Abstract
Bladder cancer is one of the most frequently diagnosed cancers in men. While cystectomy remains the primary treatment, advances in radiotherapy and chemotherapy have highlighted the value of bladder-preserving strategies, which can also enhance patients’ quality of life. Despise these advances, around 20% of patients may still require salvage cystectomy due to tumor radioresistance. This underscores the need to develop radiosensitivity predictive assays. Radiotherapy acts by inducing DNA damage, primarily through DNA double-strand breaks, which can significantly affect treatment outcomes if left unrepaired. In addition to activating DNA repair pathways, the response to radiation also involves the tumor microenvironment, cell death pathways, immune responses and different types of cell death and proliferation receptors. In recent years, personalized medicine, which tailors treatments to individual patients, has gained increasing attention in cancer care. The development of chemo- and radiosensitivity predictive assays has become a key focus of cancer research. Despite the potential impact of such assays on bladder cancer treatment, there is still no reliable test that can help clinicians and informs patients in choosing the best treatment. This review aims to highlight studies that attempted to characterize bladder cancer radiosensitivity and to discuss the potential biomarkers that could be used to develop bladder cancer radiosensitivity predictive assays.
Introduction
Bladder cancer (BC) is one of the most common cancers worldwide, particularly among males. In 2022, it was the sixth most diagnosed cancer in this group, with 471 293 new cases, and the ninth most fatal, resulting in over 165 672 deaths. 1 This malignant disease is categorized into non-muscle invasive (NMIBC) and muscle-invasive (MIBC), with the latter requiring aggressive treatments.
Radical cystectomy (RC), whether open or robotic, combined with bilateral pelvic lymph node dissection, remains the standard treatment for MIBC. However, this approach significantly reduces quality of life, leading to reduced bladder capacity, testosterone depletion, sexual dysfunction, bowel incontinence, and increased risks of colon and colorectal cancers, as well as chronic kidney.3,4 Therefore, a bladder-preserving strategy was developed. It is based on maximal transurethral removal of bladder tumor (TURBT) followed by the combination of radiotherapy (RT) and chemotherapy (CT).2-4
This highlights the need to both radiobiologically characterize MIBC and develop MIBC-specific radiosensitivity predictive assays.5,6
In recent years, many studies have attempted to enhance personalized cancer therapy by focusing on molecular and cellular signatures. Assays that evaluate tumor response are already in use is some cases, such as those applied in the preselection of patients for immunotherapy or some types of CT in breast cancer treatment.7,8 However, assays for radiosensitivity prediction remain limited and under development.9,10 These existing tests examine both tumor and normal tissue toxicities, with those developed for tumors targeting cancers for which RT is a primary treatment option.11-13 In this review, we explore the different mechanisms activated following exposure to ionizing radiation, as well as highlight the corresponding biomarkers with known BC radiosensitivity predictive capabilities.
Bladder Cancer Diagnosis and Treatments
Diagnosis of BC is primarily executed by cystoscopy and cytology, which are considered the first-line approaches for the detection of this disease. 14 Additional diagnostic modalities feature biopsy and imaging techniques such as magnetic resonance imaging (MRI), computed tomography scan (CT scan), positron emission tomography (PET) and PET-computed tomography (PET-CT) scans, ultrasonography and radiographic techniques. 15 Being an invasive procedure, cystoscopy has limited sensitivity for detecting carcinoma in situ. Also, urine cytology, a non-invasive and highly specific procedure, lacks sensitivity for low-grade urothelial tumors. 16 As substitutes for these diagnostic tools, non-invasive and highly specific detection tests based on molecular biomarkers for BC are being explored as new diagnostic approaches.17,18 In their study, Harsanyi et al identified several promising biomarkers that significantly enhance BC diagnostics through proteomic and glycomic analyses. Based on their sensitivity (SN) and specificity (SP) thresholds, biomarkers such as BLCA-4 and HTRA1, demonstrated high SN/SP thresholds of 90% indicating their effectiveness in detecting and monitoring BC. 19
BC is a heterogeneous malignant condition, and its treatment modalities also show much variation depending on patient characteristics and other factors such as cancer stage. Broadly, the treatment modalities can be classified into 3 categories: surgery, CT, and RT. These treatment modalities can be used independently or in combination with each other, termed a multimodality treatment approach. 20
Based on the presence of muscle invasion, there are 2 major stages of BC: NMIBC and MIBC. NMIBC holds a more favorable prognosis than MIBC. In this context, BC can also be categorized according to the clinical or histopathological characteristics of the tumor: Ta, T1, T2, T3, and T4. NMIBC stages comprise Ta, T1, and Tis, while T2, T3, and T4 belong to MIBC. 21
Non-muscle invasive bladder cancer treatments
The bladder wall consists of 4 layers, the urothelium, the lamina propria, the muscularis propria (or detrusor muscle) and the adventitia. Bladder carcinomas, accounting for approximately 90% of all BCs, originate in the bladder’s inner lining, the urothelium. 22 It is made up of 3 to 7 layers of stratified transitional cells. Its outermost layer is composed of umbrella cells with a distinct morphology and role in maintaining the bladder epithelium’s barrier function. 23 The adoption of specific BC treatment strategies depends on the severity of the cancer, including its type and stage, as well as the condition of the patient. 24
NMIBC is stratified to 3 main risk groups: low risk, intermediate risk and high risk. There is an additional very high-risk sub-category. 25 High-risk patients diagnosed with NMIBC face significant prognostic challenges, including high rates of recurrence and progression. The criteria for identifying high-risk NMIBC have been standardized and adopted by the European Association of Urology (EAU) and the American Urological Association (AUA) guidelines. High-risk features include tumors classified as T1 stage and/or high-grade. Additional criteria encompass having more than 3 tumors, tumor size exceeding 3 cm, the presence of carcinoma in situ (CIS), and/or a history of recurrence within 3 months. Variant histologies are excluded from these criteria. 26
The first line of treatment for NMIBC include intravesical therapy and TURBT. Intravesical therapy involves injecting medicine directly into the bladder to eradicate any remaining cancer cells, while TURBT entails removal of the tumor from the bladder using a cystoscope. 27 Giving CT directly into the bladder soon after TURBT surgery reduces the risk of recurrence. 28 The 2 most frequently utilized CT drugs in intravesical treatment, are mitomycin C and gemcitabine, but the latter has been shown to be more efficient and better tolerated.29,30 Furthermore, several meta-analyses have demonstrated that Bacillus Calmette-Guerin (BCG), administered once a week for 6 weeks after TURBT, is superior to TURBT alone, or TURBT with CT, in preventing recurrences of high-grade Ta and T1 tumors. 31 In recent years, new therapies including immune checkpoint inhibitors (eg, anti-PD-1/PD-L1 therapy) and photodynamic therapy have produced encouraging outcomes in clinical trials (KEYNOTE-057 Trial) and might ultimately be incorporated into the treatment for NMIBC. 32 The KEYNOTE-057 Trial investigated pembrolizumab, an anti-PD-1 therapy that works by inhibiting PD-1 protein which normally suppresses the immune response, allowing cancer cells to evade immune detection. Patients with high-risk NMIBC who were unresponsive to BCG treatment were included and demonstrated a complete response (CR) rate of 41% (95%CI: 32%-51%) at 3 months, with 46% of these responses lasting 12 months or longer. Pembrolizumab was generally tolerable, with manageable side effects. This trial marks an important step toward incorporating immunotherapy into NMIBC treatment protocols. 32
