Abstract
Prior to the advent of the HER2-targeted monoclonal antibody trastuzumab, HER2+ breast cancer (BC) was considered an aggressive disease with a poor prognosis. Over the past 25 years, innovations in molecular biology, pathology, and early therapeutics have transformed the treatment landscape. With the advent of multiple HER2-directed therapies, there have been immense improvements in oncological outcomes in both adjuvant and metastatic settings. Currently, 8 HER2-targeted therapies are approved by the Food and Drug Administration (FDA) for the treatment of early-stage and/or advanced/metastatic disease. Nonetheless, approximately 25% of patients develop recurrent disease or metastasis after HER2-targeted therapy and most patients with HER2+ metastatic breast cancer (MBC) die from their disease. Given the many mechanisms of resistance to HER2-directed therapy, there is a pressing need to further personalize care for patients with HER2+ MBC, by the identification of reliable predictive biomarkers, and the development of novel therapies and combination regimens to overcome therapeutic resistance. Of particular interest are established and novel antibody-drug conjugates, as well as other novel therapeutics and multifaceted approaches to harness the immune system (checkpoint inhibitors, bispecific antibodies, and vaccine therapy). Herein, we discuss standard-of-care treatment of HER2+ MBC, including the management of breast cancer brain metastases (BCBM). Furthermore, we highlight novel treatment approaches for HER2+ MBC, including endeavors to personalize therapy, and discuss ongoing controversies and challenges.
Introduction
In 2023, approximately 300 590 persons were diagnosed with metastatic breast cancer (MBC) in the United States, 1 and approximately 15% to 20% were human epidermal growth factor receptor 2 (HER2) positive. 2 This breast cancer (BC) subtype is characterized by overexpression of HER2, a ligand orphan receptor tyrosine kinase that forms heterodimers with other HER family members (HER1, HER3, and HER4), amplifying their signal. 3 Human epidermal growth factor receptors are composed of an extracellular ligand-binding domain, a transmembrane domain, and an intracellular tyrosine kinase domain. When external ligands bind to HER proteins, homo- or heterodimerization of the receptor occurs, activating downstream signaling (including the mitogen-activated protein kinase [MAPK] and phosphatidylinositol-3-OH kinase [PI3K] pathways), promoting cell division and growth while inhibiting apoptosis (Figure 1). Overexpression or amplification of the HER2 oncogene plays an important role in development and progression of this BC subtype and correlates with chemotherapy resistance and aggressive disease. 4 Per the American Society of Clinical Oncology–College of American Pathologists guidelines, HER2-positivity is defined as tumors that exhibit 3+ positive staining via immunohistochemistry (IHC) in ⩾10% of tumor cells, or tumors with 2+ or equivocal IHC staining where HER2 gene amplification may be detected by fluorescence in situ hybridization (FISH).5,6 Historically, HER2+ and triple-negative BC subtypes carried the poorest prognosis, with the highest recurrence and mortality rates. 7 HER2+ BC also has a higher propensity for intracranial metastasis, which is associated with a worse prognosis. 8 The discovery that HER2 was a major oncogenic driver and treatment target led to transformative therapeutic advances over the last 25 years. 7 Specifically, the HER2-targeted monoclonal antibody trastuzumab, combined with chemotherapy, improved disease-free survival (DFS) and overall survival (OS) in adjuvant and metastatic settings, 9 with a 50% reduction in recurrence and 30% improvement in survival. 3 Since then, other HER2-directed therapies have added to this benefit. 4 Currently, most patients diagnosed with stage I to III HER2+ BC are cured. Of patients who initially present with de novo MBC, 40% to 50% of cases are HER2+, 7 which reflects the high cure rates. However, despite remarkable advances, intrinsic and acquired resistance to HER2-directed therapies remains a vexing challenge, and prevention and treatment options for patients with HER2+ breast cancer brain metastases (BCBM) remain suboptimal. 10 Recently, a new category of patients with “HER2-low” BC were identified (~45%-55% of all BCs, that is, HER2 IHC 1+ or 2+ in the absence of HER2 gene amplification by FISH); 11 this subtype can respond well to HER2-targeted antibody-drug conjugates (ADCs). 12 Reviews detailing management options for HER2-low MBC are available elsewhere.13,14

HER2 signaling pathways. Mechanism of action of standard and novel HER2-directed therapies. Approved HER2-directed therapies include trastuzumab, which binds to the juxtamembrane extracellular subdomain IV of human epidermal growth factor receptor 2 (HER2) resulting in the inhibition of HER2 signaling; pertuzumab, which binds to the HER2 subdomain inhibiting homodimerization and heterodimerization of HER2 and HER3; ado-trastuzumab emtansine, T-DM1, which is an antibody-drug conjugate (ADC) that includes trastuzumab linked to emtansine, a microtubule inhibitor which is released after ADC phagocytosis from intracellular lysosomes; trastuzumab deruxtecan, T-DXd, which is another ADC comprised of trastuzumab and deruxtecan and is considered a potent topoisomerase I inhibitor; margetuximab, a monoclonal antibody (mAb) ADC that combines trastuzumab with an altered FC-γ domain causing T-cell activation and antibody-mediated cell cytotoxicity; lapatinib, a reversible tyrosine kinase inhibitor (TKI) of HER1 and HER2 that inhibits further downstream pathway signaling (AKT, PI3K, RAS/RAF/MEK/MAPK); neratinib, which is a TKI that inhibits HER1, HER2, and HER4; and tucatinib, which is a HER2-specific TKI. Other targeted therapies shown include the PI3K inhibitor alpelisib; and cyclin-dependent kinase 4 (CDK4) and CDK6 inhibitors (CDK4/6i) (palbociclib, ribociclib, and abemaciclib), which have or are currently being studied in clinical trials in combination with HER2-targeted agents . Olaparib and talazoparib (PARP inhibitors) ultimately result in DNA breaks and impair DNA repair pathways. Bispecific antibodies are currently being studied in clinical trials, linking various domains of HER2 to specific ligands on T cells, resulting in T-cell-mediated death.
