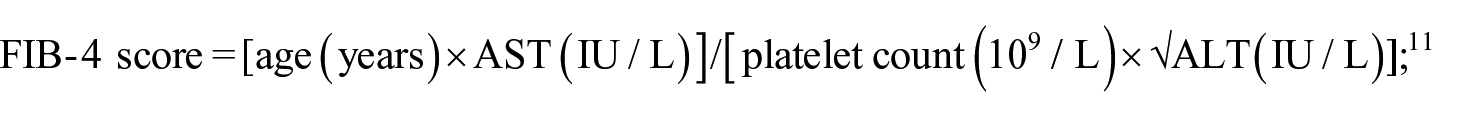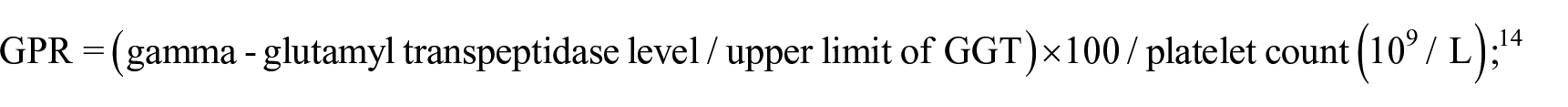Abstract
Background:
Noninvasive and cost-effective markers are needed to replace esophagogastroduodenoscopy in the screening for severe esophagogastric varices (EGVs) and portal hypertensive gastropathy (PHG).
Objective:
This study evaluated the performances of several commonly used fibrosis markers in assessing EGVs and PHG in cirrhosis patients.
Design:
Retrospective cohort study.
Methods:
A series of 323 patients with cirrhosis were consecutively enrolled and endoscopically followed up until variceal eradication was achieved. The Fibrosis-4 (FIB-4) score, albumin–bilirubin (ALBI) index, aspartate aminotransferase (AST)-to-alanine aminotransferase (ALT) ratio (AAR), AST-to-platelet ratio index (APRI), gamma-glutamyl transpeptidase-to-platelet ratio (GPR), and Lok score were calculated for each patient upon first admission. The performances of these markers in assessing EGVs and PHG were determined.
Results:
In the screening for clinically relevant esophageal varices (CREVs), none of the markers showed a significant ability to differentiate CREVs from non-CREVs (
Conclusion:
The studied markers revealed poor to no ability to assess EGVs or PHG. Hence, they cannot be used to substitute EGD in the screening for EGVs. Furthermore, endoscopic eradication of EGVs did not affect the severity of PHG.
Introduction
Portal hypertension is the root cause of major end-stage liver cirrhosis complications, such as esophagogastric varices (EGVs) and portal hypertension gastropathy (PHG), which are responsible for upper GI bleeding in the majority of cirrhosis patients. Despite the significant advances observed recently in the management of gastrointestinal bleeding, up to 30% of the first EGV-related bleeding is still fatal.1,2 The Baveno VI consensus recommends that esophagogastroduodenoscopy (EGD) screening be performed in every cirrhosis patient to assess for EGVs. For those without varices at the initial screening, EGD should be performed at intervals of 2 to 3 years, whereas those with small varices should be screened at 1- to 2-year intervals. 3 However, owing to the invasive nature of EGD and its risk of causing bleeding, many patients are reluctant to comply with frequent periodical screening management. From this perspective, many studies have suggested noninvasive methods, such as platelet counts, the spleen diameter, Child‒Pugh classification, FibroTest, FibroScan, and CT scan, either alone or in combination, to screen for cirrhosis patients who are at high risk of bleeding.4 -6 However, these techniques are not routinely available in every facility, and some are not cost-effective. The FIB-4 score, APRI, ALBI index, AAR, GPR, and Lok score are few commonly used biomarkers for detecting liver fibrosis and its severity. These markers are composed of basic laboratory parameters such as the platelet count, age, alanine transaminase, aspartate aminotransferase, and gamma-glutamyl transpeptidase levels, serum albumin level, bilirubin level, etc. Since the severities of EGVs and PHG correlate with the severity of fibrosis, we hypothetically assumed that fibrosis biomarkers could reflect the severities of EGVs and PHG in patients with liver cirrhosis. Thus, the aim of this study was to evaluate the performances of these markers in assessing the presence of EGVs and PHG and the correlation between the 2 entities.
