Abstract
Objective: We evaluated the efficacy, safety, and drug survival rate of tocilizumab in the elderly patients with rheumatoid arthritis (RA). Methods: This study was conducted in 108 RA patients who started tocilizumab between 2008 and 2018. The patients were divided into a young group (<65 years) and an elderly group (≥65 years). The efficacy, safety, and drug survival rate of tocilizumab were compared between the two groups. Results: At baseline, there were no significant differences between the elderly (
Introduction
The population of Japan is aging most rapidly among the developed countries. The country has entered a super-aged society, with senior citizens aged 65 years and above accounting for more than 28% of its population in 2020. The average age of Japanese patients with rheumatoid arthritis (RA) is also increasing, with the reasons including an increase of the elderly population in Japan, an increase of patients developing new-onset RA at an older age, and improved outcomes of RA due to advances in treatment. 1 Along with growth of the elderly RA population in recent years, interest in management of RA among elderly patients is also increasing. Elderly RA patients have more comorbidities than younger RA patients, including renal impairment, lung disease, infections, and malignant tumors. When a biologic drug is used concomitantly with methotrexate (MTX) to treat elderly RA patients, it can be difficult to administer MTX at an adequate dose and efficacy may be decreased. 2 Tocilizumab (TCZ) is a biological disease-modifying anti-rheumatic drug (bDMARD) that targets the interleukin-6 (IL-6) receptor. Because TCZ has a different target from that of tumor necrosis factor (TNF) inhibitors, it is expected to be effective for RA patients who respond poorly to TNF inhibitors. It has been reported that TCZ monotherapy is better than methotrexate monotherapy, and that TCZ monotherapy was more effective than TNF inhibitor monotherapy.3,4 These reports suggest that TCZ could be an effective option for elderly RA patients in whom it is difficult to use MTX concomitantly. On the other hand, post-marketing surveillance in Japan has shown that old age is a risk factor for serious infections among patients using TCZ. 5 Because the steroid dose is generally higher in elderly RA patients than in younger RA patients, the risk of infection is increased among the elderly patients. 6 Characteristically, C-reactive protein (CRP) becomes negative early after the start of TCZ administration due to inhibition of IL-6. This means that a typical inflammatory response, including fever and elevation of CRP, may not occur when infection develops in patients using TCZ, and that making difficult to detect and treat infectious diseases early. Therefore, the drug survival rate of TCZ therapy can be lower in elderly patients than younger patients because of discontinuation due to infections and other adverse events. In this study, we evaluated the efficacy, safety, and drug survival rate of TCZ therapy among elderly RA patients treated in the real-world clinical setting by comparing elderly RA patients with younger RA patients.
Materials and methods
Patients
This study was conducted as a single-center retrospective cohort study at Saitama Medical University Hospital in Japan. We included 108 RA patients who started TCZ therapy between July 2008 and January 2018 at Saitama Medical University Hospital. We excluded the patients with unknown outcome for the first year of TCZ therapy. Consecutive eligible patients older than 18 years of age who met the 1987 criteria of the American College of Rheumatology (ACR) or the 2010 criteria established by ACR/European League Against Rheumatism (EULAR) were enrolled in this study, and they were followed for 36 months.7,8 These 108 patients were classified into two groups based on their age at the start of TCZ administration, which were a young group (<65 years old) and an elderly group (≥65 years old). The following items were retrospectively compared between the two groups: changes of disease activity and the severity of physical dysfunction, MTX-reducing and steroid-reducing effects of TCZ therapy, drug survival rate of TCZ, and incidence of adverse events up to 36 months after the start of TCZ administration.