Current Standard of Care
There are 8 FDA-approved HER2+ targeted treatments for HER2+ MBC: monoclonal antibodies (mAb) (trastuzumab [Herceptin], pertuzumab [Perjeta], and margetuximab [Margenza]); antibody-drug conjugates (ADCs) (ado-transtuzumab emtansine [T-DM1], fam-trastuzumab deruxtecan [T-Dxd]), and tyrosine kinase inhibitors (tucatinib [Tuksya], lapatinib [Tykerb], neratinib [Nerlynx] and pyrotinib [approved for use in China, but not in the United States]). Table 1 shows approved therapies for HER2+ MBC, including toxicity and efficacy data from seminal trials.
Approved treatments for HER2+ MBC.

Abbreviations: ab, antibody; AC, doxorubicin + cyclophosphamide; ADCC, antibody-dependent cell-mediated cytotoxicity; AE, adverse events; AKI, acute kidney injury; BCBM, breast cancer brain metastases; BM, brain metastasis; C, capecitabine; CI, confidence interval; DP, disease progression; DB01, Destiny-Breast01; DB03, Destiny-Breast03; DoR, duration of response; EBC, early breast cancer; EC, epirubicin + cyclophosphamide; Erβ, estrogen receptor beta; FDA, Food and Drug Administration; HFS, hand foot syndrome; HR, hazard ratio; ICP, intracranial pressure; IRR, infusion-related reaction; L, lapatinib; LVSD, left ventricular systolic dysfunction; MBC, metastatic breast cancer; mPFS, median progression-free survival; mos., months; mOS, median overall survival; N, neratinib; OR, objective response; ORR, overall response rate; OS, overall survival; P, pyrotinib; PFS, progression-free survival; PE, pulmonary embolism; POD, progression of disease; PPES, palmar-plantar erythrodysesthesia syndrome; pts, patients; RRR, relative risk reduction; SAH, subarachnoid hemorrhage; TCP, thrombocytopenia; TKI, tyrosine kinase inhibitors; TTP, time to progression; 1L, first line; 2L, second line; 3L, third line.
HER2-Targeted Therapies
Monoclonal antibodies
In 1998, trastuzumab was FDA-approved in combination with paclitaxel for treatment of HER2+ MBC. Notable side effects of trastuzumab included cardiomyopathy, pulmonary toxicity, and infusion reactions.3,28 Pertuzumab was FDA-approved in 2012 in combination with trastuzumab and docetaxel for the 1L treatment of HER2+ MBC based on the phase III CLEOPATRA trial. 29 For patients with HER2+ MBC treated with docetaxel, trastuzumab + pertuzumab (THP) versus docetaxel, trastuzumab + placebo (TH) in the 1L metastatic setting, 8-year OS rates were 37% in the pertuzumab arm versus 23% in the placebo arm, conferring an approximately 16-month OS advantage favoring THP.15,30 Notable adverse effects included diarrhea, rash, and cardiomyopathy. Several trastuzumab (±pertuzumab) biosimilars have been approved for use in HER2+ BC. 31 Benefits include the option of subcutaneous administration in some cases, which is quicker and convenient, as well as reduced costs for patients and oncology infusion centers. 32 However, widespread adoption has been slow, due to concerns regarding efficacy and insurance issues.33,34
Margetuximab is a novel Fc-engineered HER2-targeted mAb that triggers innate and adaptive immunity. 35 The Fc fragment targets the same epitope as trastuzumab and was manufactured to increase affinity for activating CD16A (FcγRIIIA) and decrease binding to inhibitory CD32B (FcγRIIB).16,36 In the SOPHIA trial, patients with HER2+ MBC who had received ⩾2 lines of HER2-targeted therapies were randomized 1:1 to investigator’s choice of chemotherapy + trastuzumab or chemotherapy + margetuximab. The median OS (mOS) was 21.6 months with margetuximab versus 21.9 months with trastuzumab (P = .620). A preplanned, exploratory analysis of CD16A genotyping noted a possible OS improvement for margetuximab in CD16A-158FF patients versus trastuzumab (mOS, 23.6 vs 19.2 months) and a possible OS improvement for trastuzumab in CD16A-158VV patients versus margetuximab (mOS, 31.1 vs 22.0 months). Margetuximab and trastuzumab had comparable safety profiles. Final overall OS analysis was not significantly increased in the margetuximab arm.16,17 The most common AEs in the margetuximab arm were fatigue, nausea, vomiting, diarrhea, and LV dysfunction. 37 Going forward, margetuximab studies in HER2+ BC with different CD16A allelic variants could be conducted.
