Abstract
The aim of this study was to perform green synthesis of silver nanoparticles (AgNPs) from the leaf extract of
Introduction
Research on nanoparticles is inevitable today because of their applicability in various fields as they possess catalytic, optical, magnetic, biological, and distinct physiochemical properties.1,2 These properties are strongly influenced by degree of temperature, polarity of solvent, concentration of precursors, and reduction agents. 3 On a special note, silver nanoparticles (AgNPs) display remarkable applications in the field of medicine for therapeutics and diagnostics as they are eco-friendly. 4 Moreover, biological activities of AgNPs results in enhanced treatment quality, reduced side effects, and improved quality of life which in turn significantly influences the overall performance of the system. 5 Silver nanoparticles can be fabricated by different methods, which include physical, chemical, and biological.6–8 Even though physical and chemical methods yield high amount of NPs with specific size and shape, usage of microorganisms or enzymes or plant extracts related to biological practices are considered to be environment friendly, reliable, nontoxic, and prudential.9,10
Synthesizing of nanoparticles using plant extracts is considered as green synthesis and various medicinal and aromatic plant extracts of neem (
Materials and Methods
D. bipinnata (Db) leaf extract

Photograph of the
Preparation of silver nanoparticles
A total of 1.5 g of high-analytical grade silver nitrate (0.1 M AgNO3, ACS reagent, ⩾ 99.0%, Sigma-Aldrich, Merck, USA) was dissolved in 90 mL of High performance liquid chromatography (HPLC) grade water to form AgNO3 solution. Approximately 15 mL of the
Characterization of the silver nanoparticles
Synthesized AgNPs were analyzed by using dispersive methods, such as UV-Vis spectrophotometer (Perkin-Elmer, Lambda 35, USA), FTIR Spectrometer (Perkin Elmer Spectrum 2, wavelength range of 8000-350 cm−1, USA), X-ray diffractor (EDX, Siemens, Model D8, Aubrey, Texas, USA), Scanning electron microscope (SEM; TESCAN/VEGA, Model 3, USA), and EDX (Siemens, Model D8, Aubrey, Texas, USA). Fourier transform infrared spectrometer is useful in characterizing functional groups, bonding types, and nature of the compounds. The AgNP solution was centrifuged at 15 000 r/min to obtain AgNPs in powdered form and loaded in a sample holder and detected in the range of 4000 to 400 cm−1 using the KBr method. The scanning electron microscope was used to observe the morphology and composition of the AgNPs coupled with EDX to identify the elemental composition of the materials. Data generated by the EDX analysis consisted of the spectra of various peaks. The TEAM EDS point analysis software was used for the EDAX analysis to identify the element present in the sample. The phase variations and trace dimension of AgNPs were determined using X-ray diffractometer with Cu Kα radiation and Ni filter. The following Scherer’s equation was used to calculate the size of the prepared samples.

where, D is the crystal size, λ is the wavelength of X-ray,
In vitro antimicrobial properties of the silver nanoparticles
In vitro antimicrobial activity was investigated using the test samples (Db extracts/AgNPs) dispersed in 1 mL of sterilized distilled water and autoclaved (7440PAD, Medical Instruments, Mumbai, India) for 30 minutes. The bacterial strain cultures of
Radical scavenging ability of silver nanoparticles
DPPH radical scavenging assay
Reducing ability of Db extract and AgNPs were assessed using the 1, 1-diphenyl-2-picryl hydrazyl radical (DPPH) assay method. For each concentration (1, 10, 15, and 25 µg/mL), 1 mL of the sample was added with 3 mL of DPPH solution (0.004%). The prepared solution was left for 15 to 20 minutes in a dark room and read at 517 nm by UV-Vis spectrophotometer (PerkinElmer, Lambda 35, Germany). The percentage of inhibition was calculated using the formula
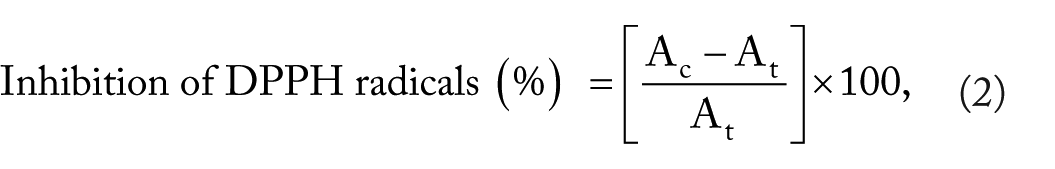
where Ac is absorbance of control sample (DPPH solution without test sample), and At is the absorbance of test sample (DPPH solution with test sample).
Reactive oxygen species (H2O2) scavenging assay
Reactive oxygen species (H2O2) was mixed in buffer with a known concentration and pH (30 mM; 7.4). Measures of 1.5 mL of samples (NPs) in different ratios or standards (α-Tocopherol and Ascorbic acid) were added to 2.5 mL of H2O2 and read at 233 nm after incubation.
