Abstract
Introduction:
This study evaluated a chlorhexidine-coated peripherally inserted central catheter (PICC) and the incidence of associated complications within both inpatient and outpatient populations.
Methods:
This IRB-approved, multicenter, prospective observational study was performed at three large teaching hospitals in the US. All adults who required a PICC for ⩾14 days were considered. Patients were monitored throughout entire catheter dwell. Duplex venous ultrasounds were performed before insertion, after 10 to 14 days of dwell time, and upon removal. Data was collected from the hospital, outpatient clinic, and patient PICC diary records.
Results:
A total of 103 patients, 56% male, with mean BMI 29 ± 8.8, were enrolled. The majority (79%) of patients were from high-risk groups—cancer, infectious diseases, transplant, and trauma. Primary treatment indications were antibiotics (66.99%) and chemotherapy (25.24%). Double lumen PICCs (59.2%) were favored clinically, as was basilic vein placement (71.84%). Mean catheter dwell was 47.01 ± 25.82 days. Three (3, 2.9%) central line-associated bloodstream infections (CLABSI) were reported. Four patients (4.6%) reported symptomatic catheter-related thrombosis (CRT), confirmed with ultrasound. Three patients (3.4%) had ultrasound-confirmed fibroblastic sleeve (FS). Eight patients (9.2%) who entered the study with pre-existing superficial thrombosis, had complete resolution at the time of catheter removal. The incidence of CLABSI was 0.82/1000 days. The combined CRT and FS rate was 6.9%.
Conclusion:
Based upon the observational findings of this study, chlorhexidine-coated PICC technology may be considered for use in patient populations who are at moderate to high-risk for catheter-related complications in both inpatient and outpatient settings.
Keywords
Introduction
Placement of a peripherally inserted central catheter (PICC) is recommended when central venous access is required for greater than 14 days. 1 PICCs are used for a variety of clinical indications, such as the administration of antibiotics, fluids, parenteral nutrition, and chemotherapy. 2 Industry analysts estimate that over 2.7 million PICCs are placed in the United States every year. 3
Central line-associated bloodstream infection (CLABSI) and catheter-related thrombosis (CRT) are common PICC-associated complications that occur despite employment of best practices during insertion and ongoing care.4,5 The downstream sequelae of CLABSI and CRT, such as catheter exchange, administration of additional medication therapies, or escalation in level of care, can add both to morbidity and to healthcare expense.4,5 Infection and thrombosis may have an underlying reciprocating relationship, and their association is a topic of ongoing clinical interest and technical research.6,7
An important technical innovation which mitigates the risk of catheter colonization, a major precursor to infection, is the impregnation of catheters with chlorhexidine, a broad-spectrum biocide, which in addition to being bacteriostatic and bactericidal, has a direct thrombin inhibitor effect. 8 Centrally inserted central catheters (CICCs) impregnated with chlorhexidine, first introduced in 1990, have since shown in numerous clinical studies, to significantly reduce microbial colonization, a precursor to CLABSI.9–11
A chlorhexidine-coated PICC (Arrowg+ard® Blue Advance® PICC, Arrow International, Morrisville, NC, referred to as “AGBA-PICC” hereafter), obtained Food and Drug Administration (FDA) 510(k) clearance for human use in 2011. In vitro studies of chlorhexidine-coated catheters have previously demonstrated reductions in microbial colonization, thrombus accumulation, and in bacterial growth inhibition on catheter surfaces for up to 30 days, when compared to plain or untreated catheters. 12 Chlorhexidine-coated catheters have also demonstrated smaller fibroblastic sleeves (FS) and improved bacterial inhibition when compared to the other studied devices.12,13 A recent study (Bludevich et al.) 14 reports an association between CRT and CLABSI in post-surgical children, with the strength of the association varying with catheter type, centering on tunneled CICCs only. In cases of CRT associated with CLABSI (21%), the diagnosis of CRT preceded diagnosis of CLABSI by a median of 7 days. Some publications, including a 2020 systematic review by Huang et al. 15 report potential association between CLABSI and CRT, also citing pathophysical changes 16 and inflammation as associated causes. 17 A 2017 systematic review and meta-analysis by Kramer et al. 18 also determined that antimicrobial PICCs had a significant effect on CLABSI reduction when compared with non-coated PICCs (albeit without finding a significant difference between antibiotic vs chlorhexidine-coated devices).
