Abstract
Background
To determine the safety and performance of an antimicrobial/antithrombogenic peripherally inserted central catheter, a retrospective cohort study was conducted.
Methods
Data were collected using chart audits by clinicians in the US, UK, and EU in 2021 and 2022 for patients requiring an antimicrobial/antithrombogenic peripherally inserted central catheter. The primary outcome endpoint was successful completion of treatment without elective device removal due to adverse events. The secondary outcome endpoint was the incidence of device-related adverse events. Following the initial analysis of the index study, which included adult and pediatric patients, data from additional pediatric cases were collected. Charts lacking primary endpoint data were excluded. Standard descriptive statistics and analyses of categorical variables were performed without adjustment and with minimal stratification.
Results
A total of 38 healthcare providers collected data for 175 evaluable cases, of which 54.3% were male. The duration of use ranged from less than 1 day to 253 days. Treatment was successfully completed without elective device removal due to adverse events in 90.3% of cases. In 18 cases (10.3%), the patients experienced adverse events, with a total of 21 device-related adverse events reported, corresponding to 5 events per 1000 catheter days. None of the reported events were deemed serious or unexpected.
Conclusions
These findings suggest the potential for safe use of the antimicrobial/antithrombogenic peripherally inserted central catheter in the pediatric patient population, with no elective device removal due to adverse events in 90% of cases. No cases of central line–associated bloodstream infection, thrombosis, or anaphylaxis were reported; however, these findings should be interpreted with caution given the moderate sample size and the rarity of such events in general practice. Owing to the limitations of this study, including the risk of bias inherent to retrospective studies, prospective controlled trials with rigorous outcome assessments are needed to confirm these observations.
Introduction
Background and rationale
Central venous catheter (CVC) placement in pediatrics is required for several diagnoses, including cancer, infection, and malnutrition. Previous studies have estimated that 5 million pediatric CVCs are placed annually. However, CVC complications, including infection and thrombosis, contribute to increased morbidity and mortality. 1 Peripherally inserted central catheters (PICC) are long-term central venous access devices commonly used for intravenous therapy, blood sampling, infusion, pressure injection of contrast media, and central venous pressure monitoring in various patient populations, including pediatrics. Common risks associated with PICCs include central line–associated bloodstream infections (CLABSI) and thrombosis. To reduce these risks, catheters with antimicrobial/antithrombogenic (AM/AT) technologies can be used, particularly in patients at higher risk of developing these conditions.
Under the EU Medical Device Regulation (MDR), manufacturers of CE-marked medical devices are required to proactively conduct post-market clinical follow-up (PMCF) to substantiate the clinical safety and performance of the devices, including all patient populations specified in the device labeling. 2 To this end, a retrospective observational study was conducted to collect real-world data to demonstrate the safety and performance of a pressure injectable PICC with AM/AT properties in the general population. Following analysis of the original dataset, it was determined that the number of pediatric cases was insufficient to satisfy MDR PMCF requirements. Moreover, there is a perceived knowledge gap regarding the use of AM/AT PICCs in pediatrics and an academic need for evidence supporting their use in this population. Thus, the decision was made to collect data from additional pediatric cases. This article reports the subset of data related to AM/AT PICC use in pediatric patients. The objective was to determine the general safety and performance of the device in this subset of the population. The hypothesis was that AM/AT PICC would perform as intended, with an acceptable rate of adverse events (AEs) in pediatric patients.
Methods
Study device description, study objective, and hypothesis
AM/AT PICCs (Arrowg+ard Blue Advance, Teleflex Medical, Morrisville, NC, USA) are single-use intravascular catheters intended for short-term (up to 30 days) and long-term use (>30 days) for intravenous therapy, blood sampling, infusion, pressure injection of contrast media, and central venous pressure monitoring. The catheters are coated intraluminally and extraluminally with chlorhexidine to reduce microbial colonization and thrombus accumulation on the catheter surfaces. Their safety and efficacy are well reported in the literature.3–11 This retrospective study and subset analysis were conducted to determine the safety and performance of AM/AT PICCs in patients who required a PICC. The primary outcome endpoint, a composite measure of safety and performance, was successful use of the device without elective removal due to device-related AEs, as adjudicated by the clinicians who regularly use the devices. Safety was measured by the overall rate of device-related AEs. If a device-related AE occurred, it was reported as such; however, it was the healthcare provider’s (HCP’s) prerogative to remove or not remove the PICC and to attribute or not attribute the removal to the AE. The composite outcome endpoint strategy was employed to minimize the required sample size and duration of the study. Safety and performance are of similar clinical importance to the patient.
