Abstract
Ankylosing spondylitis (AS) is an immune-mediated inflammatory arthritis and enthesitis involving the spine and peripheral joints. In recent years, specific antagonist of tumor necrosis factor (anti-TNFα, etanercept) 50 mg weekly therapy has rapidly gained popularity for the treatment of AS. However, the dose of etanercept has not been determined in Asian, particularly Chinese populations. The purpose of the study was to evaluate the efficacy and safety of dose reduction of etanercept (50 mg/week in 4 weeks followed by 25 mg/week in 8 weeks) in the treatment of AS with synovitis of the hip, as against the conventional dose (50 mg/week in 12 weeks) in a Chinese population. Forty-three Chinese AS patients with synovitis of the hip were involved in this study. Seventeen of them were randomized to receive conventional dose of etanercept treatment and 26 were given a dose reduction regimen for 12 weeks. The primary efficacy endpoint was disease activity of response for AS at week 12, including Bath AS Disease Activity Index (BASDAI), the serum erythrocyte sediment rate (ESR), C-reactive protein (CRP), and assessment of synovitis of the hip by ultrasonography. At 12 weeks, all of the patients had responses to some extent and the efficacy variables improved significantly over time, but not between treatment groups. Nine patients experienced at least one adverse event (generally, infections and injection site reactions), most of them mild or moderate. In sum, the dose reduction of etanercept regimen in the 12-week AS treatment was confirmed as a safe and effective therapy as the conventional dose was given.
Introduction
Ankylosing spondylitis (AS) is known as a chronic immune-mediated disease characterized by inflammatory axial and peripheral arthritis and enthesitis, 1 which may contribute to significant disability and greatly reduced quality of life. It affects 0.15–1.4% of adults in the world 2 as well as 0.2–0.54% of the Han ethnic Chinese population (92% of the whole population). 3 The etiology of AS is still largely unknown, however a number of linkage studies have implicated an interaction of environmental insults and genetic susceptibility like HLA-B27. 4 About 20% of patients with AS are involved in the hip and shoulder arthritis always portend a worse outcome. 5 Thus, targeting the specific inflammatory processes of the disease may essentially influence disease progression. In recent years, of the treatment options available, non-steroidal anti-inflammatory drugs (NSAIDs) have been widely used for relieving joint pain and inflammation. However, many of the adverse effects of NSAIDs make their use controversial in some patient populations and around 20% of patients have no responses for NSAIDs. 6 Some other classical alternatives such as disease-modifying anti-rheumatic drugs (DMARDs) also do not achieve a satisfied control of disease activity. 1 More recently, antagonists of tumor necrosis factor (TNF)α have been proved by European League Against Rheumatism as a safe and effective therapy for AS patients who are refractory to conventional treatments. 7 Anti-TNFα therapy has a rapid effect on disease symptoms, normalizing acute-phase reactant levels and reducing inflammation in sacroiliac and spinal joints, and their effects are prolonged. 8
Etanercept is the 75 kD TNF receptor fusion protein linked to the Fc portion of human IgG1 (TNFr:Fc), which treats autoimmune diseases by acting as a TNF inhibitor. The safety and efficacy of etanercept in the treatment of AS have been proved in a large number of studies and its use in AS treatment is approved in a regimen of 50 mg/week, both in Europe and the USA.9–11 Yisaipu®, a recombinant TNFr:Fc with the same structure with etanercept, is a biosimilar subsequent version of etanercept manufactured and marketed in China, which is also commercialized as Etanar in Colombia, as Etart in Mexico, or as Etacept in India. 12 Clinical trials have evidenced Yisaipu® to be effective in a Chinese population. 13 However, recommendations on which patients with AS should be given anti-TNF treatment are especially needed concerning possible side effects and high costs of these drugs. 1 In 2005, a Japanese post-marketing survey of etanercept for rheumatoid arthritis (RA) reported that 25 mg/week dose was nearly as effective as 50 mg/week although the effectiveness of the 50 mg/week dose was statistically different from that of 25 mg/week. 14 Due to the ethnic and geographical similarity, Chinese people may show similar results of dose reduction therapy of etanercept for AS. Thus, the purpose of the present study is to evaluate the effect of etanercept reduction therapy to treat AS patients with synovitis of the hip as well as its safety and tolerability profile.
