Abstract
Background and objectives
This study validated the antimalarial efficacy of Laportea aestuans extract using chemosuppressive and curative models, identified its most active fraction, and performed chemical profiling of the active fraction.
Methodology
The aerial part of L. aestuans (LA) was extracted with 80% methanol and assayed for its chemosuppressive and curative antimalarial activities through the oral administration of the extract (100, 200, 400 and 800 mg/kg) to groups consisting of five mice each. The extract was thereafter suspended in water and partitioned with dichloromethane to afford the dichloromethane (DLA) and aqueous (ALA) fractions. The fractions (10, 20, 40, and 80 mg/kg) were thereafter tested for their antimalarial activity followed by GC-MS analysis of the most active fraction.
Results
The toxicity level was found to be above 2000 mg/kg. The activity recorded for the four-day chemosuppressive antimalarial tests showed a dose-dependent activity up to 400 mg/kg. The activity was significantly lower than that of chloroquine. The antimalarial activity of the extract at the lowest dose of 100 mg/kg was 42%. In the curative test, the extract of LA gave a dose-dependent activity that was significantly higher than the negative control. The DLA and ALA fractions gave a comparable curative activity across all doses. At 10 mg/kg, ALA and DLA gave percentage clearance of 81.25 ± 1.84 and 87.45 ± 1.35, respectively, which were significantly higher than that of the negative control (0.00 ± 0.00) and significantly lower than that of the positive control (94.93 ± 0.26). The chemical profiling revealed the presence of non-polar and medium-polar constituents in the dichloromethane fraction.
Conclusion
The good antimalarial activity elicited by LA extractives supports its usage in ethnomedicine as an antimalarial agent.
Introduction
Parasitic diseases are most prevalent in developing countries, particularly those situated in warm and humid areas of tropical, subtropical, and temperate regions. Seven of the 10 globally important tropical diseases are parasitic diseases, including malaria, schistosomiasis, lymphatic filariasis, onchocerciasis, leishmaniasis, African trypanosomiasis and Chagas disease.1–3 Malaria is a disease in humans caused by Plasmodium parasites, which is transmitted to humans via bite from infected female Anopheles mosquitoes. 4 Mild symptoms include fever, chills and headache while severe symptoms include fatigue, confusion, seizures, and difficulty breathing. 5 Infants, children below 5 years, pregnant women, travelers, and people with HIV or AIDS are at higher risk of severe infection.6,7
The latest World Malaria report indicates a slight increase in malaria cases, with 249 million reported in 2022 compared to 244 million in 2021. 7 The WHO African Region bears the overwhelming majority of the global malaria burden, with an estimated 94% of all malaria cases and 95% of deaths occurring there in 2022. Children under 5 years of age accounted for about 78% of all malaria deaths in the region. 7
Malaria is preventable by using insecticide-treated nets or taking prophylactics. Treatment typically involves antimalarial medications. 8 However, resistance to these medications has emerged as a major challenge over time. 9 A prime example is the emergence of partial artemisinin resistance, which has significantly threatened global malaria control efforts in the past decade. 10 While new studies explore the development of malarial vaccines,11,12 there remains a pressing need to investigate natural sources for the discovery of more effective antimalarial lead molecules with minimal side effects.
Plant-derived medicines have been an integral aspect of traditional health care in many regions of the world for thousands of years. There is a rising interest in them as sources for the treatment of diseases.13,14 Medicinal plants biosynthesize numerous secondary metabolites that are sources of bioactive organic compounds, which help defend against infection or disease. 15 These substances may also act as attractants, insecticides, antifeedants, oviposition inhibitors, repellents, growth inhibitors, and juvenile hormone analogs. 16 Medicinal plants have shown potential use in the treatment of malaria. Scientific studies and traditional evidence have helped establish the antimalarial activities of African medicinal plants, including Fagara zanthoxyloides, Monodora myristica, Piliostigma thonningii, Cassia nigricans, Dracaena manni, Stachytarpheta cayennensis, and Newbouldia laevis. 17
Phytochemicals like benzophenanthridine alkaloids, sesquiterpenes, triterpenoid, saponins, glycoside/glucoside, and pheophytins are some of the identified active compounds isolated from these medicinal plants. 17 There are also predictions of the mechanism of action of some of these plant metabolites. For example, androstan-17-one,3-ethyl-3-hydroxy-, (5 alpha) identified in the fruits of Piper betle has been predicted to interact with plasmepsin 1 and 2, which are employed in the degradation of hemoglobin upon infection by Plasmodium falciparum. 18 Since medicinal plants have shown potential as a veritable source of antimalarial compounds, this study evaluates the antimalarial properties of Laportea aestuans, which is traditionally used in the treatment of malaria.