The PD-1 pathway is a widely recognized target for treating both NMIBC and MIBC with several monoclonal antibodies approved for use. Despite this, response rates to these T cell-focused checkpoint inhibitors (CPIs) remain low, indicating a need for alternative therapeutic approaches. 33 CAR-T cells are engineered to target specific tumor antigens, boosting the immune response against cancer cells. 34 Similarly, CAR-NK cells, which naturally recognize and destroy malignant cells, have shown potential due to their cytotoxic activity and fewer adverse effects. 34 These therapies are especially promising in combination with RT, as radiation can increase antigen presentation, making cancer cells more susceptible to immune attack. 35 Emerging data suggest that combining these approaches may help overcome the challenges of the tumor microenvironment (TME), providing more effective treatments for high-risk and severe cases of BC.33,35 CAR-T cell therapy has revolutionized cancer treatment, particularly in hematological where it has shown promising results and even led to complete cures. 35 However, its success in treating solid tumors, including BC, has been limited due to challenges like tumor-antigen heterogeneity, impaired CAR-T cell trafficking, and the immunosuppressive TME. 35 In urinary system tumors, CAR-T cell therapy is feasible due to the presence of tumor-associated antigens like PSCA, PSMA, and EGFRs. 36 As such, BC expresses several tumor-associated antigens, offering potential for CAR-T therapy, but ongoing clinical trials, such as HER2-targeting NCT03740256, have yet to produce significant results specifically for BC. 36
On the other hand, the very high-risk category is characterized by BCG unresponsiveness, variant histologies, lymphovascular invasion and prostatic urethral invasion. The current recommendation is to treat those patients with cystectomy. 25
Muscle invasive bladder cancer treatments
Cystectomy
MIBC is considered more advanced than NMIBC as the cancer spreads to the muscle layer of the bladder. As a result, it typically requires more aggressive treatment options like surgery, CT, or RT. Approximately 20% of newly diagnosed cases of BC are classified as muscle-invasive. 37 In the MIBC setting, the standard therapy is RC with pelvic lymph node dissection, in combination with CT treatment.38,39 However, approximately 50% of patients relapse within 2 years, and experience significant decrease in their quality of life following surgery. 40
For patients who have undergone RC, computed tomography scans of the chest, abdomen, and pelvis should be done every 6 months for the first 3 years, annually up to 5 years, and thereafter only when clinically necessary. Imaging intervals may vary depending on whether lymph node involvement was detected during surgery. In cases of node-positive disease, scans every 3 to 6 months are advisable, while in node-negative cases, intervals of 6 to 12 months are more appropriate. This approach aligns with guidelines from major urological and oncology organizations, which recommend flexible imaging schedules based on individual patient factors.25,41
Additional laboratory tests, including urine cytology, liver and kidney function assessments, and electrolyte checks, every 3 months during the first year, every 6 months in the second and third years, and annually thereafter up to 5 years, should be performed. Metabolic disturbances, such as electrolyte imbalances and acidosis, are common, particularly in patients with chronic kidney disease or obstructive uropathy. Chronic acidosis is also linked to bone health concerns, making regular electrolyte monitoring important for detecting patients who might need pharmacological treatment. 25
Trimodal treatments and bladder preservation
Trimodal therapy (TMT) is a treatment strategy consisting of a maximal TURBT followed by concurrent chemoradiotherapy (CTRT). This approach emerged in the aim of preserving the bladder and postponing cystectomy until the response to bladder-sparing therapy has been evaluated.4,42 After surgery and initial CTRT treatment, a cystoscopy is performed to assess the tumor response. In the case of poor response, the patient undergoes RC followed by adjuvant CT. Otherwise, CTRT is resumed and another cystoscopy is performed to reassess the tumor response. 4 Eligibility for bladder preservation typically includes patients without hydronephrosis, those with fully resected tumors following TURBT, or those with small tumors (<5 cm) without pelvic lymph node involvement or associated in situ. 43 The trimodality approach is a well-tolerated and viable initial alternative for healthy individuals who prefer not to undergo RC.
In TMT, radiation is typically administered as external beam RT at a dose of 60 to 66 Gy following a complete TURBT. RT may be combined with 2 doses of radiosensitizing CT drugs, given during weeks 1 and 4, or administered on a weekly schedule. An alternative approach involves using an induction dose of 40 to 45 Gy after TURBT. In contrast, as it was previously discussed, monotherapy typically consists of RC, the complete removal of the bladder.
A multicenter randomized phase III trial conducted in 2009, with a 10-year-follow-up, provided insights into recommendations for CTRT in MIBC.44,45 In this study, after undergoing TURBT, patients were randomly assigned to receive either RT alone or concurrent CTRT consisting of Fluorouracil (5-FU) and mitomycin. The results at follow-up revealed that the group receiving concurrent CTRT had better control over local disease (63% versus 49%) and a lower rate of cystectomy at the 5-year mark (14% versus 22%). However, there was a higher incidence of grade 3 or 4 toxicity during treatment (36% versus 28%). 45 The preferred CT regimens in TMT include cisplatin, as recommended following the Radiation Therapy Oncology Group (RTOG) trials,46,47 or combinations of drugs like 5-FU and mitomycin.
Additionally, a multicenter retrospective study with 287 patients demonstrated no significant difference in overall survival (OS) (P = .76) or progression-free survival (PFS) (P = .12) between patients receiving bladder-sparing treatment with RT and those undergoing RC. These findings suggest that RT can be an effective alternative to RC for selected patients with MIBC. 48 Despite the failure of some randomized controlled trials to accrue sufficient data (eg, NCT 02716896 and SPARE), combined modality CTRT as an alternative to immediate cystectomy for MIBC is endorsed by international organizations as a viable alternative to immediate cystectomy, including guidelines from the International Consultation on Urologic Diseases-European Association of Urology and the UK’s National Institute for Health and Care Excellence. 49 If a good tumor response is observed, the patient will be able to continue his RT treatment. Patients often favor bladder-sparing treatment (BST) because it is less invasive and generally has manageable side effects. 50
Adopting TMT rather than surgery complemented with either RT alone or CT alone, offers many advantages. Undergoing bladder-preserving treatments significantly improves patients’ quality of life, with side effects that are generally well tolerated.40,51 However, the impact of RT on normal tissues hinders its effectiveness in achieving desired therapeutic outcomes. Radiation exposure triggers a cascade of biological processes such as tissue repair, inflammation, and fibrotic remodeling, which can have long-lasting consequences. These chronic pathological responses pose a significant challenge, particularly in elderly patients diagnosed with BC, as they are more susceptible to tissue toxicity. Side effects resulting from RT can manifest shortly after treatment or even years later, with late toxicity leading to persistent and life-altering changes in the affected tissues. The severity of these side effects and the time it takes for them to manifest can vary greatly among individuals, highlighting the importance of careful consideration and personalized treatment approaches. The side effects can be burning pain during urination, frequent need to urinate, fatigue, blood in the stool or in the urine. In some cases, incontinence, cystitis and nearby nerve damage can also happen.52,53
TMT outcomes remain comparable to those of cystectomy.3,54-56 However, the choice of this treatment is highly dependent on the radiosensitivity of the patient and their tumor. If a CR is not achieved, the patient may undergo salvage cystectomy. 57 According to a systematic review published in 2021, approximately 20% of patients undergoing bladder-sparing treatments require a salvage cystectomy. 50 This highlights the need to characterize MIBC radiobiologically, and to develop MIBC radiosensitivity predictive assays. The use of such BC predictive assays following TURBT can assist both clinicians and patients in making informed decisions, helping to avoid unnecessary side effects and complications (Figure 1).
