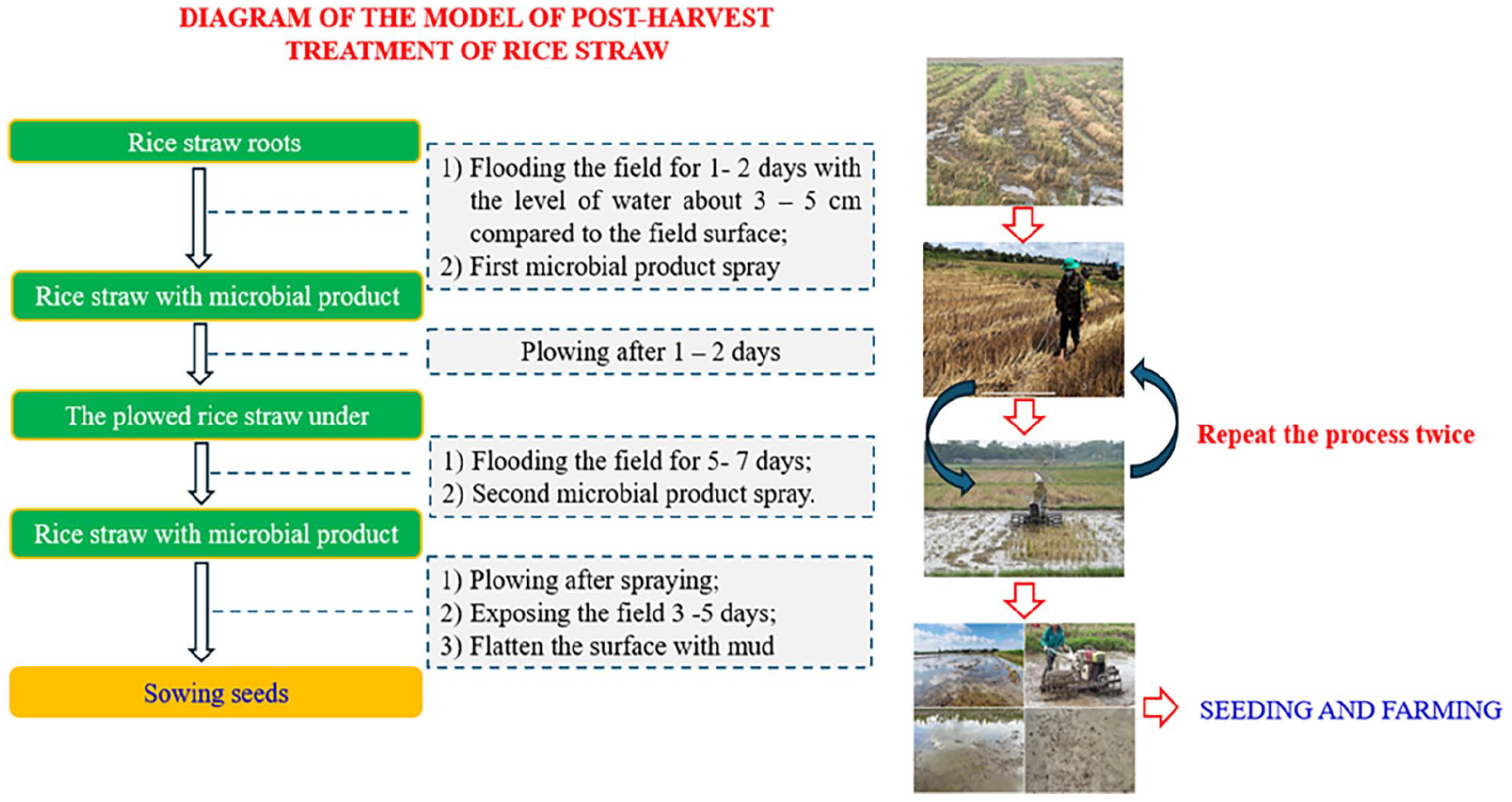
Flow chart of straw treatment using microbial preparations.
Formula and method of used microbial products used.

Meteorological Conditions During Experiment
During the experimental period (April to July 2023), the climatic conditions in the study region were characterized by high temperatures and humidity, corresponding to the summer season in southern Vietnam. The recorded average temperature ranged from 28°C to 32°C, providing an optimal environment for microbial activity and enzymatic degradation processes. Previous studies have demonstrated that cellulolytic microorganisms, such as Trichoderma spp., Bacillus subtilis, and actinomycetes, exhibit peak enzymatic activity, particularly cellulase production, within the temperature range of 30°C to 35°C (Bhattacharyya et al 2 ). The favorable temperature likely accelerated the breakdown of lignocellulosic components in straw residues during the experiment.
Furthermore, total precipitation during the 4-month period reached approximately 600 mm, averaging 150 mm per month. This moderate rainfall was sufficient to maintain optimal soil moisture levels for microbial colonization and metabolic activity, without causing prolonged waterlogging that could inhibit aerobic microbial processes. High relative humidity, recorded at an average of 80%, further contributed to maintaining surface soil moisture, facilitating microbial proliferation and straw decomposition. Elevated soil moisture and humidity are known to enhance the microbial degradation of complex organic materials, promoting faster conversion of cellulose into simpler organic compounds. Collectively, these meteorological conditions—optimal temperature, moderate rainfall, and high humidity—created a highly conducive environment for microbial-driven straw decomposition, supporting efficient organic matter transformation and nutrient cycling in the field.
Methods of Sampling and Monitoring Soil Samples
The monitoring method to determine the values of the parameter content in the soil is carried out according to the technical regulations for environmental monitoring issued by the Ministry of Natural Resources and Environment. Sample processing after collection and before analysis follows the standards US EPA Method 3051A; TCVN 6649:2000 (ISO 11466:1995); US EPA Method 3050B; US EPA Method 3060A.
➢
➢
➢
➢
➢
Sampling Location and Sampling Frequency
Samples are taken on the principle of random sampling. Each point within the study area has an equal chance of being selected. This method helps eliminate bias and accurately reflects the characteristics of the sample area. Therefore, there are a total of 4 experimental treatments, including 1 control treatment and 3 treatments using microbial preparations. Figure 3 illustrates the sampling locations in the actual field. In each experimental field (650 m²/treatment), samples are taken from 5 points (4 corners and the center). Sampling is carried out in 3 phases: before treatment, during treatment (7 days after spraying), and after treatment (14 days after spraying). Each sampling phase collects approximately 2 kg of surface soil (ranging from 0 to 20 cm deep). Therefore, the total number of samples is 4 (treatments) × 5 (samples) × 3 (phases) = 60 samples.