Patients and Methods
Study Design
This was a single-center retrospective cohort study conducted at the Third Affiliated Hospital of Sun Yat-sen University, China. A series of cirrhosis patients were consecutively enrolled, regardless of the etiology, and followed up through endoscopic procedures, including if needed, variceal banding and/or sclerosing agent injection, until complete variceal eradication was attained. The aim of this study was to investigate some easily accessible serum markers that are commonly used in clinical practice for their accuracy in assessing the severities of esophagogastric varices and portal hypertension gastropathy (PHG) and, finally, examine whether endoscopic variceal eradication affects the severity of PHG. Information such as patient age, sex, etiology of cirrhosis, laboratory results, and esophagogastroduodenoscopy results was obtained. This study was conducted in accordance with the Declaration of Helsinki and was approved by the ethics board of the Third Affiliated Hospital of Sun Yat-Sen University (registration number: [2021]02-176).
Inclusion and Exclusion Criteria
Patients aged 18 years and above who were diagnosed with cirrhosis on the basis of either clinical presentations or liver biopsy findings at our hospital from January 2011 to December 2020 and who received sequential endoscopic variceal treatment were initially enrolled in this study. Those with active gastrointestinal bleeding, acute or chronic liver failure, hepatocellular carcinoma, and other etiologies of thrombocytopenia; patients with incomplete data; and those with a history of portal hypertension interventions such as splenectomy, a transjugular intrahepatic portosystemic shunt (TIPS), or balloon-occluded retrograde transvenous obliteration (BRTO) were excluded from this study.
Endoscopic Study
Esophagogastroduodenoscopy was performed on each patient by 2 qualified endoscopists at our center. Each patient was assessed for the presence of esophagogastric varices and PHG, initially before the first endoscopic treatment and subsequently throughout the sequential endoscopic treatments. The last recorded endoscopy was 4 weeks after complete eradication of the varices was achieved. The frequency and number of treatments needed were decided by the endoscopists on the basis of the severity of each patient’s varices. The Japanese grading criteria for varices were used to record their severity. 7 On the basis of the Baveno I consensus recommended classification, we stratified varices into small varices (grade 1, also referred to as nonclinically relevant varices) and large varices (grade ⩾ 2, also referred to as clinically relevant varices). 8 Similarly, PHG was classified as mild or severe, as recommended by the Baveno consensus. 9 Figure 1 illustrates the endoscopic presentations of a patient before and after the eradication of varices. Moreover, except for 1 patient who discontinued the use of a nonselective beta blocker because of symptomatic bradycardia, all the other patients included in this study were put on a nonselective beta blocker as a treatment to decrease portal hypertension.

Endoscopic images of a patient before and after eradication of varices. (A) shows severe esophageal varices, with color blue, large-sided varices occupying more than one-third of the esophageal lumen, with positive red color sign. (B) shows an endoscopic variceal ligation of the esophageal varices. (C) shows scarred esophagus after eradication of the varices (post sequential EVL presentation). (D) shows a large gastroesophageal varices extending below the gastroesophageal junction along the lesser curvature of the stomach, mild portal hypertension gastropathy (PHG) could be observed (mild reddening, congestive mucosa, with no mosaic-like pattern). (E) shows tissue adhesive injection of the gastric varices. (F) shows post-tissue adhesive injection hyperplasia of the mucosa, with mild PHG.
Non-invasive Markers and Diagnosis Criteria
The following markers, which were generated and validated in previous studies to assess liver fibrosis, were calculated for each participant: the Fibrosis-4 (FIB-4) score, the AST-to-platelet ratio index (APRI), the albumin‒bilirubin (ALBI) index, the aspartate aminotransferase-to-alanine aminotransferase ratio (AAR), the gamma-glutamyl transpeptidase-to-platelet ratio (GPR), and the Lok score. 10 Blood samples were obtained 24 to 48 hours prior to the endoscopy procedures.
Liver cirrhosis was clinically diagnosed in patients with clinical features of esophagogastric varices, ascites or hepatic encephalopathy. Furthermore, cirrhosis was confirmed through imaging findings, such as a coarse parenchyma with shrinkage or enlargement of the liver, combined with laboratory findings, such as a low platelet count, low serum albumin level, high bilirubin level and/or prolonged prothrombin time. 16 In the case of uncertainty, liver biopsy was required to confirm the presence of cirrhosis. Additional information on the diagnosis of the main etiologies of cirrhosis is reported in the Supplemental Document.
