Abstract
Pseudomonas peritonitis is often severe and associated with less than 50% complete cure rate, often requiring catheter removal, and transfer to HD. International guidelines recommend that peritoneal catheter should be removed if peritoneal dialysis (PD) effluent does not clear after 5 days of appropriate antibiotic therapy defining the episode as refractory peritonitis. To avoid the shift to hemodialysis (HD), the simultaneous removal and replacement of the peritoneal catheter (SCR) has been employed to treat recurrent peritonitis or tunnel infections associated with peritonitis, obtaining satisfactory outcomes. However, the use of SCR is still controversial in refractory episodes. At present there is growing evidence that refractory peritonitis can be sustained by bacterial adherence along the intraperitoneal portion of the catheter, especially when Pseudomonas species are involved. We describe a case of refractory peritonitis sustained by P. aeruginosa that after a partial response to antibiotics has been successfully treated by SCR.
Keywords
Introduction
In patients on peritoneal dialysis (PD) peritonitis continues to be the main cause of PD-related death, and the leading reason of transfer to hemodialysis (HD). 1
Usually, the initiation of appropriate antibiotic treatment leads to clinical improvement within 72 hours. Therefore, ISPD guidelines recommend catheter removal if PD effluent does not clear after 5 days of adequate antibiotic therapy defining the episode as “refractory peritonitis.” 2
Instead of mandatory PD catheter removal, it is also suggested that observation for antibiotic effect longer than 5 days is appropriate if PD effluent white cell count (WBC) is decreasing toward normal, but a definite cut-off time has not been established. 2
Pseudomonas peritonitis is a severe infection that in more 50% of the cases requires catheter removal, the interruption of PD, and the transfer to HD with the creation of a temporary vascular access. 3 As reported by retrospective studies, the insertion of a new peritoneal catheter at least 2 weeks after catheter removal is performed in less than 40% of the patients. 3
In order to avoid the shift to HD, the simultaneous removal and replacement of the peritoneal catheter (SCR) has been employed to treat recurrent peritonitis or tunnel infections associated with peritonitis obtaining encouraging outcomes.4,5 However, the use of this approach is still controversial in unresponsive episodes, particularly when Pseudomonas aeruginosa (P. aeruginosa) is isolated. 6
We describe a case of refractory peritonitis sustained by P. aeruginosa that after a partial response to antibiotic therapy has been successfully treated by SCR.
Clinical case
An 80-year-old white man suffering from end-stage renal disease (ESRD) due to undetermined cause started three exchanges (1.36% glucose) continuous ambulatory PD in June 2020 with no complications.
His medical history is notable for two non-metastatic lumbar melanoma (2008 and 2015), arterial hypertension well controlled with medical therapy, colonic diverticulosis, and invasive squamous cell carcinoma of the lung treated with right upper pulmonary bilobectomy in 2018.
In September 2022 after a diagnosis of papillary urothelial carcinoma of the bladder, the patient underwent a surgical radical cystectomy with urinary diversion (bilateral ureterocutaneostomy).
At the end of December 2022, the patient presented at the PD clinic reporting the appearance of cloudy peritoneal fluid and complaining mild abdominal pain, nausea, and vomiting. At physical examination the abdomen was soft, tender, not distended, and at the exit-site no presence of erythema or purulent discharge was observed.
The microscopic exam (field at magnification 400× (HPF)) of the peritoneal liquid detected 80–100 WBCs every HPF (normal value 2 WBC every five HPF), 7 while the blood test results showed: WBCs 9.68 × 109/µL, CRP 24.17 mg/dL (normal range <0.5), creatinine 7.1 mg/dL, BUN 84 mg/dL, and Hb 10.5 g/dL. In addition, ultrasonography did not detect any anechoic collection along the tunnel of the catheter. A culture test was performed and empirical therapy with ceftriaxone (1 g/die) ip and vancomycin (20 mg/kg) ip was started.
The following day in the peritoneal fluid 40–50 WBCs every HPF were counted. However, the patient continued complaining diffuse abdominal pain and positive Blumberg sign appeared. Blood exams showed: WBCs 8.5 × 109/µL and CRP 20.99 mg/dL. P. aeruginosa was identified in peritoneal liquid culture. In the light of the poor clinical response, the patient was admitted to the nephrology department. Considering the antibiogram the antibiotic therapy was changed to tobramycin (0.6 mg/kg/die) ip and meropenem (500 mg/die) iv.
