Abstract
Abemaciclib, a cyclin-dependent kinase 4/6 inhibitor, is crucial in treating hormone receptor–positive, human epidermal growth factor receptor 2–negative metastatic or recurrent breast cancer. However, its association with drug-induced interstitial lung disease (DI-ILD) is concerning. We present an 82-year-old woman with breast cancer receiving abemaciclib, who developed persistent cough and malaise. Initial diagnostics suggested pneumonia, supported by ground-glass opacities and consolidations on chest high-resolution computed tomography. Suspecting DI-ILD, a transbronchial lung cryobiopsy (TBLC) was performed, revealing fibrosing organizing pneumonia and confirming abemaciclib-induced ILD. Discontinuing abemaciclib led to significant symptom improvement, supporting the diagnosis. This case report describes the clinical presentation and diagnostic approach in a patient with suspected abemaciclib-induced ILD, including the use. To our knowledge, this is the first reported case of fibrosing organizing pneumonia as a histopathological pattern in abemaciclib-induced ILD, expanding knowledge of this therapy’s pulmonary adverse events. Histopathological features included diffuse lymphocytic infiltration, polypoid intra-alveolar fibrosis, intraluminal granulation tissue plugs with dense hyalinization, hyalinized fibrotic alveolar septa lesions, and obliterative fibrotic processes affecting alveolar ducts. Our case suggests that TBLC might be useful in recognizing DI-ILD by providing detailed lung tissue examination, which can facilitate early diagnosis and guide management. Identifying fibrosing organizing pneumonia indicated a potentially corticosteroid-responsive pathology, suggesting a more favorable prognosis compared with patterns like diffuse alveolar damage. This case highlights the potential for abemaciclib-induced ILD to occur even after prolonged treatment periods, emphasizing the importance of vigilance and consideration of diagnostic intervention for patients on cyclin-dependent kinase 4/6 inhibitors presenting with respiratory symptoms. Timely recognition and appropriate management may mitigate adverse outcomes. Further studies are needed to confirm these findings and to better understand the role of TBLC and histopathological examination in diagnosing and managing abemaciclib-induced ILD.
Keywords
Introduction
One cornerstone in the treatment of hormone receptor–positive, human epidermal growth factor receptor 2–negative metastatic or recurrent breast cancer is the administration of abemaciclib, a cyclin-dependent kinase (CDK) 4/6 inhibitor. While it significantly enhances patient outcomes, its association with drug-induced interstitial lung disease (DI-ILD) is becoming an area of concern. 1 The incidence of DI-ILD with abemaciclib, as reported from Japan, is approximately 1.7%, with mortality of approximately 0.3%. 2 Furthermore, the MONARCH 2 and MONARCH 3 studies, which included patients from various regions, including Europe, North America, and Asia, reported 3 deaths with an incidence rate of 3.4% and a mortality of 0.4%.3 -5 These findings suggest that abemaciclib-induced ILD, while rare, is a significant adverse event worldwide. A range from mild to severe cases has been documented in previous case reports.6 -9 This highlights the necessity for a deeper understanding of the pulmonary adverse events of abemaciclib, particularly in light of its growing use in breast cancer therapy.
The specific mechanisms and histopathological features of abemaciclib-induced ILD remain underexplored, despite the known efficacy of abemaciclib. In the reported cases, only a transbronchial lung biopsy was conducted in one case, and no detailed histopathological examination, such as a transbronchial lung cryobiopsy (TBLC) or surgical lung biopsy, was conducted, which limits our understanding of this adverse effect. 6 This gap in knowledge is significant, considering the range of severity of abemaciclib-induced ILD, which can vary from mild to fatal. The current report aimed to provide a detailed histopathological examination of a case of abemaciclib-induced ILD using TBLC.
Case presentation
This is a case of an 82-year-old female patient who presented with persistent cough and body malaise. She presented to our hospital a week after the onset of symptoms and was promptly admitted with a presumptive diagnosis of pneumonia, corroborated by chest imaging. A plain chest radiograph (Figure 1A) demonstrated bilateral lower lung field opacities, while a high-resolution computed tomography (HRCT) scan (Figure 1B and C) revealed bilateral ground-glass opacities and consolidations, with a predominance in the lower lobes, particularly in the right lung. The abnormalities were mainly distributed in the subpleural regions and were associated with bronchiectatic changes.