Superoxide radical scavenging assay
Various concentrations of 0.5 mL AgNPs were diluted in 1 mL of basic Dimethyl sulphoxide (DMSO: 6 mM NaOH), and 0.1 mL of nitroblue tetrazolium in 2.5 mg/mL concentration was added to make a final mixture and read at 565 nm.
In vitro cell viability assay
The HepG2 (human liver cancer cell line) were grown and maintained in DMEM (Dulbecco Modified Eagle Medium) which was supplement with 10% Fetal bovine serum (FBS), 2 mM L-glutamine 100 U/mL penicillin G sodium and 100 mg/mL streptomycin sulphate. Cells were seeded in 96-well plate at a density of 1 × 104 cells/mL along with aforementioned media and maintained at 37°C and humidified with 5% CO2. HepG2 cells after reaching confluence were tested with various concentrations of synthesized AgNPs for cytotoxicity. After 24 hours of incubation, medium was discarded and the adherent cells were washed thrice with PBS and 30 µL of 3-(4,5-dimethyl-2-thiazolyl)-2,5-diphenyl-tetrazolium bromide (MTT; 10 mg/mL in PBS) was added to each well and incubated for 6 hours. Then 70 µL of DMSO was added to each well to solubilize the formazan crystals produced by viable cells. Furthermore, the absorbance was measured at 540 nm, as reference wavelength using a multiscan spectrophotometer (microplate reader) (Waltham, MA, USA). The effect of the AgNPs on the proliferation of HepG2 cells was expressed as the percent of cell viability using the following formula:
