Abstract
Purpose:
Neutrophil-lymphocyte ratio has been explored as a prognosticator in several cancer types, but its association with larynx cancer outcomes is not well known. We aimed to identify an optimal NLR cutoff point and examine the prognostic utility of this biomarker in patients with locoregionally advanced larynx cancer treated with curative intent.
Methods:
In the Veterans Affairs’ (VA) national database, we identified patients with locoregionally advanced (T3-4N0-3M0) laryngeal squamous cell carcinoma diagnosed between 2000 and 2017 and treated with curative intent. NLR cutoff points were calculated using Contal/O’Quigley’s method. Outcomes of larynx cancer-specific survival (CSS), overall survival (OS), and non-larynx cancer survival (NCS) were evaluated in multivariable Cox and Fine-Gray models.
Results:
In 1047 patients, the optimal pretreatment NLR cutoff was identified as 4.17 - 722 patients with NLR ⩽ 4.17, 325 patients with NLR > 4.17. The elevated NLR cohort had a higher proportion of T4 disease (39.4% vs 28.4%), node positive disease (52.3% vs 43.1%), and surgical treatment (43.7% vs 35.2%). In multivariable analysis, NLR > 4.17 was independently associated with worse OS (HR 1.31, 95% CI 1.12-1.54, P = .001) and worse CSS (HR 1.46, 95% CI 1.17-1.83, P < .001), but not with NCS (HR 0.94, 95% CI 0.75-1.18, P = .58).
Conclusion:
In locoregionally advanced larynx cancer treated with curative intent, we identified elevated NLR to be associated with inferior OS and CSS. Further prospective studies are needed to investigate pretreatment NLR and our identified 4.17 cutoff as a potential larynx cancer-specific marker for this high risk population.
Keywords
Introduction
After lung cancer, larynx cancer is the most common respiratory cancer with a significant oncologic burden worldwide.1,2 Locoregionally advanced larynx cancer represents a high-mortality subgroup that is traditionally managed with either surgical resection (total laryngectomy, TL) with adjuvant therapy, primary radiation (RT), or primary chemoradiation (CRT). While guidelines surrounding management of locoregionally advanced larynx cancer have been debated, a combination of factors including baseline medical fitness (eg, age, performance status) and concomitant comorbid conditions (eg, COPD, heart disease) contribute to final treatment assignment.3,4 Due to the significant morbidity of the combination of larynx cancer with available treatment options, there is a need for better prognostic markers in this population to aid clinical decision making.
Cancer-related inflammation is a hallmark of cancer and a key driver of cancer survival outcomes. 5 Chronic inflammation in the larynx is a recognized precursor state to larynx cancer and other solid tumors.6,7 One metric of cellular inflammation is the neutrophil-lymphocyte ratio (NLR), readily obtained from routine labs indicated in the workup of cancer. Previous studies have demonstrated that an elevated NLR is associated with poor outcomes in both non-oncologic, inflammatory conditions (eg, chronic obstructive pulmonary disease, coronary artery disease), and oncologic conditions (eg, pancreatic cancer, mesothelioma).8-10 Prior limited data series have presented associations between NLR and overall survival (OS) in patients with larynx cancer, but it has yet to be elucidated if NLR is predictive of oncologic (larynx cancer risk) or non-oncologic (competing mortality risk) outcomes in this setting. 11
In patients with locoregionally advanced larynx cancer treated with curative intent within the national Veterans’ Affairs (VA) database, we identify an optimal cutoff point for pretreatment NLR and performed competing risks analyses to delineate the prognostic potential of NLR on larynx cancer-specific survival (CSS), OS, and non-larynx cancer survival (NCS).