Laportea aestuans (Urticaceae), the West Indian Nettle, is a little-branched, annual plant native to tropical regions. It is now widely spread throughout both the western hemisphere and eastern hemisphere tropics and subtropics. 18 It is usually 1 metre tall, up to 3 metres. The stem is fleshy, becoming slightly woody at the base, and the whole plant is densely covered with stinging hairs up to 1 mm long. 19 It is widely used in the management of various diseases in African traditional medicine. The pulped whole plant is eaten or the plant sap is drunk as an anthelmintic and for the treatment of hernias. Externally, it is also applied in the treatment of fevers in children, malaria (personal communication), oedema, and ulcers. The sap, often mixed with palm oil or kaolin, is applied to abscesses and wounds; on the head of children to close the fontanel; on the abdomen to ease childbirth; and on the gums to relieve toothache. 20 A macerated fresh leaf of L. aestuans is used to massage the body for the treatment of intercostal pain and stitches on the side. Slightly scorched or smoked leaves are applied to burns and used against migraine. 18 A leaf decoction of the plant is applied on swellings; instilled in the eye to treat minor eye infections; and as an embrocation to strengthen rachitic children and to relieve fever. 19
Extract of L. aestuans is reported to elicit antioxidant properties, while its essential oil whose constituent is mainly methyl salicylate exhibits significant antioxidant and antimicrobial activities (Oloyede, 2011). 19 Other studies reported the phytochemical, toxicity, antimicrobial, and antioxidant screening of extracts obtained from the plant.19–21 Despite the rich pharmacological studies performed on the plant, its antimalarial activity is yet to be investigated. Therefore, this study uses chemosuppressive and curative models to examine its antimalarial efficacy. In addition, the study performed chemical profiling for the identification of compounds present in their active fractions.
Methodology
Plant Collection, Extraction, and Partitioning
The aerial parts of L. aestuans were collected from the Obafemi Awolowo University (OAU) campus. The plant material was identified and authenticated at the Forestry Research Institute of Nigeria, Ibadan, Nigeria, with the voucher number FHI 11035. Three hundred grams (300 g) of fresh-weight plant material was crushed and extracted with 80% methanol for 48 h. The extract was then filtered and concentrated to yield 3 g of residue. The residue was suspended in water and partitioned with dichloromethane, affording 1.2 g of a dichloromethane fraction and 1.8 g of an aqueous fraction.
Antimalarial Studies
Animals
Healthy Swiss albino mice, both male and female (nonpregnant), weighing between 18 to 24 g, were procured from the Animal House, Faculty of Basic Medical Sciences, College of Health Sciences, Obafemi Awolowo University, Ile-Ife, Nigeria. The mice were housed in aluminum cages with wood shavings as bedding material and were offered water freely and Growers’ mash as food, maintaining under a 12-h light/dark cycle. The animal experimental protocol received approval from the Health Research and Ethics Committee of the Institute of Public Health, Obafemi Awolowo University, Ile-Ife, Nigeria. All procedures adhered to the guidelines outlined in the NIH Guide for the Care and Use of Laboratory Animals (NIH Publication, No. 83-123, revised 1985). Also, the study was approved by the Postgraduate Committee of Obafemi Awolowo University with registration number: SCP17/18/H/1272. Acclimatization was ensured for a minimum of seven days before the commencement of experiments, and the mice were randomly assigned to groups of five for the study.
