Abstract
This work explores the antiplasmodial potential of ethanol extract of
Introduction
Based on the latest World Health Organization (WHO) estimates of December 2016, there were 212 million cases of malaria in 2015 and 429 000 deaths, with sub-Saharan Africa accounting for 90% of all malaria deaths in the world.
1
This staggering number of deaths resulting from malaria is associated with the continuous spread of
For thousands of years, in various parts of the world, traditional herbal medicines have been used to treat malaria.5–9 Two of the most important antimalarial drugs in use, artemisinin and quinine, were either derived directly from plants or synthesized using chemical structures of plant-derived compounds as templates.5,9,10 Most of the people in malaria endemic countries still depend on traditional herbal remedies, which could be linked to limited availability as well as unaffordability of existing pharmaceutical drugs. 11
In view of the significant challenges imposed by drug resistance, traditional plant extracts continue to be promising sources of new antimalarial compounds.
9
The use of traditional medicines for the treatment of malaria and other diseases continues to be a growing practice among many African families, despite the availability of orthodox method of treatment.
19
In addition, the short supply of artemisinin compounds leading to alternative production methods,
9
as well as the desire for low-cost drug delivery strategies,
7
has led to significant research efforts to decipher alternative plant products that could serve as effective antimalarials, especially in sub-Saharan Africa. In this report, the antiplasmodial effect of ethanolic extract of
Materials and Methods
Plant collection, authentication, and extract preparation
The stem bark of
Ethical consideration
Experimental procedures and protocols used in this study were in conformity with National Institute of Health’s recommendations in guide for the care and use of laboratory animals and in accordance with the principles of Helsinki Declaration.
Parasite/infection
Chloroquine-resistant
Antimalarial medication
Standard Coartem (Artemether and Lumefantrine), used for treating malaria (Mekophar Chemical Pharmaceutical Joint-Stock Co, Ho Chi Minh City, Vietnam) and obtained from a pharmacy, was used as reference for the antimalarial screening in this study.
Acute toxicity test (assessment of minimum lethal dose)
The assessment of minimum lethal dose was done in 2 phases, following published methods.
20
In phase 1, groups of 3 mice in each group were given oral doses of 10, 100, and 1000 mg/kg body weight of
In vivo antiplasmodial determination
Swiss albino mice (weight: 18-25 g), obtained from the Animal House, Department of Pharmacology, University of Ibadan, kept according to Institutional Animal Care and Use Committee (IACUC) standards, were allowed to acclimatize to the new environment for a week before study initiation. Each mouse was inoculated with 0.2 mL of infected blood containing about 1 × 107 dose of
Suppressive test (4-day test)
The 4-day suppressive test was conducted according to the method of Peters (1975). In this, 25 mice distributed into 5 groups of 5 mice each were infected with 1 × 107 of parasitized red blood cells on day 0. Animals in group 1 received normal saline, those in groups 2 to 4 received 200, 400, and 800 mg/kg of ethanol extract of

Prophylactic test
The prophylactic activity of the extract was performed as described previously.
24
In this, Swiss albino mice were randomized into 5 groups of 5 mice each: group 1 was treated with normal saline only, groups 2 to 4 were treated with 200, 400, and 800 mg/kg/d of the ethanol extract of
Curative test
This was also performed with standard protocol, 24 as previously described. Twenty-five mice distributed into 5 groups of 5 were infected intraperitoneally on the first day (day 0) with 0.2 mL of parasite inoculum. After 72 hours of infection, all animals were treated with different regimens: group 1 received normal saline only; groups 2 to 4 received 200, 400, and 800 mg/kg body weight of plant extract, respectively; whereas group 5 received 10 mg/kg body weight of the standard antimalarial drug (Coartem). Treatment was continued for 5 consecutive days, blood smears were made and microscopically examined to monitor the degree of parasitemia as well as estimating the number of parasitized cells.
Histologic procedures
These were performed with hematoxylin and eosin procedures. 25 Briefly, organs such as liver, spleen, kidney, and testes from each animal were fixed in 10% formal saline, grossed and cut longitudinally into pieces of 4 mm in thickness, and processed for histologic study using standard procedure. Microtome sections were dried on a hot plate and later stained with hematoxylin and eosin stains for examination with a light microscope.