Materials and Methods
Data source
VA Informatics and Computing Infrastructure (VINCI) is a comprehensive informatics platform that enables access to the VA’s national database comprised of patient-level electronic health records and administrative data. Tumor registry data is uploaded by trained registrars in accordance with protocols issued from the American College of Surgeons, thereby capturing an estimated 90% of incident cancers within the VA system.12,13 Cause specific mortality (ICD-10 code C32 for larynx cancer) information was obtained from the National Death Index (NDI). Our protocol and methods were performed in accordance with the relevant guidelines/regulations and were approved by the San Diego VA IRB (Project #H150169). Informed signed consent was waived by the IRB given that this is a retrospective analysis with minimal risk to the rights and welfare of subjects and one which could not practically be completed without the waiver.
Patient selection and covariables
Of the veterans with histologically confirmed, laryngeal squamous cell cancer diagnosed between January 1, 2000 and January 1, 2018, only those with locoregionally advanced disease (T3-4N0-3M0) were included. Patients were excluded if they were not treated definitively or did not have complete survival information. Definitive treatment was defined as TL + adjuvant CRT, TL + adjuvant RT, or CRT alone. For T3N0M0 cases, definitive treatment was also defined as TL alone or RT alone. Treatment was ultimately classified as surgical versus non-surgical depending on if TL was part of the treatment. The cancer treatment modality was assigned using a combination of structured data elements and free-text search of operative notes and radiotherapy/chemotherapy treatment summaries when available. The final cohort was comprised of 1047 patients (Figure 1).

Consort diagram.
Variables of interest included: age at diagnosis, gender, race, median income, and education level of zip code, marital status, hospital admission within 1 year prior to diagnosis, smoking status, diagnosis of COPD, Charlson comorbidity index, T category, N category, tumor grade, tumor subsite, treatment. Linked administrative data provided International Classification of Diseases-9 and 10 codes for comorbidities used to construct the Charlson comorbidity index score.14,15 Laboratory data was used to identify complete blood count (CBC) with differential test results. From this, NLR was calculated as the absolute neutrophil count (ANC) divided by the absolute lymphocyte count (ALC). Each patient’s pretreatment NLR was defined as the closest value within 6 months prior to starting treatment. All patients were followed until death or last follow up with a VA provider before July 1, 2017.
Statistical analysis
Transforming a continuous variable into a categorical variable to evaluate its predictive value can make a model more interpretable and clinically useful. Although several techniques are commonly employed (eg, median/quartiles, Receiver Operating Characteristic (ROC) tests), the appropriateness of such methods for time dependent endpoints (such as survival) have often been called into question.16,17 Thus, we employed the Contal and O’Quigley cutpoint method that uses the log-rank test statistic.16,18 Using the methods described by Contal and O’Quigley 18 and the SAS macro provided by Meyers et al 19 , we identified that our continuous NLR variable was eligible for dichotomization. Then, the Contal and O’Quigley statistic and Cox Wald statistic were identified and compared for a panel of candidate cutoff points to identify the optimal cutoff point(s) of pretreatment NLR on each survival endpoint.
After identification of the optimal pretreatment NLR cutoff, the cohort was dichotomized based on this value. Patient characteristics between these NLR cohorts were compared using Chi Square test and Wilcoxon’s rank sum test for categorical and continuous variables, respectively. OS was assessed with Kaplan-Meier analysis for unadjusted models and with Cox proportional hazards analysis for multivariable models. CSS between groups was evaluated using a competing risk analysis framework to account for the competing risk of non-larynx cancer mortality. Vice-versa logic was used to evaluate NCS. CSS and NCS were assessed with cumulative incidence analysis for unadjusted models and with Fine-Gray regression analysis for multivariable models. Secondary analyses of previously proposed NLR cutoffs in our multivariable models were conducted. For all survival analysis, hazard ratios (HR) and 95% confidence intervals (CI) were reported. Throughout this study, all multivariable models were conducted a priori with the aforementioned covariables of interest. All statistical analyses were performed using SAS version 9.4 (SAS Institute, Cary, NC), with 2-sided P-values less than .05 considered statistically significant.