The following data were obtained from the information provided by medical records: age at initiation of TCZ therapy, age at the onset of RA, gender, duration of RA, body weight, TCZ administration method (intravenous or subcutaneous), history of prior biologic drug use (bio-naive or bio-switch), concomitant use of oral MTX and/or oral steroids and the dosages, and evaluation of joint involvement by the attending doctor. We used the Disease Activity Score 28 (DAS28) and Clinical Disease Activity Index (CDAI) as composite indices of RA disease activity.9,10 Those indices were recommended to use as continuous measures of disease activity in patients with RA by the EULAR and the ACR. 11 We assessed the Health Assessment Questionnaire-Disability Index (HAQ-DI) which was a commonly used questionnaire for functional status of patients with RA. Scores range from 0 to 3, with lower scores reflecting better physical function and, thus, less disability. 12 (Supplementary Material). The following laboratory data were obtained: serum C-reactive protein (CRP), erythrocyte sedimentation rate (ESR), rheumatoid factor (RF), anti-cyclic citrullinated peptides (CCP) antibody, matrix metalloproteinase-3 (MMP-3), serum creatinine, and estimated glomerular filtration rate (eGFR). The following information was also obtained: CDAI at 0, 3, 6, and 12 months after starting TCZ therapy, HAQ-DI and use/non-use of oral MTX and oral steroids and the dosages at baseline and at 12 months after starting TCZ therapy, discontinuation of TCZ and the reason for discontinuation (adverse events or lack of efficacy) at 36 months after starting TCZ therapy, and the duration of TCZ administration.
Statistical analysis
We expected to have 40% of subjects who were 65 years or older. In order to detect the reduction of 0.5 SD from baseline to 12 months in each outcome variable within each age group (<65 or ≥65), at 90% power with two-sided 5% significance level, our study required to have 110 patients, where SD stands for the standard deviation of the within patient difference.
Demographic characteristics are presented as median and interquartile range (IQR) for continuous variables, and as frequencies and percentages for categorical variables. To assess differences of baseline characteristics between the elderly and younger RA patients, Wilcoxon’s rank-sum test was used for continuous variables and chi-squared test was employed for categorical variables. Changes of CDAI and HAQ-DI between baseline and 3, 6, and 12 months were analyzed with the mixed effect regression models to consider data clusters among repeatedly measured data within single patient. Changes of PSL and MTX doses in each group between baseline and 12 months were also evaluated by mixed effect regression models. Frequency of patients taking oral steroid or methotrexate were compared between baseline and 12 months with binary logistic regression models with Huber-White sandwich estimators for variance and covariance structure for the data clusters. Also, distribution of daily dose of steroid or methotrexate was compared between baseline and 12 months with ordinal logistic regression models with Huber-White sandwich estimator.
Survival curves for the elderly and younger RA groups were drawn by the Kaplan–Meier method and compared by using log rank test. Statistical significance was claimed at
Results
Baseline characteristics
Demographic and baseline characteristics of the patients.

TCZ = tocilizumab; VAS = visual analog scale; ESR = erythrocyte sedimentation rate; CRP = C-reactive protein; DAS28-ESR = Disease Activity Score in 28 joints using the erythrocyte sedimentation rate; CDAI = Clinical Disease Activity Index; HAQ-DI = Health Assessment Questionnaire Disability Index; RF = rheumatoid factor; ACPA = anticyclic citrullinated peptide antibody; MMP = matrix metalloproteinase; eGFR = estimated glomerular filtration rate; MTX = methotrexate; PSL = prednisolone. Unless indicated otherwise, data are presented as the median [95% CI]. Mann–Whitney U tests were used for comparisons between groups. † Fisher’s two-tailed exact tests were used.