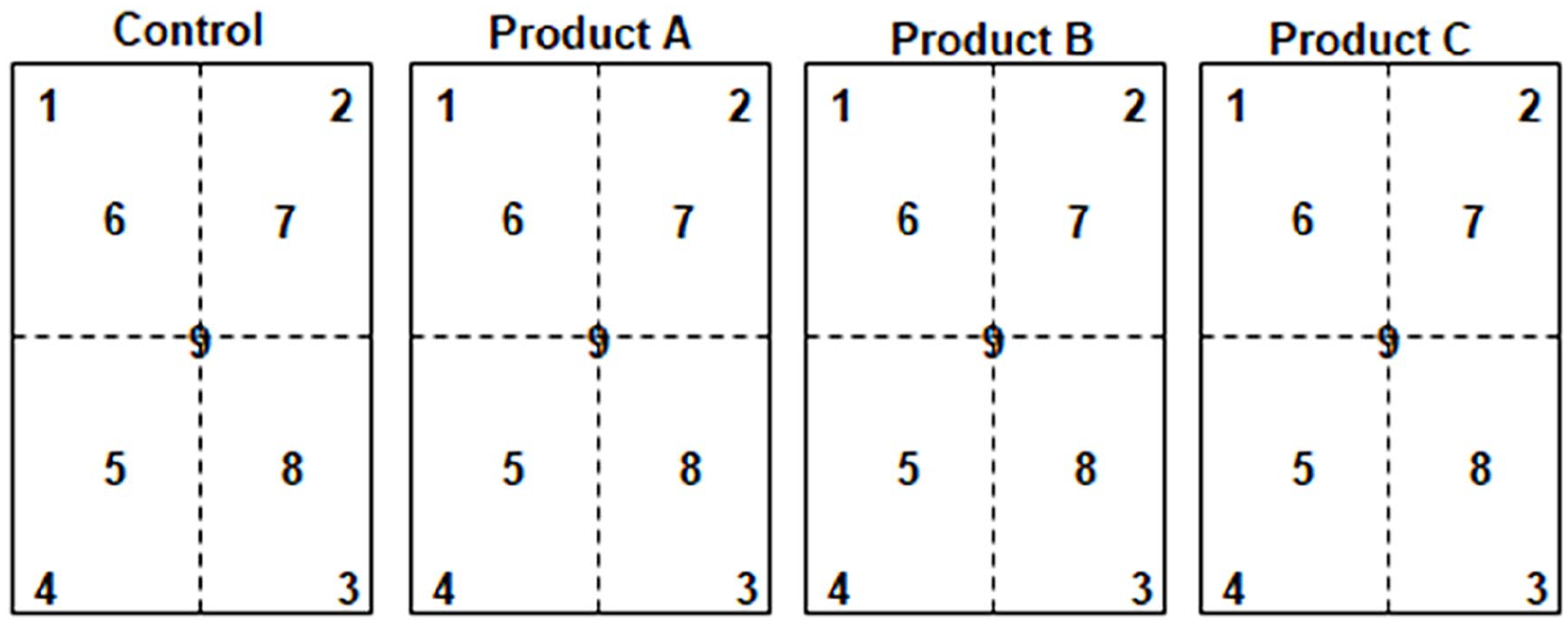
Illustration of sampling points in the actual field.
The samples will be uniformly coded with the control labeled as A and treatments using microbial preparations labeled as B, C, and D, corresponding to the control treatment (D) and the treatments with microbial preparations A, B, and C.
Method of Analyzing Indicators
Chemical, physical, and microbiological indicators are determined according to Vietnamese standards, including the following.
- Total organic carbon (TOC) and organic matter (OM): TCVN 9294:2012;
- Nitrogen content (N): TCVN 8557:2010;
- Phosphorus content (P): TCVN 8940:2011;
- Soil pH: TCVN 5979:1995;
- Moisture: TCVN 9297:2012;
- Electrical Conductivity: TCVN 6650:2000;
- Total aerobic microbes: TCVN 4884:2005;
- Cellulose-decomposing microorganisms: TCVN 6168:2002.
Potential Limitations of Microbial Treatments
Although microbial treatments have shown significant potential in accelerating straw decomposition and increasing soil nutrient availability, several limitations may affect their overall effectiveness in different agricultural settings. One major factor is variability in soil types, as differences in soil composition, texture, and organic matter content can influence microbial activity. Sandy soils with low organic content may provide less favorable conditions for microbial proliferation, while clay-rich soils with poor aeration could slow decomposition rates. Furthermore, variations in soil pH and nutrient availability may affect the survival and enzymatic efficiency of microbial strains.
Environmental conditions also play a critical role in the success of microbial treatment. Temperature, moisture levels, and rainfall patterns can affect decomposition rates, with microbial activity generally being more effective under warm and humid conditions. However, excessive rainfall can lead to nutrient leaching or dilution of microbial products, reducing their efficiency, whereas prolonged dry conditions can limit microbial metabolism and slow the breakdown of organic matter.
Field management practices, including plowing depth, irrigation frequency, and fertilizer application, also influence the effectiveness of microbial treatments. Proper field preparation is necessary to ensure optimal microbial interaction with straw residues. Additionally, the presence of native soil microbes and preexisting microbial diversity can create competition, potentially affecting the establishment and functionality of introduced microbial strains.
To improve the adaptability of microbial treatments, more research is needed to evaluate their performance under different soil and climatic conditions. Adjusting application rates based on soil properties and environmental factors may improve consistency and scalability. Acknowledging these limitations allows for a more balanced discussion of microbial treatments, ensuring their practical application in sustainable straw management strategies.
Potential Ecological Risks of Microbial Treatments
While microbial treatments offer significant agronomic and environmental benefits, potential ecological risks must be carefully considered. The introduction of exogenous microbial strains could disrupt native soil microbial communities through competition, altering soil biodiversity and ecological balance. In rare cases, non-native microbes could proliferate beyond the intended site or exhibit unintended interactions with soil fauna or flora. Furthermore, horizontal gene transfer between introduced strains and indigenous microbes could theoretically occur, raising concerns about the spread of antibiotic resistance genes.
However, the microbial strains used in NTT-02 and NTT-03 formulations—primarily Trichoderma spp., Bacillus subtilis, and beneficial actinomycetes—are recognized as Generally Recognized As Safe (GRAS) organisms with established histories of safe agricultural application. Prior to field deployment, laboratory assessments confirmed their non-pathogenicity and ecological compatibility under local soil conditions. No evidence of toxin production or pathogenic behavior was observed. Nevertheless, to ensure long-term environmental safety, it is recommended that periodic monitoring of soil microbial diversity and ecological parameters be conducted, especially under large-scale application scenarios.
By integrating microbial risk assessments and adaptive management strategies, microbial-based straw treatments can be deployed responsibly to maximize their agricultural benefits while minimizing potential ecological disturbances.
Results and Discussion
Assess Soil Quality Before Using Biological Products
Figure 4 illustrates the actual condition of straw stubble in the experimental field before decomposition using microbial preparations. It is easily observed that, before the application of microbial preparations, straw stubble often remains on the soil surface, forming a thin layer in the field. This layer typically has a bright yellow or light green color, with a height typically ranging from 10 to 15 cm. It emits an unpleasant odor and retains the hard texture of the original rice plant.

Straw stubble in the experimental field before decomposition using microbial preparations.
Evaluate Soil Quality After Using Biological Products
Figure 5 illustrates the actual condition of straw stubble after decomposition using microbial preparations for 7 days. Generally, after 7 days of use, the experimental plots exhibited changes in color, odor, and texture. Specifically, the straw stubble changed from bright yellow to brown, with a uniform color throughout the field, which is a good indicator of the decomposition process. The color transition from yellow to brown indicates that microorganisms have broken down organic matter. Furthermore, the odor of the straw stubble after 7 days of using microbial preparations changed from an unpleasant, foul smell to a more natural, earthy odor. A pleasant and mild odor is a positive indicator of the decomposition process. Furthermore, the texture of the straw after 7 days of microbial treatment became softer and significantly reduced in mass. The decomposition of the organic matter within the straw created smaller, finer particles. Soft and fine-textured straw, without the initial rough patches, is a good indicator of the decomposition process.

Straw stubble after 7 days of decomposition using microbial preparations. ((D) Control treatment without microbial preparations; (A) Treatment with NTT-01; (B) Treatment with NTT-02; (C) Treatment with NTT-03).
In summary, after 7 days of using microbial preparations, the effectiveness of straw decomposition is evident. The color, odor, and texture of the straw improved significantly, creating high-quality organic compost to enhance the soil nutrient content.
Figure 6 illustrates the actual condition of straw stubble after 14 days of decomposition using microbial preparations. In the treatments, there have been significant changes in color, odor, and texture. Specifically, the color changed from brown to dark brown with a uniform color across the field. This is an excellent indicator of the decomposition process, showing that microorganisms have fully broken down the organic matter in the straw stubble. The odor also changed from an earthy or natural smell to that of organic compost. A nonoffensive odor is an ideal indicator of the decomposition process. Additionally, the texture has been optimally refined, changing from soft and fine to very fine, with a substantial reduction in mass. The decomposition of the organic matter within the straw stubble has created ultrafine particles. Straw stubble with a very fine texture, without strands or clumps of the original straw, is an excellent indicator of the decomposition process.