Data analysis
Percentage parasitemia and mean chemosuppression were determined using 1-way analysis of variance; Student-Newman-Keuls test was used for analysis and comparing of results at 95% confidence interval;
Results
About 11.10 g of brown crude extract was obtained from the extraction procedure, a percentage yield of 22%. The acute toxicity test revealed that the extract appeared to be nontoxic at the highest dose (5000 mg/kg) tested. No toxic symptoms or mortality was observed in any of the 3 animals in 3 groups both in phase 1 and phase 2 acute toxicity test study. All animals were still alive at the end of the acute toxicity test, with LD50 value greater than 5000 mg/kg body weight, showing the extract to be nontoxic. Histology sections of the liver, spleen, kidney, and testes reveal normal tissue morphology at all doses, except at 5000 mg/kg (black ring), where we observed a mild perivascular infiltration of liver inflammatory cells (Figure 1); otherwise, hepatocytes appeared normal.
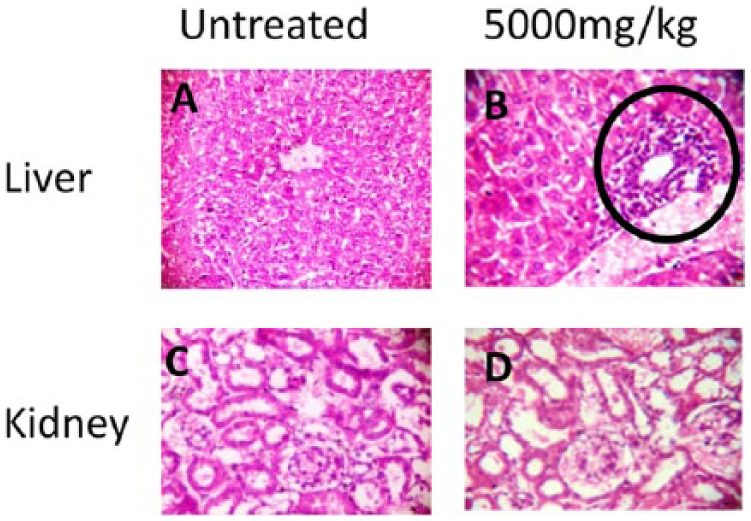
Photomicrographs of liver and kidney at the end of acute toxicity assessment. (A) Normal liver morphology. (B) (Black ring) mild perivascular infiltration of the liver inflammatory cells after treatment with 5000 mg/kg of the extract. (C) Normal kidney morphology. (D) Kidney remains normal after treatment with 5000 mg/kg of the extract. Haematoxylin and Eosin, 400x original magnification.