Despite the targeted therapy along with the reduction of inflammation indexes (blood exam results after 1 week from the onset: WBCs 12.4 × 109/µL, CRP 5.15 mg/dL) and WBCs in the peritoneal liquid (20–30 WBCs every HPF) the patient remained frankly symptomatic. The liquid peritoneal culture became negative. To exclude unknown abdominal foci of infection, a CT scan was performed, which did not identify any abscess collections.
At the beginning of January, the clinical conditions were exacerbated by an episode of hyperthermia (t° 38.5) and worsening of the abdominal pain. Surprisingly, the day after the patient experienced a spontaneous regression of the acute symptoms.
After 14 days of adequate antibiotic therapy, there were still 10–20 WBCs in the peritoneal liquid and the blood exams showed: WBC 13.7 × 109/µL, CRP 2.25 mg/dL. The patient expressed the immovable will to not be transferred even temporarily to HD. Thus, it was decided to perform SCR. For the fear of the presence of abdominal adherences due to the recent urological operation, it was preferred to insert the new catheter through the old peritoneal access. In addition, a double purse-string between the deep-cuff and the posterior rectus sheath was crafted (Figure 1). The maneuver took place without complications and PD exchanges were resumed soon after the procedure (Figure 2). 8 Since the first day after the replacement of the catheter the WBC count in the peritoneal liquid decreased to 0–1 every HPF and the inflammatory index started to normalize (blood exams 5 days after the SCR: WBC 7.67 × 109/µL, CRP 0.8 mg/dL) along with the resolution of peritonitis signs. The microbiological exam carried out on the superficial and deep cuff as well as the tip of the catheter resulted negative.
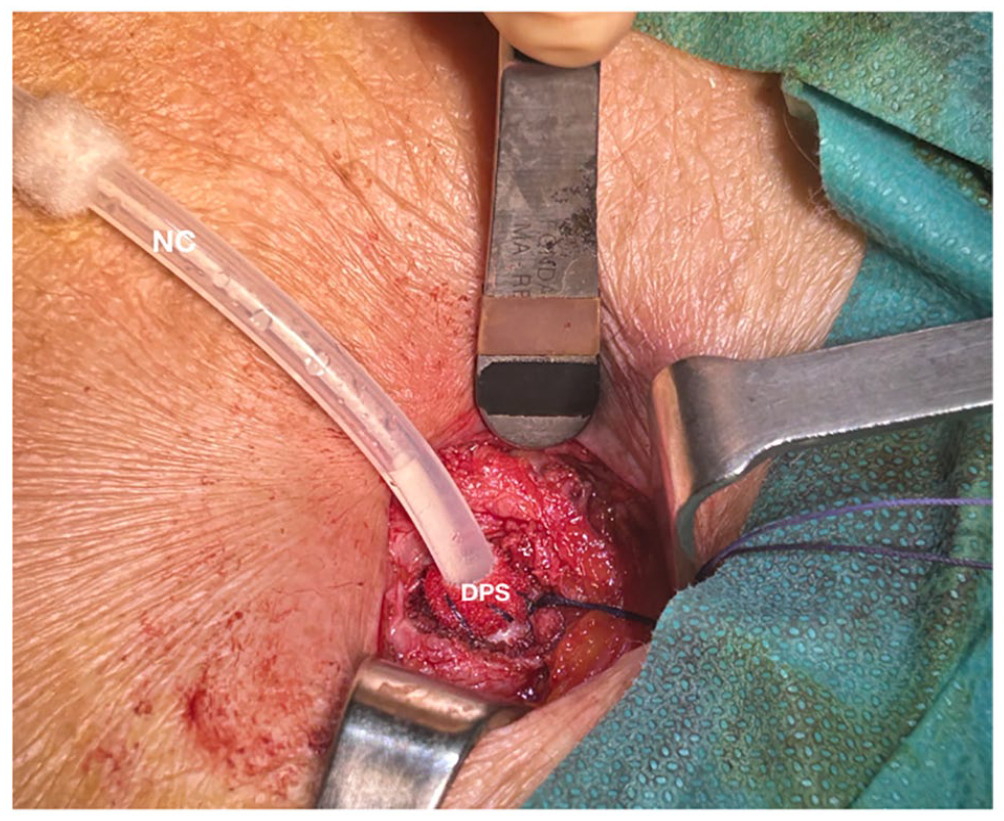
The new catheter was inserted in the peritoneal cavity through the hole of the old catheter considering the recent major intervention; A double purse-string was crafted between the posterior rectus sheath and Dacron material of the deep cuff to resume peritoneal exchanges soon after catheter replacement and minimize the risk of early pericatheter leakage.