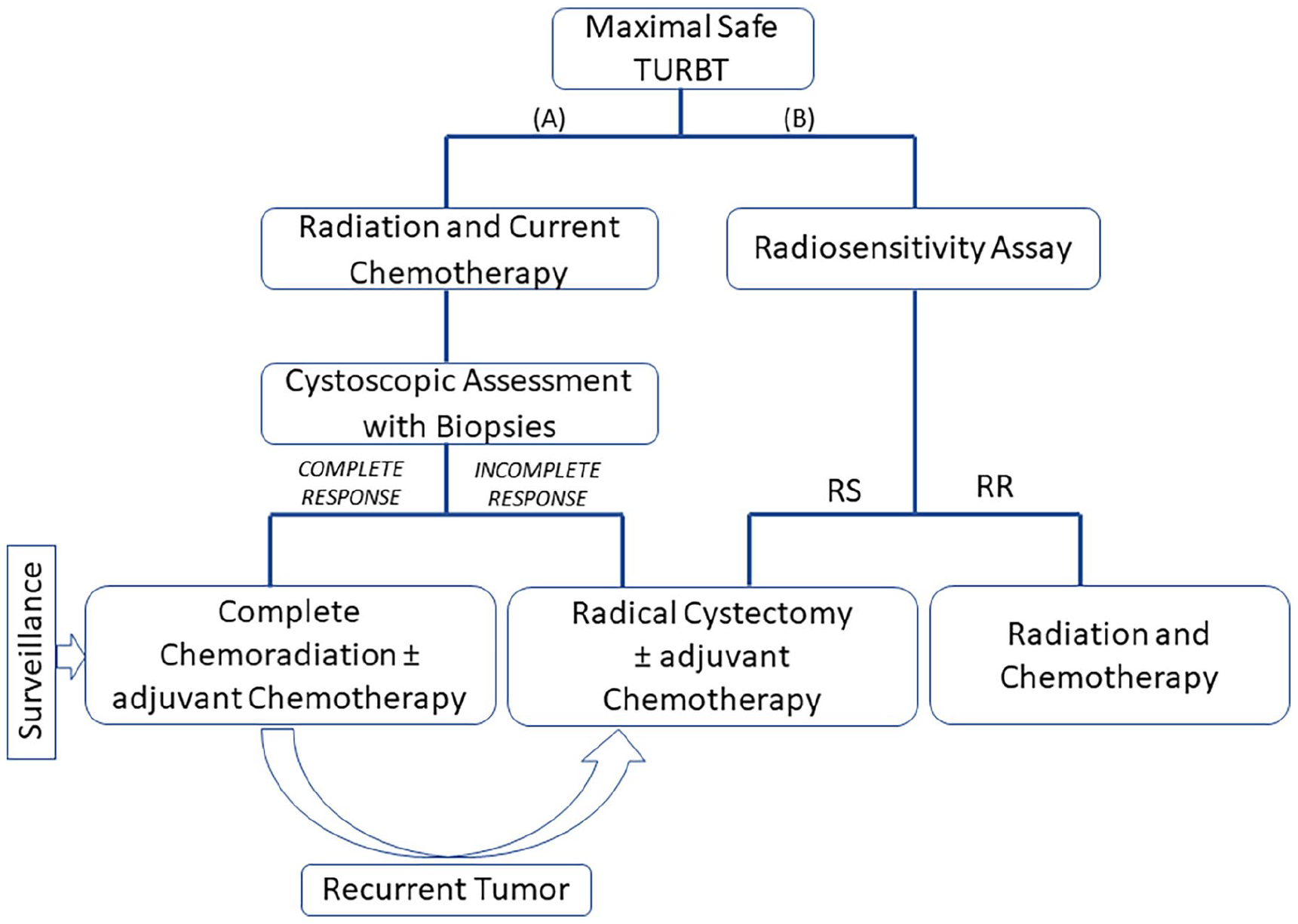
Bladder preservation procedures for the treatment of muscle-invasive bladder cancer (MIBC): (A) current scheme for trimodality treatment of MIBC with selective bladder preservation and (B) personalized treatment strategy using radiosensitivity predictive biomarkers. Abbreviations: TURBT, transurethral removal of bladder tumor; RS, radiosensitivity; RR, radioresistance.
Biological Effects of Ionizing Radiation
Radiation acts by indiscriminately inducing ionizations and excitations in the targeted cells or tissues. The damage induced to the DNA remains the main factor in determining the cell’s fate. In fact, after irradiation, different types of DNA damage are induced, including base damage, DNA single-strand breaks (SSBs) and DNA double-strand breaks (DSBs), with DSBs being the most deleterious type of DNA lesion. 58 The cell activates different mechanisms of DNA repair, depending on the cell cycle phase and the damage type. This includes base excision repair (BER), homologous recombination (HR) or non-homologous end-joining (NHEJ) with the latter being the major mammalian DSB repair pathway. Studies have shown that the capacity of the cell to repair DSBs is directly correlated with cellular and clinical radiosensitivity.59-61
Radiation can also promote apoptosis or senescence, stimulate proliferation by mimicking ligand binding to cell-surface receptors, and coordinate signaling through inflammatory cytokines such as interleukin-1 (IL-1) and tumor necrosis factor (TNF). Furthermore, radiation has immunomodulatory effects that can promote innate and adaptive immune responses. Therefore, the response to radiation is complex and involves a number of genes and proteins that can affect the radiosensitivity of tumors and normal tissues. 9 All these different pathways involve the interplay of biomarkers that can predict BC radioresponse (Tables 1 and 2 and Figure 2)
Clinical studies on bladder cancer radiosensitivity biomarkers.

In vitro studies on bladder cancer radiosensitivity biomarker.


Graphical summary of bladder cancer radiosensitivity biomarkers.
Tumor Microenvironment Challenges
Tumor microenvironment
TME plays a crucial role in tumor growth, progression, and treatment resistance by impairing the immune system’s effectiveness against cancer. It contains the extracellular matrix (ECM), tumor blood vessels, cancer cells, stromal cells (including endothelial cells and fibroblasts), immune cells and various molecules like chemokines and cytokines. 81 Its hallmark features include nutrient deficiency, hypoxia, and solid tumor metabolism. 82
Alterations in the TME consequently result in changes in tumor progression and aggression. In advanced tumor stages, the TME becomes exceptionally complex and varied. Monocytes and macrophages are key components of the TME, essential in tumor initiation and the secretion of pro-inflammatory cytokines, growth factors such as vascular endothelial growth factor (VEGF) and EGF, reactive oxygen species (ROS), and proteases. 83 The effect of RT on both the TME and immune response largely depends on several factors including the delivery and fractionation of radiation doses, as well as the type and location of the tumor. Initially, RT triggers an anti-tumor response, which subsequently leads to chronic inflammation and heightened immune suppression. Over time, pro-tumorigenic macrophages are recruited to the tumor in a dose-dependent manner, creating an immunosuppressive TME that promotes tumor regrowth and resistance. 84
The TME of BC is complex and consists of various cell types which operate under the influence of cancer cells. BC cells alter the TME by secreting soluble factors and extracellular vesicles, which influence surrounding cells. For instance, MB49 cells secrete the chemokine CCL2, which promotes the polarization of tumor-associated macrophages (TAMs) into the M2 phenotype. M2 macrophages support tumor growth and suppress immune responses, especially upon exposure to radiation. This complex interplay suggests that solely targeting cancer cells might not be sufficient for a complete tumor response. In this context, BC stem cells (CSCs) originate from mutations in either stem or differentiated urothelial cells. Dormant CSCs, that evade cell death by CT and RT which typically target rapidly dividing cells, may contribute to BC recurrence and metastasis. Given that BC cells and their interactions with the TME add further layers of complexity to treatment choices and contribute to recurrence, understanding all the factors involved is essential for developing more effective therapies. 85
Inflammatory cytokines
Within the vast complexity of the TME, cytokines are key components. Pro-inflammatory cytokines, especially from the IL-1 family, IL-6, and TNF-α, play an essential role in driving chronic inflammation in tumors. These molecules may exhibit either pro- or anti-tumorigenic effects, that depend on numerous factors, mainly the type of cancer and the TME. They also induce epigenetic changes, which contribute to the upregulation of oncogenes and the downregulation of tumor-suppressor genes, further affecting tumor progression. Within the tumor’s inflammatory environment, macrophages and neutrophils, in conjunction with cytokines, trigger DNA damage by generating ROS and reactive nitrogen species (RNS). 86 Radiation amplifies this process by stimulating a network of interconnected cytokines, adhesion molecules, ROS/RNS, and damage-associated molecular pattern (DAMPs) proteins. This process initiates a self-amplification cascade, fostering a pro-inflammatory and pro-oxidant TME that ultimately results in tumor cell death.