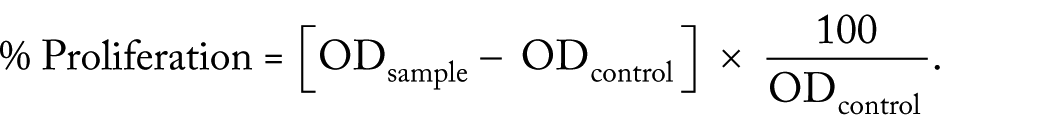
Results and Discussion
Preparation and optimization of nanoparticles
The Db extract was mixed to silver nitrate solution and incubated for 24 hours, thereby resulting in the change in the intensity color from light brown to dark due to reduction of Ag+ to Ag0. This attributes to the phenomenon of surface plasmon resonance (SPR) absorption band which is found in noble metals as a result of vibrations that are in tune with the light wave of both electrons in metal NPs. The maximum degradation of the Ag+ ions was observed after 4 hours of adding the grass leaf extract to the AgNO3 solution. The absorbance was measured from 300 to 700 nm using UV-Vis spectrophotometer which showed SPR hike at 433 nm confirming the formation of AgNPs as shown in Figure 2A. Mie theory suggested that the absorption spectra of a single spherical NP SPR band are possible; in addition, the peak increases with increased anisotropy.
36
The parameters measured in the AgNPs extract of
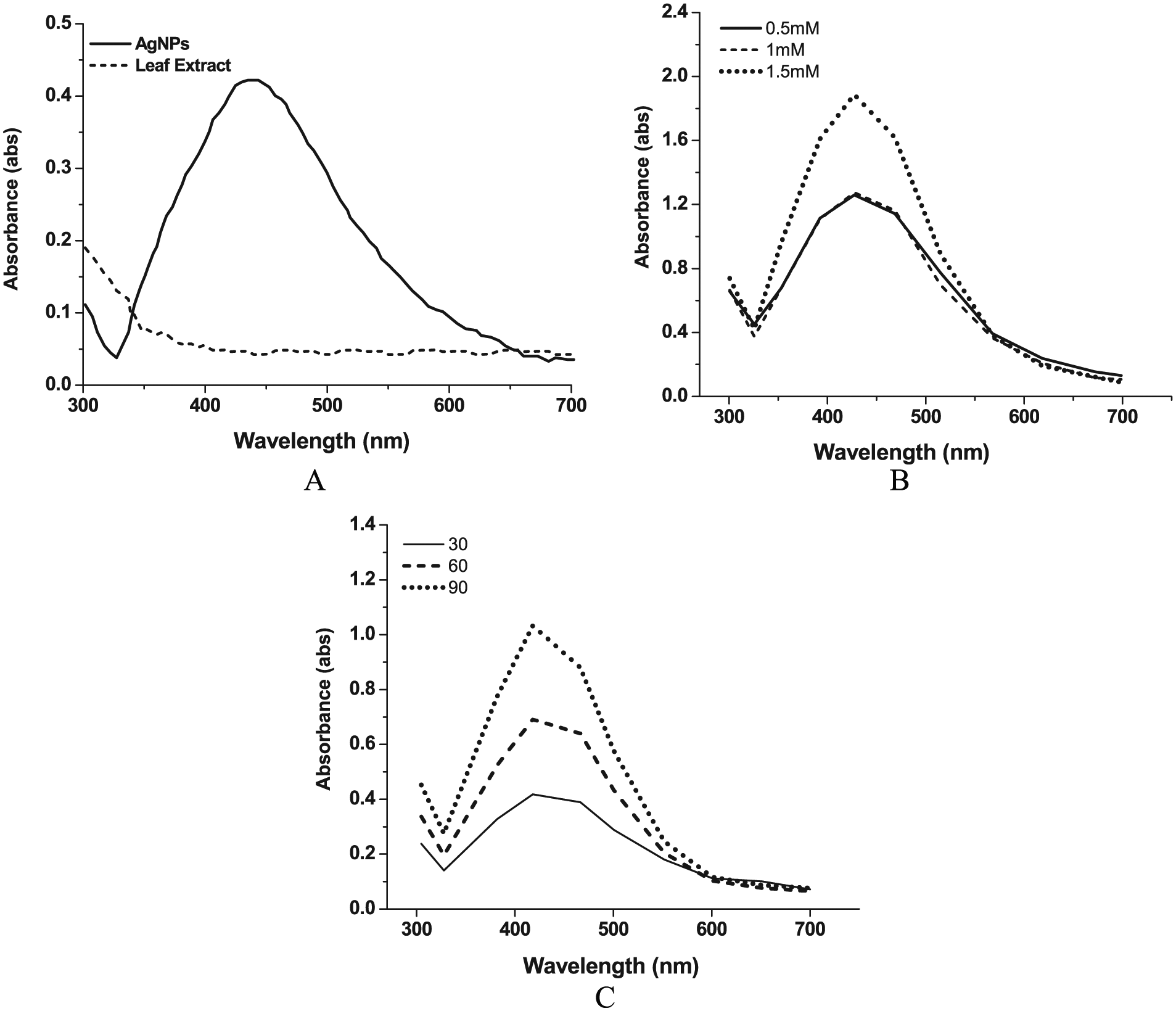
(A) Typical UV-Vis absorption spectra of the
Physicochemical characterization of nanoparticles
Fourier transform infrared spectrum (Figure 3) for the Db extract and NPs was performed to examine the stabilization of nanoparticles through biomolecules as capping agents. Grass leaf extract showed several spectral peaks such as, 