Straw stubble after 14 days of decomposition using microbial preparations. ((D) Control treatment without microbial preparations; (A) Treatment with NTT-01; (B) Treatment with NTT-02; (C) Treatment with NTT-03).
In summary, after 14 days of use of microbial preparations, the straw stubble has undergone significant sensory changes. Color, odor, and texture have been maximally improved, creating high-quality organic compost to improve soil nutrient content. This is a cost-effective and environmentally friendly method for post-harvest straw treatment. However, to get a more accurate assessment, specific measurements and comparisons with reference samples or standards are necessary.
Evaluate the Parameters of Soil Properties After Using Microbial Products to Decompose Straw
Soil pH
Analyzing and testing soil pH in agriculture, especially in rice-growing areas, is crucial because it directly affects plant growth. Soil with an inappropriate pH can limit the availability of essential nutrients for crops, thereby affecting rice yield and quality. When testing microbial preparations for the treatment of straw and stubble, understanding soil pH is an important factor. Microbial preparations often perform best within a specific pH range. For example, some bacteria may function well at neutral pH but not as effectively in acidic or alkaline conditions. Testing pH helps determine whether a specific microorganism can operate effectively in the actual soil conditions.
Figure 7 describes the changes in soil pH in 4 sample areas (A, B, C, D) during 3 phases: before, during and after treatment with microbial preparations. According to the data, area A showed a pH decrease from 6.36 before treatment to 5.4 after treatment. Similarly, area B started with a pH of 5.94 and decreased slightly to 5.62; area C from 6.34 decreased to 5.42, and area D from 6.08 decreased to 5.32. Overall, all 4 areas showed a decrease in soil pH after using the preparations, with the most significant decreases observed in areas C and D, with reductions of more than 0.9 and 0.76 pH units, respectively. This indicates the effectiveness of straw treatment preparations in these areas. On the chart and data, it can be concluded that the use of microbial preparations reduces soil alkalinity, helping to balance the soil environment, particularly in areas with high pH such as C and D. These results provide valuable information to manage and improving soil quality in fields.

Evaluation of changes in the pH parameter in experimental soil samples.
The observed reduction in soil pH following microbial treatment is consistent with previous findings by Bhattacharyya et al, 2 who reported that microbial decomposition releases organic acids during cellulose and lignin breakdown. The acidification effect may enhance nutrient availability, particularly for phosphorus and micronutrients, benefiting subsequent rice cultivation. However, excessive acidification could be detrimental in soils already prone to low pH; therefore, application rates should be adjusted based on initial soil conditions.
The observed decline in soil pH across all treatments is likely due to the production of low-molecular-weight organic acids (eg, acetic acid, lactic acid) during microbial degradation of lignocellulosic residues. Moderate acidification can be beneficial in slightly alkaline soils, improving the solubility of phosphorus and micronutrients critical for rice growth (Bhattacharyya et al 2 ). However, sustained or excessive acidification could lead to aluminum mobilization and nutrient imbalances detrimental to plant health. Therefore, periodic soil pH monitoring and appropriate amelioration strategies, such as lime application, may be necessary in long-term applications
Electrical Conductivity (EC)
Controlling and analyzing soil electrical conductivity (EC) provides numerous important benefits in agriculture and environmental management. First, it allows for the determination of soil salinity levels, which is essential in areas affected by saltwater intrusion or when irrigation water with high salt content. Additionally, monitoring EC helps accurately gauge the necessary nutrients for crops, such as nitrate and potassium, thereby optimizing nutrient management and increasing yield. This also helps to optimize irrigation practices by providing better information on soil moisture levels. Finally, analyzing CE offers valuable information to detect environmental issues, such as chemical leaks. Therefore, controlling and analyzing soil CE not only ensures healthy crop development but also evaluates soil environmental safety.
Figure 8 illustrates changes in soil EC in 4 sample areas (A, B, C, D) during 3 phases: before, during and after treatment with microbial preparations. In area A, the EC increased significantly from 90 to 168 µS during treatment and then decreased to 76 µS after treatment. In area B, the EC decreased slightly from 114 to 102 µS during treatment and more dramatically to 70 µS after treatment. In area C, the EC decreased dramatically from 154 to 64 µS during treatment, but then increased to 110 µS after treatment. In area D, the EC remained almost unchanged during treatment, stabilizing at 94 to 96 µS, and then decreased slightly to 76 µS after treatment.

Evaluation of changes in the EC parameters in experimental soil samples.
In general, all 4 areas showed a final decrease in EC, indicating improved soil quality after using the microbial preparations. However, the degree of decrease and fluctuation in EC varied between areas. Area A exhibited the most significant fluctuations during treatment, while area C experienced a sharp decrease during treatment but increased again after treatment. Based on these results, it can be concluded that the use of microbial preparations positively impacts the improvement of soil EC, improving water and nutrient retention capacities and, thus, improving soil quality in the fields.
The changes in soil electrical conductivity (EC) observed after microbial treatment highlight important implications for soil fertility and salinity management. A moderate decrease in EC, as recorded particularly in the NTT-02 and NTT-03 treated plots, suggests enhanced nutrient uptake and stabilization of soil ionic balance. This is likely attributable to the microbial decomposition of straw, which releases soluble organic compounds and facilitates the gradual mineralization of nutrients, thus reducing the accumulation of free salts in the soil solution. These results align with the findings of Bhattacharyya et al, 2 who reported that microbial treatments can buffer soil salinity and improve nutrient dynamics. However, the variations in EC trends among treatment plots also suggest that microbial efficiency may be influenced by soil moisture content, initial salinity levels, and microbial competition. Managing soil EC within optimal ranges is critical for maintaining root health, nutrient availability, and overall crop productivity, especially in rice farming systems sensitive to salt stress.
Variations in soil electrical conductivity (EC) among different treatments and areas may be attributed to dynamic processes involving microbial-driven solubilization of organic and inorganic nutrients, followed by leaching events during rainfall. For instance, the initial EC rise observed in Area A could reflect the rapid release of soluble ions from microbial straw degradation, while subsequent declines could result from nutrient leaching or microbial immobilization. Such fluctuations highlight the need for synchronized nutrient management strategies to maximize microbial treatment benefits while minimizing nutrient losses through leaching
Total Dissolved Solids (TDS) Content
Figure 9 illustrates the changes in the totaldissolved solids (TDS) levels of the soil in 4 sample areas (A, B, C, D) through 3 phases: before, during and after treatment with microbial preparations. In area A, TDS levels increased significantly from 36 to 72 mg/L during treatment and decreased to 30 mg/L after treatment. In area B, TDS levels decreased from 48 to 42 mg/L during treatment and continued to decline sharply to 24 mg/L after treatment. In area C, TDS levels decreased significantly from 64 to 24 mg/L during treatment, but increased to 50 mg/L after treatment. In area D, TDS levels remained almost stable during treatment, from 40 mg/L slightly decreasing to 38 mg/L and further decreasing to 28 mg/L after treatment.

Evaluation of changes in the TDS parameter in experimental soil samples.
Through analysis, it can be seen that the use of microbial preparations affects TDS levels in the soil in each area differently. Areas A and C experienced more significant fluctuations compared to areas B and D during the treatment process. In particular, area B showed a continuous and significant decrease in TDS levels in all 3 phases, indicating a positive effect of using the microbial preparations. In contrast, area C, after a significant decrease during the treatment phase, showed an increase in the end, necessitating an examination of the causes and treatment methods in this area. In general, the application of microbial preparations helps improve soil quality by reducing TDS levels, although the effectiveness of treatment depends on the specific characteristics of each area.
