Abstract
Background:
Real-world outcomes for patients with human epidermal growth factor receptor-2 (HER2)-positive metastatic breast cancer (MBC) treated with pertuzumab in combination with taxane chemotherapy plus trastuzumab (TaxTP) in the first line setting and trastuzumab emtansine (TE) in any line of treatment are lacking.
Methods:
Cohorts of patients treated with (1) TaxTP and (2) TE from January 1, 2013 through December 31, 2016 were retrospectively obtained from a population-based database. Cohorts were described according to age, hormone receptor (HR) status, prior systemic therapies, event-free survival (EFS) defined as time from start of treatment to start of next line of treatment or death, and overall survival (OS).
Results:
A total of 122 patients were treated with TaxTP and 104 with TE. In the TaxTP cohort, EFS was significantly longer in the trastuzumab-naïve group compared with the adjuvant trastuzumab group (median EFS = 27.0 vs 12.4 months; P = .002). In the TaxTP cohort, median OS was not reached. In the TE cohort, EFS was significantly longer in the pertuzumab-naïve group compared with pertuzumab-exposed group (median time to treatment failure [TTF] = 18.7 vs 5.5 months; P < .001). Overall survival was also significantly longer in the pertuzumab-naïve group compared with the pertuzumab-exposed group (median OS = 23.2 vs 14.1 months; P = .022). In multivariable analyses, adjuvant trastuzumab and prior pertuzumab exposure in the metastatic setting remained significant predictors of inferior EFS for patients treated with TaxTP and TE, respectively.
Conclusions:
New anti-HER2 therapies appear to be clinically relevant in the real-world.
Background
Prior to the introduction of human epidermal growth factor receptor-2 (HER2)–targeted therapies, HER2 overexpression was associated with poor prognosis in metastatic breast cancer (MBC).1,2 Slamon et al 3 demonstrated that trastuzumab plus first-line chemotherapy significantly improved progression-free survival (PFS) and overall survival (OS). Ensuing clinical trials and a subsequent body of literature4-16 have established further benefit and definitive roles for adding pertuzumab into trastuzumab plus taxane chemotherapy in the first-line setting (CLEOPATRA) 10 and use trastuzumab emtansine (TE) in the second-line setting and beyond (EMELIA13,14 and TH3RESA15,16).
Patients in clinical trials are a highly selected group and may not be representative of the general MBC population. Important to the current era, few patients enrolled on CLEOPATRA had prior adjuvant chemotherapy plus or minus trastuzumab, 10 and no patients on EMELIA or TH3RESA had prior pertuzumab.13-16 However, guideline recommendations do not exclude such patients from pertuzumab or TE eligibility.17,18 The aim of this study was to examine outcomes associated with new treatments for HER2-overexpressing MBC within a contemporary population-based cohort.
Methods
Patient selection
Treatment cohorts were retrospectively obtained from the Alberta Health Services (AHS) CancerControl Breast Data Mart (BDM). The BDM is a repository of information on all breast cancer patients diagnosed from January 1, 2004 onwards in the province of Alberta, Canada, and contains information on patient demographics, tumour characteristics, surgical intervention, CancerControl Alberta clinic visits, systemic therapies administered, and vital status. Information is prospectively collected from the Alberta Cancer Registry, the Cancer Centre Electronic Medical Record (ARIA MO), the Discharge Abstract Database (DAD), and the National Ambulatory Care Reporting System (NACRS).
Eligible patients were female, adult patients with pathologically confirmed HER2-overexpressing breast cancer, metastatic disease. Human epidermal growth factor receptor-2 overexpression was defined as per standard immunohistochemistry staining (IHC) 3+ or positive by in situ hybridization (ISH). Cohorts included all women who started (1) taxane chemotherapy/trastuzumab/pertuzumab (TaxTP) in the first-line setting (may have had one line of hormonal therapy for MBC but no chemotherapy or anti-HER2 therapies for MBC) and (2) TE as any line of treatment, from January 1, 2013 through December 31, 2016.
