Abstract
Eosinophilic peritonitis (EP) is a well-described complication of peritoneal dialysis that occurs because of an overreaction to constituents that are related to the catheter or tubing, peritoneal dialysate, pathogenic infection, or intraperitoneal drug use. EP caused by antibiotic use is rare. We present the case of a patient with cefoperazone and sulbactam-related EP. A 59-year-old woman who was undergoing peritoneal dialysis presented with peritonitis with abdominal pain and turbid peritoneal dialysis. Empiric intraperitoneal cefazolin in combination with cefoperazone and sulbactam was started after peritoneal dialysis effluent cultures were performed. Her peritonitis achieved remission in 2 days with the help of cephalosporin, but she developed EP 1 week later, when her dialysate eosinophil count peaked at 49% of the total dialysate white blood cells (absolute count, 110/mm3). We excluded other possible causes and speculated that cefoperazone and sulbactam was the probable cause of EP. The patient continued treatment with cefoperazone and sulbactam for 14 days. EP resolved within 48 hours after stopping cefoperazone and sulbactam. Thus, EP can be caused by cefoperazone and sulbactam use. Physicians should be able to distinguish antibiotic-related EP from refractory peritonitis to avoid technique failure.
Keywords
Introduction
Eosinophilic peritonitis (EP) was first described in 1968 by Lee and Schoen. 1 It is defined as the presence of >100 eosinophils/mm3 or >10% eosinophils in the total non-erythrocyte count in the peritoneal dialysis (PD) effluent.2–4 This happens because of an overreaction to constituents that are related to the catheter or tubing, peritoneal dialysate, pathogenic infection, or intraperitoneal drug use. EP caused by antibiotic use has been rarely reported.
Cefoperazone is a third-generation cephalosporin antibiotic with a broad spectrum of activity against most Gram-positive and Gram-negative bacteria. An
Case report
A 59-year-old Chinese woman presented with diarrhea, abdominal pain, and turbid dialysate after eating contaminated food, and she was admitted to the hospital. Her medical history showed that she developed chronic renal disease due to primary glomerulonephritis and started PD 5 years previously, in March 2014. She underwent incremental continuous ambulatory peritoneal dialysis (CAPD) to achieve the target of solute clearance and volume balance during the loss of residual renal function by exchanging the dialysate (Baxter Healthcare, Shanghai, China) four times, with the volume gradually increasing. She was diagnosed with multiple gallstones and subsequently had an episode of peritonitis due to
Her body temperature was normal. On physical examination, the patient’s abdomen was slightly tender, without signs of an exit-site infection. Laboratory findings included the following: peripheral leukocyte count, 12,460/mm3 with 89.1% neutrophils, 7.1% lymphocytes, 3.5% monocytes, and 0.1% eosinophils; Hb, 12.5 g/dL; platelets, 22,000/mL; high-sensitivity C-reactive protein (hsCRP) level, 5.13 mg/L; blood urea nitrogen, 8.09 mg/dL; serum creatinine, 8.61 mg/dL; albumin, 3.7 g/dL; and total protein, 8.0 g/dL. Analysis of peritoneal fluid revealed a total leukocyte count (TLC) of 152/mm3 and 75% polymorphs, which increased to 420/mm3 and 80%, respectively, on day 2. Abdominal ultrasound did not show any abnormal signs except for a 0.4-cm polyp in the gallbladder. No bacteria or tuberculosis in the dialysate were shown by the dialysate culture. On the basis of clinical signs and the PD fluid test, peritonitis was diagnosed and treated using empiric intraperitoneal cefazolin in combination with cefoperazone and sulbactam.
