Abstract
Background:
Although BRCA1 and BRCA2 mutations are known to be associated with different breast cancer (BC) subtypes, real-world evidence on how these genetic differences influence tumor behavior and treatment decisions remains limited, particularly in Japanese patients. With the recent expansion of PARP inhibitor indications in Japan, BRCA testing has become increasingly routine, highlighting the need for clinical data tailored to local populations.
Objectives:
To compare clinicopathological features, recurrence patterns, and surgical choices between BRCA1- and BRCA2-associated BC in Japanese patients, with a focus on ER-positive tumors.
Design:
A single-institution retrospective cohort study.
Methods:
We retrospectively reviewed 417 patients who underwent BRCA1/2 genetic testing at a single Japanese institution between April 2020 and November 2023. Of these, 38 patients (12 BRCA1, 26 BRCA2) had pathogenic variants. We compared clinicopathological features, recurrence patterns, and choices of risk-reducing surgery between BRCA1 and BRCA2 carriers.
Results:
BRCA1-associated cancers were predominantly triple-negative (75%) and diagnosed at earlier stages (T1 in 83.3%), while BRCA2-associated cancers were mainly ER-positive (69.2%) and more likely to present with multiple lymph node metastases (⩾2 nodes in 42.3%). Although Ki-67 levels were higher in BRCA1 tumors, this was largely subtype-dependent. Notably, ER-positive BRCA tumors showed a trend toward higher recurrence. Preferences for prophylactic surgery also varied by mutation type.
Conclusion:
This single-institution study highlights clinically meaningful differences between BRCA1- and BRCA2-associated BC in Japanese patients. BRCA2 cancers tended to present with more advanced features, while BRCA1 cancers were more often detected at earlier stage. These findings underscore the value of BRCA testing not only for PARP inhibitor eligibility but also for subtype-specific risk assessment and individualized preventive strategies.
Plain Language Summary
Why was this study done? Some people with breast cancer have inherited mutations in genes called BRCA1 or BRCA2. These mutations are known to affect the type of cancer that develops. However, we do not fully understand how they might also affect the way cancer behaves or how it should be treated—especially among Japanese patients. As genetic testing becomes more common in clinical care, it is important to understand the real-world impact of these mutations on treatment and outcomes. What did the researchers do? The researchers looked at 417 patients with breast cancer who received BRCA1/2 testing at one hospital in Japan. Among them, 38 people had BRCA mutations—12 with BRCA1 and 26 with BRCA2. They compared the cancer characteristics, treatment approaches, and recurrence between these 2 groups, with a special focus on people whose cancers were estrogen receptor (ER)-positive. What did the researchers find? Patients with BRCA1 mutations mostly had triple-negative breast cancer (a type that is harder to treat and lacks common hormone receptors), and their tumors were usually found at an early stage. Those with BRCA2 mutations mostly had ER-positive tumors, but surprisingly, these tumors often had more lymph node involvement and showed signs of coming back more often. The researchers also found that treatment and surgery decisions—such as whether to remove the ovaries to lower cancer risk—varied depending on the mutation. Why does this matter? Clinicians usually use tumor subtype (like ER-positive) to guide treatment. However, this study suggests that genetic mutations may affect cancer behavior even within the same subtype. For example, BRCA2-related ER-positive tumors may be more aggressive than expected. Understanding these differences can help doctors choose the best treatment for each patient.
Introduction
Mutations in the BRCA1 and BRCA2 genes are well-established as significantly increasing the risk of breast cancer (BC) and ovarian cancer. These mutations not only elevate cancer risk but also influence treatment strategies, underscoring the critical importance of their identification. Consequently, the number of patients diagnosed with BRCA1/2 mutations in Japan has been steadily increasing.