Statistical Analysis
The detailed calculation of the sample size used in this study is elaborated in the Supplemental Document. Continuous variables are expressed as medians with 25th or 75th percentiles or means ± standard deviations, as appropriate, whereas categorical variables are expressed as numbers and percentages. A
Results
Demographic and Laboratory Characteristics of the Cohort
The demographic and laboratory characteristics of the study population are summarized in Table 1. A total of 323 cirrhosis patients with a mean age of 49.52 ± 11.1 years were enrolled in this study, among whom 256 (79.3%) were males. The main etiology of cirrhosis was viral infection (74.9%), followed by alcohol-induced liver disease and other less frequent etiologies. Three hundred eight (95.4%) patients were initially diagnosed with clinically significant esophageal varices, defined as Grade II or Grade III esophageal varices on upper gastrointestinal endoscopy, whereas 248 (76.8%) patients were diagnosed with clinically significant gastric varices. Severe portal hypertension gastropathy (PHG) was initially observed in 56.0% of the patients. After eradication of the varices, mild and severe PHG was observed in 45.5% and 54.5% of the patients, respectively.
Demographic and laboratory characteristics of the study population.
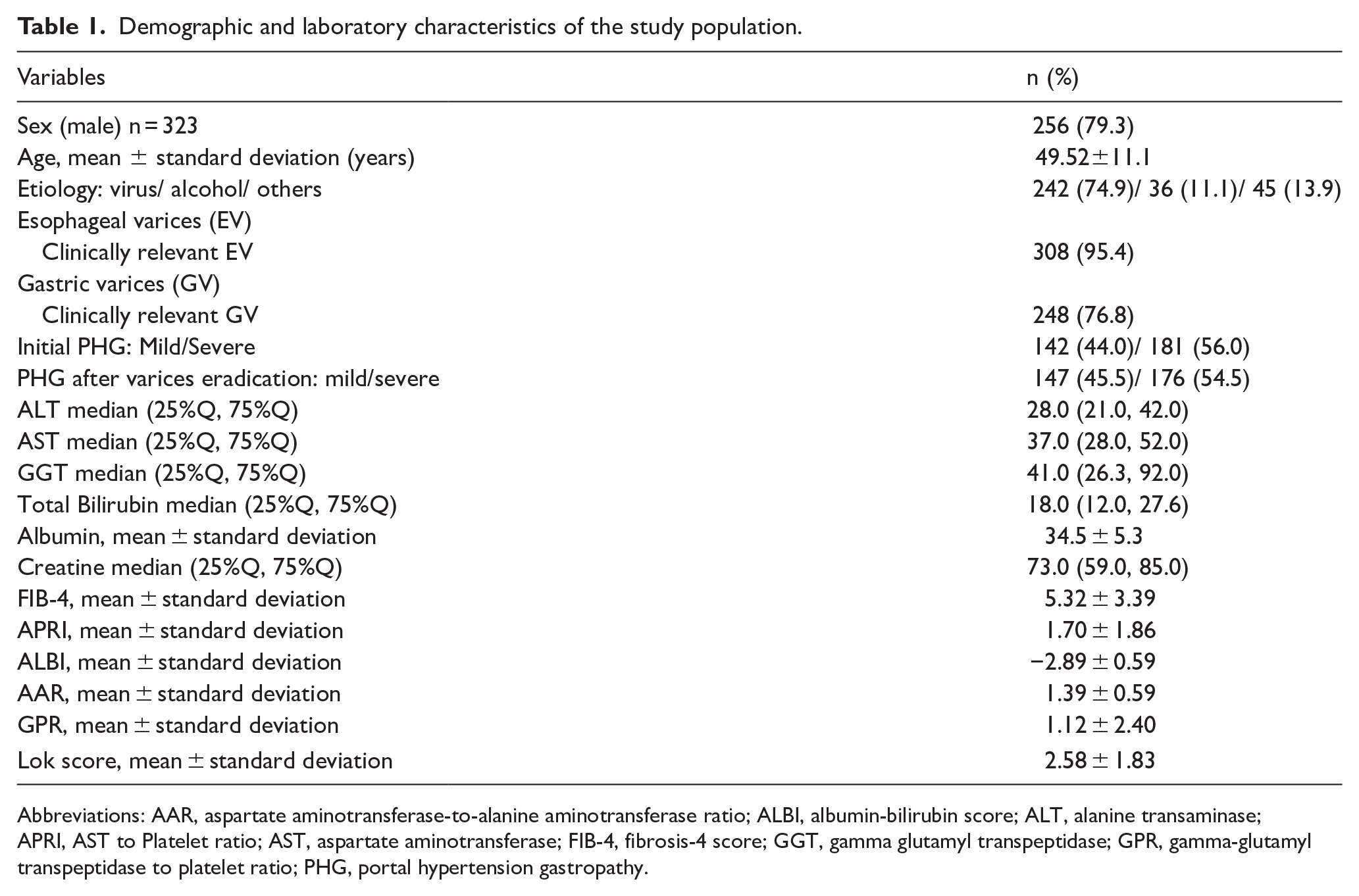