3422 cm−1, which corresponds to the oxygen-hydrogen stretch to the Hydrogen-bonded OH groups and phenols which indicates complex nature of biomolecules. The peak obtained at 2390 cm−1 confirms O–H and C–H stretching bonds, which corresponds to carboxylic acids, primary amines, and alkanes. 30 The findings are further supported by a peak at 1635 cm−1 representing amines through H-N bond. Another peak (1052 cm−1) represents the C–O and C–H stretching bonds, which indicated the availability of functional groups. 36 The FTIR spectrum of the AgNPs with respect to the percentage of transmittance as the function of wavelength showed a few similar peaks corresponding to the presence of phenolic and alkanes interaction. 27 Other peaks at 1587 cm−1 and 1383 cm−1 corresponding to the C–C stretching bond indicates the presence of aromatic compounds and the N–O symmetric stretching that demonstrated the presence of the nitro compounds, respectively. 28 The peak at 1055 cm−1 represented the C–O and C–H stretching bonds, which corresponded to the presence of alcohols, carboxylic acids, esters, ether, and aliphatic amines. 39 Consequently, the synthesized AgNPs were bound by various proteins and metabolite functional groups containing terpenoids/flavonoids must have improved the ability to form a capping agent. 29
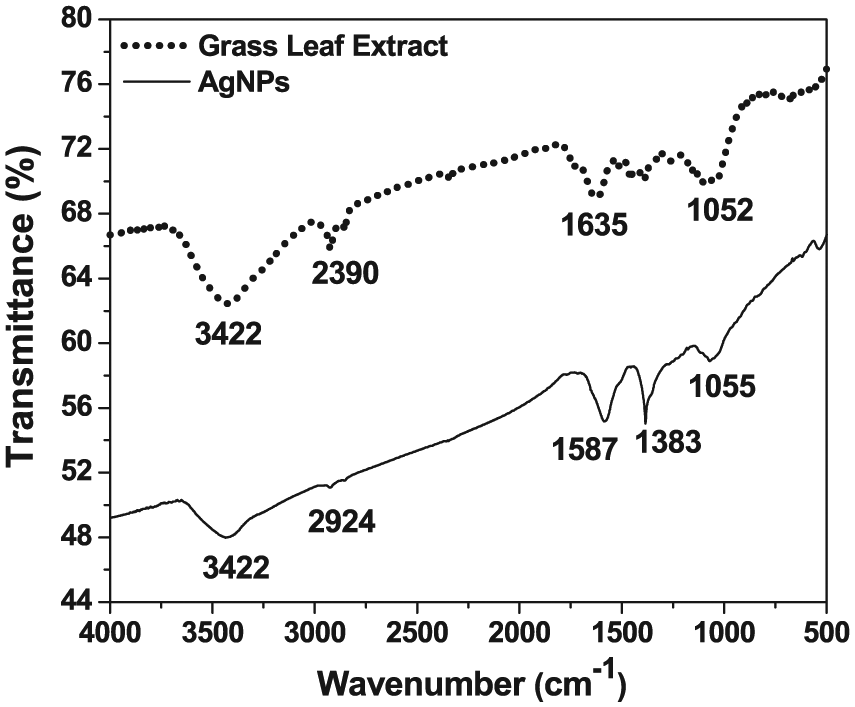
Typical Fourier transform infrared spectrum of the