Parasite
Plasmodium berghei strain NK65, susceptible to Chloroquine, was kindly donated by Professor O. G. Ademowo of the Institute of Advanced Medical Research and Training (IMRAT), University College Hospital, Ibadan. This strain was employed for evaluating the in vivo chemosuppressive and curative antimalarial activities. The parasite strain was preserved by successively transferring blood from infected mice to uninfected mice. The donor mouse was euthanized, and blood was collected via cardiac puncture into a heparinized bottle to prepare the inoculum. This inoculum was then diluted with a normal saline solution to ensure that 0.2 mL of the inoculum contained 1.0 × 107 parasitized red blood cells.
in Vivo Antimalarial Assays
The efficacy of the extract in suppressing and treating malaria was assessed by orally administering varying doses (100 mg/kg, 200 mg/kg, 400 mg/kg, and 800 mg/kg) to mice, alongside chloroquine (CQ) at 10 mg/kg for comparison, while a control group received normal saline. Each group comprised five mice. Administration commenced two hours after infection and continued daily for 3 days in the suppression model. In the treatment model, administration began on the third day post-infection and continued daily for 5 days. Prior to administering the extracts or standard drug, the body temperature of each mouse was measured using a digital clinical thermometer inserted into the rectum.
In both models, parasitemia levels were evaluated for each mouse on Day 4 (D4) and daily thereafter in the suppression and treatment models, respectively. This assessment involved cell counting in 5 fields of view on a microscope slide with a thin blood smear obtained from the tail of each mouse. The smears were fixed with methanol and stained with Giemsa. The average parasitemia in each group was calculated to determine the percentage of suppression and treatment efficacy of the extract using the formula:
Survival Times
All the animals used for this study were observed for 28 days for possible mortality, while the survival time and percentage of survivors were calculated using the formula below22,23:
Statistical Analysis
Values were expressed as mean ± SEM and analyzed statistically using One-way Analysis of Variance (ANOVA) followed by Student Newmann Keul's post-hoc for comparisons to determine the source of significant difference for all values. Values of P < 0.05 were of statistical significance.
GC-MS Profiling
The dichloromethane fraction of L. aestuans underwent GC-MS analysis utilizing a Varian 3800/4000 gas chromatograph mass spectrometer equipped with an Agilent fused silica capillary CP-Sil 5 CB column (30 m × 0.25 mm i.d.). This system was linked to a Varian 4000 mass spectrometer operating in Electron Impact (EI) mode (70 eV; m/z 30-1000 amu; source temperature 230 °C and quadruple temperature 150 °C). The initial temperature was held at 150 °C and then gradually increased to a maximum of 300 °C. A 1 μL sample was injected in split mode (10:1). Nitrogen gas was employed as the carrier gas at a flow rate of 0.8 mL/min, and the total runtime was set at 40 min.
For the identification of phytochemical components, the National Institute of Standards and Technology MS library (NIST-MS library 2009) was utilized, comparing the obtained spectrum via GC–MS to identify compounds present in the samples. Retention indices (RI) were determined relative to a homologous series of n-alkanes (C6–C28) on the CP-Sil 5 CB column under identical chromatographic conditions. The relative concentrations of components were determined through peak area normalization, with no response factors calculated. All samples and their replicates were injected as a single batch in a random order to distinguish technical variations from biological ones. Moreover, the pooled samples prepared were employed as quality controls (QCs) and injected at regular intervals during the analytical run. This approach facilitated the generation of a dataset for assessing repeatability.