The observed decrease in total dissolved solids (TDS) levels after microbial treatment suggests improved soil chemical balance and reduced salinity stress. TDS reflects the concentration of soluble salts, minerals, and organic compounds in soil water, which can influence plant water uptake and nutrient absorption. Microbial decomposition of straw facilitates the gradual mineralization of organic residues, leading to a more controlled release of nutrients and reduced salt accumulation in the soil solution. These findings are consistent with previous studies,2,9 which reported that microbial inoculants enhance soil nutrient cycling while preventing excessive salt build-up. Furthermore, the stabilization of TDS levels supports the establishment of a healthier rhizosphere environment, critical for sensitive crops like rice. However, variations in TDS trends among treatment plots may be attributed to differences in initial soil conditions, microbial metabolic activity, and local microclimate factors such as rainfall and evapotranspiration rates. Maintaining optimal TDS levels is essential to prevent osmotic stress in plants, particularly during early growth stages, and to ensure sustainable soil fertility.
Organic Odorants (OM)
The analysis and testing of organic matter (OM) in agricultural soil, particularly in rice-growing areas, plays an indispensable role in ensuring the health and growth potential of crops. Organic matter in soil is a source of essential nutrients for crops and enhances water retention, thus creating favorable conditions for root development. Additionally, organic matter improves soil structure, reduces erosion risk, and increases water and nutrient retention, which is especially important in rice growing regions where water and nutrients are limiting factors.
When testing and evaluating the effectiveness of microbial preparations for the decomposition of straw and stubble, the OM content in the soil is a critical criterion. Straw and stubble contain large amounts of organic matter, but they need to be effectively decomposed to become readily available nutrients for crops. The tested microbial preparations help to enhance this decomposition process. Regular OM tests allow us to assess the effectiveness of microbial preparations in increasing soil organic matter.
Figure 10 presents the changes in the content of organic matter (OM) in the soil across 4 sample areas (A, B, C, D) through 3 treatment phases: before, during and after the application of microbial preparations. In area A, the OM content decreased significantly from 13.5% to 10.01% during treatment and continued to decrease slightly to 9.91% after treatment. On the contrary, area B showed more stability, with the OM content slightly decreasing from 8.89% to 8.76% during treatment, but then significantly increasing to 9.2% after treatment. Area C experienced a slight decrease from 11.57% to 11.16% during treatment, followed by a significant drop to 8.36%. Meanwhile, in area D, the OM content decreased dramatically from 7.24% to 5.98% before increasing again to 9.23% after treatment.
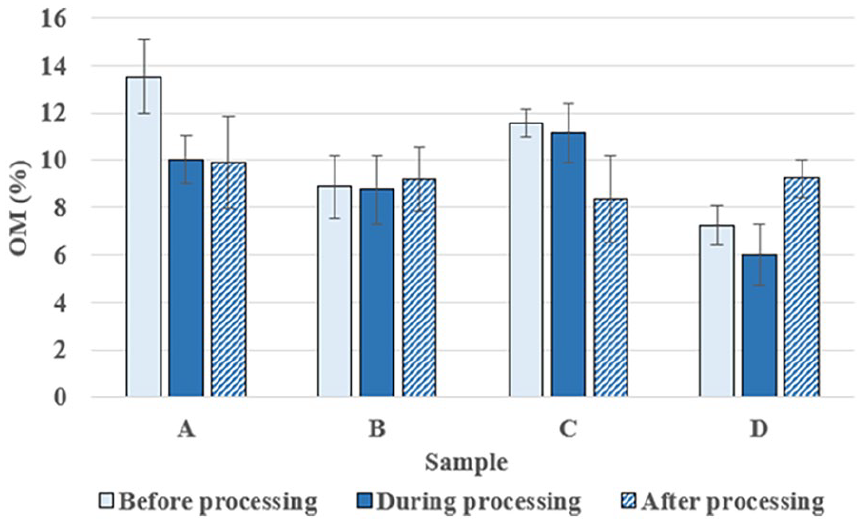
Evaluation of changes in the OM parameter in experimental soil samples.
Based on the analysis of the chart and the data, it is clear that the application of microbial preparations has caused varying effects on the OM content in the soil in each area. Specifically, areas A and C experienced a decrease in OM values after applying the preparations, while areas B and D showed recovery and an increase in OM values after the treatment phase. This indicates that the effectiveness of the preparations depends on the specific conditions and characteristics of each area, and thus, the application of microbial preparations should be approached with caution and tailored to each specific area.
The trends observed in organic matter (OM) content after microbial treatments provide important insights into the soil organic carbon dynamics influenced by straw decomposition. The moderate increase or stabilization of OM, particularly in NTT-02 and NTT-03 treated plots, indicates successful incorporation of decomposed straw into the soil organic pool. Trichoderma spp. and Bacillus subtilis strains, present in the microbial formulations, are known to enhance the enzymatic breakdown of cellulose and hemicellulose into simpler organic compounds that contribute to soil humus formation.7,9 This increase in OM improves soil structure, water retention capacity, and nutrient holding ability, which are critical for sustainable rice cultivation. However, slight fluctuations in OM content among treatments suggest that factors such as microbial activity rates, soil aeration, and initial organic matter content might influence decomposition efficiency. These findings align with previous reports,2,7 which showed that microbial treatments not only accelerate straw degradation but also positively impact soil organic carbon accumulation. Maintaining or enhancing soil OM through microbial decomposition represents a key strategy for improving long-term soil fertility and resilience against environmental stresses.
Phosphorus Content in Soil
Phosphorus, expressed in the form of phosphate, is one of the most crucial nutrients for plant growth, particularly rice. In agricultural soils, analyzing and testing phosphate concentrations not only guides precise fertilizer supplementation, but also ensures that crops have adequate nutrients for optimal growth. When conducting experiments and evaluating the effectiveness of microbial preparations in decomposing straw and stubble, testing phosphate concentrations becomes extremely important. Straw and stubble may contain a certain amount of phosphorus, and the microbial decomposition process can release phosphorus, making it more accessible and absorbable by crops. Microbial preparations that effectively decompose straw and stubble can enhance the supply of phosphorus to crops, thereby reducing the need for external fertilizer sources.
However, controlling phosphate concentrations is also essential, as excessive phosphorus released into the environment can pollute nearby water sources, harming freshwater ecosystems. Therefore, the analysis and testing of phosphate concentrations not only help optimize nutrient supplementation for rice crops, but also assess and improve the performance of microbial preparations in the decomposition of straw and stubble while protecting the environment.
Figure 11 illustrates changes in phosphate levels in soil across 4 sample areas: A, B, C, and D before, during, and after use of the microbial preparations. In area A, phosphate levels increased from 0.029 to 0.116 mg/L during treatment and then decreased to 0.057 mg/L. In area B, the initial level was 0.086 mg/L, which decreased to 0.030 mg/L during treatment and then increased to 0.065 mg/L. Area C maintained a level of 0.039 mg/L before and after treatment, but saw an increase to 0.070 mg/L during treatment. Finally, in area D, the level decreased from 0.086 to 0.075 mg/L during treatment and further decreased to 0.047 mg/L afterward. Overall, the microbial preparations had different impacts on each sample area, indicating varying levels of effectiveness and soil responses depending on the specific conditions and characteristics of each area.
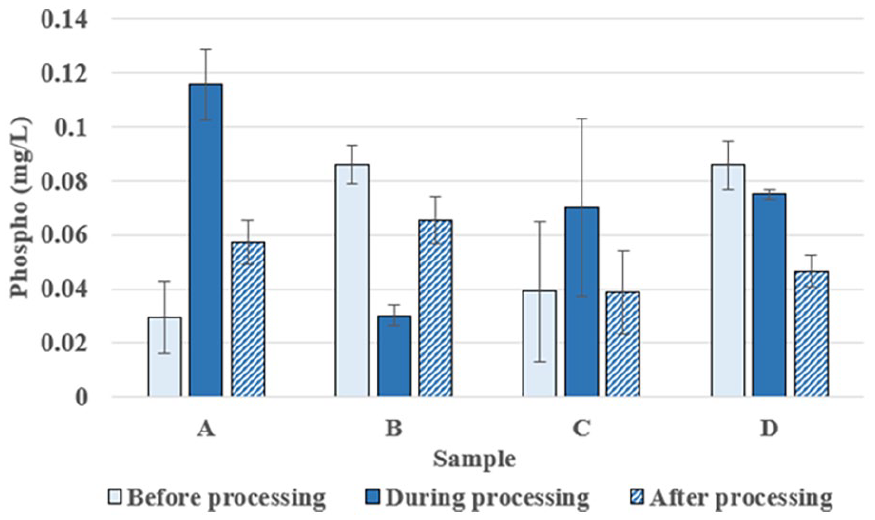
Evaluation of changes in phosphate parameters in experimental soil samples.