The Db extract showed the maximum peaks at 21.99° on XRD spectrum, which corresponded to the 011 diffraction planes indicating the microcrystalline nature of the
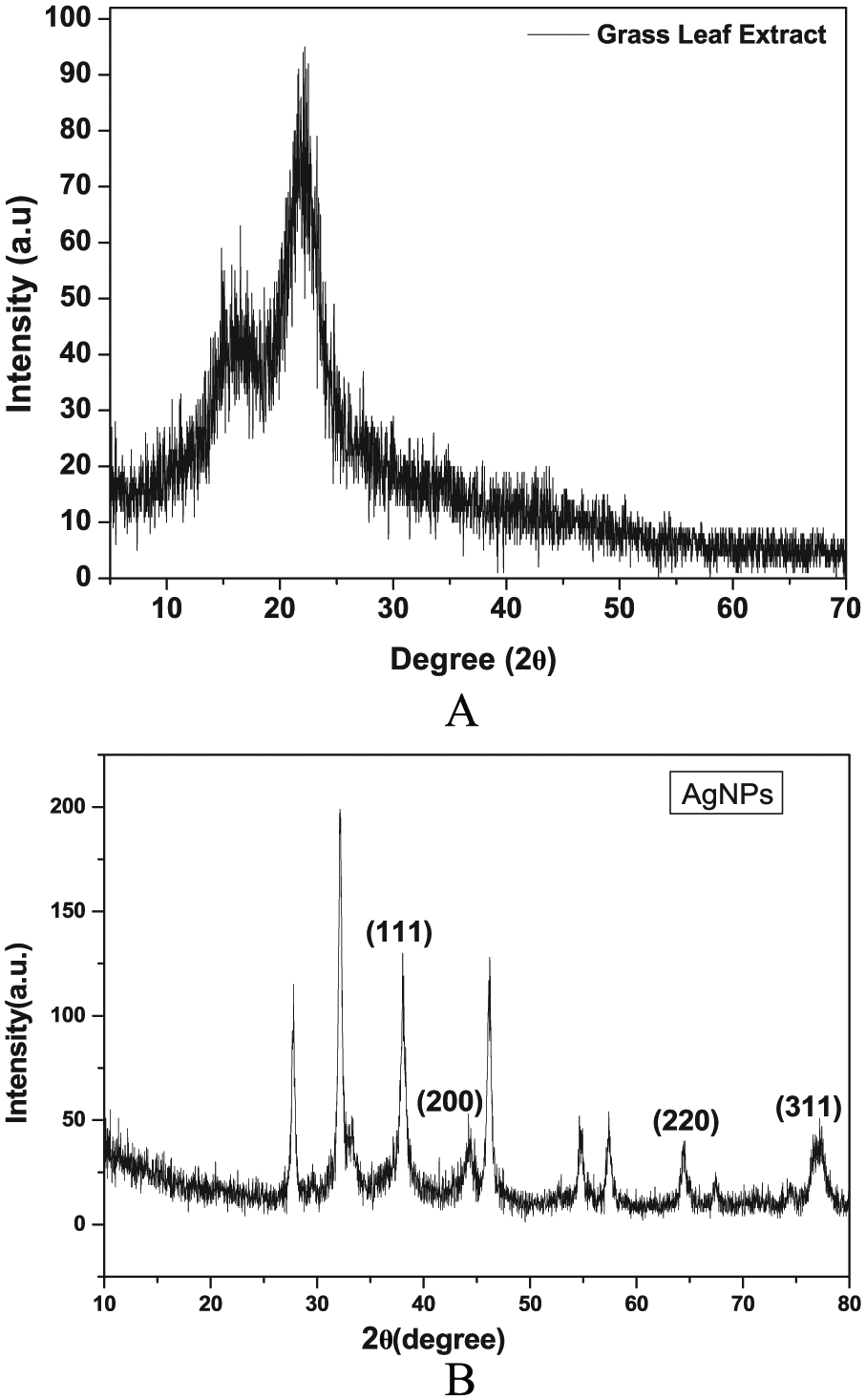
X-ray diffraction spectra: (A)
The SEM analysis verified the surface morphology and dimensions of the samples (Figure 5). The grass leaf extract showed hexagonal shape and AgNPs exhibited regular round-shaped particles within the range of 30 to 60 nm in diameter (Figure 5B). These superficial impurities may be due to the presence of biocapping agents of the unrefined fractions from the Db extract. 37 X-ray diffractor spectral analysis (Figure 6A and B) detected the presence of elements within the selected area and found 78.27% of Ag as the major element in the sample with carbon (18.06%) and oxygen (3.67%) as other elements. The significant amount of carbon and small amount of oxygen in the sample indicated the various physiochemical properties of AgNPs in the form of biocapping agent. The SEM and EDAX analyses showed no clear morphology of the formed AgNPs.42–44

Scanning electron microscopy (SEM) images: (A)
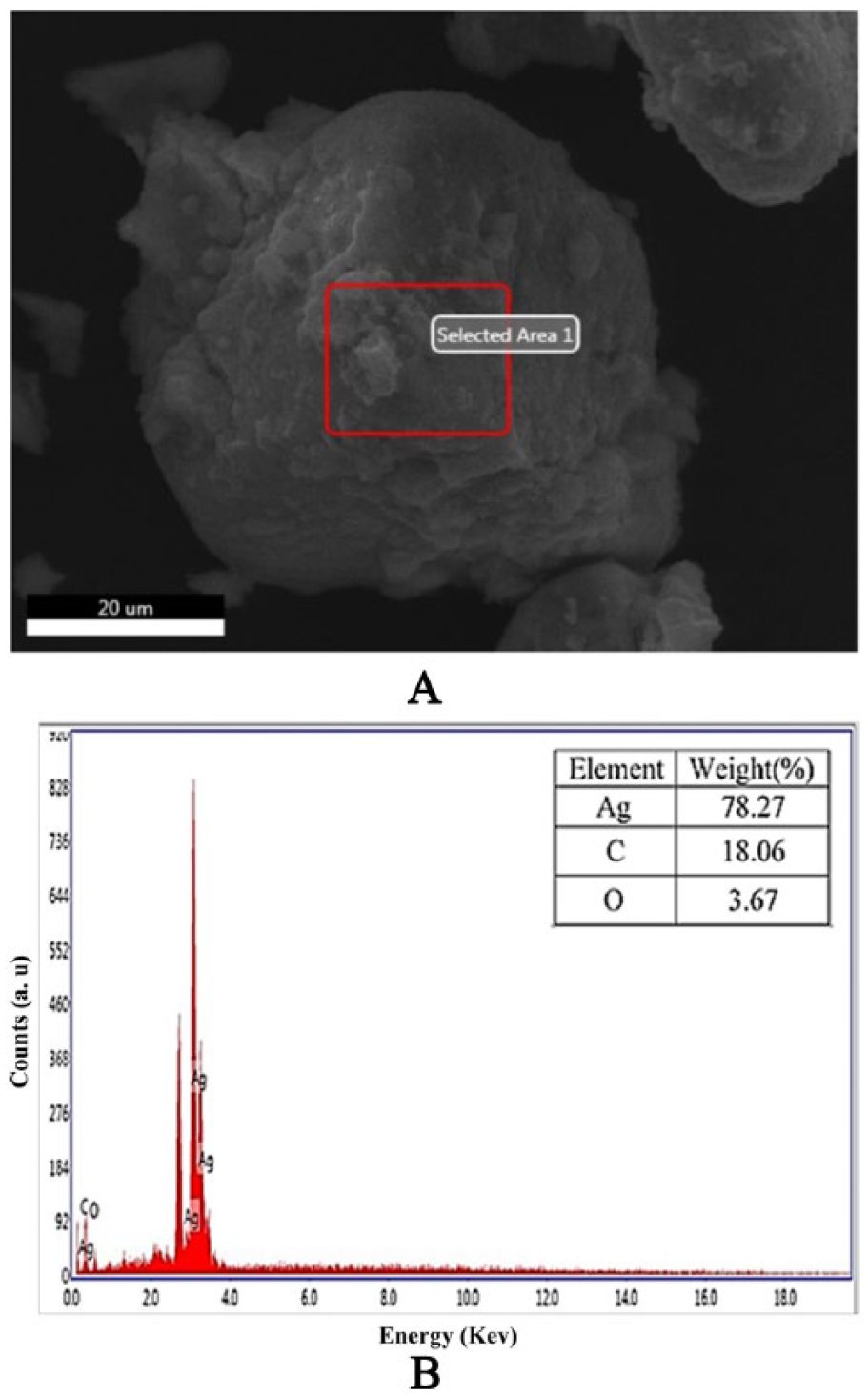
EDX spectrum: (A) Rectangular area of the AgNPs from the SEM image was selected for analysis. (B) EDX spectrum was measured from the selected area of (A). AgNP indicates silver nanoparticle; EDX, X-ray diffractor; SEM, scanning electron microscopy.