Results
Acute Oral Toxicity
Extracts that mice can tolerate at doses of up to 5000 mg/kg without exhibiting any signs of toxicity can be considered non-toxic and safe.23–27 Regarding the aqueous methanol extract of L. aestuans, doses of up to 5000 mg/kg did not result in mortality or manifest any adverse effects such as skin changes, aggressiveness, diarrhea, restlessness, dizziness, weakness, or refusal to consume food or water. Therefore, it can be inferred that the tested extract is devoid of toxicity. 28 Similar findings were also reported in studies involving Plumeria alba and Azadirachta indica.25,28
Evaluation of Chemosuppressive Effect of LA Extract
The chemosuppressive effect of LA extract varied from 42% to 67% across doses ranging from 100 to 800 mg/kg. Comparatively, chloroquine demonstrated a higher chemosuppressive effect of 80%, which was significantly greater (p < 0.05) than that of the LA extract at all tested doses. Furthermore, the mean survival time of mice treated with LA extract ranged from 11.00 ± 0.32 to 13.00 ± 0.00 days across different doses. In contrast, mice treated with chloroquine exhibited a mean survival time of 26.40 ± 1.60 days, while those in the negative control group survived for 6.60 ± 0.75 days (Table 1).
Antiplasmodial Activity of LA Leaf Extract in Mice Infected Plasmodium Berghei in 4-day Chemosuppressive Test.

Keys: Data show the mean ± SEM, n = 5. NC (negative control): Tween 80 in normal saline; Chloroquine (10 mg/kg) = positive control. Only values with different superscripts (a, b, c, d or e) within columns are significantly different (p < 0.05, one-way analysis of variance followed by the Student–Newman–Keul's post hoc test).
Evaluation of the Curative Effect of LA Extract
The findings from the analysis of the curative potential of LA extract at doses of 100, 200, and 400 mg/kg revealed a decrease in percentage parasitemia from 8.04–8.72% on day 1 to 1.76–1.92% on day 5. Additionally, the curative effects observed for doses of 800 mg/kg and chloroquine (5 mg/kg) on day 5 resulted in a reduction of percentage parasitemia to 1.19% and 0.9%, respectively (Table 2).
Percentage Parasitaemia in an in Vivo Antimalarial Activities (Curative) Test of the Methanol Extract of the LA at Different Doses at DAY 0 to 5.

Keys: LA = Data show the mean ± SEM, n = 5, NC = Negative Control (Tween 80 in normal saline); CQ = Chloroquine (10 mg/kg). Only values with different superscripts (a, b, c, d, e, f or g) within columns are significantly different (p < 0.05, one-way analysis of variance followed by the Student–Newman–Keuls’ post hoc test).
Percentage Clearance and Survival Time of LA Extract in Mice
On day 5, LA demonstrated percentage clearances of 84.28%, 85.04%, 85.65%, and 90.26% at doses of 100, 200, 400, and 800 mg/kg, respectively, in comparison to the 0.00% observed in the negative control group. In contrast, chloroquine exhibited a clearance of 92.66% (Table 3). The antiplasmodial efficacy of LA peaked at a dose of 400 mg/kg. The mean survival times, recorded in days, for the various doses, negative control, and chloroquine were 18.20, 21.40, 24.40, 26.80, 6.00, and 28.00, respectively (Table 3). The calculated ED50 and ED90 for the extract were 210.15 mg/kg and 399.21 mg/kg, respectively (Table 3).
Percentage Clearance in an in Vivo Antimalarial Activities (Curative) Test of the Methanol Extract of the LA at Different Doses at DAY 0 to 4.

Keys: LA = Data show the mean ± SEM, n = 5, NC = Negative Control (Tween 80 in normal saline); CQ = Chloroquine (10 mg/kg). Only values with different superscripts (a, b, c, d, e, f or g) within columns are significantly different (p < 0.05, one-way analysis of variance followed by the Student–Newman–Keuls’ post hoc test).
Evaluation of Curative Effect of LA Solvent Fractions
The percentage parasitemia induced by the solvent fractions of LA on day 5 at various doses of ALA10, ALA20, ALA40, ALA80, DLA10, DLA20, DLA40, and DLA80 mg/kg were 2.65, 1.83, 1.49, 3.15, 1.77, 1.24, 0.91, and 2.36%, respectively, compared to the negative control's parasitemia level of 14.27%. In contrast, chloroquine resulted in a parasitemia level of 0.72% (Table 4).