Results
Baseline characteristics and treatment exposure
The 1047 patients in our final cohort were analyzed with a median follow-up of 52 months. Median NLR of the cohort was 3.14 (range 0.14-49.28) obtained at a median of 8 days (range 1-171) before start of treatment. The optimal pretreatment NLR cutoff for our cohort was determined to be 4.17 using the Contal and O’Quigley cutpoint selection method (P < .001). This was validated to be an optimal cutoff point in the Cox Wald cutpoint selection method (P < .001). The cohort was split by this cutoff – 722 patients with NLR ⩽ 4.17 and 325 patients with NLR > 4.17.
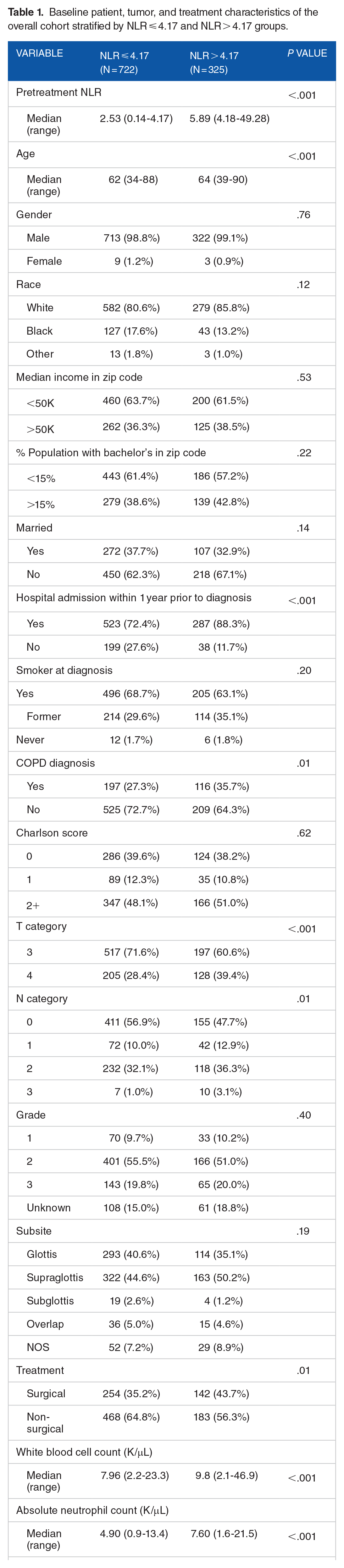
Although many variables were broadly similar between the 2 cohorts, there were a few notable differences especially in clinical factors (Table 1). A higher percentage of patients in the higher NLR cohort were hospitalized within the year prior to diagnosis (88.3% vs 72.4%) and had a diagnosis of COPD prior to their cancer diagnosis (35.7% vs 27.3%). There was a higher percentage of T4 category disease (39.4% vs 28.4%) and non-N0 category disease (52.3% vs 43.1%) in the higher NLR cohort as well. Finally, there was a higher rate of surgical treatment (43.7% vs 35.2%) in the higher NLR cohort.
Baseline patient, tumor, and treatment characteristics of the overall cohort stratified by NLR ⩽ 4.17 and NLR > 4.17 groups.
Survival
Median overall survival of the entire cohort was 29.5 months (95% CI 26.5-33.9 months). There were 770 deaths, 360 (46.8%) which were attributed to larynx cancer (cancer-specific mortality). The 5-year cumulative incidence of death from larynx cancer and any non-larynx cancer cause were 33.1% (95% CI 30.1-36.1%) and 34.1% (95% CI 31.1-37.2%), respectively. OS was better (P < .001) in patients with NLR ⩽ 4.17 (median OS 37.2 months, 95% CI 31.1-43.0 months) than in those with NLR > 4.17 (median OS 21.6 months, 95% CI 17.8-24.8 months) (Figure 2). Gray’s test found patients with NLR > 4.17 to have increased larynx cancer-specific mortality (P < .001) and similar non-larynx cancer mortality (P = .50) compared to patients with NLR ⩽ 4.17 (Figures 3 and 4).

Kaplan-Meier curves for overall survival stratified by NLR cutoff value 4.17.