While several clinical studies have reported reductions in CLABSI with the use of the ABGA-PICC, its effect on the incidence of CRT has not been reported.19,20 In this study, the authors prospectively evaluated the incidence and timing of both CRT and CLABSI associated with the use of the AGBA-PICC. The primary hypothesis was that the AGBA-PICC has a lower incidence of symptomatic and asymptomatic CRT, intraluminal occlusion, and CLABSI compared to that observed in previous trials involving non impregnated PICCs.
Methods
A prospective, observational, multicenter study was approved by the Institutional Review Boards of all participating sites, three major teaching facilities in the United States. One hundred and three subjects across all three teaching hospitals were enrolled during the study period. Inclusion criteria for study participation were: age of 18 years or greater, ability to comply with the study schedule of venous duplex ultrasound, and ability to maintain a standardized PICC diary. The following exclusion criteria included: a history of deep venous thrombus (DVT) within the preceding 3 months, stenosis or occlusion at or proximal to the elbow or knee, a hypercoagulable state (unless this diagnosis was the primary indication for AGBA-PICC placement), previous enrollment, pregnancy, breast feeding, ipsilateral axillary lymph node dissection, focal disease at or within 15 cm of catheter insertion site, and stage II or higher kidney disease (unless cleared by consulting nephrologist). All participating sites followed the same study protocol for patient recruitment. All sites utilized the same insertion techniques and post-insertion maintenance.
Patients in this study received either a single or dual lumen AGBA-PICC. PICCs were inserted proximal to the antecubital fossa using ultrasound guidance, by either interventional radiologists or interventional nurse practitioners. Device insertion followed the Institute for Healthcare Improvement (IHI) recommended strategies to prevent infection (skin antisepsis with 2% chlorhexidine and 70% isopropyl alcohol; use of maximal barrier precautions/personal protective equipment; selection of an appropriate site/vessel for venous access) and protect against CRT (ultrasound-guided puncture of the vessel; use of micropuncture, assessment of catheter to vessel ratio; intra-procedural tip location using fluoroscopy).
The patients were provided a diary to record daily information regarding their PICC interactions throughout the study enrollment period. All participants were instructed to document the following data: date of all dressing changes, any bleeding or discharge from around the insertion site, any pain, swelling or redness in the cannulated arm, all dates and times of PICC flushing procedures, and any other patient-related issues. The study investigators also contacted all outpatient study participants via telephone every other week until PICC removal, as well as 1 week after PICC removal (all ± 2 days). An outpatient participant was considered “lost to follow-up” if three attempts at contact via telephone (landline or cellular) or by certified mail failed.
During this study, the patency of each PICC lumen for hospitalized or rehospitalized patients were documented at least every 8 h and before each use. Participants receiving outpatient care had the patency of their PICC lumens documented before each use, with all dressing changes, and at routine follow-up visits with their healthcare providers. Assessment of patency consisted of a brisk free-flow blood aspiration and a forward flush with no resistance using a 10 mL syringe of 0.9% Saline. If a PICC could only be flushed (without blood aspiration), it was considered to be partially rather than fully occluded. PICCs that failed to either aspirate or flush were deemed to be completely occluded. In the absence of any mechanical cause or issue, any partially occluded PICCs were treated with administration of a thrombolytic agent, in accordance with the facility policy and manufacturers instructions for use. Patients’ medical records spanning the entire PICC dwell period were obtained for each study participant. These included hospital, outpatient clinic, infusion center, or homecare agency. These records were reviewed for all laboratory results, radiological findings, parenteral therapies, or any complications related to the PICC.
Indications for PICC placement and removal, catheter French (Fr) size and length, patient compliance, and adverse events where recorded, which included signs of infection and CRT. Clinical demographics collected included: age, sex, body mass index (BMI), all medication regimes, complete blood count (CBC), partial thromboplastin time (PTT), international normalized ratio (INR), and blood culture data (see Table 1). Bloodstream infection (BSI) without an obvious source, such as pneumonia or urinary tract infection, was automatically considered to be a PICC complication. Ultrasound of the ipsilateral upper extremity was performed pre-PICC insertion (enrollment screening), at days 10–14, and within 24 h or less of device removal (unless impracticable). Venous thrombosis was considered to be CRT if visualized in the superior vena cava or the brachiocephalic, subclavian, axillary, basilic, or brachial veins.