Percentage Parasitaemia in an in Vivo Antimalarial Activities (Curative) Test of the Solvent Fractions of LA at Different Doses at DAY 1 to 5.

Keys: DLA = Dichloromethane fraction; ALA = Aqueous phase. Data show the mean ± SEM, n = 5, NC = Negative Control (Tween 80 in normal saline); CQ = Chloroquine (10 mg/kg). Only values with different superscripts (a, b, c, d, e, f, g, h or i) within columns are significantly different (p < 0.05, one-way analysis of variance followed by the Student–Newman–Keuls’ post hoc test).
Percentage Clearance of LA Solvent Fractions in Mice
At day 5, the solvent fractions of LA showed varying percentages of clearance at different doses of ALA10, ALA20, ALA40, ALA80, DLA10, DLA20, DLA40, and DLA80 mg/kg, with values of 81.25%, 87.07%, 89.60%, 77.93%, 87.45%, 91.28%, 93.62%, and 83.30%, respectively, in comparison to the 0.00% clearance observed in the negative control. Chloroquine, in contrast, exhibited a clearance of 94.93% (Table 5). The calculated ED50 and ED90 values for ALA and DLA at day 5 were 23.14; 39.18 and 21.94; 37.20 mg/kg, respectively (Table 6).
Percentage Clearance in an in Vivo Antimalarial Activities (Curative) Test of the Solvent Fractions of LA at Different Doses at DAY 1 to 5.

Keys: DLA = Dichloromethane fraction; ALA = Aqueous phase. Data show the mean ± SEM, n = 5, NC = Negative Control (Tween 80 in normal saline); CQ = Chloroquine (10 mg/kg). Only values with different superscripts (a, b, c, d, e, f or g) within columns are significantly different (p < 0.05, one-way analysis of variance followed by the Student–Newman–Keuls’ post hoc test).
Comparative ED50 and ED90 in an in Vivo Antimalarial Activities (Curative) Test of the Solvent Fractions of LA at Different Doses at DAY 1 to 5.
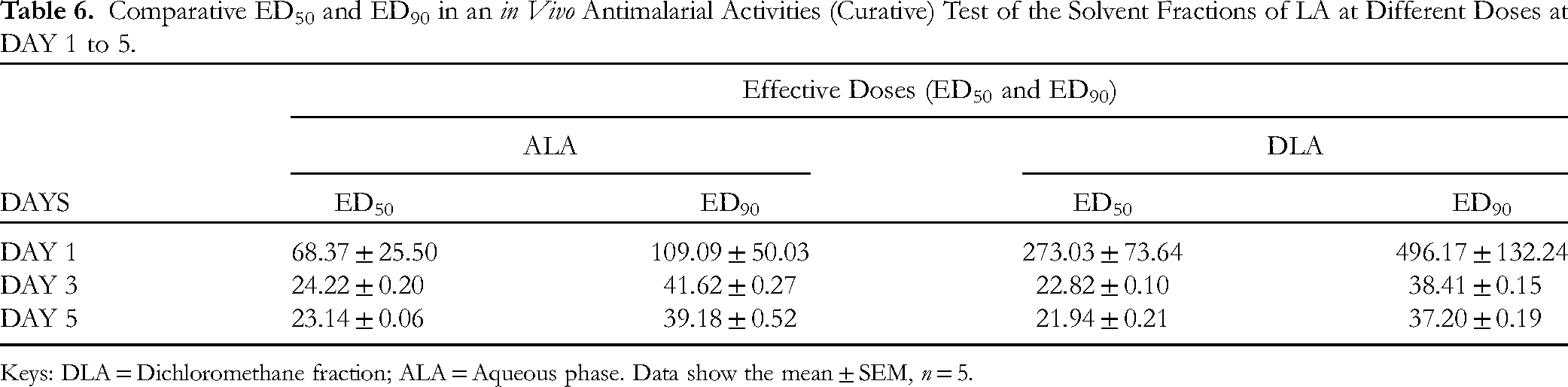
Keys: DLA = Dichloromethane fraction; ALA = Aqueous phase. Data show the mean ± SEM, n = 5.