Chest radiography at admission (A). High-resolution computed tomography (HRCT) at admission (B, C) and 3 months later (D, E). Chest radiography revealed consolidation and reticular shadow in the bilateral lower field. At admission, HRCT demonstrated nonsegmental consolidation and ground-glass opacities in both lungs, with a predominance in the lower lobes, particularly in the right lung (B, C). After 3 months, most of the ground-glass opacities had resolved, and the consolidations had significantly improved (D, E).
Pertinent medical history includes right breast cancer, surgically resected 25 years prior. She was diagnosed with estrogen receptor–positive (40% of cells stained positive), progesterone receptor–positive (20% of cells stained positive), and human epidermal growth factor receptor 2–negative breast cancer. Postoperative management comprised hormonal therapies with anastrozole or exemestane, administered during breast cancer recurrence from 21 to 11 years prior. Notably, 3 years before her current presentation, she underwent treatment with abemaciclib for ongoing breast cancer management. The initial dose was 150 mg twice daily, but due to neutropenia, it was subsequently reduced to 50 mg daily. Her medical history was otherwise unremarkable, and she was a never smoker. A thorough review of the patient’s medication history confirmed that she was not receiving any drugs known to cause DI-ILD, other than abemaciclib.
Laboratory tests demonstrated a white blood cell count of 2.6 × 109/L, C-reactive protein 1.74 mg/dL, procalcitonin 0.06 ng/mL, Krebs von den Lungen-6 898.7 U/mL, and surfactant protein-D 234.3 ng/mL. Beta-
Physical examination demonstrated an oxygen saturation of 92% on room air, representing a decline from the patient’s baseline of 96%. Breath sounds were clear on auscultation. Eastern Cooperative Oncology Group Performance Status (ECOG PS) was 0 until this episode. Chest HRCT revealed a predominantly subpleural consolidation and ground-glass opacities (GGO) in both lungs, with bronchiectatic dilatations. The HRCT pattern exhibited features consistent with an organizing pneumonia (OP) pattern, accompanied by bronchiectasis. The 2-dimensional echocardiogram showed no cardiac dysfunction. Pulmonary function tests revealed a restrictive pattern with reduced diffusing capacity: forced vital capacity (FVC) was 1.28 L (59.5% of predicted), diffusing capacity for carbon monoxide was 8.28 mL/min/mm Hg (56.5% of predicted), forced expiratory volume in 1 second (FEV1) was 1.28 L (85.9% of predicted), and FEV1/FVC ratio was 100.0%.
On the day of admission, bronchoscopy was performed due to the chest imaging findings and procalcitonin levels, as infectious pneumonia was considered unlikely. In the bronchoalveolar lavage (BAL) from the right B 5 bronchi, the cell fraction was 10% macrophages, 89% lymphocytes, 1% neutrophils, and 0% eosinophils. The CD4/CD8 ratio was 0.4. Culture and cytology of BAL fluid showed no evidence of bacterial, mycobacterial, or fungal infections. In addition, there was no indication of malignancy. The TBLC from the right lower lobe showed fibrosing OP. The TBLC specimens revealed diffuse lymphocytic infiltration accompanied by polypoid intra-alveolar fibrosis (Figure 2A). Intraluminal plugs of granulation tissue (Masson bodies) with dense hyalinization and fibrosis of the central core were observed (Figure 2B and C). In addition, hyalinized fibrotic lesions of alveolar septa and obliterative fibrotic processes affecting the alveolar ducts were observed, resulting in loss of normal alveolar architecture (Figure 2D and E). Epithelial damage was also evident.