In recent years, insurance coverage for BRCA testing and associated therapies has undergone significant changes in Japan. In July 2018, based on the results of the OlympiAD trial, 1 Poly ADP-ribose polymerase (PARP) inhibitors were approved in Japan as reimbursable therapy for the treatment of metastatic or recurrent BC. In April 2020, patients at risk of hereditary breast and ovarian cancer syndrome (HBOC) underwent BRCA testing that was covered by insurance, and prophylactic risk-reducing surgery for patients with BRCA BC, namely risk-reducing mastectomy (RRM) and risk-reducing ovarian and fallopian tube excision (RRSO), was also indicated in Japan. In August 2022, following the results of the OlympiA trial, 2 PARP inhibitors were approved as adjuvant therapies for patients with BRCA1/2 mutations of high-risk BC.
The presence or absence of BRCA mutation is important for the prevention, perioperative decision-making, and management of advanced recurrence, and an increasing number of patients are diagnosed with BRCA1/2-mutated BC. Research has revealed distinct clinical and pathological characteristics associated with BRCA1 and BRCA2 mutations. BRCA1-mutated BC (BRCA1 BC) is frequently associated with triple-negative BC (TNBC), whereas BRCA2-mutated BC (BRCA2 BC) is more commonly linked to estrogen receptor (ER)-positive BC.3-5 Differences in the prevalence of other cancers, particularly ovarian cancer, have also been described. The lifetime risks of BC and ovarian cancer among BRCA1/2 carriers have been reported in large international cohorts6,7 and further supported by a recent analysis of BRCA1/2 pathogenic variants in triple-negative versus luminal-like breast cancers. 8 In addition to BC and ovarian cancer, BRCA1/2 mutations confer increased risks of pancreatic and prostate cancer. 9 Furthermore, recent studies have further suggested a potential association between BRCA mutations and a broader range of malignancies, including gastric and esophageal cancers.9,10
Despite these insights, data from Japanese clinical settings remain limited, particularly regarding the risk of metastatic recurrence, the efficacy of chemotherapy, and the response to endocrine therapy in BRCA1 BC and BRCA2 BC. This study aims to address this knowledge gap by investigating the clinical background, clinicopathological factors, and treatment preferences of patients with BRCA-positive BC treated at a single institution in Japan. A particular focus is placed on how differences between BRCA1 and BRCA2 mutations influence treatment decisions.
Methods
This study included consecutive patients who underwent BRCA genetic testing at Nagoya University Hospital between April 2020 and November 2023. All participants provided written informed consent, and the study was ethically reviewed. The cohort comprised patients with BC or a history of BC who underwent BRCA testing covered by insurance. In Japan, the insurance criteria for BRCA testing include the following: BC diagnosed at age ⩽45; BC diagnosed at age ⩽60 and classified as TNBC, bilateral or multiple primary BCs in one breast; male BC, BC with concurrent ovarian, fallopian tube, or peritoneal cancer at diagnosis, or a family history of breast, ovarian, or pancreatic cancer within third-degree relatives. In addition, since 2020, BRCA testing has been used as a companion diagnostic tool for PARP inhibitors in advanced or recurrent BC, and since 2022, it has also been applied for perioperative treatment in high-risk BC, supporting the use of PARP inhibitors. The diagnosis for HBOC and all the tests submitted as companion diagnostics were conducted using the BRACA analysis diagnostic system (Myriad Genetics, Inc., Salt Lake City, Utah). All BRCA1/2 testing was performed as germline analysis using peripheral blood; no tumor tissue-based sequencing was included.
The study examined the age of BC onset, clinicopathological characteristics, and recurrence in patients with BRCA1/2 mutations. All histopathology information was obtained from routine diagnostic reports. Slides were not re‑examined and no new histological analyses were conducted. Pathological factors were assessed via surgical pathology for cases undergoing surgery. For cases receiving neoadjuvant chemotherapy (NAC), tumor diameter and lymph node metastases were determined based on imaging findings. In our institution, suspicious lymph nodes were evaluated by imaging and frequently confirmed by cytology/biopsy prior to NAC when feasible. Eleven patients who experienced multiple primary BCs, clinicopathological analyses focused on the most advanced or aggressive cancer targeted for treatment. Patients were followed at our outpatient clinic according to institutional practice. Outcomes and recurrences were ascertained by electronic medical records, pathology reports, and imaging studies. Follow-up time was measured from the date of definitive breast surgery to the date of last clinic visit or death; patients without events were censored at the last known contact. The data cut-off for this analysis was August 2024, and no cases were unobservable.