Efficacy of TCZ
In both the elderly and the young group, the CDAI (Figure 1(a)) and HAQ-DI (Figure 1(b)) showed decrease from baseline until 12 months after starting TCZ therapy. Low disease activity (CDAI ≤10) was achieved in 82.4% of the elderly group and 85.7% of the young group after 12 months of TCZ therapy (Figure 2). The extent of the change in CDAI during 12 months of TCZ therapy showed no difference between the elderly group and the young group (ΔCDAI: 15.1 vs 14.8). The frequency of achieving remission according to the HAQ-DI (HAQ-DI < 0.5) after 12 months of TCZ therapy was tend to be lower in the elderly group than the young group (35.1% vs. 55.8%; Evolution of mean CDAI score and HAQ-DI for 12 months after initiation of tocilizumab in the young (<65 years) and the elderly group (≧65 years) of patients with rheumatoid arthritis. *vs baseline, Frequency of patients achieving remission or low disease activity according to CDAI, and HAQ-DI remission in the young and the elderly group of patients with rheumatoid arthritis after 12 months of tocilizumab therapy. No difference was seen between the 2 groups in each index. CDAI = Clinical Disease Activity Index; HAQ-DI = Health Assessment Questionnaire-Disability Index. 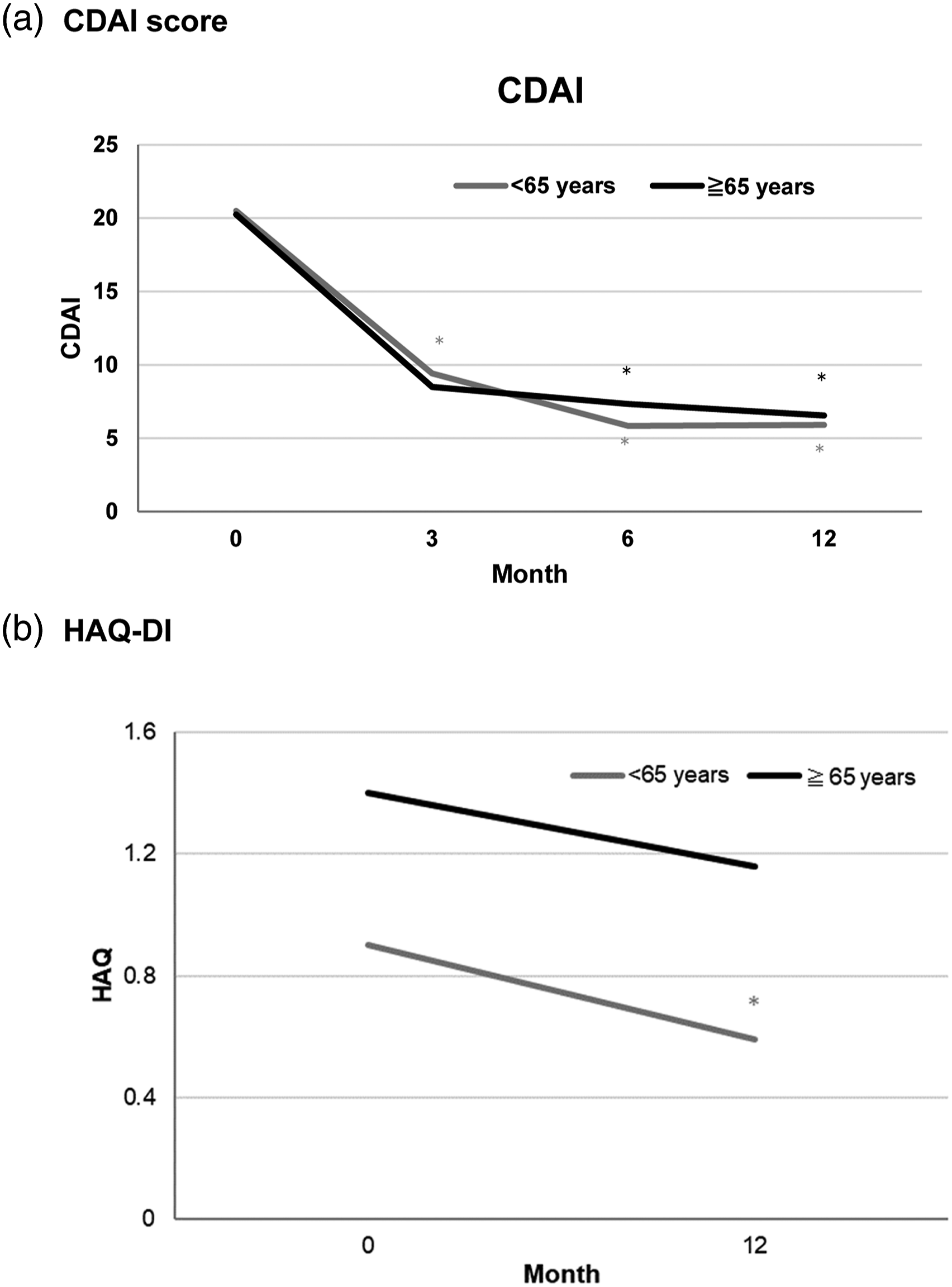

Concomitant medications
We investigated whether the frequency of using concomitant drugs (steroids and MTX) and the doses of those drugs showed a decrease at 12 months after the start of TCZ therapy. The frequency of steroid use decreased significantly from 68.9% to 39.5% in the elderly group after 12 months ( Frequency of patients taking oral steroid and the daily dose of steroid (prednisolone equivalent) among