Cumulative incidence curves for larynx cancer-specific mortality stratified by NLR cutoff value 4.17.
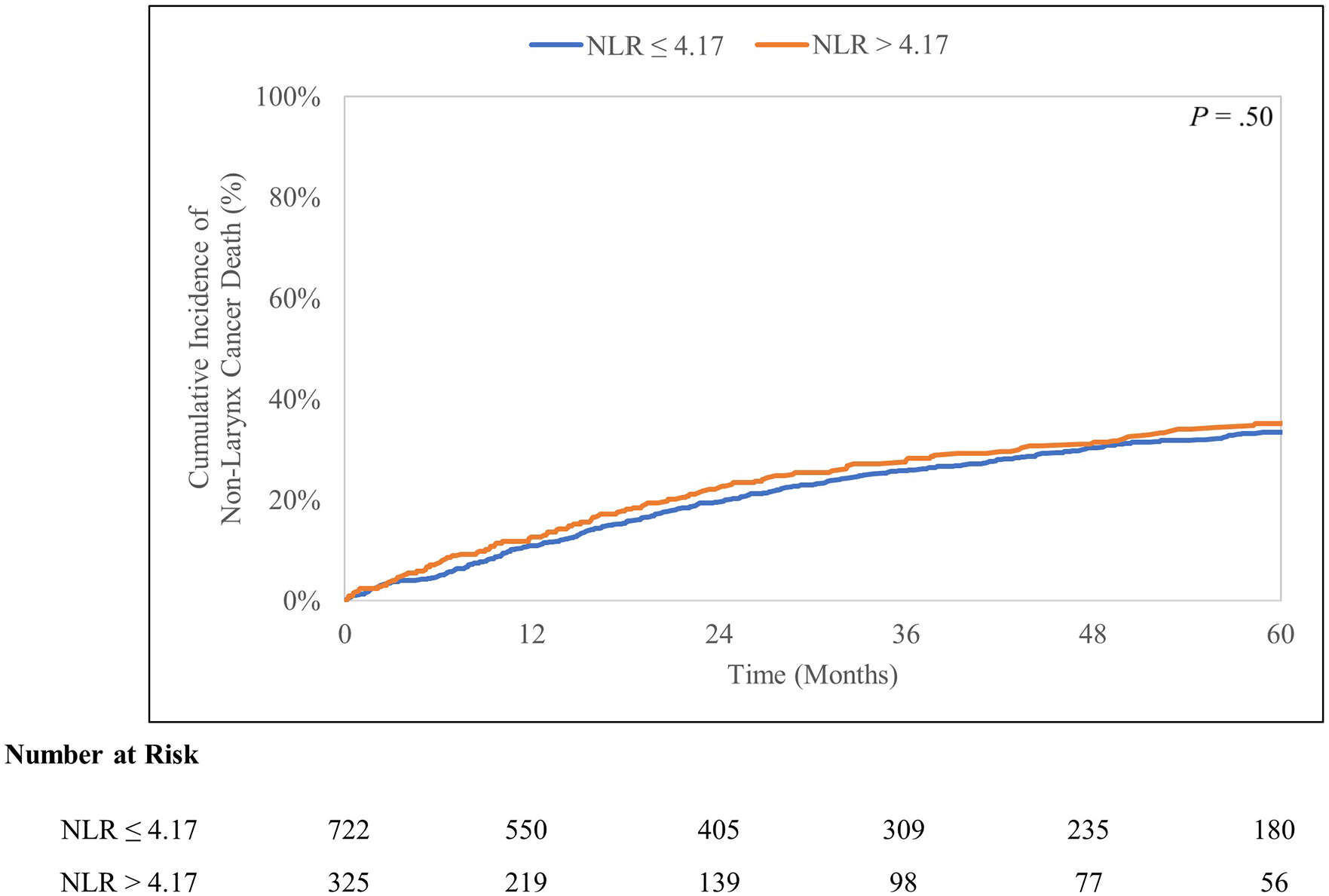
Cumulative incidence curves for non-larynx cancer mortality stratified by NLR cutoff value 4.17.