Chemical Profiling of DCM Fraction of L. aestuans
The results of the GC–MS analysis for the dichloromethane fraction are outlined in Table 7, revealing the presence of 23 compounds. Notably, four of these compounds collectively accounted for 54% of the total compounds. These prominent constituents include 9,12-Octadecadienoic acid (Z,Z)- (19.06%), 9,12,15-Octadecatrienoic acid, (Z,Z,Z)- (18.66%), and tetradecanoic acid (16.28%). The dichloromethane fraction of the LA extract contained a variety of compounds categorized as esters, fatty acids, and terpenes (see Table 7). Emeka et al 29 previously documented the effectiveness of 9,12-Octadecadienoic acid (Z,Z)- in managing hyperlipidemia and atherosclerosis.
Phytoconstituents Identified from the Dichloromethane Fraction of L. aestuans.

Molecular ion fragment.
Discussion
The endemic nature of malaria and the resistance of the parasite to current synthetic antimalarial drugs has led to the continuous search for new molecules to replace them. 30 As a result of malaria's prevalence, several in vivo, in vitro, and in silico investigations have been reported, justifying the ethnomedical use of medicinal plants as antimalarial remedies.31–33 These studies have gone beyond mere justification, with previous reports describing the antimalarial compounds found in leaves, roots, and stem bark of some antimalarial plants.25,34 Notably, some of these compounds are non-polar extracts obtained using non-polar solvents.33,35
In our search for new antimalarial leads, we considered extracts of LA due to its relevant traditional applications and reported pharmacological activities. Traditional medicine considers LA extracts safe with no adverse effects. This study confirmed the LA extract's low toxicity, with a level exceeding 2000 mg/kg. This aligns with a previous report where the LA extract showed no toxicity even at a dosage of 5000 mg/kg. 19
We then tested the extract's antimalarial effects using chemosuppressive, prophylactic, and curative models at doses of 100, 200, 400, and 800 mg/kg. The four-day chemosuppressive tests yielded a percentage chemosuppression significantly higher than the negative control. The observed activity was dose-dependent up to 400 mg/kg, but significantly lower than that of chloroquine (positive control). At the lowest dose (100 mg/kg), the extract's activity (42%) fell short of the optimal 50% threshold. Nigussie and Wale 36 define a very good in vivo antimalarial activity as one that suppresses malaria by ≥50% at 100 mg/kg. Interestingly, we observed no corresponding increase in chemosuppressive activity at doses exceeding 400 mg/kg.
Studies have shown that at a higher dosage, some plant metabolites exhibit increased biological effects with increasing doses, then plateau or reverse at higher doses, a phenomenon known as biphasic dose response. The biphasic dose-response displayed by the LA extract in the chemosuppression test could be due to the saturation of the binding sites where its constituents bind.37,38 A similar result was reported by Alehegn et al 39 in their study on the antimalarial activity of crude extract of the leaves of Bersama abyssinica Fresen. The survival time (in days) showed that the extract was able to prolong the life of the animals significantly more than that of the negative control but significantly less than chloroquine. Only at 800 mg/kg did the extract prolong the life of the mice more than 12 days.
In the curative antimalarial assay, the methanol extract of LA gave a dose-dependent activity that was significantly higher than the negative control. The activity elicited by the LA extract at the highest dose of 800 mg/kg was comparable to that of chloroquine (positive control). This suggests that further purification of the compound might yield even more active compounds if the activity is not due to synergistic action. The LA extract at all doses extended the lifespan of the mice to more than triple that of the negative control group (more than 12 days). The result showed that increasing the dose of the LA extract significantly increased the survival time of the mice compared to the negative control (6.00 ± 0.55). The extract also showed a comparable survival time (26.80 ± 0.58 days) to that of chloroquine (28.00 ± 0.00) at the highest dose of 800 mg/kg. Extracts are considered active if they can extend the survival of the animal beyond 12 days. 40 Also, Faloye et al 41 reported that the survival time for treated mice in antiplasmodial studies which doubled that of the negative control, was evidence of good antiplasmodial activities of the extract. Even at the lowest dose of 100 mg/kg, the LA extract was able to triple the survival time of the animals relative to negative control and therefore could be considered as an indication of good antiplasmodial activity.