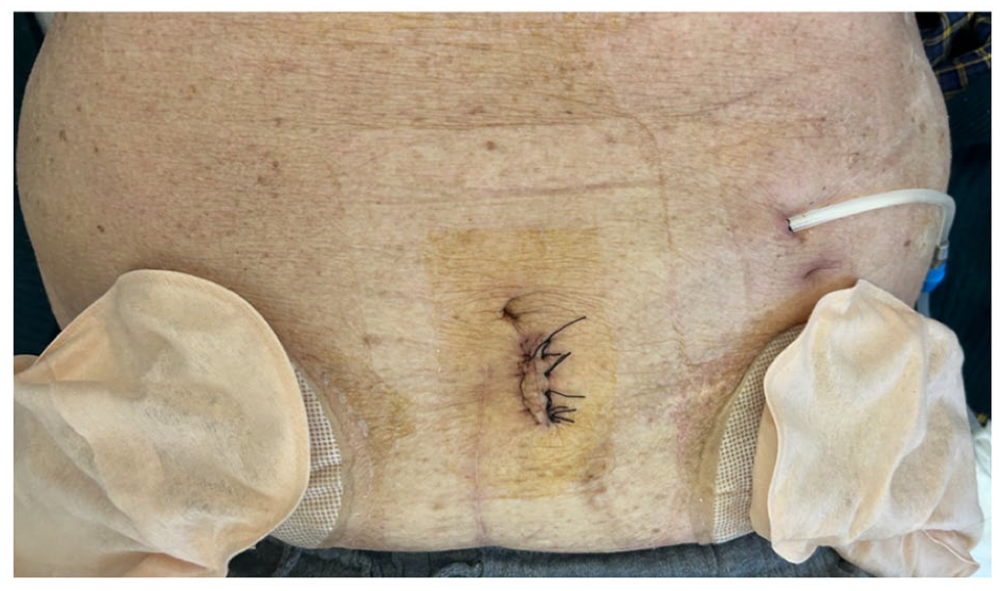
The new exit-site was created in the left side of the abdomen above the old one to increase the distance between the catheter and the ureterocutaneostomy.
The patient was dismissed 10 days after the SCR, with a WBC count in the peritoneal liquid equal to 1 every 10 HPF (Figure 3). This result was confirmed 1 week later during the ambulatory follow-up visit.
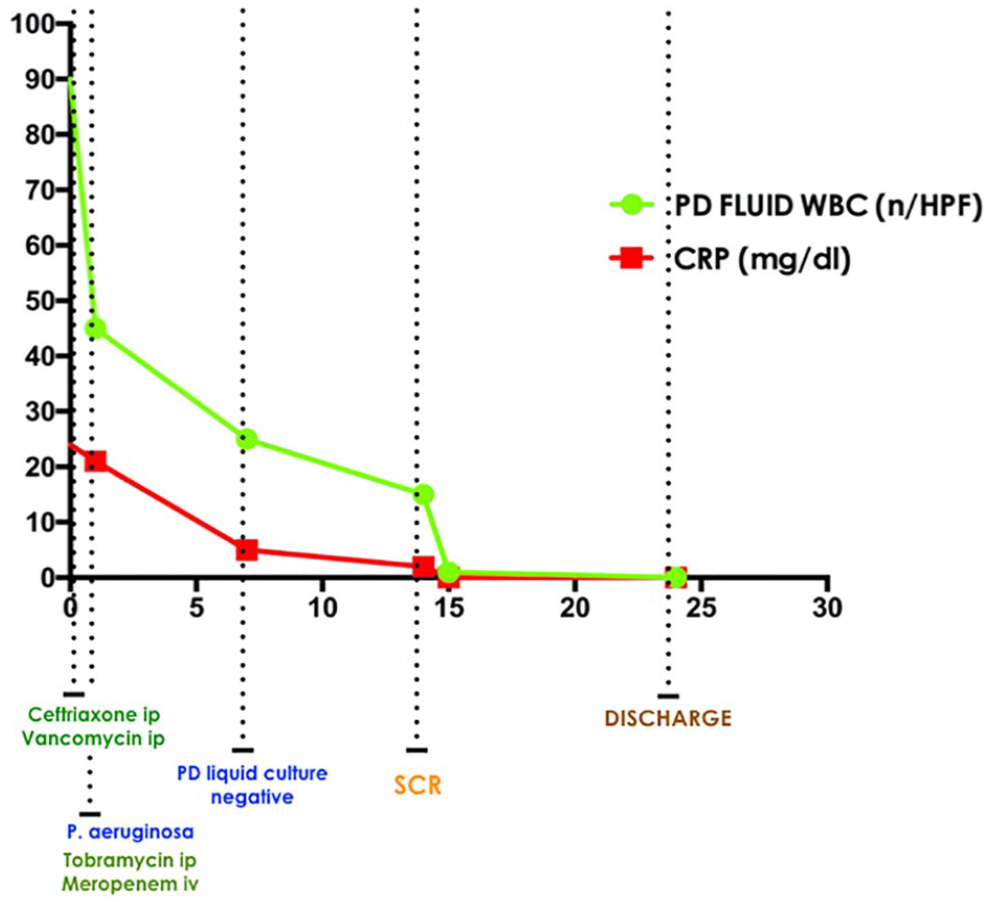
Graphical visualization of white blood cells number in the peritoneal effluent (PD FLUID WBC, number (
Discussion
This case report shows the successful use of SCR as treatment for refractory P. aeruginosa peritonitis in a patient with persistence of a cloudy dialyzate and unquestionable will to not be transferred to HD. Notably, this approach allowed to spare the patient the psychological trauma associated with a change in dialysis modality, and the placement of a central venous catheter, procedure associated with a greater probability of infection, thrombosis, stenosis of the central vessels, and increased mortality. 9
In case of mechanical complications, the employment of SCR has never been questioned. 4 On the other hand, the use of SCR to resolve PD related infections is still debated.