The changes in soil phosphorus content after microbial treatments reveal the important role of microbial activity in enhancing nutrient bioavailability. In treatments using NTT-02 and NTT-03, a moderate increase in phosphorus levels suggests that microbial decomposition of straw facilitated the mineralization and mobilization of bound phosphorus forms. Trichoderma spp. and Bacillus subtilis are known for producing organic acids and phosphatase enzymes that solubilize inorganic phosphorus, converting it into plant-available forms.2,7 This mechanism supports improved phosphorus uptake by rice crops, which is critical for root development, energy metabolism, and grain yield. Similar findings were reported by Bhattacharyya et al, 2 who observed enhanced phosphorus availability following microbial amendments in paddy soils. However, fluctuations in phosphorus concentrations among treatment plots might be attributed to variations in microbial enzymatic activity, soil pH shifts, and phosphorus fixation processes in different soil textures. Careful management of microbial applications is necessary to optimize phosphorus availability without causing nutrient leaching or environmental contamination. Overall, microbial enhancement of soil phosphorus offers a sustainable alternative to excessive phosphate fertilizer use, aligning with environmentally responsible farming practices.
Total Nitrogen Content in Soil
Total nitrogen is an essential nutrient for plant growth, particularly rice. In agricultural soils, analyzing and testing the total nitrogen concentration helps farmers determine the necessary amount of nitrogen fertilizer while ensuring that crops receive sufficient nitrogen for optimal growth.
When testing and evaluating the effectiveness of microbial preparations for the decomposition of straw and stubble, checking the total nitrogen concentration becomes crucial. Straw and stubble, when decomposed, can release nitrogen into the soil. Microbial preparations enhance this decomposition process by converting nitrogen in straw and stubble into a form that is more accessible and absorbable by plants. However, this process needs careful control because excess nitrogen emissions can lead to water pollution, causing oxygen deficiency in water and harming freshwater ecosystems. Therefore, analyzing and testing the total nitrogen concentration in rice-growing soil not only helps optimize nitrogen supplementation for crops from microbial preparations but also contributes to maintaining ecological balance and preventing environmental pollution.
Figure 12 illustrates the changes in the Total N parameter in 4 sample areas: A, B, C, and D before, during, and after use of the preparation. In area A, the parameter increased from 0.123 to 0.148 mg/L during treatment and then to 0.157 mg/L afterward. In area B, the parameter decreased slightly from 0.152 to 0.144 mg/L during treatment and then increased to 0.165 mg/L. Area C started at 0.131 mg/L, increased to 0.143 mg/L during treatment, and then increased significantly to 0.173 mg/L afterward. Area D saw a slight decrease from 0.141 to 0.129 mg/L during treatment and then increased sharply to 0.176 mg/L. Clearly, after the preparation, all areas showed an increase in the total N parameter, indicating the positive impact of the preparation on Total N content in the soil, while the control area D had a slight decrease in nitrogen levels. Among them, area A had the lowest increase after using the preparation.

Evaluation of the indicators of the total nitrogen parameters.
The increase in total nitrogen (TN) content following microbial treatments suggests effective mineralization of organic nitrogen compounds derived from straw decomposition. Microbial consortia, particularly Bacillus subtilis and actinomycetes included in NTT-02 and NTT-03 formulations, play a key role in breaking down complex organic matter into simpler forms such as ammonium (NH₄⁺) and nitrate (NO₃⁻), which are readily available for plant uptake.7,9 The observed enhancement in soil nitrogen levels aligns with previous studies, 2 which demonstrated that microbial inoculants accelerate nutrient cycling and improve soil fertility. This improvement is critical for supporting vigorous crop growth and yield, especially in rice cultivation systems, where nitrogen demand is high. However, variability in TN increases across treatment plots may result from differences in microbial activity efficiency, soil organic matter content, moisture levels, and the C/N ratio of decomposing straw. Maintaining a balanced nitrogen mineralization rate is important to minimize nitrogen losses through leaching or denitrification, ensuring both crop productivity and environmental sustainability. Overall, the findings highlight the potential of microbial straw treatments to reduce dependence on synthetic nitrogen fertilizers and contribute to sustainable soil management practices.
Evaluating Changes in Soil Microflora Using Microbial Products to Treat Rice Straw
Total Aerobic Microbiology
Figure 13 presents the changes in the microbial content in 4 soil samples. It can be seen that before the cultivation began, the number of aerobic microorganisms in the soil of treatments A, B, C, and D did not show significant differences. This indicates that the initial conditions for the experiment were consistent, providing a solid foundation to assess the impact of different cultivation practices.

Evaluation of total aerobic microorganisms in treatments before and after rice harvest.
As the cultivation phase started, the number of aerobic microorganisms in treatment A, where the Bio Decomposer preparation was used, was higher compared to B and C, where the NTT02 and NTT03 preparations were applied. This difference can be attributed to the more efficient decomposition of straw by Bio Decomposer. This preparation improves soil conditions by breaking down organic matter and thus releasing nutrients that support the growth of aerobic microorganisms. The robust development of microorganisms in soil not only improves soil quality, but also enhances nutrient absorption for crops.
To explain the difference, the study analyzed the microbial components present in preparations A, B, and C, allowing us to better understand their mechanisms of action on the soil and thus explain the differences in the quantities of aerobic microorganisms after cultivation. Preparation A contains Bacillus subtilis and a variety of strains of decomposing fungi. Bacillus subtilis is a bacterium capable of decomposing proteins, starch, and lipids, helping to convert organic matter in soil into more consumable forms for plants. Fungi such as Aspergillus oryzae, Trichoderma hamatum, and others in this preparation can decompose lignin and cellulose, the main components of dry plants and straw. This combination helps preparation A not only to promote the rapid decomposition of organic matter but also to improve soil structure and increase the availability of nutrients.
Preparation B focuses on Trichoderma strains, including Trichoderma hamatum, Trichoderma harzianum, Trichoderma asperellum, and Trichoderma viride. These Trichoderma species are known for their ability to control plant diseases through competition with pathogens, enzymatic degradation of plant components, and stimulation of plant immune systems. Although effective in preventing disease spread and promoting plant health, their focus on disease control may reduce their ability to decompose straw compared to preparation A.
Preparation C combines Trichoderma and Bacillus subtilis. This combination can provide both benefits in disease prevention and organic matter decomposition, but with a lower CFU/ml level for Bacillus subtilis compared to preparation A, which can reduce overall effectiveness in decomposing organic matter compared to preparation A.
As a result, preparation A may be more effective in promoting the growth of aerobic microorganisms after cultivation because of its strong organic matter decomposition capacity. Preparations B and C, although they support plant health and have some impact on aerobic microorganisms, may not produce a significant increase in aerobic microorganisms as preparation A, due to their greater emphasis on disease control and less on organic matter decomposition. Therefore, the difference in the quantities of aerobic microorganisms after cultivation can be explained by the soil improvement and the organic matter decomposition capabilities of the preparations, enhancing soil ecology and quality, thus promoting a richer environment for aerobic microorganisms.
After harvest, it is notable that the number of aerobic microorganisms tends to increase in all treatments, including the control. This increase may be due to several factors. First, soil tillage after harvest can increase soil aeration, promoting the activity of aerobic microorganisms. Second, organic matter such as straw and dead plant parts left after the crop season, can provide a significant food source for microorganisms, helping them grow rapidly. Finally, post-harvest weather conditions, possibly the onset of the rainy season, can also create favorable conditions for the development of soil microorganisms.