The data extracted are as follows: patient age at treatment onset, hormone receptor (HR) status, de novo versus relapsed metastatic disease, all palliative-intent systemic therapy regimens and start dates, vital status, date and cause of death if deceased, and date of last contact with AHS or CancerControl if not deceased. For patients with relapsed MBC, receipt of adjuvant therapies was also recorded.
Outcome measures
The primary breast cancer outcome of interest was event-free survival (EFS) defined as time from start of treatment to start of next line of treatment or death from any cause. Secondarily, we measured OS defined as time from start of treatment to death from any cause.
Statistical analysis
The TaxTP cohort was described and analysed by receipt of adjuvant trastuzumab, and the TE cohort was described and analysed by prior pertuzumab exposure in the metastatic setting. Patient, tumour, and treatment characteristics were compared using the Mann–Whitney U test or Pearson chi-square test. Follow-up time was from the start of treatment to data extraction on March 1, 2018. Distributions of time to treatment failure (TTF) and OS curves were calculated using the Kaplan–Meier method. Where possible, univariate analyses with log-rank tests were used to compare median survival outcomes. Multivariable analyses using Cox proportional hazards models were then applied to determine associations between survival outcomes and prior treatment exposure after adjusting for other clinically relevant variables (age as a continuous variable and HR status for both cohorts; taxane choice in the TaxTP cohort; receipt of adjuvant trastuzumab and lines of chemotherapy for MBC in the TE cohort). Neither MBC presentation (de novo vs relapsed) nor receipt of adjuvant chemotherapy was included in the Cox regression analyses, as these variables were highly correlated with adjuvant trastuzumab exposure. All tests were 2-sided, in which a P-value < .05 was considered statistically significant. Data were analysed using SPSS 22.0 (IBM Corp., Armonk, NY, U.S.A.).
The Health Research Ethics Board of Alberta (Cancer Committee) approved this study.
Results
TaxTP cohort
There were 122 patients who started on TaxTP in the first-line setting for MBC of whom 44 had received adjuvant trastuzumab and 78 were trastuzumab naïve. All patients in the adjuvant trastuzumab group had also received adjuvant chemotherapy (86% taxane containing) and most patients in this group with HR-positive disease had also received adjuvant hormonal therapy. While the trastuzumab-naïve group only contained 18% with relapsed disease, most of these patients had not received any adjuvant treatments. The groups were otherwise reasonably well balanced with respect to age, HR status, prior exposure to no more than one line of hormonal therapy in the metastatic setting and treatment by type of taxane (Table 1). Median follow-up was 33.4 (range = 14.3-51.1) months.
Characteristics of TaxTP cohort by adjuvant trastuzumab exposure.

Abbreviations: HR, hormone receptor; MBC, metastatic breast cancer; TaxTP, taxane, trastuzumab, pertuzumab.
For the TaxTP cohort, median EFS was 22.5 (95% confidence interval [CI]: 16.7-28.3) months. Figure 1 shows the Kaplan–Meier curves for EFS by receipt of adjuvant trastuzumab. Event-free survival was significantly longer in the trastuzumab-naïve group compared with the adjuvant trastuzumab group (median TTF = 27.0 vs 12.4 months; P = .002). In multivariable analysis, being trastuzumab naïve was an independent predictor of EFS (adjusted hazard ratio = 0.50; 95% CI: 0.31-0.80; P = .004). No other variables were associated with EFS (Table 2). Of note, for the 58 relapsed patients, median time from initial diagnosis to metastatic disease was 35.4 (range = 6.4-229.6) months. At the time of analysis, median OS for the entire TaxTP cohort and the trastuzumab-naïve group had not yet been reached. Median overall OS for the adjuvant trastuzumab group was 32.9 (95% CI: 17.3-48.5) months. Similar to multivariable analysis of EFS, being trastuzumab naïve was the only independent predictor of OS (adjusted hazard ratio = 0.46, 95% CI: 0.25-0.83, P = .01; Appendix 1).

EFS in the TaxTP cohort by adjuvant trastuzumab exposure.
Multivariable model EFS for TaxTP cohort.