Suppressive effect of B sapida ethanolic extract on P berghei
In the suppressive test, mean parasitemia on day 4 after infection ranged from 14.20 ± 1.17 to 18.96 ± 0.60 for 200 and 800 mg/kg, respectively (Figure 2). The value for control was 19.02 ± 1.20, whereas for Coartem was 8.38 ± 0.39. The percentage chemosuppression of the extract ranged from 6.8% to 0.3% for 200 and 800 mg/kg, respectively, whereas that for standard antimalarial Coartem was 55.9%. Percentage chemosuppression showed increases with decrease in dose levels of the extract, indicating that maximal chemosuppression could be reached at the lowest dose, although intangible. Percentage chemosuppression in Coartem was very much higher than in control and all the doses of the extract, revealing that extract had little or no suppressive effect at all doses. Percentage parasitemia in control, compared with 400 and 800 mg/kg, was not statistically significant (
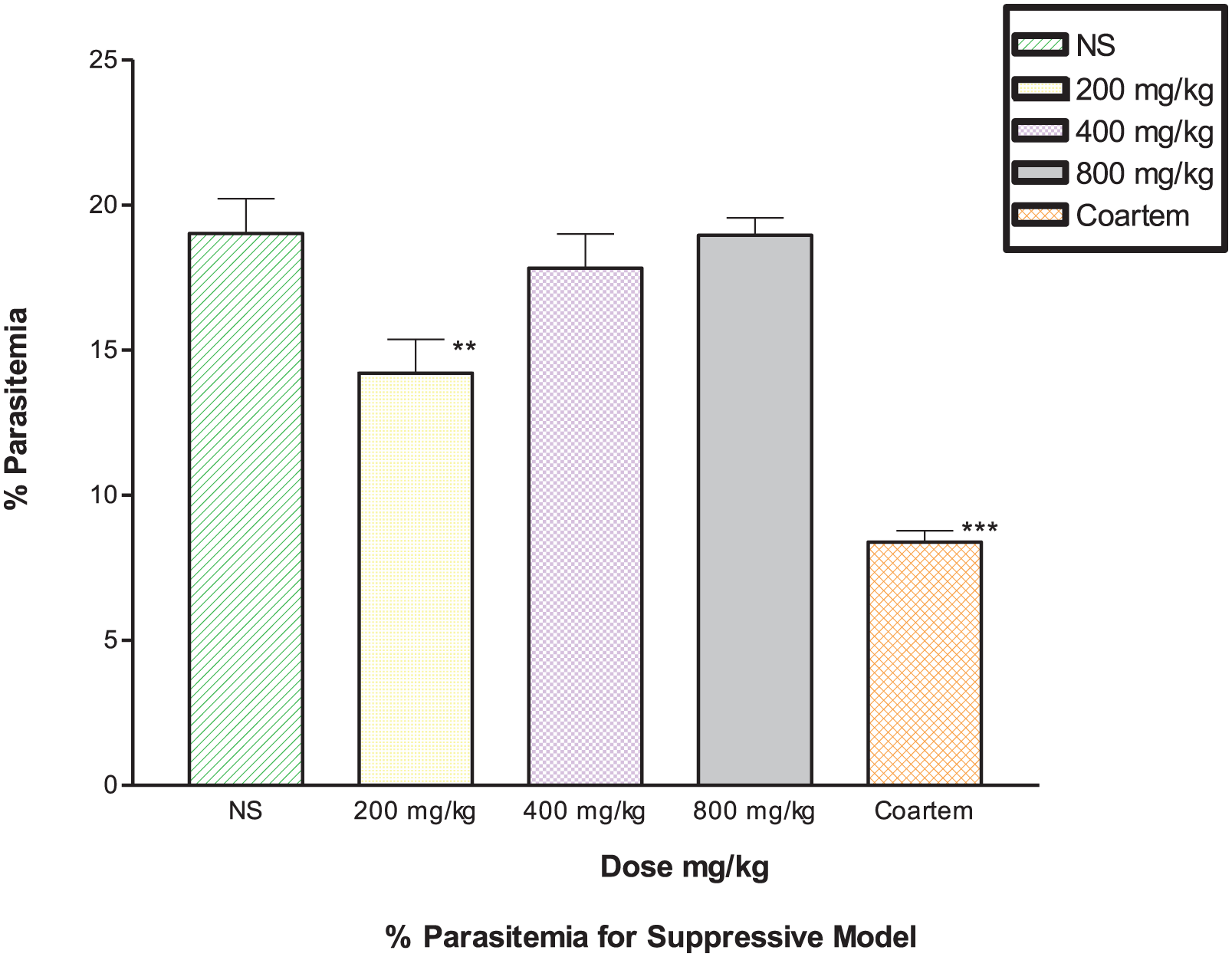
Suppressive effect of methanol extract of bark of
Prophylactic effect of B sapida ethanolic extract on P berghei