Materials and methods
Participants
Forty-three eligible patients were recruited in Henan Luoyang Orthopedic-Traumatological Hospital (HLOH) and Henan Institute of Orthopedic and Traumatology between March 2009 and July 2010. Candidates were inpatients (age range, 16–32 years) with AS with unilateral or bilateral synovitis of the hip diagnosis as defined by the modified New York criteria 1984 for AS, 15 and with active disease for >4 weeks. In patients with predominantly axial forms, inflammatory activity was defined by a Bath Ankylosing Spondylitis Disease Activity Index (BASDAI) >4, 16 and/or increase in acute phase parameters: erythrocyte sedimentation rate (ESR) and/or C-reactive protein (CRP) above normal laboratory levels. Hip joint synovitis was assessed by ultrasonography and the evaluation of synovial hyperplasia and effusions of the hip is based on measurement of the thickness of synovial membrane and the distance between femoral neck and the joint capsule. Joint effusion or synovitis distends the capsule outwards and the anechogenic distance between bone and capsule lengthens. 17
The following criteria were used to assess the eligibility of study participants: patients with complete ankylosis of the spine; patients with unilateral or bilateral synovial hyperplasia, and/or effusions of the hip assessed by ultrasonography; patients without previous history of tuberculosis, hepatitis, and other infection diseases; patients in agreement with etanercept treatment. In patients with bilateral synovitis of the hip, we mainly focused on the more severely affected side. Patients with contraindications for biologicals and previous biological use were excluded. Severe cardiovascular or nervous diseases, abnormalities in hematological profiles, psoriasis, psychiatric disease, or history of alcohol or drug abuse, were reasons for exclusion. Pregnant or breastfeeding women were also ineligible. The study was approved by the ethics committee of HLOH and Henan Institute of Orthopedic and Traumatology. The written informed consent for participation was obtained from all participants.
Randomization and treatment
Upon completion of the baseline evaluation, of 43 eligible participants, 26 patients were randomly allocated to a dose reduction treatment group and the other 17 patients in the control group received the conventional treatment. Patients remained in their groups until the end of the study. The conventional anti-TNF therapy was given according to assessment in ankylosing spondylitis (ASAS) recommendations. 18 Patients in the conventional treatment group received etanercept given as subcutaneous injections at a dose of 25 mg bi-weekly for 12 weeks. DMARDs: Sulfasalazine enteric-coated tablets for 3 weeks (0.75 g/d, three times a day for the first week; 1.5 g/d, three times a day for the second week; 2.0 g/d, twice a day for the third week) and Leflunomide enteric-coated tablets for 12 weeks (20 mg/d, once a day at night) were used to improve peripheral arthritis associated with AS, with regular monitoring of liver enzymes and blood counts indicated. The dose reduction treatment group was given etanercept as subcutaneous injection at dose of 25 mg bi-weekly for the first 4 weeks and then 25 mg weekly for the following 8 weeks. The other treatments are the same with the control group.
Efficacy endpoints
The efficacy endpoint was the proportion of participants who achieved ASAS response at week 12. 18 Patients’ inflammation was measured using the score of the morning-stiffness items of the BASDAI. ESR, CRP, and the condition of synovial hyperplasia and effusions of the hip assessed by ultrasonography were also be evaluated at the end of the treatments.
Statistical analysis
Data regarding the different association tests of which the result is discrete variables were summarized by median and range and compared using the χ2 test with Fisher’s exact test. The changes of synovial hyperplasia and effusions of the hip were performed using the t test. Two-sided significance tests (P < 0.05) were used. Statistical analysis was carried out in SPSS software, version 19.0.
Results
Baseline characteristics
Data were collected between March 2009 and July 2010. Of the 43 enrolled inpatients, 17 (14 men, 3 women) were randomly selected to receive conventional etanercept treatment and 26 (20 men, 6 women) were given dose reduction etanercept treatment. The mean age was 22 years (age range, 16–32 years), and the mean duration of disease was 7 months (range, 2–13 months). Demographic and other disease-related variables were not significantly different at baseline in both groups of patients.