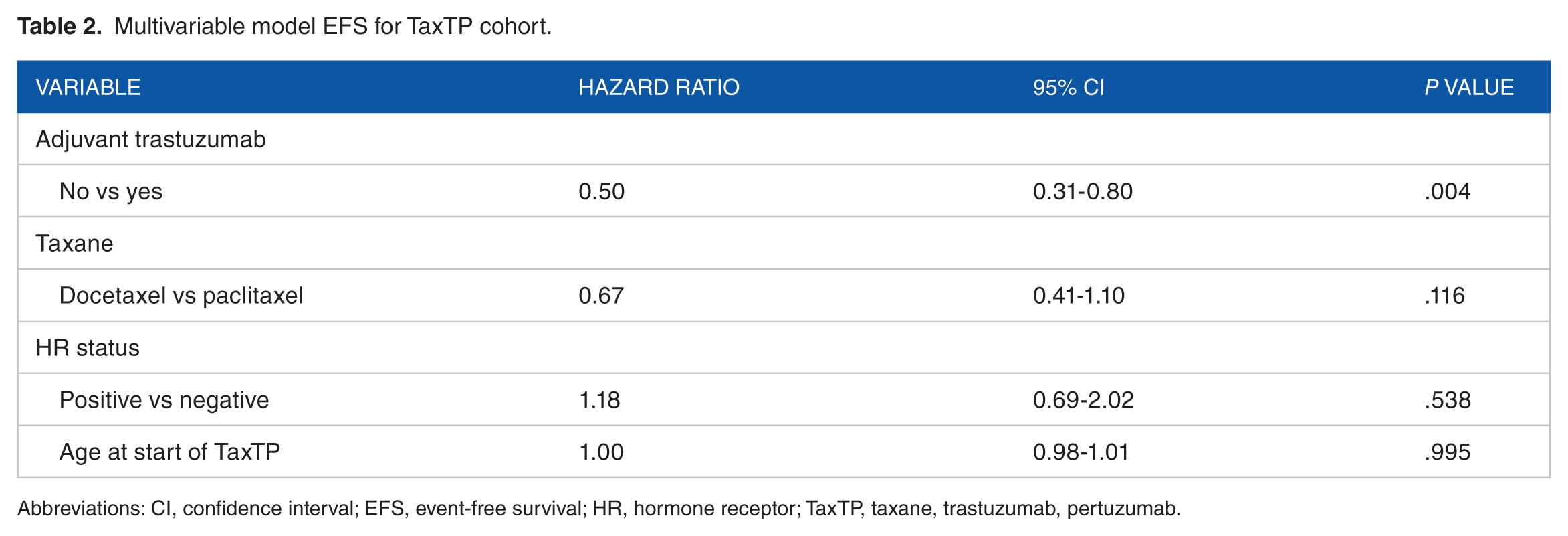
Abbreviations: CI, confidence interval; EFS, event-free survival; HR, hormone receptor; TaxTP, taxane, trastuzumab, pertuzumab.
Of the 122 patients who had first-line TaxTP, 53 (43%) started second-line therapy during the follow-up period. Fourty-four patients started TE, 6 patients had lapatinib plus capecitabine, and 3 patients had other treatments without an anti-HER2 agent.
TE cohort
In total, 104 patients received TE in any line and of these, 55 had prior pertuzumab for MBC and 49 were pertuzumab naïve. The pertuzumab-treated and pertuzumab-naïve groups were similar in age, HR status, receipt of adjuvant therapies, and metastatic presentation (ie, de novo or relapsed). In addition, exposure to hormonal therapy in the metastatic setting was similar; however, the pertuzumab-naïve group was more heavily pretreated with chemotherapy (Table 3). Median follow-up was 29.7 (range = 14.0-57.5) months.
Characteristics of trastuzumab emtansine cohort by pertuzumab exposure.

Abbreviations: HR, hormone receptor; MBC, metastatic breast cancer; TE, trastuzumab emtansine.