Tyrosine kinase inhibitors
Tyrosine kinase inhibitors (lapatinib, neratinib, tucatinib, and pyrotinib) are an orally administered treatment option for HER2+ MBC. They differ by HER protein specificity, molecular weight, binding reversibility, and side-effect profiles. 38 All are FDA-approved in HER2+ MBC, except pyrotinib. GI, cutaneous, and hepatotoxicities are commonly encountered. In 2007, lapatinib, combined with the 5-fluorouracil prodrug capecitabine, was the second HER2-targeted agent FDA-approved for HER2+ MBC. A phase III trial showed that for patients with HER2+ MBC previously treated with taxane and trastuzumab, lapatinib + capecitabine improved progression-free survival (PFS) versus capecitabine alone (8.4 vs 4.4 months; P < .001). 39 This regimen was the preferred 2L option until superseded by T-DM1 in 2013, based on the EMILIA trial. 25
In 2020, neratinib + capecitabine was FDA-approved based on the NALA trial (n = 621), wherein patients with HER2+ MBC post ⩾2 prior lines of HER2-directed therapy for MBC were randomized 1:1 to neratinib + capecitabine or lapatinib + capecitabine. 19 Approximately one third of participants had received prior T-DM1, trastuzumab and pertuzumab. A 2.2-month PFS gain (P = .0003) and a 1.8-month OS gain (P = .208) were noted in the neratinib arm, which led to FDA approval of neratinib + capecitabine in 2020. Regarding CNS outcomes, there was delayed time to intervention for and reduced cumulative incidence of BCBM in the neratinib arm. Regarding toxicities, 24% of patients in the neratinib arm had grade 3 or 4 diarrhea; however, treatment discontinuations due to treatment-related AE were comparable. 19 The CONTROL trial subsequently showed improved neratinib tolerability with dose escalation over the initial 2 weeks of therapy. 40
Tucatinib + trastuzumab + capecitabine was FDA-approv-ed for 3L treatment of HER2+ MBC in 2020 21 based on results from the HER2CLIMB study (NCT02614794), 20 wherein 612 patients were randomized 2:1 to capecitabine, trastuzumab + tucatinib versus capecitabine, trastuzumab + placebo. All participants had received prior T-DM1, pertuzumab, and trastuzumab. A median of 3 prior lines of treatment had been administered in the metastatic setting. Nearly half of the enrollees had BCBM and ~40% had untreated or previously treated and progressing BCBM, which was unique, as stable BCBM were usually required for study enrollment. Median PFS (mPFS) was 7.6 versus 4.9 months for the tucatinib versus placebo arm (P < .00001). 41 Median overall survival (mOS) was 24.7 versus 19.2 months in the tucatinib versus placebo arm (P = .004), respectively. Tucatinib-based therapy was well tolerated with low discontinuation rates. In BCBM, addition of tucatinib to trastuzumab + capecitabine improved intracranial objective response rate (RR) (47.3% vs 20.0%, P = .03), reduced risk of intracranial progression or death by 68%, and reduced risk of death by 42%, with prolongation of OS by ⩾6 months. 21 An updated analysis showed mOS was 9.1 months longer in the tucatinib arm (21.6 months; 95% CI, 18.1-28.5) versus the placebo arm (12.5 months; 95% CI, 11.2-16.9). 22 The risk of developing new BCBM as a site of first progression or death was reduced by 45.1% in the tucatinib arm versus the placebo arm (hazard ratio [HR], 0.55; 95% CI, 0.36-0.85). 22 Currently, tucatinib, capecitabine + trastuzumab is a 2L or 3L treatment for HER2+ MBC.
HERCLIMB-02 (NCT03975647) was a double-blind phase III trial of tucatinib + T-DM1 which included patients with HER2+ MBC previously treated with trastuzumab and a taxane. A significant improvement in mPFS was noted in the T-DM1 arm (9.5 months) versus 7.4 months in the placebo arm (P = .0163), with a decreased risk of progression or death by 24.1% in the tucatinib arm. 42 Furthermore, mPFS in patients with BCBM was 7.8 months (tucatinib arm) versus 5.7 months (placebo arm) (HR, 0.64). Although a higher rate of G3 hepatic AEs was seen in the tucatinib arm (AST/ALT elevations; 28.6% vs 7.3%), AEs were typically short-lived and manageable. If T-DM1 + tucatinib is FDA-approved, incorporation into treatment algorithms for HER2+ MBC as a joint third- or fourth-line treatment option is likely. Limitations include immature OS data at writing and the inability to compare the HER2CLIMB-02 regimen to T-DXd or the HER2CLIMB regimen. Importantly, HERCLIMB-02 is the second randomized trial showing that tucatinib-based therapy delays progression in HER2+ BCBM.
In 2020, pyrotinib + capecitabine was approved for 2L treatment of HER2+ MBC in China, based on results from PHOEBE which enrolled patients with HER2+ MBC who had received ⩽2 lines of chemotherapy for metastatic disease, including trastuzumab and taxanes. Patients were randomized 1:1 to pyrotinib or lapatinib + capecitabine. Pyrotinib + capecitabine improved PFS versus lapatinib + capecitabine (12.5 vs 5.6 months; P < .0001). mOS was not reached in the pyrotinib group and was 26.9 months (22.4-not reached) in the lapatinib group. 24 Pyrotinib-based therapy is an option in regions of China that do not have access to ADCs. It is unclear whether pyrotinib will be used internationally in the future.