To evaluate subtype-specific differences, tumors were reclassified into 3 groups: Luminal (ER-positive/HER2-negative), Triple Negative, and HER2-positive. This analysis included only ER-positive HER2-positive cases; no ER-negative HER2-positive cases were identified. Accordingly, Table 2 was revised to display these 3 groups separately, and statistical analyses were performed based on this classification.
The study also evaluated the personal and family history of other cancers in probands, corroborated through self-reports and information provided by relatives. Choices for primary prevention, such as risk-reducing surgeries, were also analyzed. For patients with more than one BC diagnosis, the age of onset was defined as the age at first diagnosis.
Statistical analyses were performed using JMP Pro 17 (SAS Institute Inc., Cary, North Carolina). Categorical variables were analyzed using Fisher’s exact test, and nonparametric statistical tests were used for other analyses. Given the limited sample size, we performed univariable descriptive analyses without multivariable adjustment; thus, results should be interpreted as exploratory.
Reporting guideline
The reporting of this observational study conforms to the STROBE statement 11 ; the completed STROBE checklist is provided as Supplementary File 1.
Results
Comparison of clinicopathological features
Among the 417 patients tested, 38 (9.1%) had pathogenic BRCA1/2 mutations and 11 (2.6%) had mutations of uncertain significance. BRCA1 mutations were identified in 12 patients (2.9%) and BRCA2 in 26 (6.2%). Subtype analysis showed significant differences between the groups: Luminal subtypes were more common in BRCA2 BC (69.2%) than in BRCA1 BC (16.7%; P = .0010), whereas TNBC was more frequent in BRCA1 BC (75%) than in BRCA2 BC (11.5%; P < .0001). Luminal subtypes and HER2-positive subtypes showed no significant differences (P = .5363).
The median age of onset was 46.5 years in BRCA1 carriers and 45.5 years in BRCA2 carriers, with no statistically significant difference between the 2 groups (P = .4330). Tumor size did not differ significantly (P = .3241), but the T-stage exhibited differences; BRCA1 BC had a higher proportion of T1 tumors (83.3%), whereas BRCA2 BC showed a greater incidence of T2 (30.8%) and advanced tumors (T3/T4) (11.5%). Lymph node (LN) metastasis rates approached significance (P = .0707), with BRCA1 BC showing fewer LN metastases (75%) than BRCA2 BC (53.8%). BRCA2 BC had a higher incidence of ⩾2 LN metastases (42.3%) than BRCA1 BC (8.2%). Tumor grade did not differ significantly (P = .6007). However, Ki-67 levels, a proliferation marker, was higher in BRCA1 BC than in BRCA2 BC (P = .0271). No significant differences were observed during chemotherapy administration (P = .768; Table 1).
Clinical and pathological characteristics of 38 patients with BRCA1 (n = 12) and BRCA2 (n = 26) mutations.

Variables include age, tumor stage, lymph node metastasis, molecular subtypes, grade, Ki-67 index, surgery type, and chemotherapy. Significant differences are observed in molecular subtype distribution (luminal, triple-negative) and Ki-67 index, highlighting distinct profiles between BRCA1 and BRCA2 mutation carriers.
Eight cases without information were excluded.
Significant P-values (P ≤ 0.05) are shown in bold.