Antimicrobial properties of the silver nanoparticles
Green synthesized AgNps were planned to use as a substitute for conventional antibiotics to fight against intracellular pathogenic bacteria, especially for the drug-resistant bacteria.
39
Figure 7 illustrates the antimicrobial activity against
Measured antimicrobial activity of
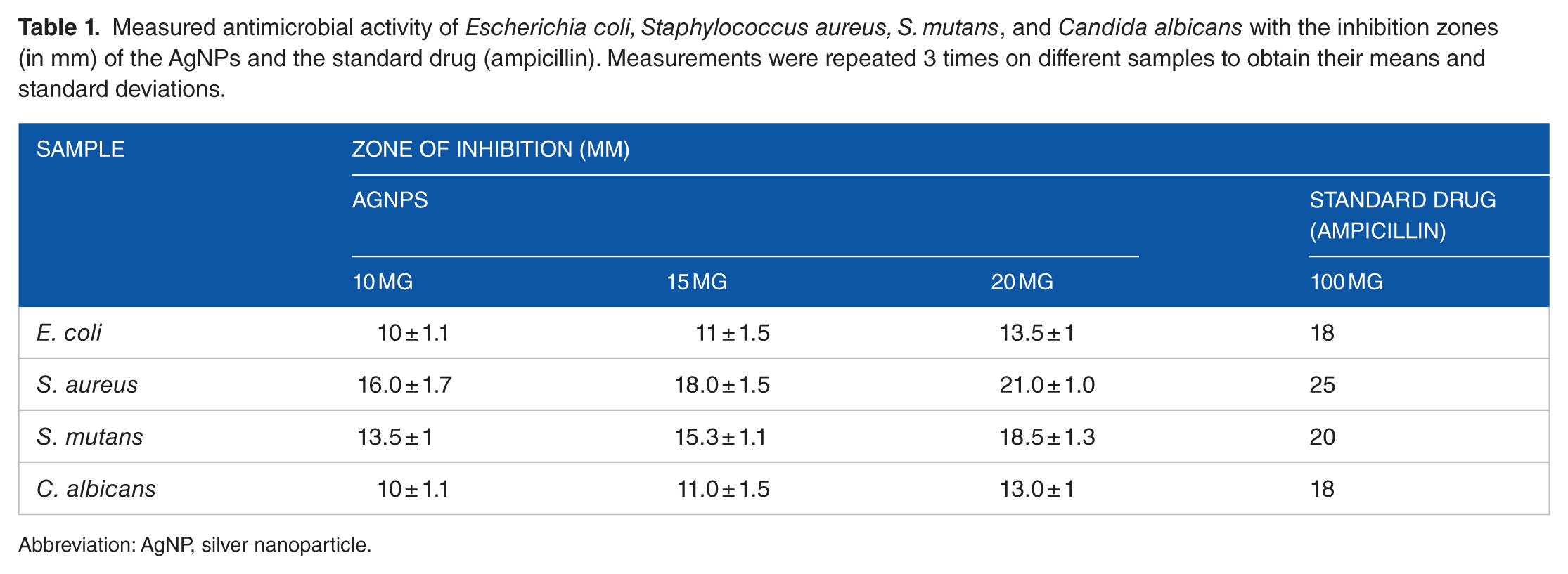
Abbreviation: AgNP, silver nanoparticle.
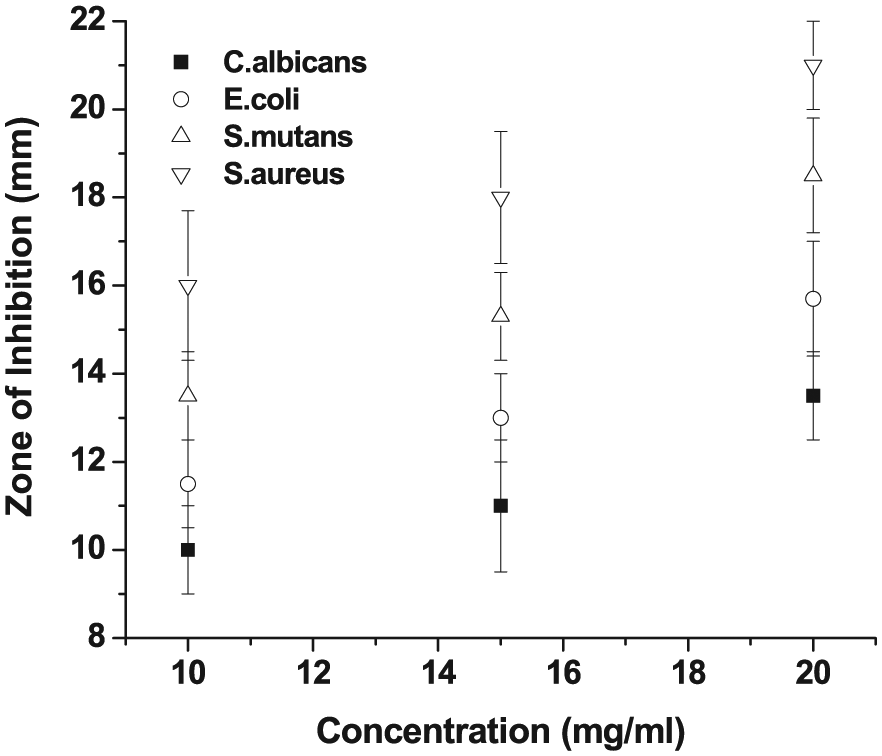
Antimicrobial activity of
Radical scavenging ability of silver nanoparticles
Neutralizing free radicals and other oxidants causes harmful effects to the human body because it consists of complex system of natural enzymatic and non-enzymatic antioxidant resistances.23–25 The free radicals are responsible for initiating various diseases like Parkinson disease, neural disorder, mild cognitive impairment, and aging.21,27 These free radicals can be protected by the intake of necessary dietary antioxidants. 29 The antioxidant intake improves the quality of life by preventing the occurrence of various degenerative diseases cost-effectively. 22 Antioxidant properties of Db extract and synthesized AgNPs was assessed against various free radicals such as DPPH, Hydrogen peroxide, superoxide and compared the same with standards, α-Tocopherol, and Ascorbic acid (Figure 8A to C). All the assays confirmed significant antioxidant activity of green synthesized AgNPs which indicates