For the TE cohort, median EFS was 8.3 (95% CI: 6.1-10.5) months. Figure 2 shows the Kaplan–Meier curves for EFS by prior exposure to pertuzumab in the metastatic setting. Event-free survival was significantly longer in the pertuzumab-naïve group compared with the pertuzumab-exposed group (median TTF = 18.9 vs 5.5 months; P < .001). In multivariable analysis, being pertuzumab naïve was an independent predictor of EFS (adjusted hazard ratio = 0.41; 95% CI: 0.26-0.66; P < .001). No other variables were significantly associated with EFS (Table 4). For the entire TE cohort, median OS was 19.0 (95% CI: 12.5-25.5) months. Overall survival was also significantly longer in the pertuzumab-naïve group compared with the pertuzumab-exposed group (median OS = 23.2 vs 14.1 months; P = .02). In multivariable analysis, being pertuzumab naïve relative to pertuzumab exposed was associated with better OS, but this did not meet statistical significance (adjusted hazard ratio = 0.60; 95% CI: 0.35-1.03; P = .065), while being trastuzumab naïve in the adjuvant setting was significantly associated with OS (adjusted hazard ratio = 0.56; 95% CI: 0.33-0.97; P = .04; Appendix 2).

EFS in the TE cohort by prior pertuzumab exposure.
Multivariable model EFS for trastuzumab emtansine cohort.

Abbreviations: CI, confidence interval; EFS, event-free survival; HR, hormone receptor; MBC, metastatic breast cancer; TE, trastuzumab emtansine.
Discussion
This is the first Canadian, provincial population-based study evaluating outcomes of patients with HER2-overexpressing MBC treated with new anti-HER2 therapies, pertuzumab in combination with taxane chemotherapy plus trastuzumab in the first-line setting, and TE in any line of therapy.
For the TaxTP cohort, EFS (time to new treatment or death) was similar to PFS as reported for the docetaxel/trastuzumab/pertuzumab arm in CLEOPATRA (median = 22.5 vs 18.5 months). 10 However, in our study, just over one-third of patients had adjuvant trastuzumab and experienced significant and clinically relevant inferior EFS compared with the trastuzumab-naïve patients (median = 12.4 vs 27.0 months). Age group, HR status, and choice of taxane chemotherapy were not associated with EFS. The latter finding is consistent with favourable outcomes reported for weekly paclitaxel plus trastuzumab and pertuzumab in a single arm, phase II study. 19 Ongoing follow-up is required to establish median OS in our TaxTP cohort and the trastuzumab-naïve group.
The real-world literature demonstrating inferior outcomes among patients exposed to adjuvant trastuzumab who relapse and are then treated with trastuzumab plus chemotherapy in the metastatic setting, in comparison to trastuzumab-naïve patients with MBC, is well established.20-23 It has been less clear as to whether the addition of pertuzumab to taxane chemotherapy plus trastuzumab mitigates the differences in prognosis for patients who had adjuvant trastuzumab and those who did not. Results of this study are not supportive of this hypothesis. In a US community oncology-based cohort of 266 HER2-positive MBC patients, treated almost exclusively with docetaxel/trastuzumab/pertuzumab, median PFS was 16.9 months with 61.5% of patients having prior adjuvant or neoadjuvant trastuzumab. 24 The trastuzumab-pretreated and trastuzumab-naïve patients were not reported separately. Further real-world data are expected from the SystHER registry study 25 and the Australian HER2-positive breast cancer population. 26
In the TE cohort, median EFS was 8.3 months, which sits between the PFS estimates for the TE arms in TH3RESA (6.2 months) 15 and EMELIA (9.6 months). 13 Overall survival in our real-world cohort was inferior when compared with the TE arms from pivotal clinical trials (19.0 months vs 22.7 months in TH3RESA 16 and 30.9 months in EMELIA). 14 Prior pertuzumab exposure was significantly associated with worse EFS (5.5 vs 18.9 months) and OS (14.1 vs 23.2 months). In multivariable analysis, age, HR status, adjuvant trastuzumab, or lines of chemotherapy for MBC were not associated with EFS. As pertuzumab and TE became available in our province at a similar time, caution is advised when interpreting results for the TE cohort. Patients who had missed the window for first-line TaxTP but were still alive on other therapies and became eligible for TE may have already been selected out for more-responsive and less-aggressive HER2-positive MBC. However, other real-world studies have suggested inferior outcomes for patients treated with TE after pertuzumab exposure compared with pertuzumab-naïve patients. In a study by Fabi et al, 27 34 patients with prior trastuzumab/pertuzumab had significantly worse PFS compared with 73 patients with prior trastuzumab only (5 vs 11 months). In a multicenter, Italian cohort of 250 patients, PFS and OS were numerically less for patients with prior trastuzumab/pertuzumab in comparison to patients with prior trastuzumab only. 28 These results were significant when the analysis was restricted to patients treated with TE in the second-line setting. 28