The percentage parasitemia in mice infected and treated with 200, 400, and 800 mg/kg was 8.22 ± 0.32, 9.7 ± 0.31, and 12.12 ± 0.54, respectively (Figure 3). Untreated saline group had percentage parasitemia of 19.10 ± 0.33, whereas that of Coartem-treated positive control was 7.24 ± 0.91. Percentage chemosuppression of parasites in mice treated with 200, 400, and 800 mg/kg was 57%, 50.8%, and 36.5%, respectively, whereas that of Coartem was 62.1%. Unlike our observation in the suppressive model, percentage parasitemia decreased with a decrease in extract dose levels, showing that a tangible optimal chemosuppression can be obtained at the lowest dose in this model and is statistically significant at all doses when compared with nontreated group (
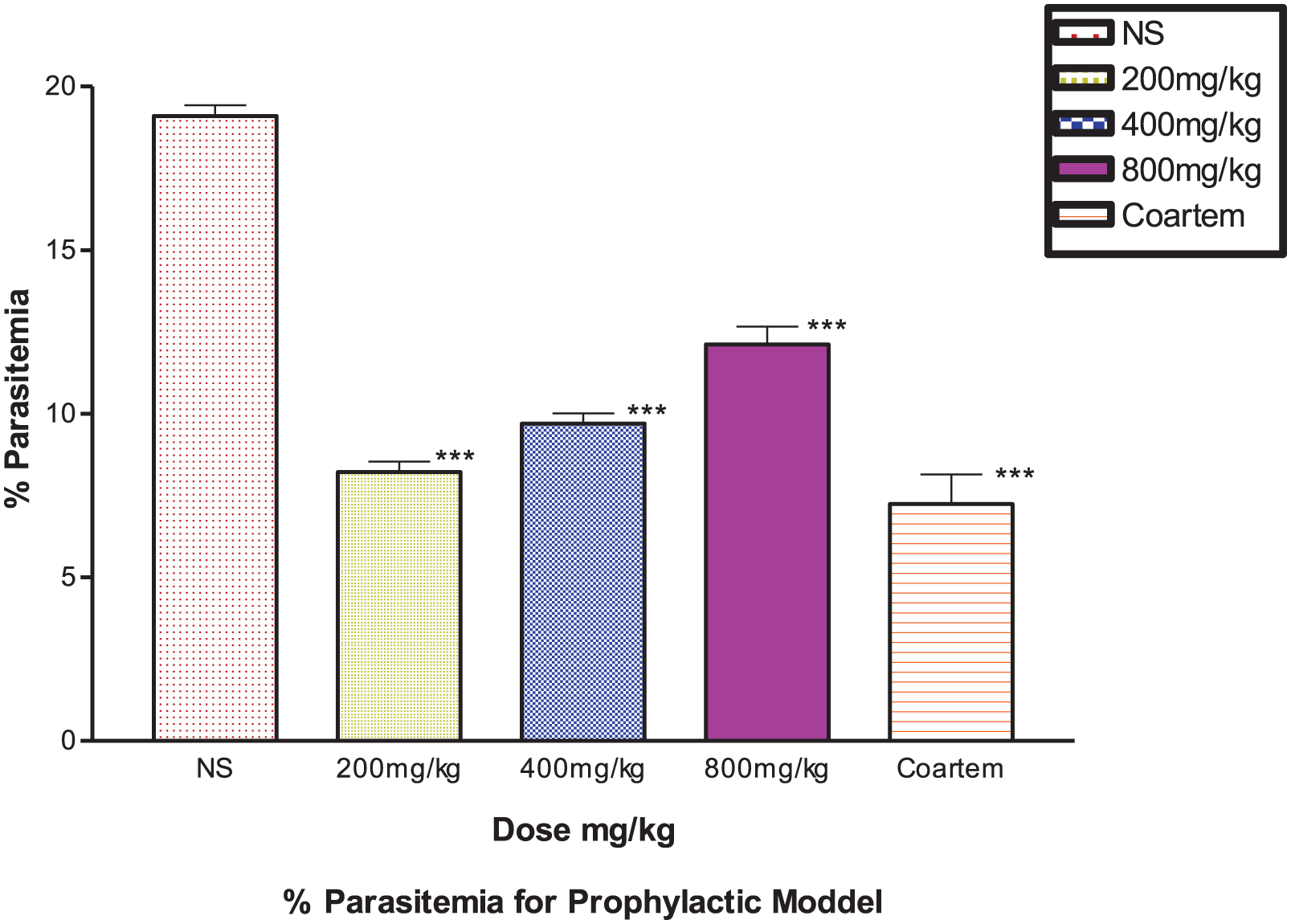
Prophylactic effect of ethanol extract of
Curative effect of B sapida ethanolic extract on P berghei
Figure 4 shows curative effect of the extract at different graded doses. Unlike the suppressive and prophylactic models, none of the extract doses tested showed significant activity. The lowest dose (200 mg/kg) that had shown significant activity in other models showed no activity in our model. For day 3, the values for Coartem versus control versus 200 mg/kg of the extract was 1.74 ± 0.29 versus 7.58 ± 0.44 versus 7.18 ± 0.82, with similar findings on day 7 (21.48 ± 0.68, 17.00 ± 0.58, and 2.06 ± 0.39 for control), 200 mg/kg of extract and Coartem, respectively, revealing the extract had no significant activity against the parasite when compared with control.