Additionally, RT alters coagulation factor levels, which are part of the innate immune response. Radiation-induced coagulation stimulates the immune system by activating caspases. These enzymes cleave pro-inflammatory cytokines and pro-apoptotic molecules, thereby initiating cell death pathways and generating an extensive immune response. Typically, a radiation dose of 7 to 10 Gy is needed to induce the production of pro-inflammatory cytokines. Conversely, fractionated radiation promotes the persistence of these cytokines within the TME, leading to prolonged upregulation of cytokine gene expression. 87
BC cells contribute to their own growth and metastasis through autocrine signaling pathways, involving the production of tumor-promoting and pro-inflammatory cytokines such as IL-6, TNF, and IL-1. IL-6, a key pro-inflammatory cytokine produced mainly by T-lymphocytes and macrophages, has been particularly linked to increased metastasis in BC. Studies conducted in vitro and in vivo have shown that reducing IL-6 production can inhibit BC progression. IL-6 is also linked to poor prognosis in BC, as its high levels correlate with reduced treatment efficacy, shorter survival times, and higher disease recurrence rates. Similarly, both IL-8 and TNF-α are associated with tumor progression and adverse outcomes. The effects of these cytokines, including IL-6, highlight the critical importance of targeting pro-inflammatory cytokines to potentially enhance treatment efficacy and improve patient outcomes. 88
RT and DNA Damage Repair Biomarkers
After irradiation, DNA damage is induced, and the DNA damage response (DDR) signaling pathway is activated. The DDR either promotes cell cycle arrest and attempts to repair damaged DNA to enable cell survival and replication, or it triggers apoptosis.89,90 DSBs, the most dangerous type of DNA lesions, are repaired by either the NHEJ or HR repair pathways. In eukaryotic cells, NHEJ is the dominant repair pathway of DSBs, as it is the main repair pathway in cells that are in the G0/G1 cell cycle phase, while HR is the main pathway in G2/S, when a sister chromatid is available.91,92
After irradiation, ataxia telangiectasia mutated (ATM) dimers get phosphorylated into phosphorylated ATM (pATM) and monomerized, before transiting to the nucleus in a process called radiation-induced ATM nucleoshuttling (RIANS).61,93 ATM kinase is the major physiological mediator that acts upstream of the signal transduction pathway initiated by ionizing radiation (IR). 94 pATM phosphorylates different target proteins including the histone variant H2AX, at position Ser139.95,96 A cascade of phosphorylation events then follows, based on the cell cycle phase and repair pathway.
HR starts with the recognition of DSBs by MRE11, RAD50 and NBS1, individual proteins forming the MRN complex, along with ATM. The choice between repair pathways, is governed by 2 main proteins, p53-binding protein 1 (53BP1) and BReast CAncer gene 1 (BRCA1). Some studies have shown that BRCA1 can suppress the activity of 53BP1, thereby promoting HR.97,98 Other proteins, such as Bloom syndrome protein (BLM), exonuclease 1 (EXO1), and DNA replication helicase/nuclease (DNA2), are recruited to DSB sites to complete the resection. The exposed single-stranded DNA (ssDNA) is then coated by the replication protein A (RPA) complex, which activates ATM and the ataxia telangiectasia and Rad3 related (ATR) protein, leading to the phosphorylation of checkpoint kinase 1 (Chk1). RAD51 subsequently constructs a displacement loop by locating a homologous sequence and displacing one of its strands. Following strand invasion and synthesis, a Holliday junction is formed, which can be resolved with either a strand crossover or no crossover.
The NHEJ process starts with the monomerization of ATM dimers and the phosphorylation of H2AX into γH2AX on DSB sites. A heteromeric complex consisting of the Ku proteins, Ku70/Ku80, is also recruited to the damage sites to recognize and bind to broken DNA ends with high affinity, thereby promoting DNA end bridging.99,100 The DNA protein kinase catalytic subunit (DNA-PKcs) interacts with the Ku proteins to induce a series of phosphorylation steps that regulate the end-processing activity of NHEJ. 101 This process is carried by exonucleases such as Artemis and the Werner protein (WRN), which also inhibit the activity of HR by blocking MRE11. As some nucleotides may be lost in the end termini, the DNA polymerases μ and λ replace the missing nucleotides. Finally, DNA ligation is completed by a complex formed by XRCC4, DNA ligase IV, and XRCC4-like factor (XLF). 102
Many studies have correlated the expression and kinetics of DDR proteins with tumor and normal tissues radiosensitivity. These include, but are not limited to, pATM, MRE11, BRCA1, BRCA2 and Rad51.94,103-106 In BC specifically, Ananya Choudhury et al demonstrated that high MRE11 expression was associated with improved cause-specific survival (CSS) in 179 MIBC patients treated with radical RT compared to those who underwent cystectomy. 62 This finding suggests that MRE11 may serve as a predictive marker for survival in patients undergoing radical RT. These results are consistent with those of Laurberg et al, who also found that high MRE11 expression is associated with improved CSS in patients treated with radical RT. 63 However, further studies on a larger cohort of MIBC patients are required to confirm the predictive capacity of MRE11 expression. 9 With regard to pATM, no association was found with CSS. Nevertheless, mutations in ATM may predict worse OS. 64 On the other hand, some studies suggest that ATM mutations may lead to a favorable response to RT.107-109 .