Abbreviations: AAR, aspartate aminotransferase-to-alanine aminotransferase ratio; ALBI, albumin-bilirubin score; ALT, alanine transaminase; APRI, AST to Platelet ratio; AST, aspartate aminotransferase; FIB-4, fibrosis-4 score; GGT, gamma glutamyl transpeptidase; GPR, gamma-glutamyl transpeptidase to platelet ratio; PHG, portal hypertension gastropathy.
Performances of Different Markers in Grading the Severity of Esophageal Varices (EVs)
Based on their severity, esophageal varices were stratified into clinically relevant EVs (CREVs) and nonclinically relevant EVs (non-CREVs). As shown in Figure 2 and Supplemental Table 1, the differences in the mean or median FIB-4 scores, APRIs, ALBI indices, AARs, GPRs, and Lok scores were not statistically significant (all

Biomarkers level in different esophageal varices severity subgroups. (A) shows comparison of different biomarkers level between clinically relevant esophageal (CREV) varices and non CREV; (B, left) shows an example of CREV at the lower esophageal segment, presenting as large varices with positive red color sign. (B, right), shows an example of non-CREV at the lower esophageal segment, presenting as small (<1 cm) linear varices, with no red color sign.
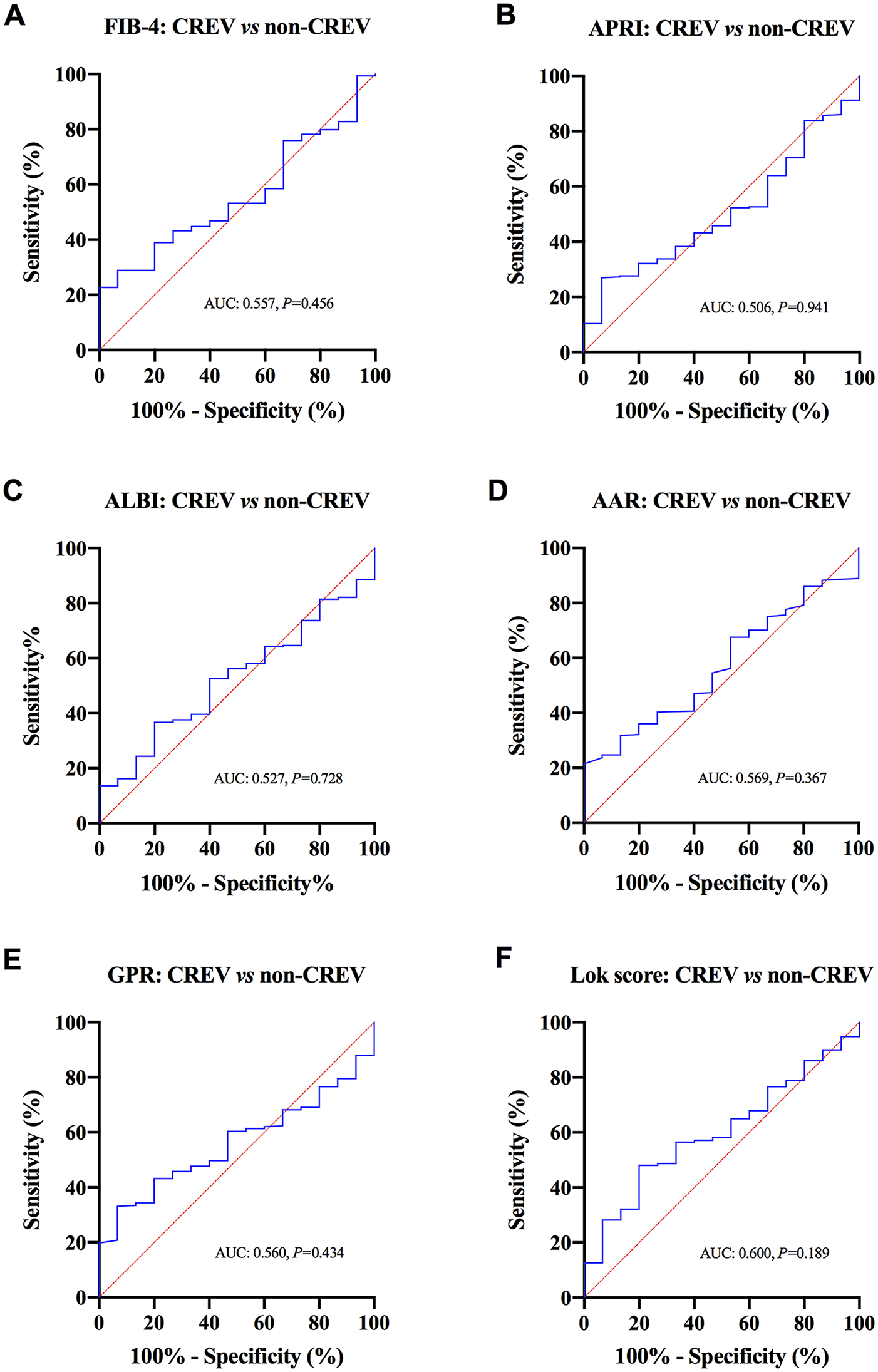
Receiver operating characteristic (ROC) curve analysis of the markers in differentiating clinically relevant esophageal varices (CREV) from non-CREV. (A-F) respectively showcases the ROC curve of FIB-4, APRI, ALBI, AAR, GPR, and Lok score.
Performances of Different Markers in Grading the Severity of Gastric Varices (GVs)