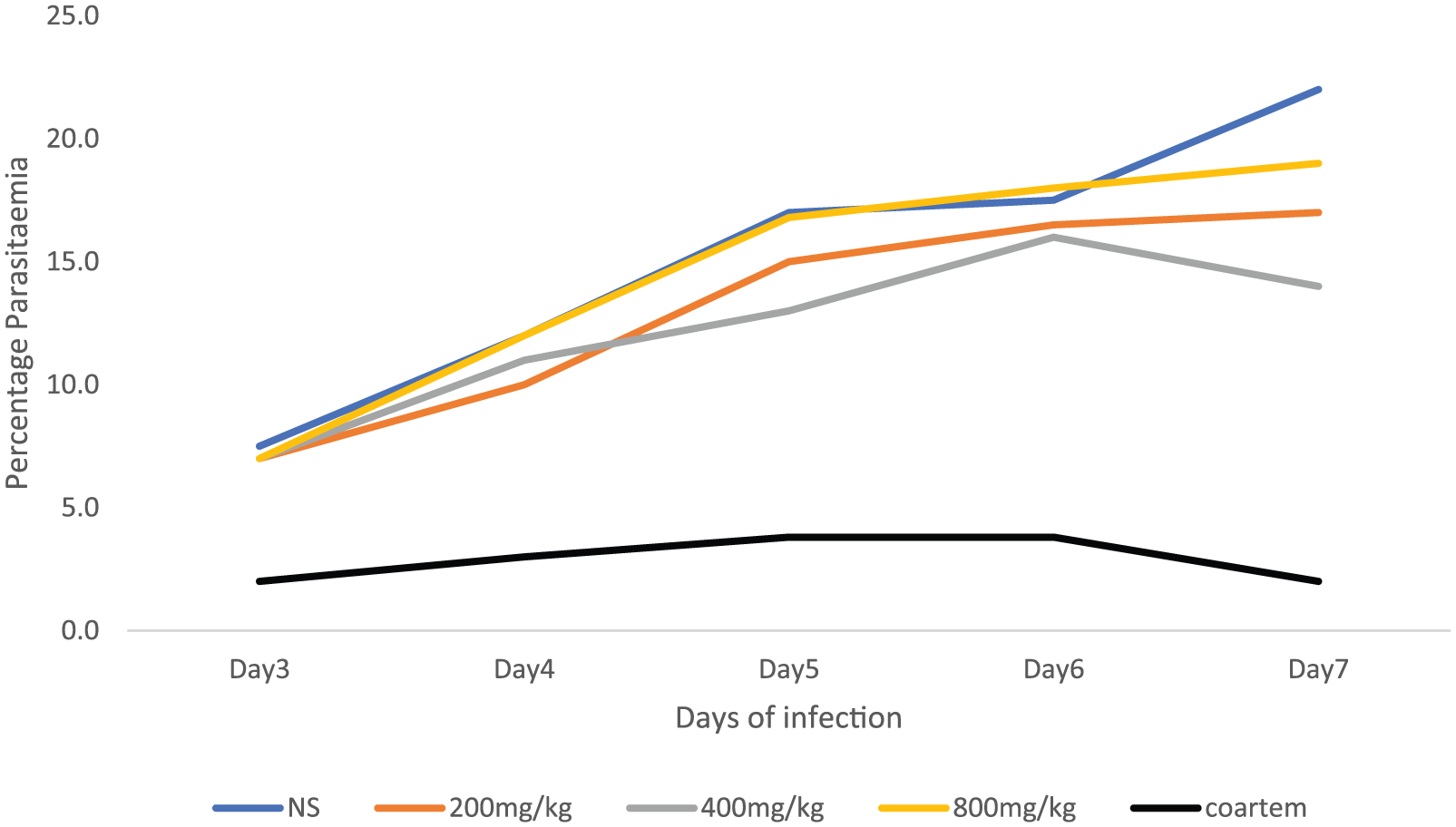
Curative effect of ethanol extract of
Effect of malaria on weight of malaria-induced mice
The weights of all test animals decreased 24 hours after infection, with this decrease continuing among the suppressive, curative, and untreated groups 72 hours after treatment. However, by day 5 (21.40 ± 1.20), this progressive weight loss was turned around for the prophylactic group when compared with the weights obtained at the same dose on day 0 (20.78 ± 0.92) and day 7 (21.70 ± 1.08) (Table 1).