Antibody-drug conjugates
Antibody-drug conjugates combine an antigen-specific antibody backbone with a potent cytotoxic payload, resulting in an improved therapeutic index. T-DM1 combines trastuzumab with the maytansinoid, DM1 (a potent microtubule-disrupting agent), joined by a stable linker.25,43 T-DXd has a higher drug: antibody ratio (~8:1) versus T-DM1 (~3.5:1) and inhibits topoisomerase I versus microtubule inhibition. 44 The high membrane permeability of DXd facilitates local bystander effects, inducing malignant cell death in the tumor microenvironment. 44 Side effects of HER2-targeted ADCs include cytopenias, nausea, and interstitial lung disease (ILD). In 2013, T-DM1, the prototype ADC, was FDA-approved for 2L treatment of HER2+ MBC based on EMILIA (NCT00829166) which showed that T-DM1 increased PFS in HER2+ MBC post trastuzumab and a taxane, versus lapatinib + capecitabine (9.6 vs 6.4 months, respectively; P < .001). 25 mOS was also superior in the T-DM1 arm (30.9 vs 25.1 months, [P < .001]). T-DM1 was the standard 2L option until 2021 when superseded by T-DXd, based on striking results from the phase III DESTINY Breast03 trial (DB03). DB03 randomized patients with HER2+ MBC +/- BCBM, previously treated with taxane and trastuzumab, to T-DM1 versus T-DXd. 27 Updated results showed that PFS was 28.8 months with T-DXd and 6.8 months with T-DM1 (HR, 0·33; P < .0001); mOS not reached in either arm. 45 The incidence of ⩾grade 3 AEs was similar. Drug-related ILD occurred in 15% patients on T-DXd versus in 3% on T-DM1, with no grade 4 or 5 events. Following accelerated FDA approval in the 3L HER2+ MBC setting, 46 T-DXd was subsequently approved in the 2L in 2022. Results from DESTINY-Breast09 (NCT04784715) will determine whether T-DXd will replace THP as 1L treatment for HER2+ MBC.
Triple-positive MBC
Targeting both ER and HER2 is relevant in patients with HR+ HER2+ MBC, as there is signaling overlap between the pathways and dual inhibition is more effective. 47 Combination of an aromatase inhibitor + HER2-targeted therapy (ie, trastuzumab + pertuzumab) as evaluated in the PERTAIN trial is an option for older patients who are not chemotherapy candidates. 48
Evolving treatment and sequencing considerations
The treatment landscape for HER2+ MBC is crowded. Treatment decisions are influenced by patient comorbidities, potential toxicities, and activity against BCBM, PFS, and OS data. Evaluation of novel compounds and regimens is underway and will further impact treatment sequencing in the future. Currently, standard 1L therapy remains THP based on the 16-month OS advantage noted in CLEOPATRA. 15 For patients relapsing ⩽6 months after completion of adjuvant therapy, and for those relapsing ⩽12 months from completion of pertuzumab-based adjuvant treatment, T-DXd is the preferred 1L option. For patients relapsing 6 to 12 months after non-pertuzumab-based adjuvant treatment, 1L THP is recommended. 49 T-DXd is standard 2L therapy; however, tucatinib, capecitabine + trastuzumab is an acceptable 2L option for patients unable to tolerate T-DXd or for isolated CNS progression. Apart from tucatinib, trastuzumab + capecitabine, other regimens used in the 3L and beyond include (1) neratinib + capecitabine, (2) margetuximab + chemotherapy, (3) T-DM1 (if not previously administered), (4) lapatinib + trastuzumab, (5) chemotherapy + trastuzumab, and (6) endocrine therapy (ET) + trastuzumab (HR+ disease). Other questions relate to activity of (1) neratinib after a tucatinib-based regimen, and (2) T-DM1 after progression on T-DXd. As T-DM1 and T-DXd have different chemotherapy moieties, response is theoretically possible. Ongoing trials of FDA-approved targeted therapies in HER2+ MBC are shown in Table 2.
Selected trials of FDA-approved HER2-directed therapies in HER2+ MBC currently recruiting participants.

Abbreviations: ADC, antibody-drug conjugate; AST, advanced solid tumors; BM, brain metastases; Cmax, maximum plasma concentration; CNS, central nervous system; cORR, confirmed overall response rate; EBC, early breast cancer; ET, endocrine therapy; HP, Herceptin + Perjeta; LMD, leptomeningeal disease; MBC, metastatic breast cancer; MTD, maximum tolerated dose; N/A, not applicable; pCR, pathologic complete response; PFS, progression-free survival; T-DXd, trastuzumab deruxtecan; TKI, tyrosine kinase inhibitor; Tx, treatment; 1L, first line; 2L, second line.
The current treatment paradigm for HER2+ MBC is shown in Figure 2. In the future, predictive biomarkers may identify which patients can discontinue therapy safely. Other patients may exhibit intrinsic or acquired resistance to HP based therapy in the 1L and could benefit from therapy change or escalation. For example, mutation activation within the PI3K pathway with ERBB3 mutation, PIK3CA mutation, and PTEN loss alters HER2-targeted efficacy. MAPK mutations ultimately can lead to variations in pathway dependence (eg, a PI3K/AKT to ERK/MEK switch) causing HER2 resistance and MEK inhibitor sensitivity. 50

Treatment paradigm for HER2+ MBC. Abs indicates antibodies; ADCs, antibody-drug conjugates; CDK4/6i, cyclin-dependent kinase 4/6 inhibitors; MBC, metastatic breast cancer; PI3Ki, PI3K inhibitors; T-DM1, trastuzumab emtansine; T-DXd, trastuzumab- deruxtecan; TKIs, tyrosine kinase inhibitors.
De-escalation strategies
Personalization of therapy as it relates to de-escalation versus escalation is an important principle. Some patients are exceptional responders to 1L HER2-targeted therapy and remain in remission on maintenance trastuzumab ± pertuzumab for years after induction therapy. An important question is the optimal selection strategy and timeline for discontinuation of maintenance therapy in a subset of patients with HER2+ MBC.