As illustrated in Figure 4, when the clinically relevant gastric varix (CRGV) group was compared with the nonclinically relevant gastric varix (non-CRGV) group, only the median AARs [1.3 (1.0, 1.6) vs 1.4 (1.1, 1.9),

Biomarkers level in different gastric varices severity subgroups. (A) shows comparison of different biomarkers level between clinically relevant gastric varices (CRGV) and non CRGV; (B, left) shows an example of CRGV presenting as tumorous color blue varices, with positive red color sign; (B, right) shows an example of non CRGV presenting as small tortuous varices with no red color sign.
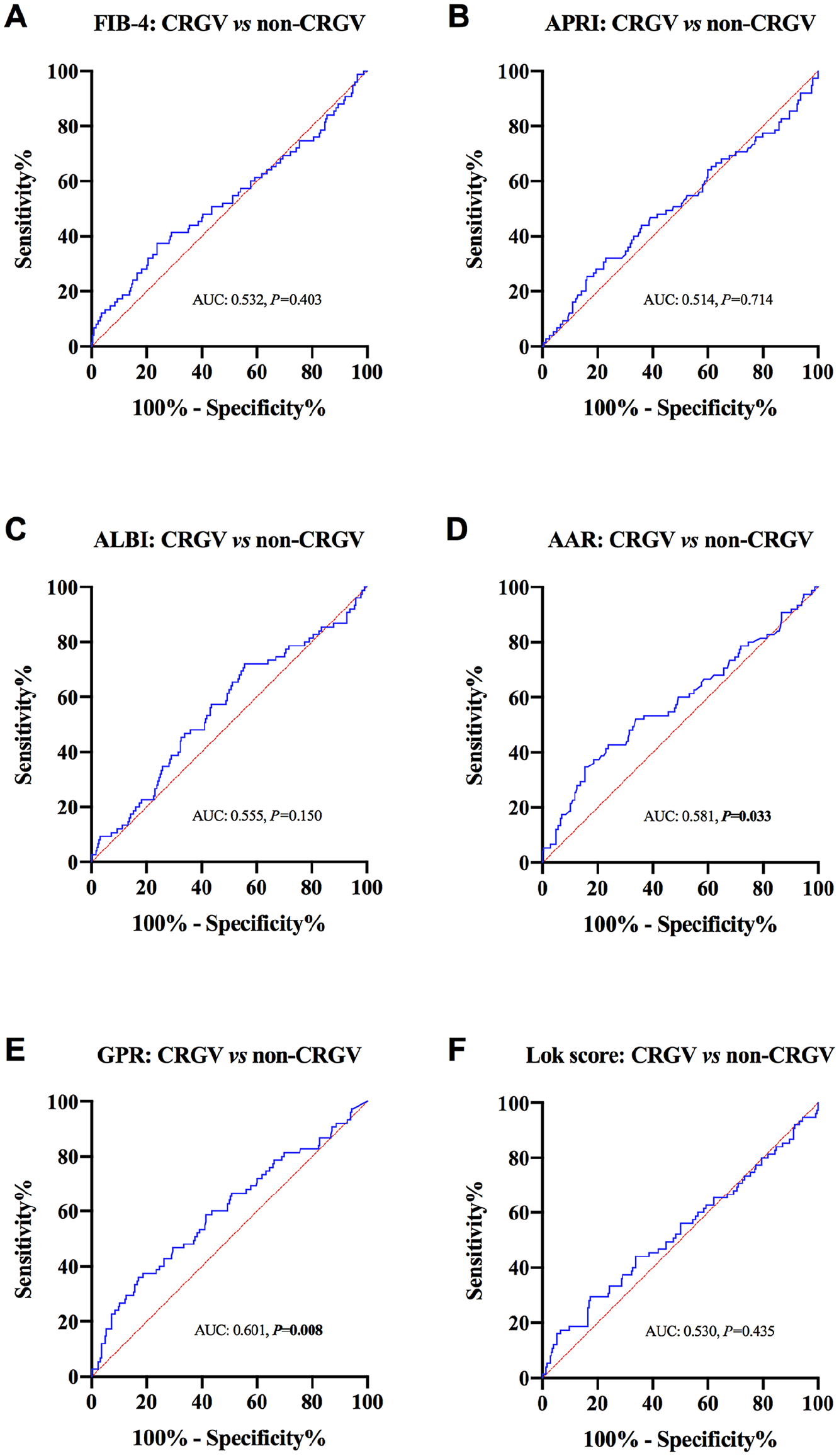
Receiver operating characteristic (ROC) curve analysis of the markers in differentiating clinically relevant gastric varices (CRGV) from non-CRGV. (A-F) respectively showcases the ROC curve of FIB-4, APRI, ALBI, AAR, GPR, and Lok score.
Marker Levels in Different PHG Severity Groups
In this study, the patients were also divided into 2 groups according to the severity of their portal hypertension gastropathy (mild or severe PHG). As depicted in Figure 6, only the median FIB-4 scores were significantly different between the 2 groups as follows: mild PHG: 3.8 (2.7, 6.2) versus severe PHG: 4.9 (3.3, 7.1),

Biomarkers level in different portal hypertensive gastropathy severity groups before endoscopic treatment. (A) shows comparison of different biomarkers level between mild and severe portal hypertensive gastropathy (PHG). (B, left) shows mild PHG, presenting as isolated mosaic pattern with no red markings; (B, right) shows severe PHG, presenting as diffuse mosaic pattern with cherry red spots.
Relationship Between the Severities of PHG and Varices
Chi-square analysis between the severities of PHG and varices revealed no difference in the severity of esophageal varices between the mild and severe PHG groups (
Relation between initial PHG and esophageal varices.

Abbreviations: CREV, clinically relevant esophageal varices; PHG, portal hypertensive gastropathy.
Relation between initial PHG and gastric varices.

Abbreviations: CRGV, clinically relevant gastric varices; PHG, portal hypertensive gastropathy.
Discussion
Early detection of large esophagogastric varices and severe PHG through noninvasive methods is highly important because of the reluctance of patients to use EGD as a screening tool. 17 Several biomarkers, such as transient elastography, the FIB-4 score, the Forns index, the Lok score, the King score, the APRI, the AAR, the Fibro index score, have been proposed in the past to predict large varices in cirrhosis patients.11,18 -20 In this study, the included markers not only failed to distinguish the diverse severities of esophageal varices but also the diverse severities of PHG. However, the AAR and GPR had moderate capacities to distinguish between severe and mild gastric varices. Furthermore, no correlation was found between PHG and EGV severities, and the eradication of EGVs did not affect PHG in cirrhosis patients.