Histological background, perioperative treatment, and recurrence by subtype
Analysis by the 3-group classification (Luminal, Triple Negative, HER2-positive) is summarized in Table 2. Median age was 45.5 years in Luminal, 49.5 years in Triple Negative, and 39 years in HER2-positive tumors (P = .1749). BRCA1 mutations were most frequent in the Triple Negative group (75%), whereas BRCA2 mutations predominated in the Luminal (90%) and HER2-positive (80%) groups. Other clinicopathological variables are shown in Table 2. The median invasive tumor size was 18.5 mm in Luminal, 17.0 mm in Triple Negative, and 13.0 mm in HER2-positive cancers (P = .6739). Lymph node involvement of 2 or more nodes was observed most frequently in Luminal tumors (50.0%), compared with 8.3% in Triple Negative and 20.0% in HER2-positive cancers (P = .0787). Tumor grade also differed across groups: grade 3 tumors accounted for 25.0% of Luminal, 66.7% of Triple Negative, and 20.0% of HER2-positive cancers (P = .0371). The Ki-67 proliferation index was significantly higher in the Triple Negative group (median 50%) compared with Luminal (15%) and HER2-positive (15%) cancers (P = .0021).
Clinical characteristics of BRCA mutation carriers stratified into Luminal (n = 20), Triple Negative (n = 12), and HER2-positive (n = 5) groups.

One case of Stage 0 has been excluded. Triple Negative tumors were enriched for BRCA1 mutations, showed higher Ki-67 indices, and were more often high grade.
Eight cases without information were excluded.
Significant P-values (P ≤ 0.05) are shown in bold.
Treatment patterns varied accordingly. Chemotherapy was administered to 45.0% of Luminal patients, but to 83.3% of Triple Negative and all HER2-positive patients (P = .0219). Endocrine therapy was prescribed in 90.0% of Luminal cases. The use of adjuvant PARP inhibitors was limited during the study period; they were administered to 10.0% of Luminal patients, 5.9% of Triple Negative patients, and none of the HER2-positive patients (P = 1.0000). Recurrence rates were highest in the Luminal group (45.0%), while it was lower in triple-negative tumors (8.3%), and no recurrence was observed in HER2-positive tumors (P = .437).
Family and personal cancer histories
Regarding the family history of BC, 91.7% (11 patients) of patients with BRCA1 BC reported having a family history of BC, whereas 8.3% (1 patient) reported no family history. In contrast, 69.2% (18 patients) of patients with BRCA2 BC had a family history of BC, and 30.8% (8 patients) reported no family history (Table 3). BRCA1 BC tends to have a family history of ovarian cancer and pancreatic cancer, whereas BRCA2 BC tends to have a family history of other cancers such as stomach, esophageal, and prostate cancers (Table 4).
Patient with family history of breast cancer.

Within the third degree of proximity.
Number of family members with cancers other than breast cancer.

Within the third degree of proximity. Including cases with more than 2 types of cancer.
The occurrence of multiple BC diagnoses within individual patients showed no significant difference between groups: 33.3% (4 patients) for BRCA1 BC and 26.9% (7 patients) for BRCA2 BC (Table 5).
Patients with more than 2 breast cancer.

Regarding the incidence of cancers other than BC within the cohort, BRCA1 BC patients (n = 12) reported 3 cases of ovarian cancer, 1 case of gastric cancer, and 2 cases of other cancers, with 75% (9 patients) reporting no history of other cancers. Conversely, BRCA2 BC patients (n = 26) reported no ovarian cancer cases, but 2 cases of gastric cancer, 1 case of esophageal cancer, and 3 cases of other cancers. A majority (84.6%, 22 patients) of BRCA2 BC patients had no history of cancers other than BC (Table 6).
Patient who experience with other cancers.

Including cases with more than 2 types of cancer.