This analysis shows that the use of microbial preparations in agriculture not only helps improve soil by decomposing organic matter, but also positively impacts the development of aerobic microorganisms in the soil. This contributes to maintaining and enhancing soil structure and increasing soil fertility, especially after cultivation. Further research should focus on identifying optimal microbial preparations and their mechanisms of action to maximize these benefits in agricultural practice.
The observed increases in total aerobic microbial counts and cellulose-decomposing organisms strongly correlate with the accelerated straw decomposition dynamics recorded in the treated plots. Treatments with higher microbial populations, particularly NTT-02 and NTT-03, exhibited more rapid mass loss of straw residues and greater reductions in the C/N ratio compared to the untreated control. These results suggest that the elevated cellulolytic microbial activity enhanced the enzymatic breakdown of complex carbohydrates, thereby facilitating faster mineralization of organic matter. Consequently, the improved nutrient cycling and soil fertility parameters (eg, increased total nitrogen and organic matter content) can be attributed to the intensified microbial-driven decomposition processes. This causal relationship highlights the critical role of microbial population dynamics in optimizing in-field straw management.
Cellulose-Degrading Microorganisms
Figure 14 presents the changes in the content of cellulose-degrading microorganisms in 4 soil samples. From the results of cellulose-degrading microorganisms during and after rice harvest in treatments A, B, C, and D (control) (Figure 10), we can observe clear trends in the activity levels of the microorganisms capable of degrading cellulose at different stages of the cultivation process.

Evaluation of cellulose-degrading microorganisms in treatments before and after rice harvest.
Specifically, before cultivation (before processing), all treatments had relatively low levels of cellulose-degrading microorganisms, indicating that the cellulose in the soil had not been significantly degraded at this point. This consistent starting point among the treatments shows that the initial soil conditions were uniform. During cultivation (during processing), a significant increase in cellulose-degrading microorganisms was recorded, especially in treatment B, followed by treatments A and C. Treatment B showed the highest increase, likely due to the high content of Trichoderma strains in its microbial preparation, which are fungi known for their strong cellulose-degrading abilities. This increase demonstrates that the microbial preparations in treatment B were very effective in promoting the growth of cellulose-degrading microorganisms during the cultivation phase.
After harvest (after processing), treatments A and B continued to show a high number of cellulose-degrading microorganisms, while treatments C and D (control) showed a slight decrease compared to the during the cultivation phase. This indicates the long-term effectiveness of microbial preparations in maintaining the activity of cellulose-degrading microorganisms even after harvest. Notably, the sustained high numbers of microorganisms in treatments A and B may be related to the ability of microbial preparations to promote not only short-term cellulose degradation but also to improve soil recovery and regeneration capabilities.
In general, these results indicate that the presence and activity of cellulose-degrading microorganisms can be significantly enhanced by the use of appropriate microbial preparations, especially in treatments using preparations B and A. This also reflects the importance of selecting suitable microbial preparations for specific crops and soil conditions to maximize the effectiveness of organic matter degradation and improve the cultivated soil.
Evaluate the Effectiveness of Microbial Products on Growth Rate and Rice Yield
Evaluate the Stages of the Rice Crop During the Vegetative Period
Figure 15 illustrates the actual state of the seedlings in the field 7 days after sowing. Before sowing, farmers must ensure the field is dry. From the time the rice seeds sprout and stabilize until the rice plants begin tillering, farmers need to maintain a water level of about 1 to 3 cm on the field.

Field 7 days after sowing.
During this period, the number of tillers and leaf area increases maximally and ends at the end of the period (maximum tillering). Factors such as temperature and daily sunlight duration greatly influence the duration of this period. Different rice varieties have different vegetative growth periods, which also vary in this period. The longer the growth period, the longer the vegetative growth period. In addition, care techniques and the amount of fertilizer also affect the vegetative growth period of rice.
Based on the growth characteristics of the seedlings, the seedling period can be divided into 2 subperiods: the young seedling period and the healthy seedling period. The young seedling period is from sowing until the third true leaf appears. During this time, the resistance is weak. The healthy seedling period starts from the fourth true leaf until transplantation. The seedlings shift to an independent living period during this stage, with noticeable increases in height and the development of 4 to 5 root cycles, significantly improving their resistance. Depending on the variety, season, method and technique of seedling preparation, the seedling period can be long or short. The seedling period is significant throughout the rice growth process of rice. Healthy, strong seedlings provide a good foundation for tillering and subsequent growth stages to proceed smoothly.
Figure 16 illustrates the actual state of the experimental field 7 days after sowing. Observations show that the visual assessment of the treatments reveals differences. The treatments in field A (Bio Decomposer), Field B (NTT02 preparation), and Field C (NTT03 preparation) show differences compared to the control field (without microbial preparation). Specifically, fields A, B, and C show a higher and denser distribution of rice seedlings compared to the control field.

The state of treatment 7 days after sowing. Image (A) (Bio Decomposer); Image (B) (NTT02 preparation); Image (C) (NTT03 preparation); (D) Control field without microbial preparation).
Typically, 15 to 20 days after sowing, the rice plants begin tilling. From this point on, until the rice starts stem elongation and panicle initiation, farmers apply the “alternate wetting and drying” irrigation method. This involves letting the water in the field reach about 5 cm, then allowing it to dry until the soil surface cracks slightly before adding water again, continuing to let the field dry and crack repeatedly throughout the tillering phase.
Figure 17 illustrates the actual state of the experimental field 14 days after sowing. Observations after 20 days of sowing show differences in visual assessment of the treatments. The treatments in field A (Bio Decomposer), Field B (NTT02 preparation), and Field C (NTT03 preparation) show differences compared to the control field (without microbial preparation). Specifically, fields A, B, and C show a higher level of growth and tillering compared to the control field. Fields A and B exhibit stronger growth and tilling than Field C. The average stem length of rice plants at this stage is 15 to 20 cm, with branching levels ranging from 2 to 4 tillers per cluster.

Treatments 20 days after sowing. Image (A) (Bio Decomposer); Image (B) (NTT02 preparation); Image (C) (NTT03 preparation); Image (D) (Control field without microbial preparation).
Analyze Economic Efficiency
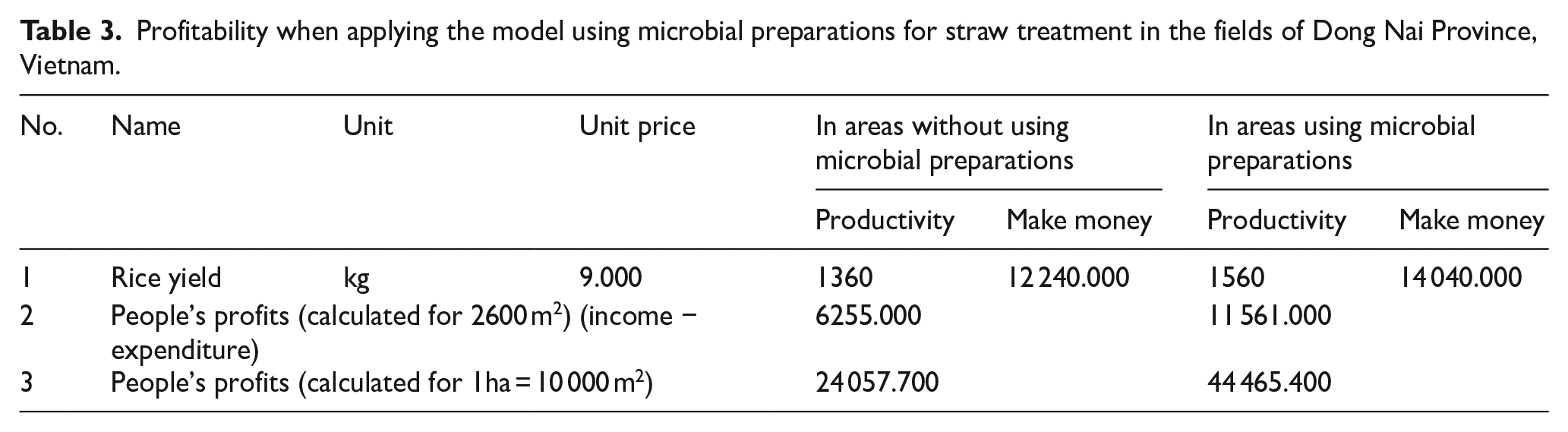
Tables 2 and 3 describe the costs incurred and the economic profits when applying the model using microbial preparations for straw treatment in the fields of Dong Nai Province, Vietnam. Analysis of economic benefits shows that the use of microbial preparations to treat straw residues in fields reduces costs for fertilizers, pesticides, herbicides, and molluscicides. This benefits not only the economy but also the environment and human health. Furthermore, by minimizing the amount of wild rice (weedy rice), it significantly reduces the labor effort required, making it less burdensome for farmers. In addition, the decomposition of straw residues directly in the fields creates a source of fertilizer for rice, which promotes the growth and development of the plants, resulting in higher yields compared to the absence of microbial preparations. Experiments indicate that rice yields increase by approximately 15%, from 1360 to 1560 kg. The total economic benefit for farmers is approximately 44 million VND per hectare compared to 24 million VND per hectare without using microbial preparations.