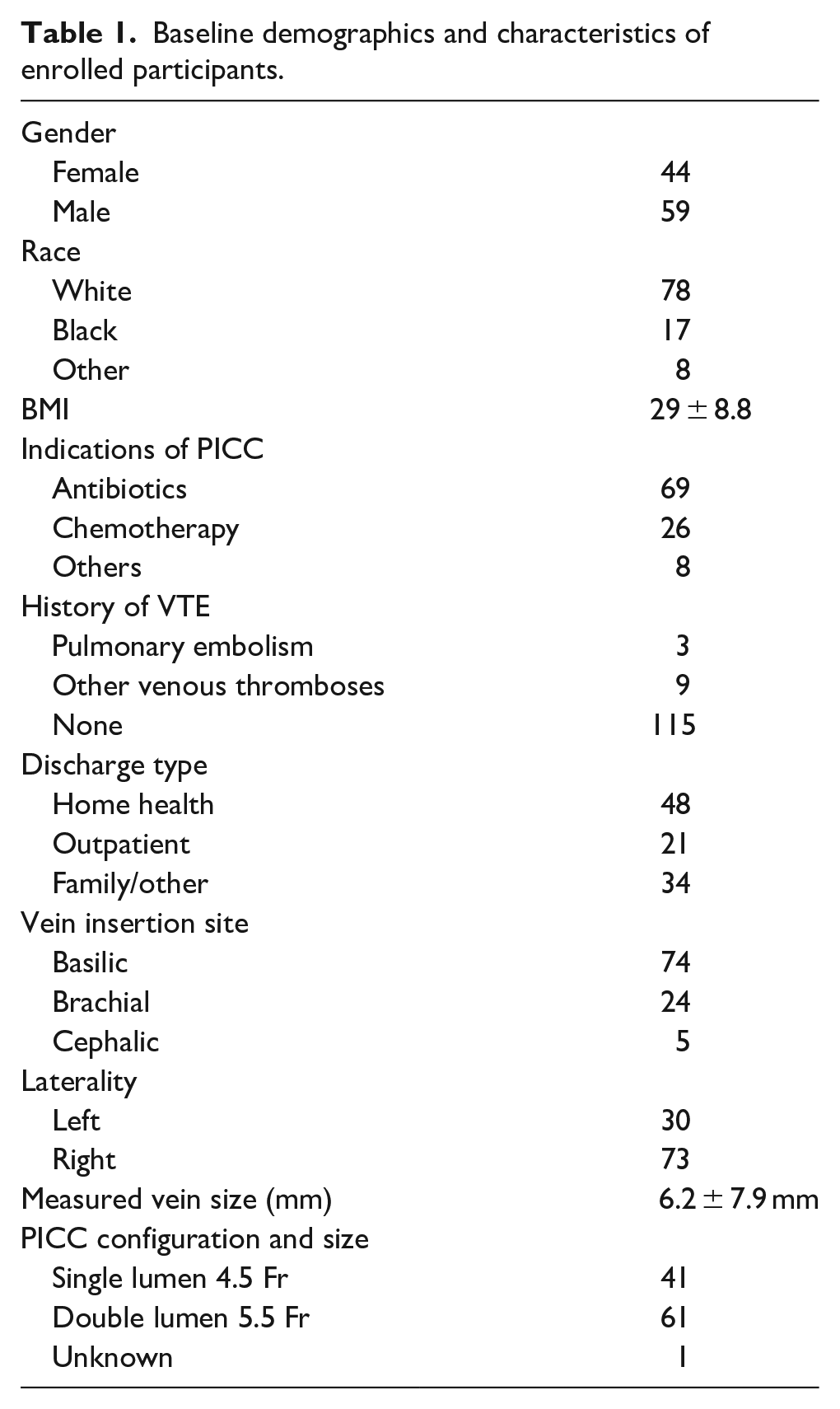
Baseline demographics and characteristics of enrolled participants.