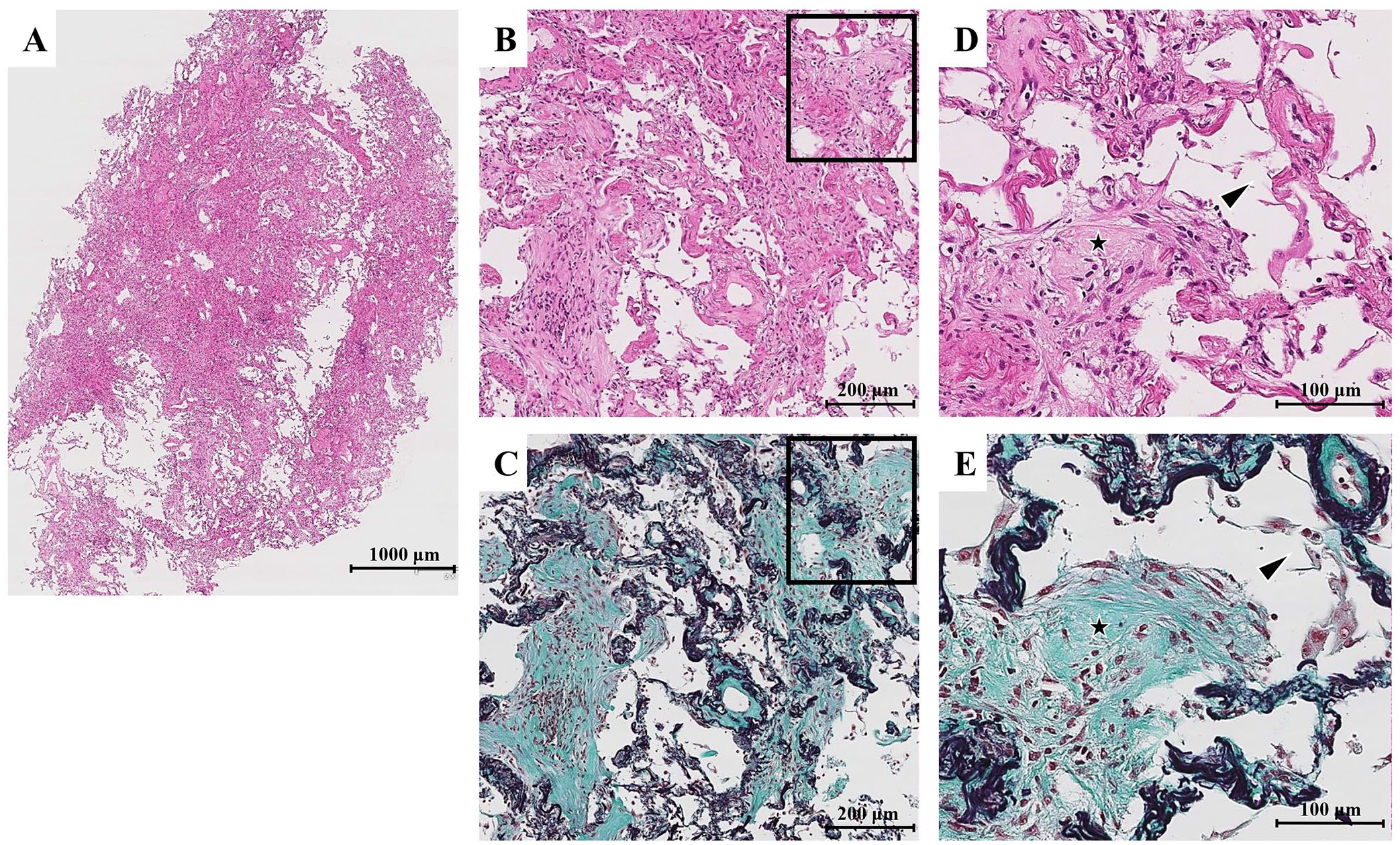
Histologic findings from a transbronchial lung cryobiopsy. Low-magnification (A) and high-magnification (B, C, D, E) views with hematoxylin and eosin (H&E) staining (A, B, D) and Elastica Masson-Goldner (EMG) staining (C, E) are shown. The high-magnification images (D, E) provide an enlarged view of the area enclosed by the square in the low-magnification images (B, C). Diffuse lymphocytic infiltration was observed, accompanied by polypoid intra-alveolar fibrosis. Intraluminal plugs of granulation tissue (Masson bodies) with dense hyalinization and fibrosis of the central core were noted (B, C). Hyalinized fibrotic lesions of alveolar septa and obliterative fibrotic processes affecting the alveolar ducts were also observed (black star in D, E), resulting in loss of normal alveolar architecture. Epithelial damage was also evident (arrowhead in D, E).