In addition to clinical studies, there are several preclinical publications that highlight potential biomarkers for BC radiosensitivity. The alkaline comet assay (ACA) is one such technique that measures and quantifies RT- and CT-induced DNA damage. Many studies have shown the potential of this technique in assessing BC radiosensitivity in vitro,110-112 and many DNA DSB repair proteins were identified as radiosensitivity biomarkers, such as γH2AX, pATM and others. 113
Apart from DSB repair proteins, biomarkers pertinent to other types of DNA damage were also examined. Kawashima et al investigated the impact of the excision repair cross-complementing group 1 protein (ERCC1), a major factor in the nucleotide excision repair (NER) pathway, on in vitro and in vivo RT- and CT-sensitivity. They found that a higher expression of ERCC1 mRNA is correlated with a higher radioresistance. Their results were clinically confirmed by quantifying ERCC1 through IHC in 22 patients with MIBC.65,114 In another study, Sei C. Sak, et al assessed the expression of Apurinic/apyrimidinic endonuclease 1 (APE1) and X-ray repair cross-complementing protein 1 (XRCC1), both involved in the BER pathway, in 90 MIBC patients treated with RT. They suggested that there is a significant correlation between high-level expression of APE1 and XRCC1 and improved CSS among patients. 66
Cell Death: Apoptosis Biomarkers
Apoptosis, also known as programed cell death, is a highly regulated process of cellular destruction. It plays a significant role in eliminating abundant cells during various stages of embryonic development, growth and differentiation. 115 This process maintains tissues homeostasis by regulating the balance between cell proliferation and cell death, thereby ensuring continuous tissue renewal.116,117 Cells undergoing apoptosis exhibit distinct morphological and cellular changes, including chromatin condensation, DNA fragmentation, and cellular shrinkage, which ultimately lead to the disintegration of the cell into membrane-bound particles known as apoptotic “blebs.” These particles are quickly cleared from the body by phagocytic cells, preventing cell death-associated inflammation. 118
The initiation of apoptosis is mainly activated by 2 pathways, intrinsic and extrinsic. The intrinsic pathway is activated by signaling pathways from inside the cell that govern mitochondrial integrity. In contrast, the extrinsic pathway is initiated by extracellular signals transduced by transmembrane “death receptors.” 119 Both pathways ultimately lead to the completion of apoptosis by activating the execution phase. As they progress, they converge in the activation of central activator caspases like caspase-3, -6, and -7.118,120
Extrinsic apoptosis is triggered by extracellular ligands, when transmembrane “death” receptors (DRs) such as CD95/Fas, tumor necrosis factor receptor 1 (TNF-R1), DR3, DR6 and tumor necrosis factor-related apoptosis-inducing ligand (TRAIL) receptors DR4 and DR5 are activated. 121 Binding of a death ligand to a death receptor triggers the formation of the death-inducing signaling complex (DISK) containing the adaptor protein Fas-associated death domain (FADD) protein and procaspase-8 and -10. 122 Therefore, the DISK represents the standard solution used by cells to transduce signals from outside to within. Procaspase-8 is subject to autocatalytic cleavage within the DISK. This results in the formation of active caspase-8, the main initiator caspase of extrinsic apoptosis, equivalent to caspase-9 in the intrinsic pathway. Caspase-8 directly triggers the activation of the executioner caspases-3 and 7 . 121
On the other hand, the intrinsic or “mitochondrial” apoptosis pathway is controlled by Bcl-2 proteins. These proteins, classified into both pro-apoptotic and anti-apoptotic families, have between 1 and 4 Bcl-2 homology (BH) domains. The pro-apoptotic Bcl-2 proteins can be subdivided into distinct categories: the “BH3-only activators,” such as BIM, BID, and PUMA, which trigger apoptosis; “BH3-only sensitizers,” including BAD, NOXA, HRK, BIK, and BMF, which enhance cellular sensitivity to apoptosis; and the “pore-forming effectors,” namely Bax and BAK, which directly form pores in the mitochondrial membrane. Counteracting these are anti-apoptotic or “pro-survival” Bcl-2 proteins, such as MCL-1, Bcl-2, and Bcl-XL, which inhibit apoptosis by sequestering the pro-apoptotic Bcl-2 family members. 123 The activation of BH3-only proteins by apoptotic signals can trigger Bax/BAK homo-oligomerization, resulting in mitochondrial outer membrane permeabilization (MOMP). 124 MOMP leads to the release of a variety of proteins into the cytosol, that are typically located in the intermembrane space between the outer (OMM) and inner (IMM) mitochondrial membranes. 125 Cytochrome-c (Cyt-c), protein released as a result of MOMP, subsequently interacts with apoptotic protease activating factor 1 (Apaf1) and procaspase-9 to assemble the apoptosome. This complex promotes the activation of initiator caspase-9 and executioner caspase-3. 126 MOMP is the step often described as a point of no return in apoptosis, as it triggers the release of cysteine proteases, caspases that cleave numerous specific cellular substrates into the cytosol, leading to an irreversible progression to cell death. 125
IR-induced apoptosis is believed to primarily occur through the intrinsic pathway, 122 which integrates intracellular signals triggered by different stressors, including DNA damage. 125 This involves p53, often referred to as the “guardian of the genome.” p53 plays a dual role in cellular function; it primarily functions as a tumor suppressor by inducing cell cycle arrest to allow DNA repair or apoptosis thereby preventing the propagation of cells with significant DNA damage. 127 This complexity is evident in RT, where p53 can have paradoxical effects. Specifically, after exposure to radiation-induced DNA damage, the p53 pathway can be triggered to initiate DNA repair processes. However, in cases where DNA damage is severe and irreparable, p53 directly influences the expression of apoptotic genes by modulating Bcl-2 and Bax.122,128-131
p53 is one of the biomarkers that can indicate a tumor’s aggressiveness and responsiveness to RT.132-135 Ong et al. found that MIBC patients with p53 mutations were more likely to have poor local control and decreased survival after undergoing radiation and transurethral resection. 78 In their research, Rotterud et al. examined a group of 59 patients with pathological stage T2 to T4a transitional cell carcinoma (TCC) of the bladder who underwent adjuvant RT followed by cystectomy. In the subgroup of patients who received a higher dose of radiation (46 Gy), a significant association was observed between p53 nuclear overexpression, the absence of residual tumor in the surgical specimen, and CSS. This indicates that p53 overexpression may confer radiosensitivity. 67 Additionally, Rodel et al showed that, while p53 expression alone was not correlated with BC radiosensitivity, high p53 expression was identified as a potential prognostic indicator for BC progression. 74 Overall, while certain investigations suggested a correlation between p53 overexpression and adverse clinical outcomes, others report improved survival or increased radiosensitivity. This highlights the complex role of p53 in response to radiation. 136 While it shows potential as a biomarker for radiation-sensitivity, its effectiveness may be influenced by the underlying molecular mechanisms at play. More research is needed to investigate whether p53 expression can be used to predict the response of patients to RT.
Other apoptosis biomarkers can also serve as predictors of BC radiosensitivity. Matsumoto et al. investigated the correlations between Bcl-2 and Bax expression levels and the response to CTRT in 62 patients with bladder TCC by IHC. According to their findings, the Bax to Bcl-2 ratio serves as a valuable predictor in neoadjuvant CTRT for BC as it showed a significant association with the CR rate. 68 In another study conducted by Rodel et al, the apoptotic index (AI), Bcl-2 levels, and p53 expression were examined as potential predictors for response to CTRT in a cohort of 70 patients with invasive bladder carcinoma. The AI is defined as the proportion of apoptotic cells and apoptotic bodies in tumor samples.137,138 The results showed a significant correlation between the AI and the response to CTRT, suggesting that a high AI is predictive of radiosensitivity and highlights its potential as a predictive biomarker. Moreover, the study indicated that decreased Bcl-2 levels may be indicative of an improved response to CTRT. 74
Radiation and Immune Checkpoint Biomarkers
After irradiation, dead tumor cells express tumor antigens on their surface, which activate antigen-presenting cells, such as dendritic cells (DC). These activated DCs initiate an adaptive immune response by recruiting cytotoxic T lymphocytes (CTLs) to the TME to eliminate tumor cells.139,140 Additionally, the process also enhances the secretion of various DAMPs and proinflammatory cytokines, such as type 1 interferons (IFNs), leading to an overall enhancement of the immune response.141,142 Moreover, radiation-induced DNA damage activates a cyclic guanosine monophosphate–adenosine monophosphate (cGAMP) synthase (cGAS)-mediated innate immune response. The DNA sensor cGAS detects DNA DSBs in the cytosol and becomes activated. Following the synthesis of cGAMP by the activated cGAS, STING is also activated by direct binding to cGAMP, which in turn stimulates the production of type I IFNs by phosphorylating the transcription factor IRF3. Consequently, type I IFNs mediate a specific anti-tumor immune response, contributing to tumor control. Although RT primarily targets local tumors, it can also affect distant, untreated tumors by inducing a systemic anti-tumor response known as the abscopal effect. 140 Forner et al investigated the abscopal effect in metastatic head and neck squamous cell carcinoma (HNSCC). A sustained response in metastatic disease outside the RT field after receiving palliative RT for a single progressing site was observed in a patient with sinonasal SCC who had oligometastatic progression on nivolumab. 143