Statistical analysis
The primary study endpoints were the occurrence of CLABSI and CRT, including any removal or exchange of a PICC related to a complication. Secondary endpoints included PICC patency and need for use of a thrombolytic or declotting agent. Descriptive statistics were used to summarize all baseline clinical characteristics, technical details, and outcomes. Continuous outcome variables are presented as mean, medians, and standard deviation. Categorical outcome variables are presented as relative frequencies. Chi-square and t-tests were used to compare categorical and continuous data respectively. Time-to-event endpoints where defined as time to first intervention for thrombosis, catheter occlusion, or CLABSI. These endpoints were analyzed using Kaplan–Meier methods. Log-rank tests were used to identify clinical variables associated with complications. Incidence rate of interventions per 1000 catheter-days was calculated by dividing the total number of interventions by the total number of days of catheter dwell time for all subjects and multiplying the result by 1000. Statistical analysis was performed with STATA 15.1 statistical software (STATA Corp., College Station, TX, USA).
Results
A total of 103 patients were enrolled and received an AGBA-PICC. Demographic data displayed the following information; female (44, 42.7%), male (59, 57.3%). Mean patient BMI was 29 ± 8.8. Indications included infectious disease (43, 41.7%), cancer (30, 29.1%), recent surgery (12, 11.7%), and other diagnoses (28, 27.2%). Antibiotics (69, 67.0%) and chemotherapy (26, 25.2%) administration were the most common indications for PICC use. Double lumen 5.5 Fr PICCs (61, 59.2%) were most frequently placed. The basilic vein (73, 71.6%) was the most common deep vessel used for PICC insertion. Post-PICC insertion, study participants were discharged to home health (48, 46.6%), to outpatient therapy (26, 20.38%), into family care (15, 19.4%), and the remaining patients (14, 13.5%) continued with inpatient therapies for the entire course of treatment. Six (5.8%) of patients were lost to follow-up. Ten (13%) PICCs remained indwelling and fully functional at day 90. Overall, the mean catheter dwell was 39 days.
Central line-associated blood stream infection (CLABSI)
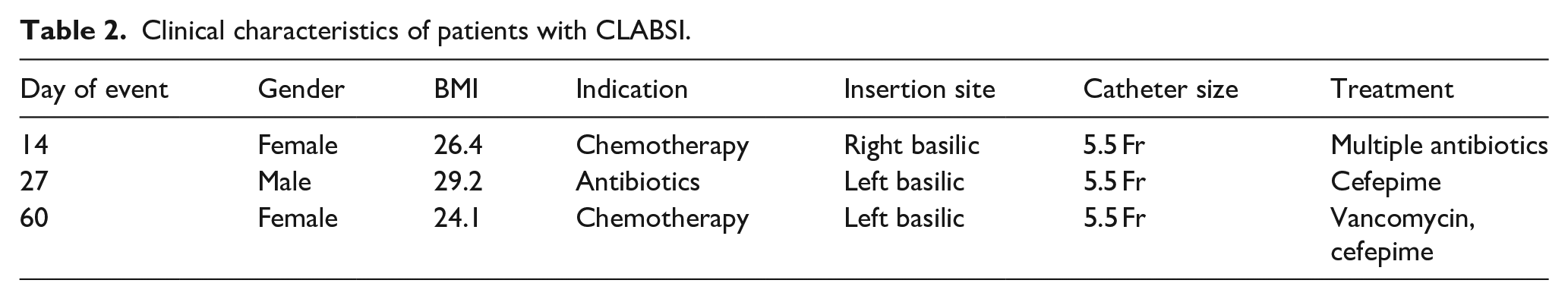
Three (2.9%) patients had laboratory confirmed CLABSI at day 14, 27, and 60 respectively (see Table 2). In each of these instances, the PICC was inserted via the basilic vein. None were discovered to have concomitant DVT diagnosed, and all cases resolved with short courses of antibiotics. The incidence rate for CLABSI with clinical manifestations was 0.82/1000 days.
Clinical characteristics of patients with CLABSI.