The primary working impression was drug-induced ILD due to abemaciclib based on the clinical presentation, laboratory findings, imaging studies, and histopathological evidence. Hypersensitivity pneumonitis and collagen vascular disease–associated interstitial lung disease were considered in the differential diagnosis; however, these conditions were deemed inconsistent with the patient’s clinical history, physical examination findings, and serological test results. Given the patient’s clinical stability, we decided to discontinue abemaciclib without administering steroids to observe if the condition would improve with drug cessation alone. The possibility of lung-dominant collagen vascular disease was considered; however, the diagnosis of DI-ILD was further supported by the marked improvement in HRCT findings and clinical symptoms after discontinuation of abemaciclib without antibiotic or corticosteroid use (Figure 2C and D). While distinguishing drug-induced ILD from cryptogenic OP (COP) can be challenging, a key factor supported our diagnosis: the marked improvement after abemaciclib discontinuation alone. This clinical course is more consistent with drug-induced ILD than COP, which often requires corticosteroid treatment.
Discussion
This article reports a case of DI-ILD associated with abemaciclib diagnosed via TBLC. First, we identified fibrosing OP as one histopathological manifestation of abemaciclib-induced ILD, providing new insights into the pulmonary consequences of this targeted cancer therapy. The concept of a fibrosing variant of OP has been previously described in the literature. In 1997, Yousem et al conducted a comparative study of steroid-responsive and steroid-resistant cases of idiopathic bronchiolitis obliterans OP/COP to identify histopathologic predictors of outcome. They highlighted poor prognostic indicators, including background remodeling of the pulmonary parenchyma (scarring/interstitial fibrosis) and dense eosinophilic hyalinization of the fibromyxoid plugs within the alveolar spaces. 10 Subsequently, in 2013, the international multidisciplinary classification of the idiopathic interstitial pneumonias recognized a “fibrosing variant of OP” in the context of COP. It was noted that some patients exhibiting this mixed pattern of fibrosis and OP have underlying conditions such as polymyositis or antisynthetase syndrome. 11 Building on these observations, Beardsley and Rassl proposed the term fibrosing OP to encompass the histologic features of dense hyalinization and fibrosis of the central core of the Masson bodies, hyalinized fibrotic lesions of alveolar septa, and obliterative fibrotic processes primarily affecting the alveolar ducts, resulting in loss of normal alveolar architecture. 12 Fibrosing OP is characterized by the presence of OP with additional features such as dense hyalinization and fibrosis of the central core of the Masson bodies, hyalinized fibrotic lesions of alveolar septa, and obliterative fibrotic processes mainly in the alveolar ducts with loss of normal alveolar structure. 12 These findings suggest that fibrosing OP represents a more chronic and potentially treatment-refractory process compared with classic OP. The presence of fibrosing OP has important implications for prognosis and treatment strategies, as it may indicate a more aggressive clinical course. Therefore, the identification of fibrosing OP carries significant clinical relevance in the management of patients with OP. Second, our case suggests that TBLC may have diagnostic value in recognizing DI-ILD, potentially providing a basis for future investigations.
Diagnosing abemaciclib-induced ILD in this case was challenging for 2 main reasons: the long treatment duration and the negative Drug Lymphocyte Stimulation Test (DLST) result. Although the patient had been on abemaciclib for 3 years before developing ILD, which is longer than previously reported cases, drug-induced ILD can occur even after prolonged exposure. A postmarketing study by Chen et al 2 reported cases occurring up to 11 months after treatment initiation, though our case represents an unusually long duration. Our case suggests that abemaciclib-induced ILD may potentially occur even after prolonged exposure, highlighting the need for continued vigilance throughout the treatment course. Furthermore, the DLST was negative in our case. However, the DLST has limitations, including low sensitivity and specificity, and a negative result does not exclude the possibility of drug-induced ILD. 13 Negative DLST results have been reported in other cases of abemaciclib-induced ILD, including the case reported by Okayasu et al. 6 Despite these challenges, the marked improvement after abemaciclib discontinuation alone strongly supported our diagnosis.