In HER2+ EBC, pathologic complete response to preoperative systemic therapy is an established tool to guide therapy de-escalation or escalation. In HER2+ MBC, a subset of patients with HER2+ MBC on 1L maintenance therapy (trastuzumab + pertuzumab) have extended PFS and OS (8-year OS 37% in CLEOPATRA). 15 Therefore, some patients may be cured but nevertheless continue HER2-directed therapy, which, although well tolerated, could be associated with unnecessary expense and patient inconvenience. As minimal residual disease assays are correlated with response in HER2+ exceptional responders, 51 the hypothesis of STOP-HER2 (NCT05721248) is that selected patients with HER2+ MBC can safely stop HER2-directed treatment. STOP-HER2 is enrolling patients who are progression free after ⩾3 years on 1L HER2-targeted therapy. Participants stop therapy with close follow-up and extensive biomarker assessment. A parallel, observational cohort enrolls patients who prefer to continue therapy. Primary goals are to evaluate 1-year PFS in patients with HER2+ MBC and exceptional response to HER2-targeted therapy who either continue or stop the same. SAPPHO is a phase II study in development which will test efficacy of a sequential regimen of non-cross-resistant HER2-targeted treatments in de novo HER2+ MBC. The primary objective is to estimate the probability of enrollees being off anti-cancer treatment (except ET as applicable) and free of disease progression 4 years from study entry. Results from both trials will advance our knowledge regarding optimization of treatment strategies for HER2+ MBC.
Mechanisms of resistance to HER2-directed therapy
Patients with HER2+ MBC may live many years, and exceptional responders exist, particularly those with oligometastatic disease. 52 However, HER2+ MBC is usually incurable, and mOS is <5 years . 53 A barrier to improved OS is primary or acquired resistance to HER2-directed therapies (22%-25%).54,55 Continued research focusing on unraveling novel and established mechanisms of resistance is ongoing; these include impaired drug binding to the HER2 receptor, which triggers compensatory mechanisms within the HER family (eg, HER3), including activation of different receptor tyrosine kinases (IGF-1R, MET), overactivity of the HER2 downstream signaling cascade (ie, the PI3K/AKT/mTOR pathway), and PTEN suppression. 56 Others include masking or loss of the HER2 epitope, HER2 heterogeneity, and host and tumor immunity. 57 Additional downstream pathways implicated in carcinogenesis include bidirectional crosstalk between HER2 and ER, and the cyclin D1-CDK4/Rb axis. 54 Reviews outlining resistance mechanisms to HER2-directed therapies are available elsewhere. 58
Biomarkers
Given the evident heterogeneity of HER2+ MBC, clinica-lly validated biomarkers could identify primary and acquired resistance to HER2-directed therapies, allowing better refinement of systemic therapy. However, biomarker discovery for HER2+ BC has been elusive. Currently, the use of HER2-targeted therapies is largely determined based on HER2 status. Hormone receptor (HR) expression is a notable biomarker as ~ 60% to 70% of HER2+ BC co-express HR and HER2, which modulates response to both HER2-directed and endocrine therapy due to “crosstalk” between the ER and HER2 pathways. 59 Combined HER2 and ER targeting can, therefore, be an effective treatment strategy in selected clinical settings.
Biomarkers evaluated to date include PIK3CA, HER2 co-ligands, PTEN, programmed death ligand 1 (PD-L1), tumor-infiltrating lymphocytes (TILs), and FcdeltaR polymorphisms. 60 Biomarkers most likely to inform prognostic stratification and treatment in the short to medium term include heterogeneity and quantification of HER2 levels, HER3 expression, intrinsic molecular subtypes (PAM50 analysis), DNA mutations, and other immune-related factors. 61 In CLEOPATRA, while HER2, HER3, and PIK3CA were prognostic, only HER2 accurately selected patients for trastuzumab + pertuzumab-based treatment in HER2+ MBC. 15 In EMILIA, T-DM1 was effective in both PI3KCA-mutated and wild-type tumors, 25 and in TH3RESA, a phase III trial of T-DM1 versus treatment of physician’s choice in pretreated HER2+ MBC, T-DM1 prolonged mPFS in all biomarker subgroups analyzed. 62
Future Directions
Novel HER2-targeted antibodies
Bispecific antibodies under evaluation in HER2+ MBC (Table 3) include zanidatamab, a HER2-targeted bispecific antibody that binds 2 nonoverlapping extracellular domains of HER2 (ECD4 and ECD2) and KN026, which simultaneously binds 2 distinct HER2 epitopes. 63 In a phase 1b/2 trial (NCT04276493), zanidatamab + docetaxel was tolerable and active in 1L HER2+ MBC (ORR, 86.4%; 95% CI, 65.1-97.1). In another trial evaluating zanidatamab + palbociclib + fulvestrant in pretreated HR+ HER2+ MBC, responses were seen in most participants (NCT04224272). Furthermore, in a phase I trial in HER2+ MBC, KN026 was well tolerated, with an ORR of 28.1% and mPFS of 6.8 months. 64 Interestingly, co-amplification of HER2/CDK12 may select patients who benefit most. Bispecific antibodies which simultaneously target both HER2 and various immune targets are also under clinical evaluation (Table 3) and are discussed in the section on novel immunotherapies.
Selected trials of HER2-directed therapies under investigation in HER2+ MBC (completed, ongoing and pending activation).