Catheter related thrombosis (CRT)
Ultrasound at 10–14 days post-PICC-insertion was completed in 87/103 (84%) patients. Vessel narrowing or non-occlusive thrombosis was observed in 36/87 (41%) patients at day 10–14. Ultrasound within 24 h prior or post PICC removal were performed in 56/87 (64.4%) patients. Whereas 26/56 (46.4%) had non-occlusive thrombosis, none (0%) had complete occlusive thrombosis. Four patients (4.6%) reported symptomatic catheter-related thrombosis (CRT), confirmed with ultrasound. Interestingly, eight (9.2%) patients with asymptomatic deep venous thrombosis at the initial enrollment screening had complete thrombosis resolution on follow-up scanning at PICC removal. Logistic regression analyses did not reveal any associations between CRT and the clinical variables studied (see Table 3).
Univariate logistic regression analysis of suspected CRT based on clinical variables.
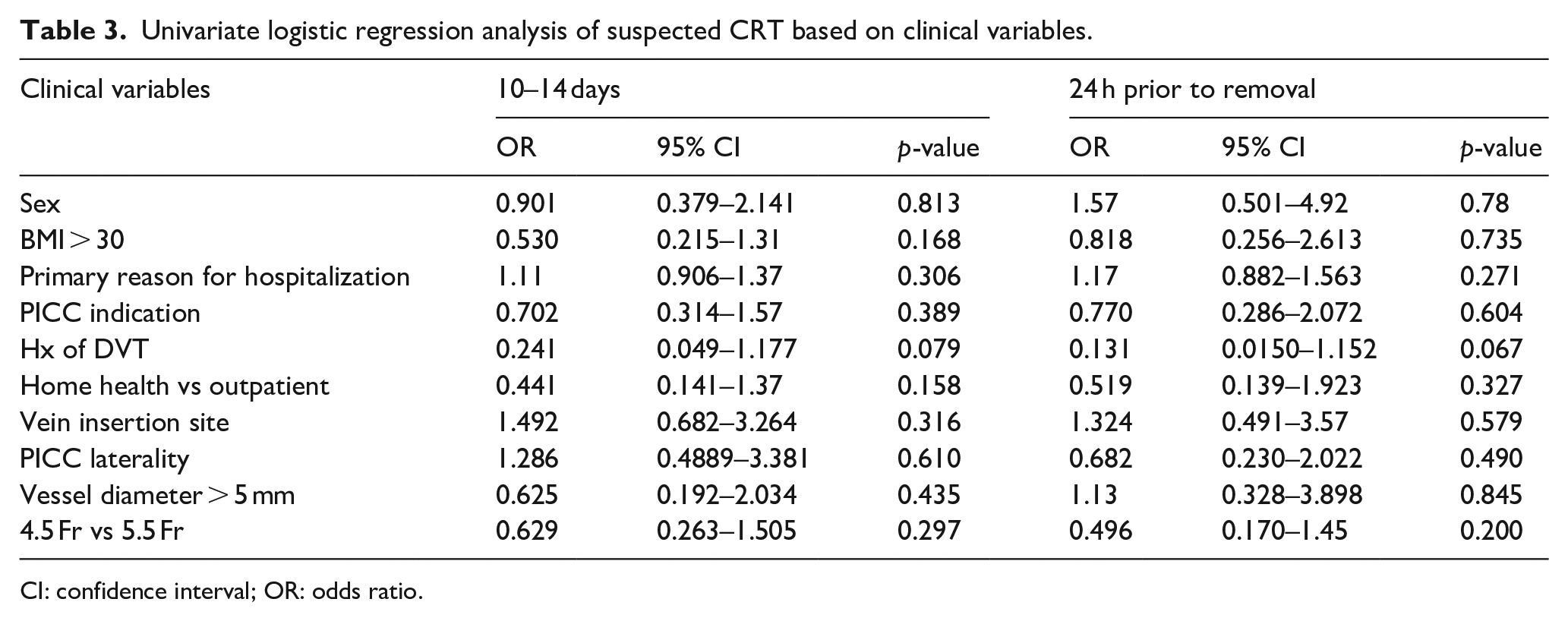
CI: confidence interval; OR: odds ratio.