However, excellent results by using SCR as treatment either for tunnel infections with secondary peritonitis or in recurrent peritonitis after resolution of the clinical signs and normalization of WBC count were reported in several studies. 4
In the first case the peritonitis is secondary to the extraluminal spread of the infection along the subcutaneous tunnel until the peritoneal cavity; while in the latter case, an unidentified bacterial colonization of the device determines new infective episodes as soon as the antibiotics are discontinued. In both conditions the removal of the catheter permits the elimination of the cause that sustains the infectious process permitting its complete resolution.
In case of refractory peritonitis, the use of SCR has been discouraged as reported by Cancarini et al. 6 who observed a successful rate less than 20%.
Several causes were hypothesized for these failures, such as differences from in vitro to in vivo effect of antibiotics, presence of organisms that rapidly colonizes the new catheter, intra-abdominal source of infection other than PD catheter (e.g. diverticulosis), or biofilm formation along the intraperitoneal portion of the catheter.
At present there is growing evidence that refractory peritonitis is commonly associated with either bacterial adherence to the catheter silastic or biofilm formation along the tunnel/intraperitoneal portion of the catheter. 10 In the case of P. aeruginosa refractory peritonitis without tunnel infection, it is important to distinguish the episodes that are sustained mainly by catheter colonization. In fact, P. aeruginosa forms confluent and mature biofilms on silastic that could be eradicated only by the removal of the peritoneal catheter. 10
As observed in our case, the initial laboratory response associated with the partial improvement of the clinical signs could represent the good antibiotic efficacy in vivo.
However, the persistence of a cloudy dialyzate after 14 days of an appropriate antibiotic therapy prompt us to suspect a diagnosis of peritonitis associated with biofilms along the catheter rather than a bowel source of infection. As a fact, the removal of the catheter led to the clearance of the WBC into the dialyzate. Furthermore, the occurrence of acute exacerbation of the infectious process could indirectly indicate the results of continuous shedding of microorganisms from the device into the peritoneal cavity corroborating the presence of catheter contamination. 10
In case of infectious complications, we previously recommended the insertion of the new catheter in the opposite side of the old one and to perform the “clean step” (insertion) before the “dirty step” (removal). 5 However, in the light of the recent major urological intervention, we considered more reasonable to use the same peritoneal opening to place the catheter tip in a position as close as possible to the original one and preserve the excellent catheter function.
In conclusion, our case suggests that in case of refractory peritonitis sustained by P. aeruginosa, SCR could spare the patient the temporary shift to HD and allow to continue PD treatment. However, it is important to underline that the procedure was performed, albeit with the persistence of a cloudy dialyzate, in the presence of a negative culture of the PD effluent and in absence of clinical signs (e.g. abdominal pain, rebound tenderness, fever).
Footnotes
Acknowledgements
None.
Authorship
Research idea and study design: LN, AS; data acquisition: LN, MS, FT; data analysis/interpretation: LN, AS, CA, VS; supervision or mentorship: GC, AS. Each author contributed important intellectual content during manuscript drafting or revision and accepts accountability for the overall work by ensuring that questions pertaining to the accuracy or integrity of any portion of the work are appropriately investigated and resolved.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
No required (retrospective case report undertaken through a case note review).
Informed consent to participate
Informed consent obtained by the patient.
Informed consent to publish
No doubt that anonymity can be maintained.