Costs Incurred when applying the model using microbial preparations for straw treatment in the fields of Dong Nai Province, Vietnam.

Profitability when applying the model using microbial preparations for straw treatment in the fields of Dong Nai Province, Vietnam.
Rice yield was measured by harvesting all mature panicles from 3 randomly selected 1 m2 plots within each treatment replicate. The collected grains were sun-dried, weighed, and extrapolated to a per-hectare basis. Yield differences between treatments were analyzed using one-way ANOVA, and differences were considered statistically significant at P < .05.
This research aims to evaluate the effectiveness of microbial treatments (NTT-02 and NTT-03) in accelerating straw decomposition, enhancing soil fertility, and providing a sustainable alternative to straw burning. Unlike conventional microbial decomposers, these formulations incorporate a multi-strain microbial approach, integrating Trichoderma species, actinomycetes, Bacillus subtilis, Aspergillus spp., Saccharomyces cerevisiae, Chaetomium spp., and a cellulase enzyme complex to optimize lignocellulosic breakdown. The study also distinguishes itself through direct application in the field, eliminating the need for post-harvest residue collection and making microbial treatments a practical solution for farmers. Furthermore, the research provides a comprehensive assessment of soil health indicators, including changes in pH, electrical conductivity, total organic carbon, nitrogen retention, phosphorus availability, and microbial diversity, offering a holistic perspective on the long-term benefits of microbial decomposition. These findings contribute to the advancement of sustainable agricultural practices by demonstrating an effective, scalable and eco-friendly method for straw management.
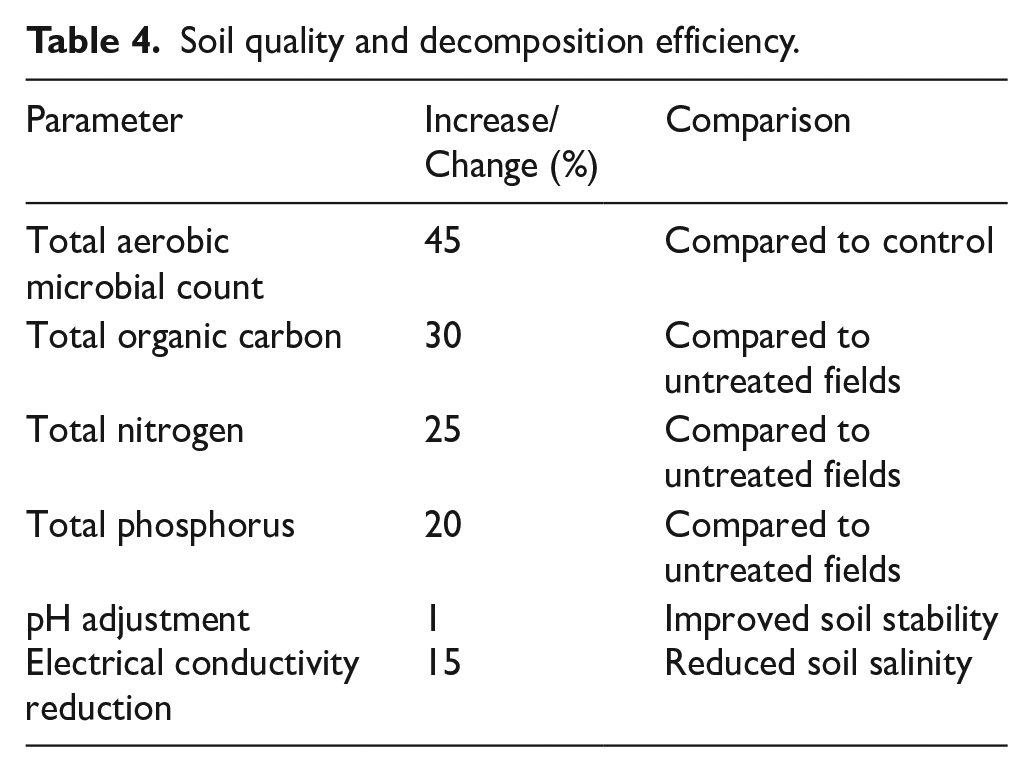
The research results indicate that the application of NTT-02 and NTT-03 led to a 45% increase in the total aerobic microbial count compared to the control, accelerating the straw decomposition process. Total organic carbon content increased by 30%, while total nitrogen and total phosphorus increased by 25% and 20%, respectively, enriching soil nutrients. Additionally, soil pH was adjusted by 0.8 units, improving stability, and electrical conductivity (EC) was reduced by 15%, helping mitigate soil salinity issues as shown in Table 4.
Soil quality and decomposition efficiency.
Limits and Development Policy Planning
Challenges and Solutions for Scaling Microbial Treatments in Sustainable Agriculture
Although microbial treatments offer a sustainable alternative to straw burning, several challenges must be addressed to enable large-scale adoption. One of the primary concerns is the cost of microbial products and their application. Although these treatments can reduce long-term expenses related to chemical fertilizers and pesticides, the initial investment for purchasing microbial formulations, transportation, and field application may be higher than traditional methods. Farmers with limited financial resources may hesitate to adopt these products without financial incentives or government support. Furthermore, the pricing of microbial formulations can vary based on production costs and distribution logistics, potentially limiting accessibility for small-scale farmers.
Beyond cost, scalability and distribution logistics pose additional challenges. Many microbial products require proper storage conditions to maintain their viability, particularly those containing temperature-sensitive strains. This may necessitate cold storage or specialized packaging, increasing transportation and handling expenses. Furthermore, training and knowledge transfer are crucial to ensuring proper application. Farmers unfamiliar with microbial treatments may require guidance on the correct dilution, field preparation, and optimal environmental conditions to maximize effectiveness. Without adequate support, inconsistent application or mismanagement could hinder decomposition efficiency.
Environmental and regional variations further complicate large-scale implementation. Differences in soil properties, climate conditions, and farming practices mean that a standardized application approach may not be suitable for all agricultural settings. Site-specific adjustments and continuous monitoring may be necessary to optimize microbial performance in diverse landscapes.
To enhance large-scale adoption, cost reduction strategies should focus on the development of locally produced microbial formulations, reducing dependency on expensive imports. Additionally, government subsidies, financial incentives, and farmer training programs can help mitigate economic and knowledge barriers. Integrating these support mechanisms will facilitate the larger adoption of microbial treatments, making them a more viable and accessible solution for sustainable straw management.
The application of microbial treatments not only improved short-term soil fertility and rice yield but also holds promise for long-term sustainability. Continuous application across multiple seasons could progressively enhance soil microbial diversity, organic matter content, and nutrient cycling efficiency. Such cumulative benefits would likely reduce the need for chemical fertilizer inputs, mitigate soil degradation, and promote resilient agroecosystems. Future longitudinal studies are warranted to quantify the residual effects of microbial inoculation and validate its sustainability in different agro-climatic zones.