On day 3 after the onset of peritonitis, the patient returned with the complaint of persistent symmetrical pruritis in her palms and the soles of her feet, although her dialysate white blood cell (WBC) count decreased to 56/mm3. Allergic reaction to antibiotics was suspected, and thus, oral moxifloxacin at 400 mg per day was started instead of cefazolin, and cefoperazone and sulbactam were continued. Ebastine and fexofenadine hydrochloride were administered to relieve her symptoms. On day 4, the patient’s dialysate culture revealed
A dialysate differential WBC count with a manual eosinophil count was performed on both peripheral blood and the dialysate because of suspected EP. The dialysate showed a WBC count of 110/mm3 with 49% eosinophils, which confirmed the presence of EP, with a normal blood eosinophil count. Dialysate organism examination including bacteria, fungi, and tubercle bacillus was repeated, but there were no positive findings. No parasites or cysts were found upon fecal examination. Her immunoglobulin (Ig)E level was within the normal range, and serological tests for cytomegalovirus (CMV) and Epstein–Barr virus (EBV) showed that the IgM level was within the normal range, and the IgG level was elevated almost four-times the upper limit of normal. Therefore, cefoperazone and sulbactam-associated EP was suspected. Because EP is often self-limited, the patient’s daily ultrafiltration volume was not influenced by EP, and we continued treatment with cefoperazone and sulbactam for 14 days. During this time, intermittent itching persisted and fluid analysis revealed a fluctuation in the patient’s WBC count in the peritoneal effluent from 110 to 220/mm3, and her eosinophil proportions were 18% to 49%. Pruritis was finally alleviated 12 hours after cefoperazone and sulbactam were stopped. Repeat dialysate cell count showed an obvious decrease in eosinophil levels from 25% to only 9% (Figure 1).
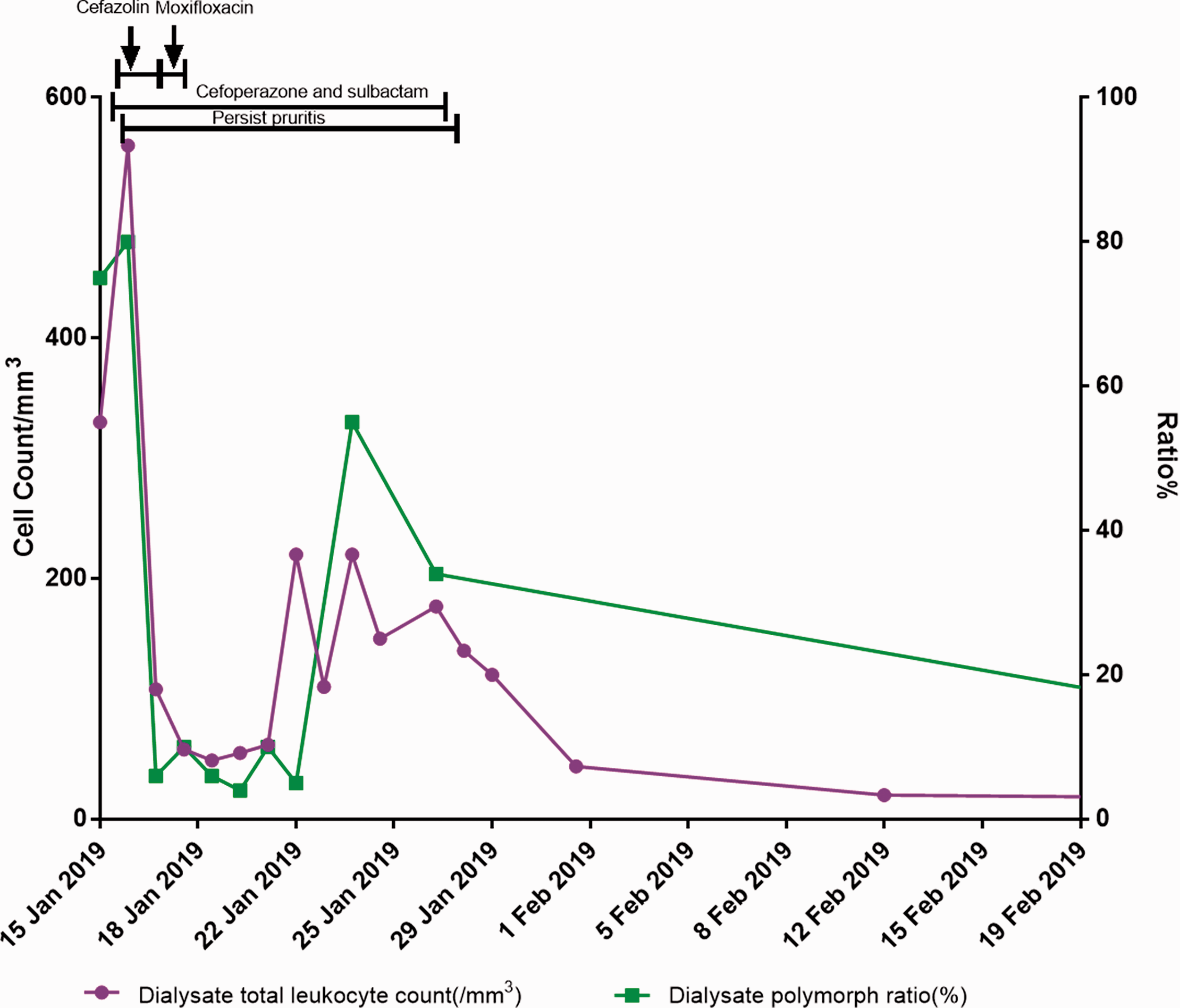
Timeline of dialysate leukocyte change related to antibiotic use.
Discussion