Our findings show that abemaciclib-induced ILD histopathologically manifests as fibrosing OP. A postmarketing study by Chen et al in Japan reported 4 main imaging patterns in abemaciclib-induced ILD: diffuse alveolar damage (DAD), OP, GGO, and nonspecific interstitial pneumonia. However, these were based solely on radiological findings, with no histopathological confirmation, leaving the underlying pathology unclear. Four detailed case reports of DI-ILD attributed to abemaciclib treatment have been documented in prior studies.6 -9 These include a case manifesting with elevated inflammatory cells in the BAL fluid, another diagnosed as eosinophilic pneumonia due to increased eosinophils in the BAL fluid, and a case of alveolitis accompanied by epithelial cell damage as identified through transbronchial lung biopsy. Our case is different in that we identified fibrosing OP as the histopathological pattern using TBLC. This suggests that abemaciclib-induced ILD can manifest with a variety of histopathological patterns, and fibrosing OP may be one of them. While some agents induce specific lung reactions, such as eosinophilic pneumonia by minocycline, others, including amiodarone, induce various responses, including DAD and various interstitial pneumonia types.14,15 The pathogenesis of CDK4/6 inhibitor-associated DI-ILD remains speculative, although in vivo studies indicate that palbociclib could augment inflammatory cell mobilization in the BAL due to cell cycle arrest and consequent cellular senescence, implicating inflammatory cell infiltration as the disease’s primary component.16,17 The histopathology of our case, featuring fibrosing OP, supports these hypotheses. To elucidate the histopathology of abemaciclib-induced ILD, further cases are required. Moreover, the predominance of case reports from Asia suggests a potential racial predisposition to abemaciclib-induced ILD, warranting further investigation into ethnic differences in susceptibility and manifestation of this adverse event.
Interestingly, TBLC allowed a detailed examination of drug-induced ILD histopathology. The TBLC’s utility was reported as a relatively safe procedure with a diagnostic yield of approximately 80% in ILD. 18 Although current review articles suggest that histological confirmation is not required for a DI-ILD diagnosis, TBLC may aid in recognizing interstitial pneumonia and excluding infection, potentially contributing to early DI-ILD diagnosis.19,20 While BAL findings, particularly the lymphocyte fraction, may provide some insights into the potential response to corticosteroid treatment, it is important to recognize that BAL cellular profiles may not always accurately reflect the extent and nature of lymphocytic infiltration in the lung parenchyma.21,22 In our case, the identification of fibrosing OP on TBLC narrowed the differential diagnosis to conditions such as polymyositis and antisynthetase syndrome. 12 However, the absence of clinical features characteristic of these conditions increased the likelihood of drug-induced ILD. Currently, reports on the histopathological findings of abemaciclib-induced ILD are limited. As more data accumulate, the diagnostic confidence based on histopathology is expected to increase. The discussed case report featured mild DI-ILD without desaturation; however, epithelial damage that was histologically observed required corticosteroid treatment, culminating in a positive outcome. 6 In our case, TBLC enabled early disease diagnosis based on the histopathological findings, which suggested a potentially corticosteroid-responsive pathology. However, instead of initiating corticosteroid therapy, we opted for close monitoring after discontinuing abemaciclib. The patient’s condition improved significantly with drug cessation alone, without the need for corticosteroids. This favorable outcome contrasts with cases presenting with DAD, a histopathological pattern associated with acute lung injury and high mortality. 23 The absence of DAD on histology, combined with the patient’s improvement after abemaciclib discontinuation alone, justified our cautious approach of close monitoring without immediate corticosteroid treatment. This case would have satisfied the severity predictors reported in prior studies without a definitive diagnosis of interstitial pneumonia on histopathology: age >70 years, ECOG PS ⩾ 1, and pre-existing ILD, which may have led to unnecessary corticosteroid administration. 2
In summary, our case report presents fibrosing OP as a histopathological pattern observed in this instance of abemaciclib-associated pulmonary adverse event. This observation may contribute to the understanding of lung injury mechanisms in abemaciclib-induced ILD. Furthermore, the use of TBLC in this case highlights its potential usefulness in obtaining adequate lung tissue for accurate diagnosis. Notably, this case demonstrates that abemaciclib-induced ILD can occur even after prolonged treatment periods, suggesting the need for continued vigilance throughout the course of therapy. Further studies could potentially help accumulate more histopathological data and refine management approaches for this condition. However, it is important to note that this study is based on a single case report, which limits the generalizability of our findings. Further research involving a larger number of cases is necessary to determine whether fibrosing OP is a consistent histopathological manifestation of abemaciclib-induced ILD.
Declarations
Footnotes
Acknowledgements
Ethics approval and consent to participate
This case report did not require ethical approval as per institutional policies. However, written informed consent for participation and publication was obtained from the patient.
Consent for publication
Written informed consent for publication was obtained from the patient.
Author contributions
Funding:
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Competing interests:
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Availability of data and materials
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