Weights of mice for prophylactic model.
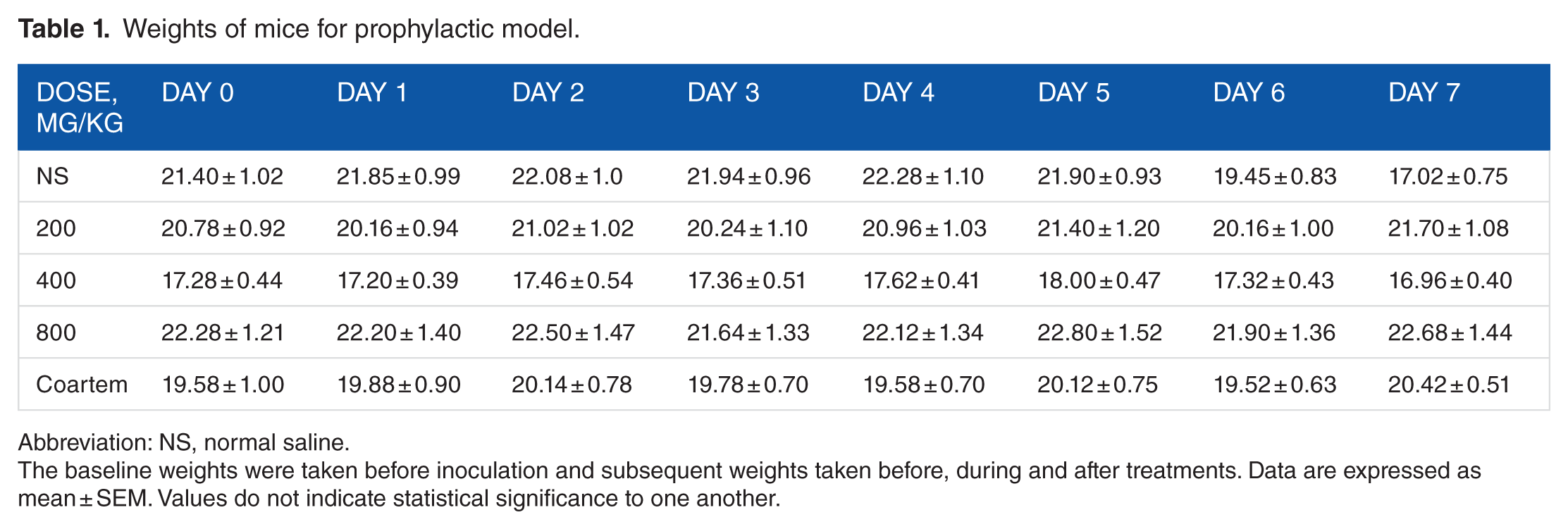
Abbreviation: NS, normal saline.
The baseline weights were taken before inoculation and subsequent weights taken before, during and after treatments. Data are expressed as mean ± SEM. Values do not indicate statistical significance to one another.
Effect of B sapida ethanolic extract on organs of P berghei–infected mice
The kidney, liver, and spleen of the mice revealed normal tissue architecture after treatment, demonstrating that this extract appears safe on the major organs of the mice (slides not shown).
Survival time of mice in the 3 models
The survival time for mice in the prophylactic group was longer than those in suppressive and curative groups (Figure 5). Mice in prophylactic group started dying on day 9, whereas most were still alive at day 20 and beyond. However, animals in curative group started dying on day 5 and were all dead by day 9, whereas those in the suppressive group started dying on day 5 with 98% dead by day 11. These results clearly demonstrate the prophylactic effect of this extract, at different doses, on malaria-infected animal.