Catheter patency
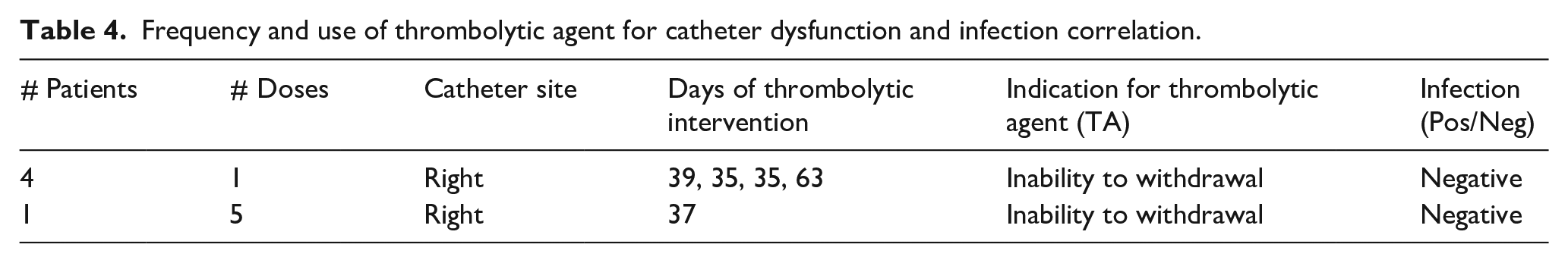
A clinical assessment of either catheter occlusion (CO) or partial withdrawal occlusion (PWO) occurred in one and in five patients respectively (total of 6/79, 7.6%) after an average dwell time of 36.3 days (see Table 4). The patient with the complete catheter occlusion had their PICC removed on day 9. The five patients with PICCs with PWO received a thrombolytic agent to restore lumen patency. Three patients with PWO (3.4%) had ultrasound-confirmed FS. Of these five patients who required the administration of a thrombolytic agent to treat PWO, the average time to first dose was 41.8 days.
Frequency and use of thrombolytic agent for catheter dysfunction and infection correlation.
Discussion
Microbial colonization and contamination of a PICC on both intra- and extra-luminal surfaces may occur at any time during insertion, or through subsequent use from environmental factors, patient skin, or healthcare providers.1,4,5 Introduction of a PICC into a vein also triggers a series of pathophysiological interactions, including activation of contact and complement factors, platelet activation, and leukocyte adhesion at the venous intima and the luminal and non-luminal surfaces of the PICC, creating a potential opportunity for both CLABSI and thrombosis development, where progression of one of these phenomena could possibly promote the progression of the other.8,9,22–24
Clinical studies have demonstrated impactful microbial colonization reduction with use of chlorhexidine-coated PICCs,19,20 It has also been demonstrated in animal studies catheter impregnation with chlorhexidine may reduce FS accumulation and reduces rates of catheter occlusion. Sylvia et al. 12 examined the use of a chlorhexidine-coated PICC and demonstrated a reduction in FS weight (64%) and length (66%) when compared to plain/uncoated PICCs in an ovine model, even though such difference was found to be not statistically significant. Presumably, this effect occurs prior to complete transformation of thrombus into a mature FS. It may be related, in whole or part, to the bacteriocidal effects that chlorhexidine offers, but requires further investigation. 8 An important exception to the above is a randomized study by Storey et al. 20 that found no statistically significant difference in incidence of CLABSI with impregnated PICCS relative to plain/uncoated PICCs. However, this study was unblinded and underpowered: out of 167 line insertions only three CLABSI occurred. 21 Furthermore, all patients with difficult vascular access were excluded, removing many patients with compromised immunity from consideration.
Multiple risk factors have been associated with a higher risk of CRT, including older age, hospitalization, insertion in the subclavian vein, left-sided insertions, longer duration of catheter, catheter-to-vein ratio >0.45.22–30 Balsorano et al. 31 attributes insertion technique (ultrasound guidance, appropriate catheter size choice, proper verification of tip location and catheter securement) as significant preventative factors, and determined that PICC-related complications had been reduced by the adoption of evidenced-based interventions (i.e. ultrasound-guided vein puncture, catheter to vessel ratio <30%, micro-introducers, novel materials, sutureless securement devices, healthcare professionals training)—strategies that were also used within this study.
Much published literature related to thrombosis in the setting of PICCs has been based on qualitative clinical criteria, and likely underestimates the true incidence of CRT or FS formation.7,23–25 In this study, the authors endeavored to use ultrasound to assess and standardize criteria for documentation of CRT, and to compare our results with those of similar ultrasound based studies.32,33 This study identified asymptomatic CRT in 41% of patients using frequency interval venous duplex ultrasound. Three patients with PWO had ultrasound-confirmed FS. Two of the patients with PWO were treated successfully with a thrombolytic agent and had no evidence of FS and a functional catheter immediately prior to their removal US. Evans et al. 34 previously identified that approximately 15.4% of catheters required a thrombolytic agent to treat withdrawal occlusion.