The median effective doses (ED50 and ED90) of the extracts were determined to establish a basis for comparison of the antimalarial activities between the crude extracts and subsequent partitioned fractions. 36 Median effective doses have been used to compare the activities of different formulation ratios and partitioned fractions of MAMA Decoction. 35 Previous studies have reported that a lower ED50 and ED90 indicate higher activity. 37
Comparing the ED50 and ED90 in the chemosuppressive and curative models, LA exhibited a lower ED50 (210.15 ± 1.86) in the curative model than in the chemosuppressive model (328.10 ± 2.42). This suggests that LA elicits a better curative effect than a suppressive effect. Therefore, the curative model was chosen for further investigation on the solvent fractions of LA.
In the curative antimalarial studies of the partitioned fractions, the solvent fractions (ALA and DLA) displayed comparable curative activity across all doses. This suggests that the antimalarial potency of the plant is distributed equally between the polar and non-polar solvents. It also suggests that the active phytochemical constituents can be extracted from both solvents or that the antimalarial effect is produced by more than one phytochemical constituent. 42
At the lowest dose (10 mg/kg), ALA and DLA produced significantly higher percentage clearances (81.25 ± 1.84 and 87.45 ± 1.35, respectively) compared to the negative control (0.00 ± 0.00) but significantly lower than the positive control (94.93 ± 0.26) at the same dose. The ED50 and ED90 values of ALA and DLA were 23.14 ± 0.06; 39.18 ± 0.52 and 21.94 ± 0.21; 37.20 ± 0.19, respectively. This indicates that a larger portion of the antimalarial constituents of LA reside in the non-polar fraction (DLA). Previous studies have shown that active antiplasmodial compounds of some antimalarial plants reside in the non-polar fractions.25,42
The ED50 and ED90 of chloroquine were reported to be 2.19 ± 0.10 and 4.29 ± 0.10, respectively,26,43 which are significantly better than the values reported for DLA (21.94 ± 0.21; 37.20 ± 0.19). However, it is projected that purification of the dichloromethane fraction would further improve its activity. Further purification of DLA might yield compounds comparable to or even better than chloroquine in their antiplasmodial activity.
Limitations of the Study
The study focused on evaluating the antimalarial activity of the extract and fractions of L. aestuans. To identify the chemical constituent responsible for the most active fraction's antimalarial activity and elucidate its possible mechanism of action, molecular docking and molecular dynamics simulation could be performed. This would further elucidate the possible mechanism of action of the identified phytochemical.
Conclusion
This study employed chemosuppressive and curative models to evaluate the antimalarial potency of L. aestuans, with the curative model identifying the most active fraction. The curative model yielded considerably higher antiplasmodial activity compared to the chemosuppressive model in infected mice. The dichloromethane fraction exhibited the strongest antiplasmodial activity. However, both solvent fractions tested elicited good antimalarial activity, suggesting that the chemical constituents distributed between the non-polar and polar fractions largely contribute to the antimalarial efficacy of L. aestuans. The chemical profiling revealed the presence of non-polar and medium-polar constituents in the dichloromethane fraction
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
This study was approved by the Postgraduate Ethical Committee of Obafemi Awolowo University, Ile-Ife, Nigeria, with registration number: SCP17/18/H/1272.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Statement of Human and Animal Right
All procedures in this study were conducted in accordance with the Animal Care approved protocols of the Postgraduate Ethical Committee of Obafemi Awolowo University, Ile-Ife, Nigeria.
Informed Consent
There are no human subjects in this article and informed consent is not applicable.