the direct role of secondary metabolites, namely, phenolic compounds, terpenoids, and so on in removing exposed radicals. DPPH radical solution which showed a deep purple color with maximum absorbance at 517 nm turns into yellow color when it accepts electron. Discoloration of DPPH started after adding AgNPs due to the antioxidant ability; radical scavenging activity of the Db extract increased with concentration from 1 to 50 µg/mL in the percentage of inhibition ranging from 27.9% ± 2.1% to 50.12% ± 2.37% (Figure 8A). The AgNPs exhibited the highest percentage of inhibition of 44.41% ± 2.3% even with the lowest concentration of 1 µg/mL. Nevertheless, the lowest concentration of the AgNPs (1 µg/mL) exhibited the highest percentage of inhibition, which is similar to that of the 50 µg/mL Db extract. Hydrogen peroxide is the most commonly generated reactive oxygen species inside the cell and leads to the formation of in vivo hydroxyl radical (OH*). In this method, test samples decayed hydrogen peroxide on incubation and the same was spectrophotometrically measured at 230 nm (Figure 8B). The order of the antioxidant activity improved with increase in the concentration but varied between the samples (ascorbic acid > AgNPs > α-Tocopherol > Db extract). Generation of superoxide radical through addition of NaOH to DMSO is another standard and highly accepted method to assess the inhibition of reactive oxygen species. Silver