In multivariable analysis, NLR > 4.17 was independently associated with worse overall survival (HR 1.31, 95% CI 1.12-1.54, P = .001) (Table 2). For the other endpoints, similar multivariable analysis demonstrated NLR > 4.17 to be independently associated with inferior CSS (HR 1.46, 95% CI 1.17-1.83, P < .001) but not with NCS (HR 0.94, 95% CI 0.75-1.18, P = .58) (Table 2).
Multivariable a priori regressions on overall survival (OS), larynx cancer-specific survival (CSS), non-larynx cancer survival (NCS) in the overall cohort.

Secondary analyses using previously proposed NLR cutoffs
Zeng et al 20 proposed a pretreatment NLR cutoff of 3.0 which they identified using the median value of 125 patients with locoregionally advanced larynx cancer treated with CRT. In their analysis, pretreatment NLR > 3.0 was independently associated with worse OS. Implementing their NLR threshold to our cohort of 1047 patients, multivariable analysis revealed pretreatment NLR > 3.0 to be associated with OS (HR 1.23, 95% CI 1.06-1.44, P = .01), but not with CSS (HR 1.20, 95% CI 0.96-1.50, P = .18) nor with NCS (HR 1.07, 95% CI 0.87-1.32, P = .52).
Fu et al 21 proposed a pretreatment NLR cutoff of 2.59 which they identified using ROC analysis of 420 patients with locoregionally advanced larynx cancer treated with TL. In their analysis, pretreatment NLR > 2.59 was independently associated with worse CSS and OS. Applying their NLR cutoff to our dataset yielded similarly poor results as Zeng et al’s cutoff. Multivariable analysis revealed pretreatment NLR > 2.59 to be associated with OS (HR 1.29, 95% CI 1.09-1.50, P = .01), but not with CSS (HR 1.19, 95% CI 0.94-1.52, P = .24) nor with NCS (HR 1.14, 95% CI 0.92-1.41, P = .22).
Discussion
As the largest analysis of NLR in an advanced larynx cancer population, our study identified NLR to be significantly associated with OS and CSS but not with NCS. Previously proposed NLR cutoffs did not perform well in our dataset. With our identified pretreatment NLR cutoff of 4.17, this readily available and inexpensive lab marker offers potential as a valuable cancer-specific marker for the high risk population of patients with locoregionally advanced larynx cancer.
Although previous literature examining the predictive capacity of NLR in locoregionally advanced larynx cancer is limited, the majority is in agreement with our results. Fu et al proposed a cutoff of 2.59 based on ROC analysis of 420 patients with advanced larynx cancer treated with TL; Zeng et al proposed a cutoff of 3.00 based on the median value of 125 patients with advanced larynx cancer treated with CRT.20,21 Both cutoff points were evaluated to have associations with OS in our dataset, albeit weaker associations than the one resulting from our identified cutoff of 4.17. Most importantly, neither of their proposed cutoffs were associated with CSS in our dataset. This might be a reflection of their methodology not tailored to the time-dependent endpoints of survival in question or the small size and single-institution nature of their studies. A couple of other studies on heterogenous populations of larynx cancer demonstrated elevated NLR to be associated with inferior OS and progression-free survival (PFS) which further reinforces the potential for NLR to be a cancer-specific marker for larynx cancer.11,22 Another limitation of previous studies we overcome is the inclusion of numerous, possible confounding covariables in our modeling. Given the documented potential of NLR to serve as a marker for non-oncologic, inflammatory conditions and overall competing mortality risk, our inclusion of details such as diagnosis of COPD or hospitalization within the year prior to diagnosis try to overcome potential confounding and delineate NLR’s role as a cancer-specific or non-cancer-specific marker in this population. Overall, we build on previous literature to present a multi-institutional cohort that is the largest and most detailed to date in order to investigate NLR’s impact on granular survival outcomes in larynx cancer.