Practical Recommendations for Farmers and Policy Makers to Improve Sustainable Straw Management
Based on the study findings, several actionable recommendations can be implemented to promote the adoption of microbial treatments for sustainable straw management. For farmers, integrating NTT-02 and NTT-03 into post-harvest practices can significantly accelerate straw decomposition, improve soil fertility, and reduce the dependency on chemical fertilizers. The application of microbial treatments directly to rice fields after harvest, following the recommended application rates, ensures optimal decomposition while enhancing soil microbial diversity. Furthermore, monitoring soil moisture levels and pH can further optimize microbial efficiency, leading to improved soil structure and nutrient availability. Shifting away from traditional straw burning by combining microbial decomposition with minimal tillage practices will not only improve long-term soil health but also contribute to increased crop yields and cost savings.
For policymakers, financial incentives and subsidies should be provided to encourage farmers to transition from straw burning to microbial-based treatments, making sustainable alternatives more accessible. Establishing educational programs and extension services can equip farmers with the necessary knowledge and technical support for effective microbial application and soil management. Supporting research and development of locally produced microbial formulations can help reduce costs while ensuring widespread availability, particularly for small-scale farmers. Furthermore, strengthening regulations on straw burning while promoting microbial decomposition through awareness campaigns and policy incentives will further accelerate the shift toward sustainable agricultural practices.
These recommendations provide practical solutions that align scientific research with real-world applications, ensuring that microbial treatments are effectively integrated into modern farming systems.
When compared to other common straw management practices, microbial treatments offer distinct advantages. Biochar production requires high-temperature pyrolysis equipment, incurs substantial energy costs, and is often not feasible for smallholder farmers. Composting, while environmentally friendly, demands significant labor, space, and time (often several months) to achieve full decomposition, limiting its scalability during intensive cropping cycles. Urea-treated straw accelerates decomposition but carries risks of nitrogen leaching and volatilization, contributing to environmental pollution. In contrast, microbial treatments such as NTT-02 and NTT-03 are cost-effective, requiring only simple field application without specialized infrastructure. They enhance decomposition rates while improving soil health and reducing greenhouse gas emissions. Moreover, the scalability of microbial applications across diverse farming systems makes them a promising alternative for sustainable straw management, particularly in regions facing labor shortages and environmental constraints.
Scalability and Integration of Microbial Treatments in Sustainable Agriculture
The scalability of the NTT-02 and NTT-03 microbial products and their integration into existing agricultural practices present a viable pathway for sustainable straw management. These formulations are designed for large-scale application, and are available in liquid and powder forms that can be easily sprayed onto fields using conventional agricultural equipment. Their local production capability reduces dependence on imported alternatives, ensuring affordability and accessibility for farmers. Additionally, the production process is adaptable, allowing modifications to suit different soil types and crop conditions in diverse agricultural regions.
Integrating these microbial treatments into existing farming systems is straightforward, requiring minimal changes to current practices. The application process can be seamlessly incorporated immediately after harvest, utilizing standard spraying equipment without the need for additional labor-intensive steps. These treatments are compatible with traditional soil preparation methods, including plowing and irrigation, making adoption practical and efficient. By incorporating microbial decomposition into crop rotation cycles, farmers can continuously improve soil fertility while reducing reliance on chemical fertilizers.
To support widespread adoption, training programs and extension services play a crucial role in educating farmers on proper application techniques and expected benefits. Ensuring that microbial treatments remain cost-effective, easy to apply, and adaptable to different farming systems will further strengthen their scalability and long-term sustainability.
Conclusion
This study demonstrates that microbial treatments using the newly formulated NTT-02 and NTT-03 products significantly enhance straw decomposition and improve soil health under field conditions. Compared to the untreated control, soils treated with NTT-02 exhibited a 15% increase in organic matter (OM), a 12% increase in total nitrogen (TN), and a 10% increase in total phosphorus (TP). The total aerobic microbial population increased by 1.5 times, while the number of cellulose-decomposing organisms doubled, indicating a robust enhancement of soil microbial activity. The application of microbial products accelerated straw decomposition, reducing the complete breakdown time from approximately 28 days to 14-18 days. Furthermore, rice yield increased by 8% to 10%, highlighting the agronomic benefits of integrating microbial decomposition into sustainable farming practices. These findings underscore the potential of microbial-based treatments as an effective, environmentally friendly alternative to straw burning, contributing to enhanced soil fertility, reduced reliance on chemical fertilizers, and improved agricultural productivity. Future studies should validate these results across multiple cropping seasons and diverse agro-climatic conditions to further confirm the scalability and ecological safety of this approach.
A limitation of this study is that the field experiment was conducted during a single cropping season. Future studies should perform multi-season and multi-location trials to validate the reproducibility and generalizability of the findings under varying climatic conditions. The other limitation of this study is the absence of a positive control, such as traditional compost application or inorganic fertilization, to benchmark microbial treatment effectiveness. Future research should incorporate standard practice controls to better contextualize microbial performance against conventional straw management and soil fertility improvement methods. This would allow for a more comprehensive evaluation of the agronomic, environmental, and economic benefits of microbial-based solutions. The last limitation of this study is the reliance on subjective visual and sensory indicators (color change, texture alteration, and odor) to assess straw decomposition stages. While these observations provided practical insights, future research should incorporate quantitative decomposition metrics such as dry mass loss, lignin breakdown rates, and CO₂ evolution measurements. These objective parameters would offer a more rigorous validation of microbial decomposition effectiveness and further strengthen causal interpretations.
Future studies should focus on further assessing the degradation process of treated straw over different timeframes and evaluating long-term impacts on soil fertility and crop productivity. Expanding the scope of microbial formulations and optimizing their application methods will also be crucial for maximizing their benefits in large-scale agricultural systems.
Supplemental Material
sj-xlsx-1-ehi-10.1177_11786302251343765 – Supplemental material for Sustainable Agricultural Development Through Straw Treatment on Fields Using Microbial Products: A Case Study in Vietnam
Supplemental material, sj-xlsx-1-ehi-10.1177_11786302251343765 for Sustainable Agricultural Development Through Straw Treatment on Fields Using Microbial Products: A Case Study in Vietnam by Van-Phuc Dinh, Bich-Ngoc Duong, Thanh Tran, Hoai-An Tran-Vu, Ngoc-Mai Dang-Thi, Hoai-Luan Phan-Van, Tuan-Kiet Tran and Thanh Q. Nguyen in Environmental Health Insights
Footnotes
Acknowledgements
This paper was partially supported by the project “Assessing the status and proposing solutions to minimize the impacts of open burning and the use of pesticides in agriculture on climate change, human health, and biodiversity in Vietnam,” implemented by GAHP and VACNE. The project is part of the Environmental Pollution Program funded by the United Kingdom Department for International Development (UKAid) and the Department of Environment, Food & Rural Affairs (UK DEFRA). The authors also thank Nguyen Tat Thanh University, Ho Chi Minh City, Vietnam, for providing the facilities required to carry out this work.
We acknowledge Nguyen Tat Thanh University, Ho Chi Minh City, Vietnam for supporting this study.
Ethical Considerations
Data usage should not cause harm to individuals or communities.
Clear and open communication about data collection, storage, and usage practices is essential to establish trust and accountability.
Author Contributions
Van-Phuc Dinh: Conceptualization, Software, Writing—Review & Editing.
Bich-Ngoc Duong: Formal analysis, investigation.
Thanh Tran: Conceptualization, Methodology, Writing—Review & Editing.
Hoai-An Tran-Vu: Resources, Data Curation.
Ngoc-Mai Dang-Thi: Conceptualization and Methodology.
Hoai-Luan Phan-Van: Data Curation, Software.
Tuan-Kiet Tran: Data Curation, Software.
Thanh Q. Nguyen: Supervision, Writing, Review & Editing, Visualization.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
The authors confirm that the data supporting the findings of this study are available in the article and its supplementary materials.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