steroid users in the young and elderly group of patients with rheumatoid arthritis at baseline and after 12 months of tocilizumab therapy. (a) Frequency of patients taking oral steroid. (b) Daily dose of steroid (prednisolone equivalent). Frequency of patients taking methotrexate and the weekly dose of methotrexate among methotrexate users in the young and elderly group of patients with rheumatoid arthritis at baseline and after 12 months of tocilizumab therapy. (a) Frequency of patients taking methotrexate. (b). Weekly dose of methotrexate.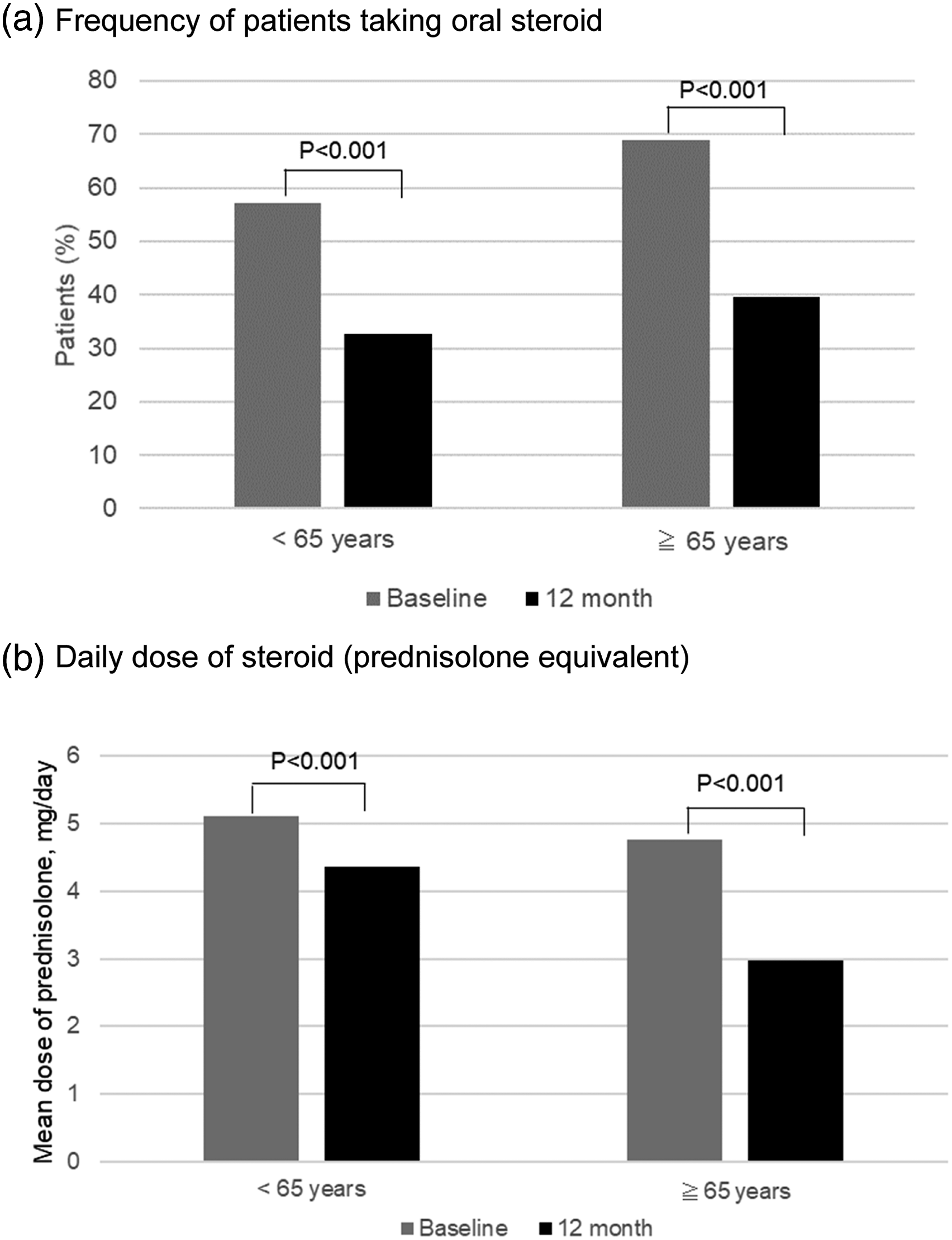

Safety and drug survival rate of TCZ
As shown in Figure 5, there was no significant difference in drug survival rate of TCZ for three years between the elderly group and the young group (62.2% vs 68.3%). TCZ therapy was discontinued due to adverse events in 24.4% of the elderly group versus 17.5% of the young group, with the difference not being significant. Patients who discontinued the drug due to lack of efficacy tended to be fewer in the elderly group than in the young group (6.7% vs 14.3%). Within 3 years of TCZ therapy, one case was death due to pneumonia in the elderly group, and 2 cases of death due to adverse events, myocardial infarction and sudden death, in the young group. In the elderly group, TCZ was discontinued due to toxic adverse events in 11 cases, which included 3 cases of infection (fetal pneumonia, cellulitis, and buccal abscess), 2 cases of fatigue, one case each of drug eruption, corneal perforation, leg edema, systemic edema, anorexia, and intestinal perforation. Eleven patients in the young group also discontinued TCZ therapy due to toxic adverse events, which