Abbreviations: ADC, antibody-drug conjugate; AE, adverse events; AST, advanced solid tumors; BAT, bispecific antibody armed activated T cells; BBB, blood-brain barrier; CBR, clinical benefit rate; CI, confidence interval; CNS, central nervous system; CR, complete response; DCR, disease control rate; DLT, dose limiting toxicities; DoR, duration of response; ER+, estrogen receptor positive; ET, endocrine therapy; G, grade; HR, hazard ratio; IS, intrinsic subtype; L, line; LVEF, left ventricular ejection fraction; mAB, monoclonal antibody; MBC, metastatic breast cancer; mos., months; MMAE, monomethyl auristatin E; MTD, maximum tolerated dose; OR, overall response; ORR, objective response rate; OS, overall survival; PD, progressive disease; PD1, programmed death; PDL1, programmed death ligand 1; PFS, progression-free survival; Ph, phase; PK, pharmacokinetics; PR, partial response; RP2D, recommended phase 2 dose; SAE, serious adverse events; SD, stable disease; SOC, standard of care; T-DM1, ado-transtuzumab emtansine; T-DXd, fam-transtuzumab deruxtecan; TEAE, treatment-emergent adverse events; TKI, tyrosine kinase inhibitor; TPC, treatment of physicians choice; TTP, time to progression.
Novel HER2-targeted ADCs
HER2-directed ADCs are key immune-targeted strategies, which increase cytotoxicity while reducing chemotherapy off-target AEs due to antibody-driven drug internalization. Compared with treatment of physicians choice, trastuzumab duocarmazine (SYD985) significantly improved PFS in HER2+ MBC after 2L+ of MBC therapy. Ocular toxicity was the most common AE, and ILD was observed in a small percentage of patients. 71 ARX788 is a novel ADC targeting HER2 with a different payload and use of microtubule inhibitor against cell lines resistant to T-DM1 with induction of greater apoptotic events.89,90 The phase III ACE-Breast-02 trial recently met its PFS endpoint. 68 Therefore, the global ACE-Breast-03 trial (NCT04829604) is ongoing in patients pretreated with T-DXd and tucatinib. Other ADCs under clinical evaluation include disitamab vedotin (RC48-ADC), MRG002, ZW49, and PF-06804103 (Table 3).
Novel HER2-targeted TKIs
HER2-targeted TKIs in development include DZD1516, poziotinib, and epertinib (S-222611) (Table 3). DZD1516 is a highly selective TKI with full BBB penetrance and very high HER2 selectivity that was evaluated in a phase I trial in HER2+ MBC. 65 Of 19 (82%) evaluable patients, stable disease was the optimal response. Combination therapy and evaluation in earlier treatment lines is planned.
Novel combination strategies
Combination therapies have been evaluated in HER2+ MBC, whereby HER2 is not the only target. Agents studied, or under study, include novel HER2-targeted TKIs, 65 CDK4/6 inhibitors (CDK4/6i), 91 PI3K inhibitors (PI3Ki),92,93 and PARP inhibitors (PARPi). 94 Specifically, there was preclinical and clinical data to support further study of CDK4/6i, 91 PARPi, 94 and PI3Ki 92 in HER2+ MBC. These agents can be combined with HER2-targeted therapy ± ET, often without chemotherapy. Ongoing and reported trials are shown in Table 3.
CDK4/6i
There is strong rationale for combining CDK4/6i with HER2-targeted therapies. In MONARCHER, patients with HR+ HER2+ MBC who had received ⩾2 HER2-directed therapies were randomized 1:1:1 to abemaciclib, trastuzumab, and fulvestrant (A), abemaciclib and trastuzumab (B), and trastuzumab with investigators choice of chemotherapy (C). A 2.6-month PFS advantage in arm A versus arm C (8.3 mos. vs 5.7 mos. P = .051) was noted. 80 However, in 2022 evaluation of abemaciclib in HER2+ MBC was discontinued. PATINA (NCT02947685) is evaluating palbociclib + HER2-directed therapy + ET versus standard therapy as 1L treatment for HR+ HER2+ MBC; results are expected shortly. PATRICIA (NCT02448420) showed that palbociclib + trastuzumab is active and safe in trastuzumab pretreated ER+/HER2+ (PAM50 Luminal A or B subtype). 95 As enrollment was stopped prematurely, a new randomized cohort aims to recruit 102 patients with HER2+/HR+ PAM50 Luminal A or B tumors. 96
PARPi
Preclinical data show that HER2+ BCs are sensitive to PARP inhibition independent of sensitivity to HER2-targeted therapies. 94 The premise behind TBCRC50 (A phase 1B/2 study of the PARPi niraparib + trastuzumab in patients with HER2+ MBC; NCT03368729) is that HER2+ BC resistant to HER2-targeted therapies is sensitive to PARP inhibition mediated by the canonical NFκB signaling pathway, increasing cell death and treatment response. TBCRC50 is enrolling patients with HER2+ MBC (2L-5L setting).
PI3K inhibitors
Mutations in PIK3CA co-occur with HER2-amplification in ~ 20% of cases. 92 Therefore, PI3Ki+ HER2-directed therapy were evaluated for the treatment of PIK3CA-mutated HER2+ MBC. However, early phase studies showed that addition of pan-PI3Ki to HER2-targeted therapies caused treatment-limiting side effects. 93 The α-specific PI3Ki alpelisib + T-DM1 was active and tolerable in persons with trastuzumab-resistant HER2+ MBC. 97 Phase III studies are required to assess the efficacy of adding α-specific PI3Ki to HER2-targeted therapies.