This study focused mainly on noninvasive markers that are composed of routinely available laboratory indicators, such as the blood platelet count, AST, ALT, GGT, albumin, and bilirubin levels, and the INR. These markers were studied on various spectra to assess their abilities to screen for severe EGVs and severe PHG, which are together responsible for the majority of gastrointestinal bleeding in cirrhosis patients.21 -24
Previous studies have revealed that the severity of EGVs is correlated with the degree of portal hypertension.9,25 Similarly, the severity of PHG has been linked in several studies to the severity of portal hypertension.26 -28 In cirrhosis-induced portal hypertension, increased splanchnic inflow results in splenomegaly and subsequent thrombocytopenia due to the splenic pooling of platelets. 29 Moreover, severe portal hypertension (PH) disrupts the normal function of the liver, which manifests as hypoalbuminemia, a long prothrombin time, a high INR, and hyperbilirubinemia. In some cases, severe PH in combination with the primary etiology of cirrhosis leads to damage to hepatocytes, which can be assessed by high levels of liver transaminases and GGT. Consequently, some of these biomarkers have been used to predict portal hypertension features, such as esophageal varices and variceal bleeding.
Hassan
Conclusions
In summary, the FIB-4 score, APRI, ALBI index, ARR, GPR, and Lok score, which are commonly used to identify cirrhosis, have poor abilities to assess the severities of esophagogastric varices and PHG. These markers alone cannot substitute for EGD in the screening for EGVs and PHG. Furthermore, endoscopic eradication of EGVs did not affect PHG. This study has the merit of being among the few studies that assessed the prediction of PHG by using noninvasive markers. The main limitations of our study lie in its retrospective nature and the small sample size, resulting in bias that could affect the accuracy of the results. Larger prospective multicentre studies are needed to confirm our findings.
Supplemental Material
sj-docx-1-bmi-10.1177_11772719251339185 – Supplemental material for Poor Abilities of Noninvasive Biomarkers to Assess Esophagogastric Varices and Portal Hypertensive Gastropathy
Supplemental material, sj-docx-1-bmi-10.1177_11772719251339185 for Poor Abilities of Noninvasive Biomarkers to Assess Esophagogastric Varices and Portal Hypertensive Gastropathy by Kodjo Kunale Abassa, Xiaoning Guo, Shuyan Tan, Zhiling Liang and Siwei Tan in Biomarker Insights
Footnotes
List of Abbreviations
AAR: aspartate aminotransferase (AST)-to-alanine aminotransferase (ALT) ratio
ALBI: albumin-bilirubin index
APRI: aspartate aminotransferase-to-Platelet ratio
AUC: area under the curve
CREV: clinically relevant esophageal varices
CRGV: clinically relevant gastric varices
EGD: esophagogastroduodenoscopy
EGV: esophago-gastric varices
FIB-4: Fibrosis-4 score
GPR: gamma-glutamyl transpeptidase to platelet ratio
PHG: portal hypertensive gastropathy
Ethical Considerations
This study was conducted according to the Declaration of Helsinki, and approved by the ethic board of the Third Affiliated Hospital of Sun Yat-Sen University (registration number: [2021]02-176).
Consent to Participate
All patients gave written informed consent to participate.
Consent for Publication
All participating patients provided written informed consent, which included consent for publication of results.
Author Contributions
Kodjo Kunale Abassa: Conceptualization; Data curation; Formal analysis; Investigation; Methodology; Resources; Software; Validation; Writing—original draft. Xiaoning Guo: Conceptualization; Data curation; Formal analysis; Investigation; Methodology; Resources; Validation; Writing—original draft. Shuyan Tan: Conceptualization; Data curation; Methodology; Resources; Validation. Zhiling Liang: Conceptualization; Data curation; Methodology; Resources; Validation. Siwei Tan: Conceptualization; Formal analysis; Investigation; Methodology; Resources; Software; Validation; Writing—original draft; Writing—review & editing.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The National Natural Science Foundation of China (82170569); the Science and Technology Planning Projects of Guangzhou City (2024A04J6565); the Natural Science Foundation of Guangdong Province (2022A1515012546; 2023A1515011204).
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
Please contact the corresponding author for available data.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.