Programed cell death ligand 1 (PD-L1) is one of the most well-characterized immunological biomarkers for predicting therapeutic outcomes. PD-L1, a 40-KDa transmembrane protein and a member of the B7 protein family, promotes cancer progression by binding to its receptors, PD-1, on tumor-specific T cells. This interaction activates proliferative and survival signaling pathways.144,145 Many studies have demonstrated that PD-L1 can potentially predict the radiosensitivity of various cancers, including esophageal squamous cell carcinoma (ESCC), head and neck cancer (HNC), and glioblastomas.146-148
Multiple studies have investigated the correlation between PD-L1 expression levels and BC radiosensitivity. Zhang Reyes et al demonstrated that BC cell-intrinsic PD-L1 signals regulate key virulence and treatment resistance pathways, such as BC cell proliferation in vitro, mammalian target of rapamycin complex 1 (mTORC1) signaling in vitro, and metastatic cancer spread. BC cell-intrinsic PD-L1 signals promote both basal and stress-induced autophagy, while inhibiting autophagy in melanoma and ovarian cancer cells. 149 Additionally, Chun-Te Wu et al reported that low PD-L1 expression is associated with improved OS and progression-free survival in MIBC patients treated with CTRT. Conversely, a study of 72 MIBC patients treated with TMT showed that high PD-L1 expression is linked to poor TMT response and increased loco-regional failure. 69
Signal Transduction Biomarkers
The exposure of cells to IR triggers complicated cellular reactions that cause modifications in proliferation and cell death. These reactions are regulated by pre-existing signaling pathways, mainly involving protein kinases, that regulate the cytoprotective, cytotoxic and cellular stress responses to radiation. After irradiation, the cytoprotective responses are activated in order to repair radiation-induced DNA damages. This involves the activation of biosynthesis machinery through the cascades of mitogen-activated protein kinase (MAPK) and phosphatidylinositol-3-phosphate kinase (PI3K) pathways. 150
The exposure to IR within the range of 1 to 5 Gy, leads to the activation of the epidermal growth factor receptor (EGFR), also known as ErbB1, which belongs to the receptor tyrosine kinases (RTKs) family. 150 There are 4 members in the family, EGFR/HER1, ErbB2/HER2, ErbB3/HER3, and ErbB4/HER4. 151 Activation of the MAPK cascade occurs as a result of stimulation of EGFR by extracellular ligands of the EGF family or by clinically relevant doses of IR. 152 MAPK pathway, consisting of the Ras-Raf-MEK-ERK signaling cascade, is responsible for the phosphorylation of transcription factors involved in cell survival and proliferation processes. 153
The importance of MAPK as a downstream effector of EGFR is validated by the inhibition of MAPK using the specific MEK1/2 inhibitor (PD98059) in combination with radiation. This approach led to increased radiosensitivity, as evidenced by reduced cell growth, increased apoptosis, and decreased clonogenicity compared to treatment alone. 154
Upon activation of EGFR by IR, PI3K activity, which plays a critical role in protecting cells from apoptosis, is also stimulated. 155 Downstream targets of PI3K include the phosphoinositide-dependent kinase 1/2 (PDK1/2), which activates the protein kinase Akt. 156 Through phosphorylation, Akt can directly inhibit key pro-apoptotic proteins such as Bad, Bax, and Bim, members of the Bcl-2 family. Additionally, the PI3K/Akt signaling pathway can also inhibit GSK3, the principal physiological substrate of Akt, responsible of triggering hypoxia-induced apoptosis. 157
In vitro and in vivo studies have demonstrated that inhibiting PI3K/Akt signaling, either through chemical or biological inhibitors, can enhance the radiosensitivity of certain types of cancer cells. These findings indicate that the PI3K/Akt pathway has a pro-survival function, promoting radioresistance in cancer cells. 158
Radiation-induced cytotoxicity or stress response stimulates the stress-activated protein kinase (SAPK) cascade, which involves certain cytokine receptors, sphingomyelinase with ceramide production, and activation of another protein kinase cascade. This cascade includes MEKK1, SEK1, and c-Jun NH2-terminal kinase 1/2 (JNK1/2), also known as MAPK8. 159 JNKs activate apoptosis by either modulating the activities of mitochondrial pro- and anti-apoptotic proteins or upregulating pro-apoptotic genes via the transactivation of specific transcription factors such as c-Jun. Therefore, JNK is a key player in the immediate response to IR-induced stress. 160
Many studies have investigated the capacity of signal transduction biomarkers to understand and predict radiosensitivity of various types of cancer. For example, the expression of HER1, HER2, HER3, and HER4 was correlated with the radiosensitivity of human breast cancer and colon carcinoma.161-163 The role of these signal transducers is further highlighted by the radiosensitizing effects of inhibitors targeting the PI3K/Akt/mTOr pathway, ERK1/2 and EFGR across a wide range of cancers.162,164-166
In the case of BC, many biomarkers have been identified as potential predictors of radiosensitivity. Chakravarti et al analyzed the correlations between erbB-1 (EGFR) and erbB-2 (HER2) expression levels and the response to CTRT in MIBC patients undergoing CTRT bladder preservation therapy. According to their findings, HER2 positivity was significantly associated with lower CR rates (50% versus 81%, P = .026) after CTRT, whereas EGFR positivity was associated with better CSS (P = .042). This correlation appeared to significantly improve outcomes in BC. 79 Also, Koga et al confirmed that the expression levels of erbB-2 in pretreatment biopsy samples from 35 MIBC patients was associated with CTRT resistance. 72 Additionally, Inoue et al validated the correlation between the overexpression of erbB-2 with resistance to CTRT in 119 MIBC patients. 70
VEGF, a potent angiogenic factor, and its receptor (VEGF-R) have been shown to play major roles in tumor and lymph angiogenesis. 167 Lautenschlaeger et al investigated the association of VEGF with CTRT response in 43 MIBC patients. According to their report, significant correlations were found between reduced OS rates and increased levels of cytoplasmic VEGF-B, VEGF-C, and their receptor VEGF-R2. 80 Furthermore, Keck et al showed that the increased expression of Neuropilin-2 or its ligand VEGF, in 247 BC patients, was associated with shorter CSS. 71
Proliferation Biomarkers
One of the key characteristics of cancer cells is their ability to sustain uncontrolled and continuous proliferation. 168 Exposure to X-rays through RT induces DNA damage, leading to cell cycle arrest and a consequent decrease in proliferation rate. 169 However, radiation also can stimulate the proliferation of surviving cells and enhance their ability to resist various forms of cell-killing stress over the long term. 170 Irradiation activates EGFR through a ligand-independent pathway, 171 which triggers the tyrosine kinase domain of EGFR through auto-phosphorylation, which in turn activates downstream signaling pathways, including the EGFR-dependent PI3K/Akt and Ras/MAPK pathways. These pathways are critical for promoting cell survival and proliferation, contributing to RT resistance. 172
In the EGFR-dependent Ras/MAPK pathway, the growth factor receptor bound protein-2 (GRB2) is recruited either directly or indirectly, which then activates Ras through the son of sevenless (SOS) protein. This activation leads to the phosphorylation of Raf, which in turn phosphorylates MEK1/2 and stimulates the extracellular signal-regulated kinase-1/2 (ERK1/2). Once phosphorylated, ERK1/2 translocates into the nucleus and activates various transcription factors that are responsible for regulating genes that promote cell proliferation. 172 In addition to the Ras/MAPK pathway, EGFR can also directly or indirectly activate signal transducer and activator of transcription proteins (STATs), including STAT1, STAT3, and STAT5, involved in cell proliferation. Once activated, these STATs translocate into the nucleus and directly regulate the expression of genes responsible for cell proliferation, survival, transformation, and oncogenesis. 172 Furthermore, IR exposure influences other signaling pathways, such as those involving NF-kB and (activator protein 1 (AP-1), through ROS-mediated mechanisms. This leads to increased activity of ERK 1/2 and p38 kinases, potentially raising the risk of malignancy. NF-kB and AP-1 are associated with cellular proliferation, and failure to halt the cell cycle at mitotic checkpoints due to unrepaired DNA damage can further elevate the risk of cancer. 173 Additionally, NF-κB enhances the production of genes like c-Myc and cyclin D1, which promote cell proliferation and prevent cell death. The activation of the NF-κB pathway is believed to contribute to the development of radioresistance in various types of cancer cells. One example is Aurora A, a serine/threonine kinase involved in cell mitosis, which activates NF-κB signaling in hepatocellular carcinoma (HCC), thereby contributing to the cancer’s radioresistance. 174 Additionally, a study that evaluated NF-kB expression in pretreatment biopsy samples from 35 MIBC patients using immunohistochemistry (IHC) found that NF-kB overexpression was linked to CTRT resistance in these patients. 72
Proliferating cell nuclear antigen (PCNA) is a widely used biomarker for the detection of tumor cell proliferation activity. Studies have shown that PCNA is often overexpressed in various cancers, including lung cancer, prostate carcinoma, and breast cancer. Elevated PCNA levels have also been linked to various digestive system tumors, such as colorectal carcinoma, ESCC, and HCC. However, limited studies in BC have shown that PCNA immunostaining could potentially help identify patients who may benefit from preoperative RT. 175 Further clinical trials are needed to confirm the predictive potential of PCNA in MIBC.