Immune-based therapies
The role of immunotherapy in HER2+ BC is relevant given the presence of higher stromal TILs, which infer immunogenic potential. 98 Therefore, novel HER2-directed mAbs, ADCs, vaccines, and adoptive T-cell therapies are being evaluated. Other immune-based therapies which have/are being studied include checkpoint inhibitors (CPIs), bispecific antibodies, and immune-stimulator Ab conjugates (ISACs). Most drug development efforts are focusing on ADCs and immunotherapies currently. Table 3 shows results of ongoing and completed studies, including AEs and safety data, for CPIs, HER2-targeted vaccines, bispecific antibodies, and other immune-based therapies.
Immune checkpoint inhibitors
Combining HER2-directed therapy and CPI in HER2+ MBC has shown modest results (Table 3). Results from PANACEA, whereby patients with heavily pretreated HER2+ MBC received trastuzumab + pembrolizumab showed limited responses; however, ORR was better in patients with PD-L1+ disease. It was advised that further studies should focus on PD-L1+ patients with less heavily pretreated HER2+ MBC. 87 In KATE2, 202 participants were randomized 2:1 to T-DM1 and atezolizumab versus T-DM1 and placebo. PFS was improved by ~4 months in patients with PD-L1+ status. 88 Rates of grade 3-5 AEs were similar between arms with a recommendation to focus on patients with PD-L1+ HER2+ MBC. 88 In the NRG-BR004 trial (NCT0319988), patients with treatment-naïve HER2+ MBC were randomized to 1L THP + atezolizumab versus THP and placebo. Due to toxicity and accrual concerns, the study prematurely closed in 2022. Follow-up continues to assess PFS, OS and monitor for delayed immune AEs. 99
HER2 targeted vaccines
Therapeutic cancer vaccines aim to treat existing malignancies by improving the antitumor immune response. 100 Several types of HER2 vaccinations are in clinical development; ongoing trials in HER2+ MBC are evaluating adoptive T-cell therapy, plasmid-based DNA vaccines, and others (peptide, protein, cell-based, and dendritic cell vaccines) (Table 3). Interestingly, in a phase I study (NCT00436254) of a plasmid DNA vaccine (n = 66), mOS was not reached at almost 10 years for patients with stage III/IV HER2+ MBC 101 (mOS 4.7 years in CLEOPATRA). 30
Other immune-based therapies
Immune-stimulator antibody conjugates (ISAC) combine tumor-targeting monoclonal antibodies with immunostimulatory agents to enable targeted delivery of immune activators into tumors (Table 3). NJH395 is a novel, first-in-class ISAC (toll-like receptor 7 agonist conjugated to a HER2-targeted antibody via a noncleavable linker payload). 102 In a phase 1 study in non-breast HER2+ metastatic cancers (n = 18), cytokine release syndrome was common, but manageable. However, antidrug antibodies and neuroinflammation posed a clinical challenge. 76 BDC-1001 is a HER2-targeted ISAC which triggers local activation of the innate immune system and generates a durable tumor-targeted adaptive immune response. In a phase 1/2 study of BDC-1001 ± nivolumab in advanced HER2+ solid tumors, the combinations were overall well tolerated, and clinical activity was noted (5 partial responses and 10 stable disease ⩾ 6 months). Further development of BDC-1001 with phase 2 expansion in HER2-expressing solid tumors at the RP2D is planned. 103 CAR-M and CAR-NK therapies, bispecific engagers, engineered toxin bodies, HER2-targeted proteolysis-targeting chimeras (PROTACs), and targeted thorium-227 conjugates are also under study; some of these agents are in clinical development (Table 3).
Special considerations: brain metastasis/leptomeningeal disease
Durable treatment options for HER2+ BCBM and leptomeningeal disease remain limited. Therefore, improving prevention strategies and treatment options is critically important. The incidence of CNS metastases as a first site of recurrence is <10% (2.2% at 4-year median follow-up in the HERA trial).104,105 Unfortunately, in adjuvant trials of pertuzumab and T-DM1, there was no reduction in the incidence of BCBM in the treatment versus control arms.106,107 Multidisciplinary management is key, as treatment options must be individualized in terms of local regional and systemic therapy. Regarding systemic therapy, an exploratory analysis from CLEOPATRA showed a longer median time of first site of CNS metastasis in the pertuzumab arm (15.0 vs 11.9 months, HR, 0.58, 95% CI, 0.39-0.85), which infers that despite reported low BBB permeability, mAbs have a role in 1L treatment of HER2+ MBC with BM. 108 More recently approved HER2-targeted therapies better penetrate the BBB (Table 4); the strongest data for CNS efficacy are for tucatinib and T-DXd. 109 In HER2CLIMB (291 patients with BCBM), the addition of tucatinib resulted in a CNS ORR of 47.3%, mPFS of 9.9 months, and an OS of 18.1 months. 20 In the T-DXd arm of DB03, subgroup analysis CNS response rates were impressive (67.4% CNS-ORR and mPFS of 15 months with T-DXd vs 20.5% and 3 months with T-DM1). 45 Furthermore, the prospective, single-arm, phase II TUXEDO-1 trial evaluated T-DXd in 15 patients with HER2+ active BCBM. In the intention-to-treat population, intracranial RR was 73.3% (11/15), and at 11 months median follow-up, PFS was 14 months. 110 Whether tucatinib and/or T-DXd can prevent BCBM is being evaluated in postneoadjuvant trials (Destiny-Breast05 [NCT04622319], A011801 [NCT04457596]). To ensure further progress, careful clinical trial design is required, including further studies of HER2-targeted TKIs, ADCs, and HER2 mAbs with optimal BBB penetrance. Furthermore, trials including relevant CNS endpoints, patient-reported outcomes, and a focus on BCBM prevention are needed. Novel preclinical models will provide detailed information regards the mechanistics of CNS tropism, the BBB, and the brain-tumor microenvironment. 111 Novel drugs, combinations, and targets (ie, BM-specific genomics, immunotherapy, HER2 CAR T-cells) are being studied. When possible, oncologists should enroll patients on clinical trials, including those with progressive/untreated BCBM.