included 5 cases of infection (two cases of bacterial pneumonia, pneumocystis pneumonia, and two cases of infectious arthritis), 2 cases of laboratory abnormality (liver dysfunction and elevation of β-D-glucan), 1 case each of breast cancer, fetal acute myocardial infarction, sudden death, and intestinal perforation. Drug survival of tocilizumab in the young and the elderly group of patients with rheumatoid arthritis based on the Kaplan–Meier curve of time to discontinuation of tocilizumab therapy whatever the causes of drug discontinuation. No difference was seen between the two groups.
Discussion
We evaluated the efficacy, safety, and drug survival rate of TCZ therapy in elderly RA patients receiving treatment in the real-world clinical setting. In the present study population, the body weight was lower, renal function and physical function were worse, use of oral MTX was less frequent, and the MTX dose was lower in the elderly group compared with the young group. These characteristics of the elderly group corresponded with those previously reported for the general elderly population and elderly RA patients. 13 Although the frequency of using MTX was lower in the elderly group than in the young group, efficacy of TCZ therapy was similar between the two groups. In addition, a steroid-reducing effect of TCZ was observed in both groups, and the drug survival rate of TCZ for 3 years was not lower in the elderly group compared with the young group. Furthermore, there was no difference between the groups in the frequency of discontinuing TCZ therapy due to adverse events. Thus, the safety of TCZ was not inferior in the elderly RA patients compared with the younger RA patients.