On the other hand, Ki-67, a protein expressed in dividing cells, is widely recognized as a biomarker of cancer cell proliferation. It is a nuclear protein present during all active phases of the cell cycle but absent in resting cells. Unlike PCNA, which can be difficult to interpret, Ki-67 is easier to detect, making it an essential biomarker for assessing the proliferative fraction of tumors. Due to its technical simplicity and reliability, the Ki-67 labeling index is commonly used for this purpose, and has demonstrated prognostic value in BC, breast cancer, oral squamous cell carcinoma, glottic cancer and small cell lung cancer.176-180
Many studies have assessed the ability of Ki-67 to predict BC radiosensitivity. In a study of 190 MIBC patients treated with a bladder-sparing CTRT protocol, Tanabe et al found a significant correlation between high Ki-67 expression and improved CSS. Among the 94 patients treated with CTRT with 40 Gy of RT combined with cisplatin, a higher Ki-67 labeling index was independently associated with a greater CR rate. 73 Similarly, Rödel et al conducted a study on 70 MIBC CTRT-treated patients, showing that a high Ki-67 index predicted BC radiosensitivity. The study suggested that patients with elevated pretreatment Ki-67 levels should be prioritized for CTRT. 74
Finally, in addition to the cited biomarkers, Yoshida et al identified the apparent diffusion coefficient (ADC) values, which quantify water molecule diffusion as a potential predictor of BC proliferation. Their study revealed correlations between ADC values and both tumor size and histologic grade, suggesting their potential as a biomarker. In an analysis of 23 BC patients treated with CTRT, lower ADC values were associated with higher sensitivity to the treatment. Furthermore, an inverse correlation between ADC values and Ki-67 labeling index in MIBC tissues was observed. 75
E3 Ubiquitin Ligases and Deubiquitinases as Biomarkers
Numerous E3 ubiquitin ligases and deubiquitinases (DUBs) modulate immune checkpoints, either directly or indirectly. More than 600 distinct E3 ubiquitin ligases playing a role in protein degradation have been identified in humans. These ligases are not only crucial for maintaining intracellular protein levels but also for various cellular processes such as intracellular transport, autophagy, DNA repair, and metabolism. Dysregulation of E3 ubiquitin ligases can lead to disrupted signaling pathways and the accumulation of defective proteins, which may contribute to the development and progression of cancer. 181 Casitas B lymphoma-b (Cbl-b) is an E3 ligase that is essential for the regulation of effector T cells. Cbl-b can influence the ubiquitination and degradation of PD-L1 following the inhibition of PI3K/Akt, Jak/STAT, and MAPK/Erk signaling pathways. Notably, Cbl-b also acts as a downstream regulator of CD28 and CTLA-4 signaling pathways. By regulating both innate and adaptive immune cells, Cbl-b contributes to creating an immunosuppressive TME. New Cbl-b inhibitors provide antigen-specific immune stimulation and hold promise as therapeutic agents in immuno-oncology. 182
BC tumorigenesis has been associated with the involvement of several E3 ligases. RNF126, a RING domain E3 ligase, is often highly expressed in BC, and is linked to tumor development. It interacts with PTEN, a tumor suppressor, promoting its polyubiquitination and subsequent degradation via proteasomes. Silencing RNF126 stabilizes PTEN, which inhibits the PI3K/Akt signaling pathway. Studies have demonstrated that RNF126 promotes DNA DSB repair through both NHEJ and HR pathways by ubiquitinating Ku80 and promoting the dissociation of Ku70/80 from DSBs. Moreover, depleting RNF126 significantly enhances the effectiveness of cisplatin in inducing apoptosis in BC cells. These findings suggest that targeting RNF126 could improve cisplatin-based CT and help manage BC development. Two additional E3 ligases, RNF114A and NEDD4, can regulate the ubiquitination of PD-L1, thereby influencing both immune suppression and the effectiveness of anticancer responses.
cIAP2, a RING-type E3 ligase, is crucial for DNA repair and has recently been associated with the regulation of radiosensitization in BC. Transfecting increased levels of cIAP2 into T24 cells leads to a progressive reduction in MRE11 levels, as cIAP2 promotes its degradation. Furthermore, silencing cIAP2 in T24 cells increases radiosensitivity. These findings suggest that further investigation into cIAP2′s role could reveal its potential as a biomarker for optimizing RT in BC.
On the other hand, DUBs are proteases that regulate ubiquitination by removing ubiquitin from substrates or cleaving ubiquitin chains. Research has shown that protein deubiquitination, mediated by DUBs, is associated with the development and progression of cancers, including BC. OTUD5, a DUB, is crucial for several cellular processes such as DNA damage repair, transcription, and innate immunity. In BC, OTUD5 is significantly overexpressed in tumor tissues compared to normal urothelial cells. It targets the mTOR signaling pathway to promote cell proliferation. Therefore, knocking down OTUD5 decreases cell proliferation, suggesting that targeting this DUB might help in BC treatment. Similarly, UCHL5, another DUB overexpressed in BC, is associated with poor prognosis and tumor progression. It promotes tumor growth by activating the Akt/mTOR pathway and increasing c-Myc expression. The UCHL5 inhibitor b-AP15 has been shown to reduce cancer stemness and overcome cisplatin resistance. An additional DUB, UCHL5, helps suppress tumor formation by stabilizing tumor suppressors such as p53 and PTEN through deubiquitination. In BC, USP13 stabilizes PTEN, thereby inhibiting tumor progression. These findings suggest that E3 ligases and DUBs could serve as potential targets for BC treatment or represent a promising therapeutic strategy. 181
Long Non-coding RNAs
Long non-coding RNAs (LncRNAs) are RNA molecules longer than 200 nucleotides that interact with DNA, RNA and proteins to regulate gene expression during transcription, translation, and protein modification. They associate with protein complexes to modify chromatin states through histone modification and DNA methylation. Despite their significant role in genetic processes, their full function remains to be understood. 183
LncRNAs play a role in regulating cell proliferation and evading growth suppressors by inactivating proteins involved in proto-oncogene suppression, leading to increased oncogene expression. They are also involved in replicative immortality by enabling telomerase activity and preventing telomere shortening after cell division, allowing cancer cells to replicate indefinitely. LncRNAs are linked to epithelial-mesenchymal-transition (EMT), thereby promoting tumor invasion and metastasis and inhibiting regulatory proteins involved in EMT. They also induce angiogenesis, contributing to cancer cells resistance to apoptosis. Multiple studies have shown LncRNAs act as molecular sponges for microRNAs, regulating their activity in processes such as embryonal development and growth control. 184