EP is defined as the presence of >100 eosinophils/mm3 or >10% eosinophils in the total non-erythrocyte count in the PD effluent. Most EP episodes develop within 3 months of starting PD and subside within 1 month after stopping treatment modification. EP is probably caused by an overreaction to constituents of the catheter or tubing, peritoneal dialysate, or intraperitoneal drugs such as iodine, heparin, fibrin, icodextrin, or even streptokinase.8–11 Various agents such as bacterial, fungal, or parasitic infection have also been reported in relation to EP.12–14 EP cases attributed to drugs are rare. Three original case reports of antibiotic-related EP in the literature were identified.15–17 (Table 1) Piraino et al.
15
described a series of patients with EP, four of whom were thought to have EP secondary to antibiotic use, but only one woman had EP twice after the initiation phase (in the first 3 months) of dialysis. Moreover, vancomycin-related EP was reported twice in the literature.16,17 All of these three patients were women. They all experienced bacterial peritonitis right before EP, and each of these three patients had a positive
Antibiotic-related eosinophilic peritonitis after the first 3 months: literature review

*, episode 1; **, episode 2
F, female; EP, eosinophilic peritonitis; PD, peritoneal dialysis; WBC, white blood cell; NA, not available.
Attributing causation to an antibiotic is based upon timing that is coincident with antibiotic administration, negative culture results and workup for other etiologies, and remission of EP that is associated with withdrawal of the suspected antibiotic. In our case, cefoperazone and sulbactam use was associated with a notable episode of persistent pruritis, followed by an increasing eosinophil count in the PD effluent despite the general improvement of peritonitis. Screening tests were performed for potential causes such as bacterial and fungal infection and pathogens such as CMV or EBV. Drugs such as cefazolin and moxifloxacin could be excluded because the timing of the drug use did not correspond to EP progression. Because cross-reactivity has been described for cephalosporins in cephalosporin-allergic patients and because intraperitoneal cefazolin was administered together with cefoperazone and sulbactam, we could not completely rule out a role for cefazoline as a trigger of the adverse event. However, because EP was completely alleviated within 48 hours after the withdrawal of cefoperazone and sulbactam, we can deduce that cefoperazone and sulbactam use was the most likely cause of EP in our patient.