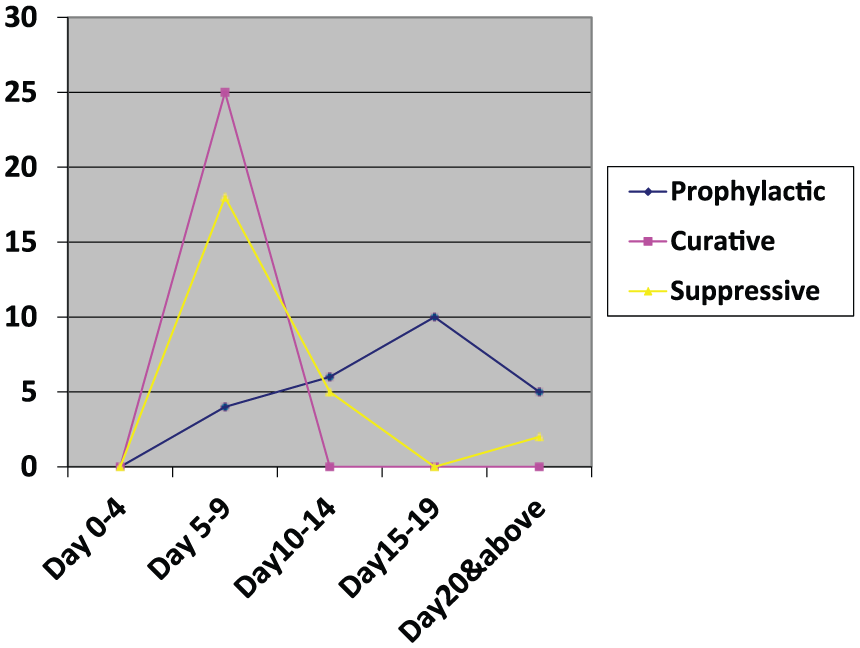
Graph showing survival time of mice in the 3 models.
Discussion
Despite the many and varying efforts at malaria control, this disease is still a global threat of enormous proportion and a significant contributor to health and economic inequities in endemic countries. Inhabitants of these countries, under the scourge of disease, face the conundrum of finding appropriate, effective, and cheap means to protect themselves against infection, whereas the parasite, however, has shown an increasing resistance to available antimalarial drugs. This resistance is also favored by population movement and human migration leading to the introduction of resistant parasite to areas previously free of drug resistance. The possibility of synthesizing new antimalarials from plants has become a major policy goal in the control arena, becoming more urgent, in light of the limited number of antimalarials in development, the potential of parasite resistance to the only available therapeutic drugs (artemisinin and its derivatives), and the poor solubility of artemisinin. These are some of the factors driving the design of alternative methods of artemisinin delivery that could potentially slow the process of resistance evolution.10,26,27 We demonstrate that
This study shows that
The absence of death after administering 5000 mg/kg body weight of
Loss of appetite, ultimately leading to weight loss, is one of the characteristics of malaria infection.
31
We observed weight loss in
The survival time for mice in the prophylactic group extended beyond day 20, whereas those in the curative model did not survive beyond day 9. About 98% of the mice in the suppressive model were dead by day 15, with only 2% surviving till day 20 but not beyond, further confirming the prophylactic activity of
Although there have been some reports of
Conclusively, our results suggest that the ethanol bark extract of
Footnotes
Acknowledgements
The authors are grateful to Dr GO Gbotosho, Malaria Research Laboratory, Institute for Medical Research and Training (IMRAT), University College Hospital, Ibadan, Nigeria, for providing the malaria parasite used for the study, also to Mrs Sakirat Braimah for technical assistance.
Peer review:
Two peer reviewers contributed to the peer review report. Reviewers’ reports totaled 428 words, excluding any confidential comments to the academic editor.
Funding:
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests:
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Author Contributions
OO conceived and designed the experiments. OOO, JAO, and DIA performed the laboratory work. JAO and DIA analyzed data. OOO wrote first draft of manuscript. JAO, DIA, and BNT contributed to writing of manuscript. BNT, OOA, and OO made critical revisions and approved final version. All authors reviewed and approved the final manuscript.
Disclosures and Ethics
The authors also confirmed that this article is unique and not under consideration or published in any other journal. Experimental procedures and protocols are in conformity with National Institute of Health’s recommendations in guide for the care and use of laboratory animals and the principles of the Declaration of Helsinki.