Selection of risk-reducing surgery
We compared the rates of RRM and RRSO in patients with BRCA1 and BRCA2 BC. Among BRCA1 BC, 18% underwent simultaneous RRM and 9% opted for heterochronous RRM, whereas most (73%) did not. Similarly, among patients with BRCA2 BC, 19% underwent simultaneous RRM, 8% underwent heterochronous RRM, and 73% did not undergo RRM; no difference in the trend was observed between BRCA1 and BRCA2 BC. In contrast, for RRSO, 34% patients with BRCA1 BC underwent simultaneous RRSO, 8% underwent heterochronous RRSO, and 58% did not perform RRSO; in BRCA2 BC, 11% opted for simultaneous RRSO, a higher proportion (31%) underwent heterochronous RRSO, and 58% did not undergo RRSO. Although the final proportion of RRSOs performed remained the same, simultaneous RRSOs were more common in BRCA1 than in BRCA2 BC preferred heterochronous RRSOs (Figure 1).
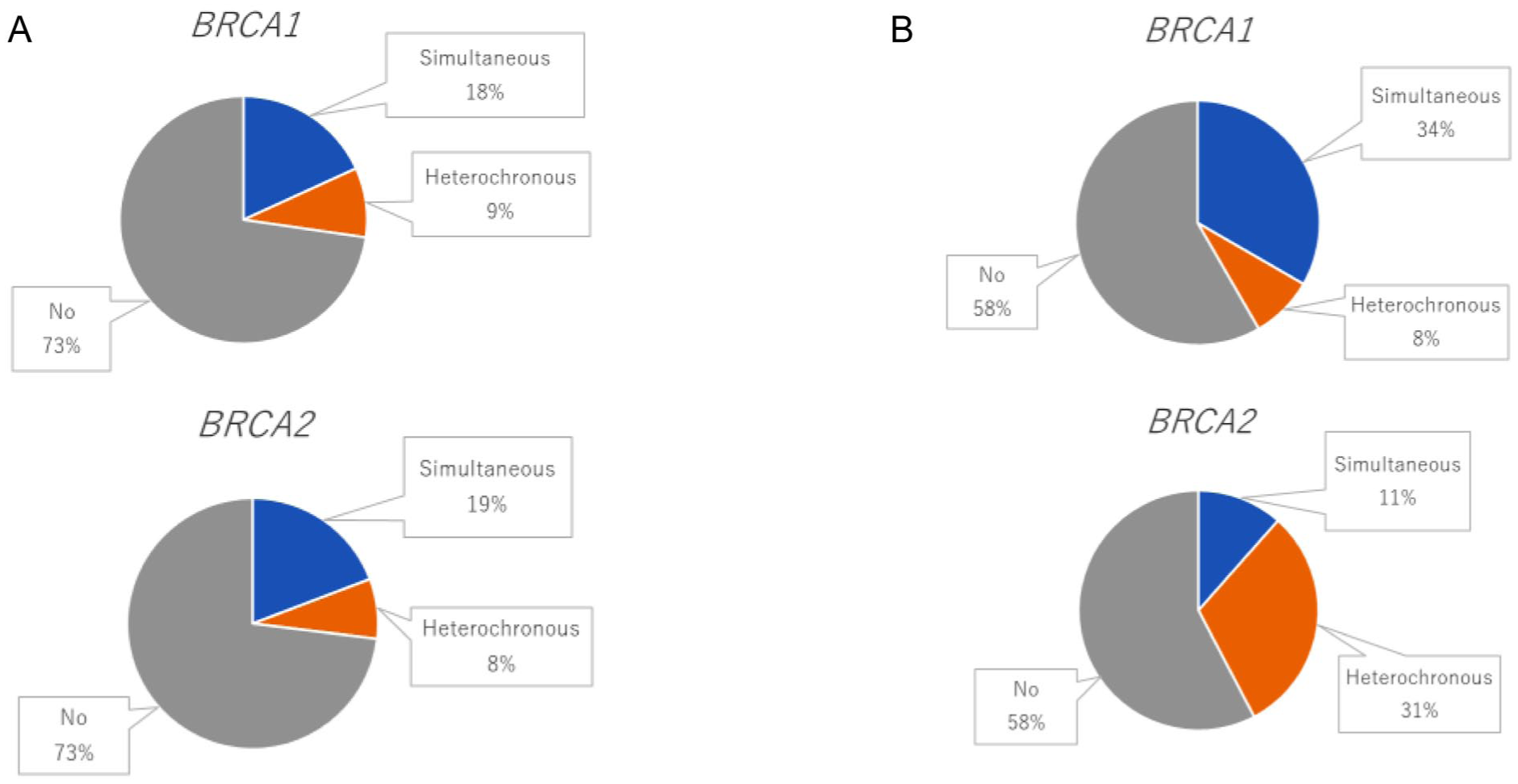
(A) RRM and (B) RRSO.