LncRNAs have been implicated in modulating radiosensitivity across various cancers by regulating proteins that either suppress or induce tumor growth.185,186 In BC, several studies have demonstrated the involvement of LncRNA biomarkers involvement in radiosensitivity. Tan et al investigated the function of LncRNA taurine-upregulated gene 1 (TUG1), showing that higher TUG1 expression correlated with shorter OS. TUG1 also promoted BC cell invasion and radioresistance by inducing EMT. Inhibition of TUG1 increased E-cadherin expression and decreased mesenchymal markers, reversing EMT and reducing metastasis. 187 Additionally, Han et al used real-time qPCR to analyze TUG1 expression in 44 bladder urothelial carcinomas patients. They found that TUG-1 silencing inhibited cell proliferation and induced apoptosis, with TUG1 upregulation more prominent in high grade carcinomas compared to normal tissues, further establishing its role in BC progression. 188
MicroRNAs
MicroRNAs (miRNAs) are small, non-coding RNA molecules that regulate gene expression. They regulate DDR, cell cycle progression, apoptosis, and influence changes in the TME. 189 Altered miRNAs expression has been linked to cancer development, with specific miRNAs classified as oncogenic miRNAs or oncomiRs. For instance, miRNA-21 is frequently overexpressed in various cancers, while Let7 is commonly downregulated in most malignancies. 190 The ability to easily identify miRNAs in tumors makes them valuable prognostic and therapeutic response biomarkers. 191 Additionally, miRNA expression undergoes radiation-induced changes, highlighting their potential role in cellular responses to IR. 189
Certain miRNAs, including miR-421 and miR-24, disrupt the DDR pathway by reducing ATM and H2AX expression. This interference compromises DDR, enhances radiosensitivity, and increases genomic instability. In their study, Meng et al aimed to identify miRNA biomarkers predicting response to CTRT and OS in 40 patients with MIBC undergoing TMT. The selection included patients with both complete and incomplete responses. Deep sequencing of miRNA levels in primary tumors revealed associations between several miRNAs, including miR-23a and miR-27a, and OS. In vitro experiments revealed that the overexpression of either miR-23a or miR-27a in BC cells induced cell proliferation, migration, invasion and sensitivity to radiation by targeting secreted frizzled-related protein1 (SFRP1), a negative regulator of the Wnt signaling pathway. The presence of these miRNAs at low levels could potentially serve as a biomarker, helping guide the selection of TMT as the preferred management strategy due to its positive response. 76
Conclusion
With the increasing practice of bladder-preserving cancer treatments, identifying BC radiosensitivity is becoming essential. Although the literature presents an array of potential biomarkers, the absence of functional assays hinders the implementation of personalized treatments for BC patients (Figure 2). This gap prevents patients from achieving optimal treatment outcomes. By building on the theoretical understanding of the biomarkers mentioned in this review, new assays should be developed to detect one or more of them, enabling more accurate assessment and prediction of BC radiosensitivity. This approach would advance medical practice toward personalized medicine, improving both diagnostic and prognostic strategies. A better understanding of individual cancer radiosensitivity could guide clinicians in adopting more targeted approaches, potentially sparing patients unnecessary treatments or enhancing treatment plans tailored to their radiosensitivity. This would ultimately improve the quality of life for BC patients and boost CSS.
Additionally, most studies on BC radiosensitivity focus on conventional RT. However, radiation delivery has advanced through techniques such as image-guided RT and FLASH RT, and particle therapy, which are revolutionizing cancer treatments. Applying these cutting-edge technologies to BC could alter tumor and normal tissue responses. More preclinical and clinical studies are needed to develop predictive algorithms for each treatment, incorporating biomarkers with sufficient predictive performances to be effectively and routinely used in patient care.
List of abbreviation
ACA: Alkaline comet assay
AI: Apoptotic index
Apaf1: Apoptotic protease activating factor 1
ADC: Apparent diffusion coefficient
APE1: Apurinic/apyrimidinic endonuclease 1
ATM: Ataxia telangiectasia mutated
ATG12: Autophagy-related 12
BER: Base excision repair pathway
BAK: BCL-2 antagonist or killer
BAX: BCL-2-associated X protein
BC: Bladder cancer
BCG: Bacillus Calmette-Guerin
BC2001: Bladder Cancer 2001
BST: Bladder-sparing treatment
BLM: Bloom syndrome protein
CIS: carcinoma in situ
Cbl-b: Casitas B lymphoma-b
CSS: Cause-specific survival
Chk1: Checkpoint kinase 1
CTRT: Chemoradiotherapy
CT: Chemotherapy
CPIs: Checkpoint inhibitors
CRC: Colorectal cancer
CR: Complete response
CSCs: Cancer stem cells
CGAS: Cyclic GMP-AMP synthase
Cyt-c: Cytochrome-c
CTLs: Cytotoxic T lymphocytes
DAMP: Damage-associated molecular pattern
DISC: Death-inducing signaling complex
DRs: Death receptors
DC: Dendritic cells
DUBs: Deubiquitinases
DDR: DNA damage response
DNA-PKcs: DNA protein kinase catalytic subunit
DNA2: DNA replication helicase/nuclease
DSBs: Double-strand breaks
ECM: Extracellular matrix
EGFR: Epidermal growth factor receptor
EMT: Epithelial to mesenchymal transition
ESCC: Esophageal squamous cell carcinoma
ERCC1: Excision repair cross-complementing group 1 protein
EXO1: Exonuclease 1
FADD: Fas-associated death domain
HNC: Head and Neck Cancer
HR: Homologous recombination
IHC: Immunohistochemistry
IMM: Inner mitochondrial membranes
IGF1R: Insulin-like growth factor 1 receptor
IFNs: Interferons
IL-1: Interleukin 1
IR: Ionizing radiation
JNK: c-Jun NH2-terminal kinase
LI: Labeling index
LncRNAs: Long non-coding RNA’s
MALAT-1: Metastasis associated lung adenocarcinoma transcript 1
MCRC: Metastatic CRC
miRNAs: MicroRNAs
MOMP: Mitochondrial outer membrane permeabilization
MAPK: Mitogen-activated protein kinase
MIBC: Muscle-invasive bladder cancer
NHEJ: Non-homologous end-joining
NMIBC: Non-muscle invasive bladder cancer
NER: Nucleotide excision repair
OMM: Outer mitochondrial membranes
OS: Overall Survival
PI3K: Phosphatidylinositol-3-phosphate kinase
PDK: Phosphoinositide-dependent kinase
PD-L1: Programed cell death ligand 1
PFS: Progression-free survival
PCNA: Proliferating cell nuclear antigen
CaP: Prostate cancer
RC: Radical cystectomy
RT: Radiotherapy
RIANS: Radiation-induced atm nucleoshuttling
RNS: Reactive nitrogen species
RPA: Replication protein A
SFRP1: Secreted frizzled-related protein1
STATs: Signal transducer and activator of transcription proteins
SAPK: Stress-activated protein kinase
TAMs: Tumor-associated macrophages
TCC: Transitional cell carcinoma
TUG-1: Taurine-upregulated gene 1
TURBT: Transurethral removal of the bladder tumor
TMT: Trimodal therapy
TME: Tumor microenvironment
TNF-R1: Tumor necrosis factor receptor 1
TRAIL: Tumor necrosis factor-related apoptosis-inducing ligand
VEGF: Vascular endothelial growth factor
VEGFR: Vascular endothelial growth factor receptor
XRCC1: X-ray repair cross-complementing protein 1
XLF: XRCC4-like factor
Footnotes
Acknowledgements
The authors are thankful for the assistance of the members of Department of Radiation Oncology of the American University of Beirut Medical Center. They would also like to thank all the members of the team of Dr. Larry Bodgi. This study was funded by the Medical Practice Plan of the Faculty of Medicine at the American University of Beirut, Beirut, Lebanon.