The first retrospective study comparing young and elderly RA patients receiving TCZ therapy was reported by Pers et al. from France in 2016, in which 222 RA patients were classified into an elderly group (65 years or older,
Mori et al. reported that age was weakly associated with the increase of hospitalized infection during the first year of follow-up in the patients treated with TNF inhibitors (
MTX is the standard first-line treatment for RA; however, old age is a risk factor for adverse effects in RA patients treated with MTX. In this study, the MTX dose decreased in the elderly group after initiation of TCZ therapy, as also occurred in the young group. The MTX dose was reduced because the patients responded well to TCZ therapy rather than due to adverse events (data not shown). The SATORI and SAMURAI studies conducted in Japan showed that TCZ monotherapy was more effective than conventional treatment for RA patients who had responded poorly to MTX or existing DMARDs.18,19 ADACTA study, which was a study conducted outside Japan, demonstrated that TCZ monotherapy was more effective than adalimumab monotherapy. 4 In addition, The ACTRA-RI study demonstrated that there was no significant difference in retentions of TCZ with and without MTX during maintenance therapy for RA patients achieving clinical improvements during the first year. 20 In the present study, both the efficacy and safety of TCZ were not inferior in the elderly RA patients compared with the younger RA patients. Therefore, it seems that TCZ can be effective for elderly RA patients in whom it is difficult to use MTX or to increase the MTX dose, allowing dose reduction or discontinuation of MTX after TCZ is added.
Because long-term steroid therapy causes progressive osteoporosis and increases the risk of fractures, steroids should be used with care, especially in elderly patients. Post-marketing all-case surveillance of TCZ showed that the risk of severe infection was increased by about 2.8 times in patients using steroids at doses of more than 5 mg per day (PSL equivalent). 5 A multicenter randomized prospective study of RA patients using TCZ showed that the steroid dose was reduced to less than 5 mg/day (PSL equivalent) in 55% of patients and steroids were discontinued in 20% of patients at one year after initiation of TCZ therapy. 21 A study investigating risk factors for progressive joint destruction in 400 RA patients showed that steroid use for three months or longer increased the risk of progressive of bone erosion by four-fold in elderly patients who had newly developed RA at an age ≥65 years but did not increase the risk in younger patients. 22 In our study, the dose of PSL was reduced significantly at one year after the start of TCZ administration in the elderly RA patients, as in the younger RA patients, and 23% of the elderly group discontinued steroids. Because the steroid dose can be reduced or steroids can be discontinued after TCZ is initiated, it is possible that use of TCZ could reduce the risk of progressive osteoporosis and joint destruction.
This study had some limitations that should be considered. First, it was a retrospective study and use of TCZ by a patient was determined by the attending doctor, so there might have been selection bias that could not be identified by reviewing baseline characteristics. In addition, the sample size was not large enough. However, this study provided evidence that TCZ was tolerable and effective even in elderly RA patients although old age is a risk factor for infection, and this information was obtained in a real-world clinical setting. In conclusion, the efficacy, safety, and drug survival rate of TCZ therapy were not inferior in elderly RA patients compared with the results obtained in younger RA patients, and TCZ also showed a steroid-reducing effect in elderly patients. These results suggest that TCZ is a useful treatment option, even in elderly RA patients.
Conclusion
The efficacy, safety, and adherence of TCZ in elderly patients were not inferior to those in younger patients. These results suggest that the utility of TCZ in elderly patients with RA.
Supplemental Material
sj-pdf-1-eji-10.1177_20587392211045790 – Supplemental Material for Efficacy, safety, and adherence of tocilizumab therapy in elderly patients with rheumatoid arthritis: A real-world observational study
Supplemental Material, sj-pdf-1-eji-10.1177_20587392211045790 for Efficacy, safety, and adherence of tocilizumab therapy in elderly patients with rheumatoid arthritis: A real-world observational study by Yoshinobu Nakao, Yu Funakubo Asanuma, Takuma Tsuzuki Wada, Mayumi Matsuda, Hiroaki Yazawa, Yoshihiro Yoshida, Akira Todriki, Ayumi Shintani and Toshihide Mimura in European Journal of Inflammation
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
Ethical approval for this study was obtained from Institutional Review Board of Saitama Medical University Hospital (Approval number: 2021-049).
Informed consent
This observational study was conducted in accordance with the ethical standards of the Declaration of Helsinki and approved by the Institutional Review Board of the Saitama Medical University Hospital. The board waived the requirement for patients’ informed consent by showing the research information on the website of Saitama Medical University because of the retrospective and observational design that used only clinical data obtained by daily clinical practice and guaranteed the opportunity for the study subjects to refuse.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