Discussion
This study revealed striking differences in clinicopathological features and treatments selection in actual clinical practice between patients with pathogenic mutations in BRCA1 and BRCA2 in BC. These findings provide insights into the influence of genetic differences on BC phenotypes and treatment approaches.
In contrast to most international reports,7 our cohort did not demonstrate a significant age difference between BRCA1 and BRCA2 carriers. The Japanese HBOC Consortium also reported younger onset in BRCA1 compared with BRCA2 carriers. 12 The discrepancy with our findings may reflect referral bias, testing criteria, or the small sample size of our single-institution cohort. Chemotherapy use, and invasive tumor size did not differ significantly between BRCA1 and BRCA2 BC, but there were differences in tumor size trends; although BRCA1 BC is generally considered more biologically aggressive, in this cohort BRCA1 cases were more frequently diagnosed at earlier T1 stage, whereas BRCA2 cases more often presented with larger tumors and multiple lymph node metastases. This may be partially influenced by earlier detection, potentially due to greater awareness stemming from strong family histories in BRCA1 carriers. 13 However, given the small sample size and retrospective design, these observations must be interpreted with caution. Tumor size and lymph node status at diagnosis are subject to patient consultation behavior and diagnostic timing, which are not standardized and introduce potential bias. Consequently, no definitive conclusions should be drawn from these comparisons without further validation. Conversely, BRCA2 BC is associated with extensive tumor size and can potentially cause advanced cancer. The higher proportion of LN metastases (⩾2 nodes) in BRCA2 BC than in BRCA1 BC also supports the observation of a more advanced disease at diagnosis in this group. Although this trend did not reach statistical significance, it suggests that BRCA2 BC may have a more invasive phenotype. The significantly higher Ki-67 index observed in BRCA1 BC than in BRCA2 BC likely reflects the predominance of triple-negative BC in BRCA1 cases, rather than an independent biological difference. As Ki-67 is known to be elevated in TNBC,14,15 this result should be interpreted in that context. Therefore, the proliferation index may not be directly attributable to the BRCA1 mutation itself, but rather to the associated subtype distribution. However, despite the high prevalence of Luminal BCs in BRCA2 BC, no difference in grade when comparing BRCA1 and BRCA2 carriers (Table 1), which is an indicator of histological malignancy, and we were concerned about the higher malignancy of Luminal BC with BRCA mutations. Therefore, we compared Luminal BC with other types of BC among BRCA1/2 BC.
When comparing Luminal BC to the other subtypes, there was a difference in the LN metastasis rate, with a higher percentage of patients in the Luminal BC group demonstrating 2 or more LN metastases than in the other groups. Although this difference approaches statistical significance, it highlights a trend that may influence prognosis and treatment decisions. The higher rate of LN metastasis in the Luminal BCs group may indicate the need for more aggressive adjuvant therapy for this subtype. Furthermore, there was no difference in grade, which is different from the general trend observed in Luminal BC (ie, lower Ki-67 and grade). 14 Despite these histological factors, we cannot eliminate that the significantly lower chemotherapy use in Luminal BC patients may be related to the lower Ki-67 levels. Furthermore, regarding recurrence, the recurrence rate in the Luminal BC group was significantly higher than in the other subtype groups. As previously reported, BRCA1/2 mutated Luminal BC also had a 3.4-times higher risk of death due to BC than due to non-Luminal BC. 16 These findings suggest the importance of BRCA testing for high-risk Luminal BC to confirm the presence or absence of mutations, and the importance of selecting drug therapy in addition to hormone therapy for BRCA1/2 mutated Luminal BC.