Elevated NLR in response to an evolving tumor microenvironment is theorized to represent two opposing forces in the oncologic state: carcinogenesis versus host immune response.23,24 Increased neutrophilic activity represents a heightened inflammatory state known to promote carcinogenesis, while dwindling lymphocyte counts are thought to represent the host’s decreasing immunological capacity to limit tumor growth and spread.24-26 Baseline differences in NLR between benign laryngeal lesions, pre-cancerous laryngeal lesions, and malignant laryngeal lesions have been established previously, reinforcing the likely cancer-specific association NLR possesses in larynx cancer.27,28 In our study, we observed the elevated NLR cohort to have higher proportions of T category and N category disease at time of diagnosis compared to the lower NLR cohort as expected. The findings of increased NLR with increased T and N category disease in the general realm of head and neck cancer has been reported as well.22,28-30
Our study retains the inherent limitations of a retrospective database analysis. There is potential for selection bias within our design as not all patients had available lab parameters available (Figure 1), but we have little reason to believe this would disproportionately affect either cohort. Additionally we are not able to present results on PFS but as mentioned earlier, other studies in the larynx realm report similar associations between elevated NLR and worse PFS.11,22 Ultimately, a key limitation to NLR as a biomarker is that it is susceptible to both internal and external forces that may alter the host’s baseline inflammatory state such as baseline rheumatologic disorders, chronic disease states, use of immunomodulatory/suppressive medications. Although we incorporated a few of these in our analysis, many of these factors are not readily available in our database and represent potential confounders. The inclusion of as many of these factors as possible in a potential prospective trial will be important to conclusively examine the effect of NLR on outcomes. Finally, there is research demonstrating NLR to have different baseline values in different populations (eg, by race or age). 31 Although we account for some of these variables in our models, caution should be maintained when generalizing the results of our study to all populations.
Overall, we posit that NLR represents a readily-available and inexpensive lab measure that offers potential as a cancer-specific biomarker for locoregionally advanced larynx cancer. This population has a significant need for additional risk stratification due to the morbidity of the disease and treatment options. An accurate and strongly predictive cancer-specific marker in this setting can significantly aid in pre- and post-treatment decision making and may even lead to future personalized treatment algorithms. Further prospective studies are needed to validate the usefulness of NLR as a clinical tool in patients with locoregionally advanced larynx cancer.
Footnotes
Funding:
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: University of California San Diego, Altman Clinical and Translational Research Institute MedGap Program.
Declaration of conflicting interests:
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Dr. Tyler Stewart has served as a paid consultant for Seattle Genetics. Dr. Abhishek Kumar has an ownership stake in Sympto Health. All conflicts of interest are unrelated to this work. All other authors have no conflicts of interest to disclose.
Author Contributions
The authors confirm contribution to the manuscript as follows. All authors had access to the data and approved the final version of the manuscript. Nikhil V. Kotha—Conceptualization/design, Data curation and formal analysis, Writing—original draft, Writing—review and editing. Rohith S. Voora—Conceptualization/design, Data curation and formal analysis, Writing—original draft, Writing—review and editing. Alex S. Qian—Data curation and formal analysis, Writing—original draft, Writing—review and editing. Abhishek Kumar—Conceptualization/design, Data curation and formal analysis, Writing—review and editing. Edmund M. Qiao—Data curation and formal analysis, Writing—review and editing. Tyler F. Stewart—Conceptualization/design, Writing—review and editing, Supervision. Brent S. Rose—Conceptualization/design, Writing—review and editing, Supervision. Ryan K. Orosco—Conceptualization/design, Writing—review and editing, Supervision.
Presentation
Accepted for Oral Presentation at 125th AAO-HNSF 2021 Annual Meeting & OTO Experience: October 5th, 2021.
Data Availability
The datasets generated during and/or analyzed during the current study are not publicly available due to secure/restricted access to the national Veterans Affairs database but are available from the corresponding author on reasonable request.