Efficacy
On the basis of the prespecified definition of a treatment response and the intention-to-treat principle, all of the patients had responses to some extent (Table 1). In the AS conventional treatment group, mean ESR was 42.12 ± 11.53 mm/h at 0 week and decreased promptly to 11.87 ± 3.64 mm/h at 12 weeks; CRP also dropped from 25.08 ± 9.87 mg/L to 5.60 ± 2.29 mg/L. Similarly, mean BASDAI was 5.12 ± 0.68 at week 0 and decreased rapidly 1.40 ± 0.35 by week 12. The condition of synovial hyperplasia and joint effusion also improved from 3.03 ± 0.74 mm to 1.01 ± 1.01 mm and 5.63 ± 2.50 mm to 1.03 ± 0.96 mm, respectively. For the dose reduction treatment patients, both of the average ESR and CRP decreased essentially from 39.80 ± 10.38 mm/h to 9.38 ± 2.11 mm/h and 26.66 ± 9.57 mg/L to 5.01 ± 1.62 mg/L, respectively. Mean BASDAI scores reduced from 4.82 ± 0.69 to 1.42 ± 0.23. Finally, the improvements of synovial hyperplasia reduced significantly from 3.18 ± 0.75 mm to 0.83 ± 1.07 mm as well as joint effusion from 5.83 ± 2.09 mm to 0.90 ± 1.10 mm. The thickness of synovial membrane and synovial fluid was obviously decreased after dose reduction treatment of etanercept after 12 weeks. Measurements were carried out on the monitor using a magnified picture, which allowed an accuracy of 0.4 mm. All parameters were significantly reduced at week 12 (P <0.001) in both treatment groups. Changes in these disease activities from baseline to week 12 are shown in Figure 1. However, there were no significant differences found in an efficacy endpoint between these two treatment groups.
Comparison of disease activity at baseline and etanercept therapy at 12 weeks.
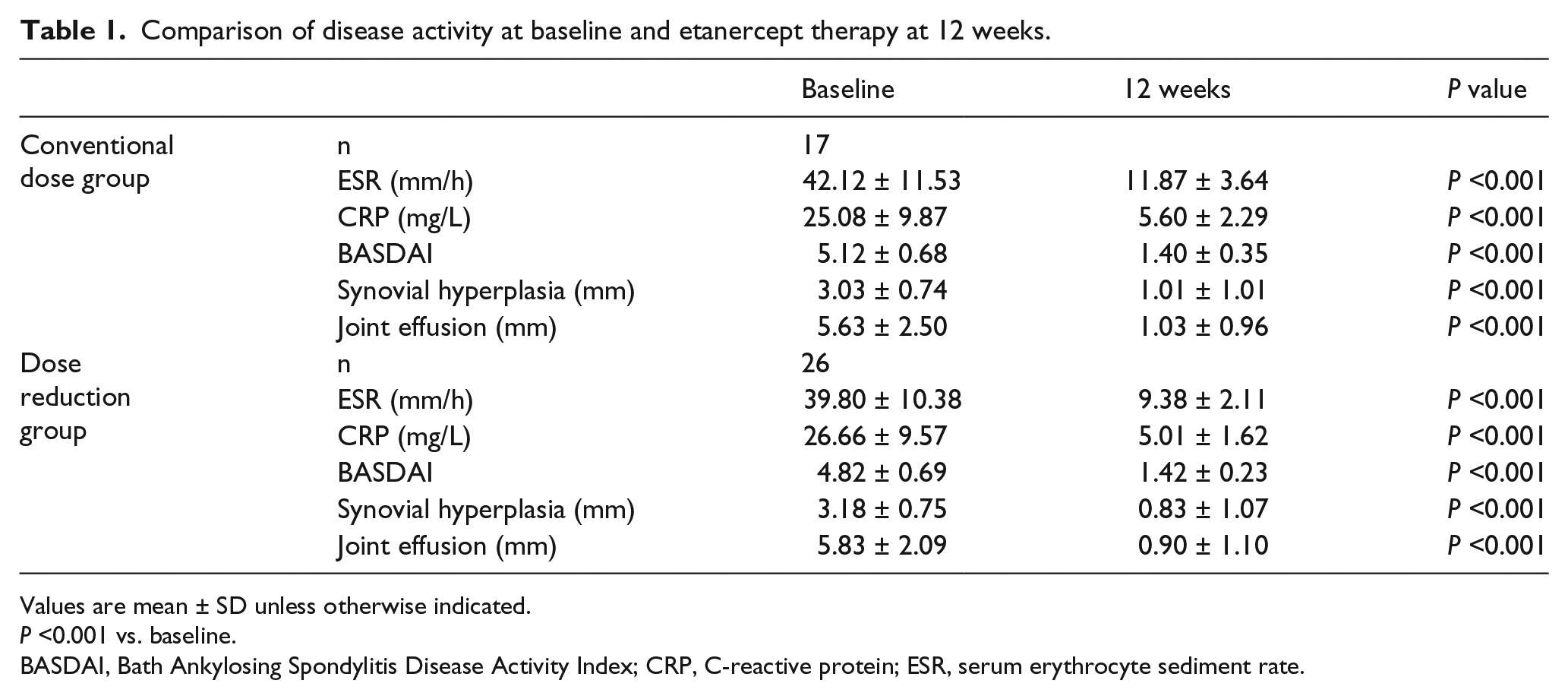
Values are mean ± SD unless otherwise indicated.