The use of adjuvant PARP inhibitors has improved the prognosis of BRCA-mutated BC. 2 In Japan, PARP inhibitor was indicated for the treatment of recurrent high-risk HER2-negative BC starting in 2022. In this study, only 3 patients were treated with PARP inhibitors as adjuvant therapy, and the reason for the low use of PARP inhibitors was that insurance coverage for PARP inhibitors as postoperative therapy did not begin until midway through the observation period. Further case accumulation and observation of the experience with PARP inhibitors are needed, but it is expected that the prognosis of BRCA1/2 mutated Luminal BC will be improved if BRCA testing as an appropriate companion diagnosis is performed in the future and linked to the use of postoperative PARP inhibitors.
Regarding family history and related issues, BRCA1 BC is more likely to report family and patient histories of ovarian cancer, which is supported by the 75.6-fold increased risk of ovarian cancer in Japanese with BRCA1 mutation and 11.3-fold increased risk of ovarian cancer with BRCA2 mutations. 9 The BRCA1 mutation was associated with a family history of BC (91.7%), and given its cancer specificity, it is possible that BRCA1 BC families have a higher awareness of BC. Conversely, BRCA2 BC were more likely to report a family history of other cancers, such as gastric and esophageal cancers. The same tendency was also observed for the patient propensity to develop other cancers. In addition to prostate and pancreatic cancers, reports on the Japanese population indicate an increased risk of gastric cancer (5.2-fold for BRCA1 and 4.7-fold for BRCA2) and a 5.6-fold increased risk of esophageal cancer for BRCA2 positivity. 9 This suggests that there is a different cancer spectrum associated with BRCA1 and BRCA2 mutations, which may guide genetic counseling, surveillance, and patient awareness.
Regarding risk-reducing surgery, RRM was performed in 27% of both BRCA1 (18% simultaneous, 9% heterochronous) and BRCA2 carriers (19% simultaneous, 8% heterochronous) in our cohort, showing no difference between the groups. These rates are at the lower end of international reports (30%-40% for BRCA1 and 20%-30% for BRCA2),6,7 and consistent with Japanese registry data (16.3% vs 8.5%), 17 likely reflecting cultural and systemic factors such as insurance coverage, timing of counseling, patient preferences, and reconstructive availability. In contrast, the rate of RRSO differed between groups: a higher proportion of BRCA1 carriers underwent concurrent RRSO (34%) compared with BRCA2 carriers (11%), possibly reflecting the higher ovarian cancer risk associated with BRCA16,7 and more frequent family history. Patients with BRCA1 BC are also more likely to have TNBC and to receive preoperative chemotherapy, which provides additional opportunities for counseling. Notably, 31% of BRCA2 carriers eventually underwent heterochronous RRSO; thus, the overall implementation rate of RRSO was ultimately similar between groups, reflecting the challenges of ovarian cancer screening and the influence of counseling timing on surgical decisions.
This study revealed differences between the BRCA1 and BRCA2 mutated BC. While BRCA1 cancers tended to be detected earlier, BRCA2 cancers tend to present with larger tumors and multiple lymph node metastases, a trend that was reinforced when focusing on the Luminal type. Differences were also observed in the choice of risk-reducing surgery and incidence of other cancers. These findings emphasize the need for individualized cancer management strategies according to BRCA status.