Efficacy of systemic therapy for HER2-positive BCBM.
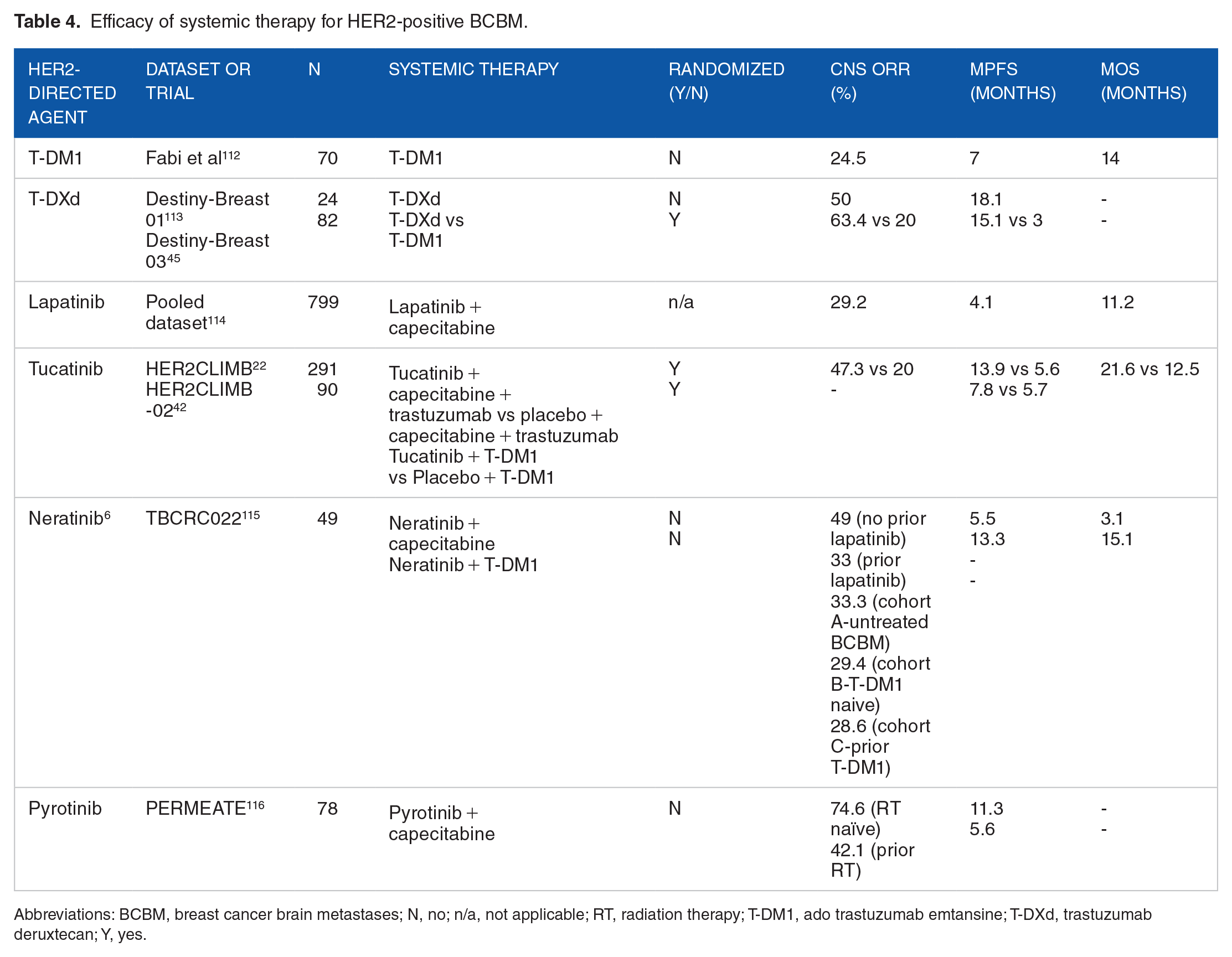
Abbreviations: BCBM, breast cancer brain metastases; N, no; n/a, not applicable; RT, radiation therapy; T-DM1, ado trastuzumab emtansine; T-DXd, trastuzumab deruxtecan; Y, yes.
Conclusions
Major progress has been made in the treatment of HER2+ MBC with the emergence of multiple targeted agents that improve PFS and OS, and possibly cure selected patients. However, challenges remain, namely, overcoming therapeutic resistance, optimal sequencing of therapies (and use of immune-based therapies), therapy (de)escalation, and the effective prevention and treatment of BCBM. Exploiting the potential of novel therapies and identifying predictive biomarkers will be crucial to ensure continued progress. Innovative research strategies addressing the aforementioned challenges will undoubtedly drive further accomplishments in the field.
Footnotes
Acknowledgements
Not Applicable