P <0.001 vs. baseline.
BASDAI, Bath Ankylosing Spondylitis Disease Activity Index; CRP, C-reactive protein; ESR, serum erythrocyte sediment rate.

Effect of treatments of different doses of etanercept on disease activity at 12 weeks. The asterisks denote P >0.05 for the comparison between the conventional treatment and dose reduction treatment groups. BASDAI, Bath Ankylosing Spondylitis Disease Activity Index; CRP, C-reactive protein; ESR, serum erythrocyte sediment rate.
Adverse events
There were no serious adverse events or withdrawals due to adverse events. Six patients (23.1%) in the dose reduction group occurred adverse event including three with reactions at the injection site: two with elevated aminotransferase (AST) levels; one with minor infections. And three patients (17.6%) in the conventional treatment group occurred elevated aminotransferase (n = 2) and minor infections (n = 1). The two study groups did not differ significantly with regard to either the overall rate of adverse events or the rates of specific events. Since the adverse events were all of mild to moderate intensity, there was no requirement of treatment discontinuation.
Discussion and conclusion
In the past few years, the introduction of anti-TNF has been the most substantial development in the treatment of AS and other spondyloarthritides. 15 Large randomized, placebo-controlled trials of etanercept given subcutaneously in a dose of 50 mg once per week or 25 mg twice per week, in patients with AS have shown impressive short-term improvements in spinal pain, function, and inflammatory markers.9,19 These trials show dramatic improvement of pain, function, and disease activity including BASDAI, Bath Ankylosing Spondylitis Functional Index (BASFI), Bath AS Metrology Index (BASMI), ERS, and CRP in patients with active disease compared with placebo.
The demonstrated efficacy of dose reduction of etanercept for the treatment of AS with synovitis of the hip in the Chinese population is a promising development in the treatment of this disease. The results of this study show that the use of etanercept, despite treatment with DMARDs, in patients with AS both in 25 mg bi-weekly doses for 12 weeks and in 25 mg bi-weekly for the first 4 weeks and then 25 mg weekly for the following 8 weeks, improves disease activity. Moreover, all the improvements of the values of disease activity have no significant differences between the two treatment groups. Cost-effectiveness is always an important element when expensive treatments are discussed. A previous Chinese cost-effectiveness RA study reported that the average costs and health outcomes using Yisaipu® was $77,815.30 per quality-adjusted life year (QALY). 20 Additionally, treatment emergent adverse events commonly occur during etanercept treatment. The most common adverse events were mild or moderate infections and injection-site reactions. 21 The dose reduction treatment may potentially decrease the risk of adverse event occurrence using etanercept. Therefore, dose reduction of etanercept regimen would substantially benefit patients both in terms of cost-effectiveness and safety.
Several limitations exist in the current analysis. Although we noticed that the primary amino acid sequence of Yisaipu® was identical to the original product, the efficacy and safety of the biosimilar compared with originally licensed biopharmaceutical still needs to be investigated further. 22 Due to the high expenses of magnetic resonance imaging (MRI), the clinical effectiveness of etanercept lacks confirmation by MRI, which has been proved especially useful for identification of early sacroiliitis and spondylitis including patients with undifferentiated spondyloarthritis. Given that the potential adverse events of etanercept may cause additional harm to patients, local prescribing information should be consulted for detailed information, including for contraindications, precautions, and use in special patient populations.
In conclusion, the administration of dose reduction of etanercept (50 mg/week in 4 weeks followed by 25 mg/week in 8 weeks) in the treatment of AS with synovitis of the hip did also significantly improve symptoms as against the conventional dose (50 mg/week in 12 weeks). In addition, such a regimen in a 12-week AS treatment was as safe as the conventional dose.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