nanoparticles showed better superoxide scavenger capability than α-Tocopherol and very close to the activity of ascorbic acid in inhibiting the formation of a red dye formazan as shown in Figure 8C. Nanoparticles showed excellent antioxidant activity compared with that of the Db extract or other medicinal plants.46,47
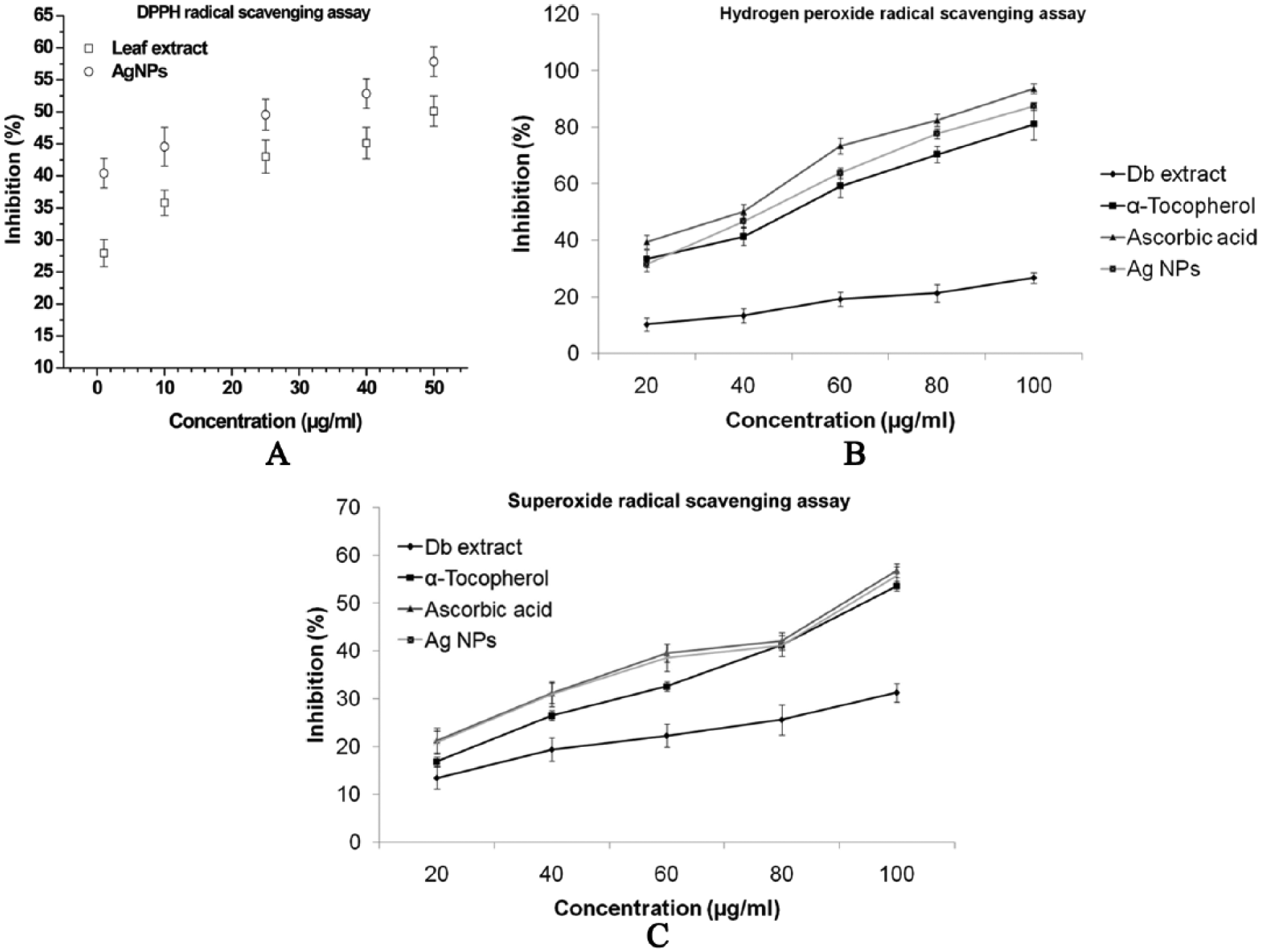
Antioxidant activity of the
In vitro cell viability assay
Toxic effects of AgNPs at 0, 1, 2, 4, 8, 16, 32, 64, and 128 μg/mL concentration for HepG2 cells was determined using MTT assay. Results have confirmed that nanoparticles up to the concentration of 128 μg/mL observed a significant decrease in viability of HepG2 cells (
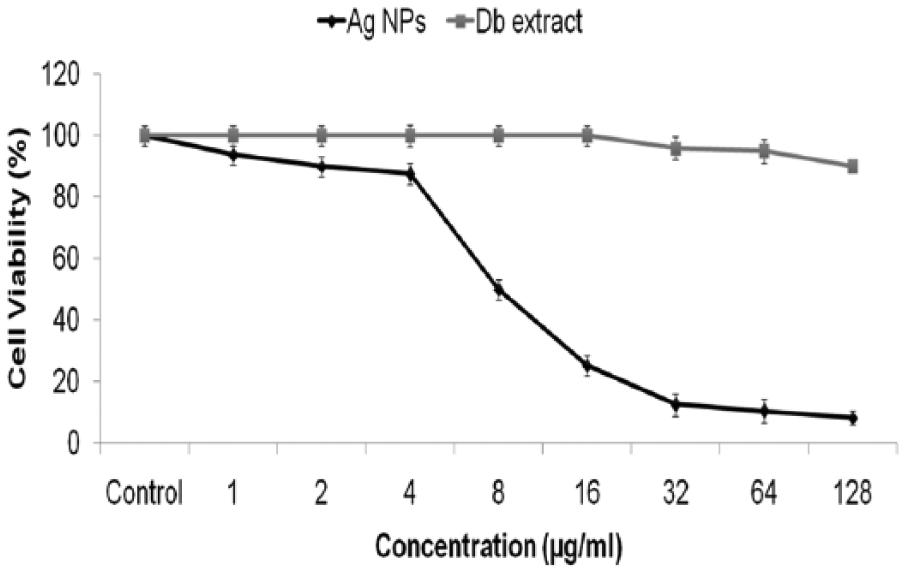
In vitro cell proliferation assay of Hep cells treated with AgNPs and
Conclusions
This work describes synthesizing of silver nanoparticles by exposing them to
Footnotes
Acknowledgements
Authors would like to thank the management, Vignan’s Foundation for Science, Technology & Research, India, for providing administrative and research support.
Funding:
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests:
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Author Contributions
SRG conceived the idea and designed the methodology. SRG, VRD, NSSK, MMH performed the assays. VRD and SRG analyzed and interpreted the results. VRD gave critical comments. SRG and VRD prepared the final draft.