Our report has several aspects in common with Rosner et al.’s case. 16 In that case, a 61-year-old woman underwent 4 years of CAPD, and she had peritonitis initially, but it quickly resolved with vancomycin use. She developed EP on day 7 of vancomycin treatment and synchronously achieved palliation with cessation of vancomycin. Oh et al. 18 presented the case of an 84-year-old man who developed self-limited EP several days after the initiation of CAPD. The cause of EP was unclear, but it was probably associated with a hypertensive reaction to some components of the PD system. In our case, however, the patient had already undergone a 5-year course of PD, which should have excluded such a hypertensive reaction. Furthermore, allergic symptoms were consistent with the course of cefoperazone and sulbactam use. A lymphocyte transformation test can theoretically distinguish an allergy cause from a pathogenic cause of EP, but this test is not recommended in the literature.
The third-generation cephalosporin was recommended by the International Society of Peritoneal Dialysis (ISPD) in 2005, 2010, and 2016 as the first-line empiric antibiotic to cover Gram-negative organisms.19–21 Compared with third-generation cephalosporins only, cefoperazone and sulbactam cover a broader spectrum of bacteria and different multidrug-resistant organisms (MDROs),22,23 and they are one of the most common first-line antibiotics. Eosinophilia is also the most frequent adverse reaction to cephalosporins, although anaphylaxis only occurs in extremely rare cases.14,24 It is useful for diagnosing the presence of EP related to cefoperazone and sulbactam, especially to distinguish it from refractory peritonitis. The absence of distinctive clinical improvement after 96 hours of antibiotic treatment is an important clue for refractory peritonitis, which must be treated by catheter removal in a timely manner to avoid technical failure in PD. Conversely, if clinical signs of peritonitis improve with a simultaneous and unexplained trend of WBC counts in the PD effluent, we should suspect the occurrence of EP. There is no consensus on whether administration of the suspected drug should be stopped.
We suggest tailoring the treatment to the individual patient, taking into account the side effects of antibiotics and the necessity for maintenance based on the initial response to the antibiotic and drug sensitivity to bacteria. To date, regular monitoring of the dialysate eosinophil count during the peritonitis episode is not routine. Because the 2016 ISPD update of the peritonitis recommendations did not recommend monitoring the dialysate eosinophil count at the start of peritonitis, we suggest that timely screening of the dialysate eosinophil count should be performed once EP is suspected. Moreover, identifying the underlying cause of EP is also valuable because multifactorial mechanisms such as many intraperitoneal drugs, specific organisms, and non-PD-related allergic reactions have been reported, which are non-antibiotic causes of EP. 14 Allergy testing should be recommended, as it is for a proven drug allergy, and future intraperitoneal and systemic administration of the involved drugs or related chemical compounds may pose a risk for more serious events in PD patients.
To the best of our knowledge, this is the first reported case where EP may relate to cefoperazone and sulbactam, which are commonly used to treat Gram-negative bacterial infections in clinical practice. Physicians should be able to distinguish antibiotic-related EP from refractory peritonitis to avoid technique failure.
Supplemental Material
sj-pdf-1-imr-10.1177_03000605211025367 - Supplemental material for Cefoperazone and sulbactam-related eosinophilic peritonitis: a case report and literature review
Supplemental material, sj-pdf-1-imr-10.1177_03000605211025367 for Cefoperazone and sulbactam-related eosinophilic peritonitis: a case report and literature review by Zi Wang, Zhiying Li, Suping Luo, Zhikai Yang, Ying Xing, Chengwei Pu and Jie Dong in Journal of International Medical Research
Footnotes
Ethics statement
This study conforms to the CARE guidelines. 25
The patient provided consent for treatment. Ethical approval was not required by the Peking University First Hospital because this manuscript only reports a case. However, written informed consent was obtained from the patient, including consent for intervention and for publishing the findings.
Declaration of conflicting interest
The authors declare that there is no conflict of interest.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Author contributions
ZW and SL collected and analyzed the clinical data. ZW performed the literature search and drafted the manuscript. ZL and ZY treated and followed up with the patient throughout the case and acquired the patient’s consent. YX and CP contributed to the acquisition of laboratory data. JD critically revised the manuscript. All authors read and approved the final version of the manuscript.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