Limitations
This study has several limitations. First, the sample size was relatively small (12 BRCA1 and 26 BRCA2 carriers), which limited statistical power, particularly for subgroup analyses such as recurrence and lymph node involvement. Second, the retrospective, single-institution design introduces potential selection and information biases, and the findings may not be generalizable to other settings. Third, data on family history and surgical choices were based on patient self-reports, which may have introduced recall bias. In addition, treatment decisions may have been influenced by institutional policies and physician discretion, further limiting external validity. Fourth, the timing of genetic testing varied according to physician judgment and insurance coverage availability, which may have affected case inclusion. Finally, the relatively short observation period made it difficult to evaluate long-term treatment efficacy and recurrence rates. Larger, multicenter studies with longer follow-up are warranted to validate and extend these findings.
Conclusion
In this single-institution retrospective study, we identified subtype-specific clinical and pathological differences between BRCA1- and BRCA2-associated BC in Japanese patients. BRCA2 cancers tended to present with larger tumors and multiple lymph node metastases, whereas BRCA1 cancers were more frequently detected at early T stage. These findings underscore the importance of incorporating BRCA status into individualized treatment and prevention strategies, while larger multicenter cohorts are needed to validate these trends.
Supplemental Material
sj-docx-1-bcb-10.1177_11782234251399355 – Supplemental material for Clinical and Pathological Characteristics and Treatment Implications of BRCA1- and BRCA2-Mutated Breast Cancer in Japanese Patients: A Single-Institution Retrospective Study
Supplemental material, sj-docx-1-bcb-10.1177_11782234251399355 for Clinical and Pathological Characteristics and Treatment Implications of BRCA1- and BRCA2-Mutated Breast Cancer in Japanese Patients: A Single-Institution Retrospective Study by Yumiko Akita, Madoka Iwase, Toyone Kikumori, Dai Takeuchi, Yuko Takano, Takahiro Ichikawa and Norikazu Masuda in Breast Cancer: Basic and Clinical Research
Footnotes
Acknowledgements
We extend our gratitude to the patients who participated in this study. We sincerely thank Maki Morikawa, Mami Morita, and Miki Hatakeyama, the genetic counselors from Nagoya University Hospital, for their invaluable involvement in providing counseling to all patients. We thank Satoko Shimada, MD, PhD (Department of Pathology, Nagoya University Hospital), for her continued contributions to routine breast pathology diagnostics at our institution.
Ethical Considerations
This study was conducted in accordance with the Ethical Guidelines for Medical and Biological Research Involving Human Subjects (Ministry of Health, Labour, and Welfare, Japan) and the Declaration of Helsinki. The study protocol was reviewed and approved by the Institutional Review Board of Nagoya University Hospital (approval no.: 2022-0244).
Consent to Participate
Written informed consent was obtained from all participants.
Consent for publication
Not applicable.
Author Contributions
Yumiko Akita: Conceptualization, Data curation, Formal analysis, Investigation, Methodology, Writing – original draft, Writing – review & editing.
Madoka Iwase: Conceptualization, Methodology, Project administration, Writing – review & editing.
Toyone Kikumori: Methodology, Writing – review & editing.
Dai Takeuchi: Methodology, Writing – review & editing.
Yuko Takano: Methodology, Writing – review & editing.
Takahiro Ichikawa: Methodology, Writing – review & editing.
Norikazu Masuda: Project administration, Writing – review & editing.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Norikazu Masuda has received honoraria (eg, lecture fees) from AstraZeneca, Chugai, Daiichi Sankyo, Eisai, Eli Lilly, and Pfizer. He has also received research funding from AstraZeneca, Chugai, Daiichi Sankyo, Eisai, Eli Lilly, Gilead Sciences, MSD, Novartis, Ono Pharmaceutical, and Pfizer. In addition, he serves on the Board of Directors for the Japanese Breast Cancer Society (JBCS; 2021–2024), the Japan Society of Clinical Oncology (JSCO; 2023–present), and the Japan Breast Cancer Research Group Association (JBCRG; 2021–present). All other authors declare no conflicts of interest.
AI Disclosure
A language model (ChatGPT, OpenAI) was used to assist with language enhancement and refinement in the manuscript. No scientific data were generated or modified using AI.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