Despite the many strengths of our data, which are population-based and prospectively collected, there are limitations to this study. First, there was potential for selection bias. The BDM includes patients diagnosed from January 1, 2004 onwards and hence, patients who were diagnosed with early-stage disease prior to this date, relapsed and received any of the treatments analysed, were not included. Hence the cohorts collected would have excluded relapses beyond 9 to 14 years from initial diagnosis. However, HER2-overexpressing early breast cancer is rarely associated with late relapse,29,30 especially in the absence of adjuvant trastuzumab which was not publicly funded in Alberta prior to August 2006. Second, we could not control for performance status, comorbidities, sites of metastatic disease, and metastatic burden. In the TaxTP cohort, inferior survival outcomes attributed to adjuvant trastuzumab exposure could be partly related to greater disease burden at metastatic presentation. It is conceivable that patients with relapsed MBC are more likely to present with symptoms and have a greater burden of metastatic disease compared with de novo patients who may have been diagnosed based on routine staging tests. Inferior median OS in our TE cohort relative to EMELIA 14 and TH3RESA 16 could be in part explained by differences in performance status, comorbidities, and sites/burden of metastatic disease. Third, for the TaxTP cohort, we could not measure and control for completion of adjuvant trastuzumab. Likewise, in the metastatic setting, we did not have data on the number of cycles of taxane chemotherapy given with trastuzumab and pertuzumab, the number of cycles of trastuzumab and pertuzumab given following deletion of docetaxel or paclitaxel, or prescription of hormonal therapy during the maintenance trastuzumab and pertuzumab phase of treatment. As the HR status was not significantly associated with the outcomes measured, it is hypothesized that few or none of the patients with HR-positive disease received hormonal therapy during maintenance trastuzumab and pertuzumab in keeping with the CLEOPATRA trial protocol. 10 Further analyses of HR-positive patients on maintenance trastuzumab and pertuzumab plus or minus hormonal therapy are warranted. Fourth, EFS (defined in our study as time to next treatment or death) was our surrogate for the more standard measure of PFS. It is reassuring that our EFS outcomes were comparable to PFS outcomes reported in pivotal clinical trials. The potential correlation of EFS and PFS is possibly explained by favourable tolerability of the anti-HER2 therapies considered. A final limitation is that we were unable to record toxicity of TaxTP and TE in our real-world cohorts.
Conclusion
New anti-HER2 therapies appear to be clinically relevant for a contemporary cohort of patients with HER2-positive MBC. Our data can inform resource planning for this under-researched population. Further studies are required to confirm or refute inferior survival outcomes for patients on TaxTP with prior adjuvant trastuzumab exposure and for patients on TE with prior pertuzumab exposure.
Footnotes
Appendix
Multivariable model OS for trastuzumab emtansine cohort.

| Variable | Hazard ratio | 95% CI | P value |
|---|---|---|---|
| Pertuzumab exposure | |||
| No vs yes | 0.60 | 0.35-1.03 | .065 |
| Adjuvant trastuzumab | |||
| No vs yes | 0.56 | 0.33-0.97 | .037 |
| Lines of chemotherapy for MBC | |||
| 0-1 vs 1-2 | 1.06 | 0.61-1.83 | .847 |
| HR status | |||
| Positive vs negative | 0.95 | 0.55-1.62 | .836 |
| Age at start of TE | 1.01 | 0.99-1.04 | .179 |
Abbreviations: CI, confidence interval; HR, hormone receptor; MBC, metastatic breast cancer; OS, overall survival; TE, trastuzumab emtansine.
Funding:
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests:
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Author Contributions
SL conceived and designed analysis, and collected data. SL, WC and DS contributed to data collection and analysis tools, performed analysis, and wrote manuscript.