Previously identified studies, where ultrasound was used to diagnosis thrombosis (symptomatic and asymptomatic), the overall rates of CRT ranged between 37% and 58%.31,32 A 2-year prospective, observational study using plain/uncoated PICCs, reported symptomatic CRT rates of 2.7–4.3/1000 days, with an average dwell-time of 5.5 days. 34 Comparatively, the AGBA-PICC demonstrated a symptomatic CRT rate of 3.2% and a 6.5% withdrawal occlusion rate, with an average dwell time of 39 days.
Furthermore Balsorano et al. 31 demonstrated symptomatic CRT rates with subgroup analyses, reporting thrombotic rates of 5.9% in onco-hematologic patients, 2.2% in oncologic patients, and 2.4% in patients representative of mixed populations. When the patient setting was considered, thrombotic events occurred in 2.5% of patients admitted to hospital, 4.3% of outpatients, and 1.5% in mixed populations. 31 The study by Piredda et al. 35 diagnosed thrombosis with ultrasound color-Doppler only when the appearance of arm or facial swelling and/or pain became symptomatically apparent. The authors current research sought to identify CRT in all patients at specified time intervals to also include post-catheter removal which may represent a more accurate CRT rate or highlight previously underreported occurrences.
As discussed above, the incidence of AGBA-PICC related asymptomatic CRT falls within the higher range of currently published research. However, the symptomatic CRT rate was 1.67–2.65-fold lower for AGBA-PICCs when compared to reports on plain/uncoated PICCs. 34 The catheter patency, as measured by withdrawal occlusions, were 2.3-fold lower with a 7.6-fold increase in catheter dwell days as compared to plain/uncoated PICCs. 34 The use of the AGBA-PICC was also associated with a low incidence of CLABSI (0.82/1000 days, 3.9%), which compares favorably with the results of prior reports,9,10,26,30 potentially lowering the infection-thrombosis relationship risks.
Limitations
The following are limitations with this study. The study sample size was limited, therefore generalizability of the results should be interpreted accordingly. Enrollment of study participants was through interventional radiology, therefore representation of other clinical models who insert and manage PICCs, may not be represented. A small number of patients (6) were lost to follow-up during the study period, as they could not be contacted either by landline or cellular phone or letter. Patients who were non-compliant when documenting their experiences in the patient diary, may not have accurately documented all procedures and interactions or related complications they may have experienced during the study period.
Conclusions
Although antimicrobial PICCs are relatively new technologies, they also come with the caveat of limited high-level clinical evidence to promote their efficacy. The incidence of CLABSI in this study with use of the AGBA-PICC was 0.82/1000 days, a 2.2–9.39-fold reduction compared to plain/uncoated PICC data of 1.8–7.7/1000 days reported previously.36,37 This study’s symptomatic CRT rate was 4.6%. Asymptomatic vessel narrowing or non-occlusive CRT was observed in 41% patients at day 10–14. Fibroblastic sleeve was observed infrequently. PWO was also infrequent and resolved with use of a thrombolytic agent in some instances.
While current interest in the reduction of hospital-acquired complications in patients with vascular access devices is high, there is still a significant amount of investigation required before the full impact of chlorhexidine impregnation technology is understood completely. However, in consideration of the observational findings of this study, chlorhexidine-coated PICC technology may be appropriate for patient populations who are at moderate to high-risk of catheter-related complications in both inpatient and outpatient settings.
Footnotes
Author contributions
The writing and development of this manuscript was performed by MW, TS, NS, DR; study design MW, JW, JG; conduct of the study MW, JW, JG, NS, HE, MI, DR; analysis of the research MW, QY, TS, DR. All manuscript revisions were performed and reviewed by TS, MW, and DR, and all authors approved the final version of this manuscript for submission.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was sponsored by Teleflex Inc., (Morrisville, NC, USA), manufacturer of the chlorhexidine-coated PICC used in this study. The primary investigator institutions involved received a research grant to perform this study.