Study design
A retrospective observational study was designed by the study sponsor, which evolved into a two-phase project. Records were examined to identify patients who had completed treatment with an AM/AT PICC over the past 60 days. The index study (first phase) included the general population requiring an AM/AT PICC, and the secondary phase focused on pediatrics only, defined as patients aged ≤18 years. Patient identities, including names and screening identifiers, were deidentified. Inclusion criteria comprised availability of medical records for patients aged ≤18 years requiring vascular access with an AM/AT PICC—the population of interest for this report. The exclusion criterion was medical records lacking data necessary to determine device performance (e.g. reason for device removal)—the primary objective of the study. The reporting of this study conforms to the Strengthening the Reporting of Observational studies in Epidemiology (STROBE) guidelines. We used the STROBE writing guide when drafting this article and the STROBE checklist to demonstrate adherence to the guidelines. 12
Setting and participants
The study was approved by the Advarra Institutional Review Board (review number Pro00076111, Advarra, Columbia, MD, USA), with an exemption from informed consent and continued review, and was conducted in accordance with the Declaration of Helsinki. 13 Participants were HCPs, comprising physicians and nurses located in the European Union, United Kingdom, and United States, functioning as a healthcare panel convened by a market research firm. The firm vetted the qualifications of each HCP panelist to ensure an acceptable level of experience in vascular access and research methodology. Prior to the initiation of the study, the HCP panelists were trained on the use of the electronic data capture system for data collection, and the process was standardized among the participating HCPs. This ensured a reasonable degree of measurement comparability. The identities of the HCPs were unknown to the study sponsor; moreover, it remained unknown whether they had paid connections to the sponsor. The first phase of the study started in May 2021, and the second phase concluded in October 2022. Data were extracted from records for consecutive cases in which AM/AT PICC had been used and removed within the past 60 days. A 60-day cutoff was selected to monitor recent use patterns and because it was expected that an acceptable number of pediatric cases could be obtained during this interval. This strategy did not exclude cases of device use >60 days; it only excluded cases in which the device had been removed more than 60 days prior to the record search. No follow-up data were collected beyond device removal. As part of training, each participant was instructed on the importance of collecting data for all qualifying cases occurring in the past 60 days. As typical for a retrospective study, there was no monitoring to verify adherence to this requirement; however, data were reviewed for plausibility by the market research firm throughout the collection process. Following data collection, analysis was conducted by biostatisticians and scientists from the study sponsor organization. Within the initial dataset of 315 cases from the general population, there were 69 pediatric cases. The sponsor did not deem this subset sufficient for PMCF purposes; subsequently, the study was extended to collect additional data for pediatric patients.
Variables
The primary outcome endpoint of the study was successful device use without elective removal due to device-related AEs. The overall device-related AE rate served as the secondary outcome endpoint. Clinical characteristics, including specific uses of the device, device specifications, placement site, duration of use, and incidence of AEs, were collected. The source of all these variables was the electronic health record for each case included in the analysis. By closely reviewing the medical record for each case, it was determined whether the device was electively removed due to a device-related AE and whether other AEs occurred.
Bias
There was a possibility of sponsor bias, given that the manufacturer of the study device conducted the analysis; however, this was subject to audit by the EU MDR authorities. Employees of the manufacturer also contributed to the manuscript preparation, but they were members of the clinical and medical scientific affairs group rather than part of the commercial teams. The use of healthcare panelists from market research firms could result in selection bias. This was minimized by collecting data from consecutive qualifying cases over a defined period in which the study device was used, regardless of clinical outcome. Information bias was reduced by targeting all records in which device placement and removal dates were recorded, along with the reason for removal. Observer bias was possible due to unverifiable data abstraction, but mitigation was attempted by involving multiple HCPs in the data collection process and standardizing training for data collection. Response bias was addressed by designing study questions on the case report form that were clearly stated and providing clear, relevant answer options that were neither open-ended nor leading. Employing both performance and safety as outcome endpoints could be perceived as classification bias. Further evaluation of the inherent biases described here is recommended, so that they can be addressed in future studies.
Sample size calculation
Sample size calculation was conducted using R Project statistical software (The R Foundation, Indianapolis, IN, USA), based on weighted average data from the clinical literature describing the historical success rate for similar PICCs. For the first phase of the study, a sample size of 315 cases was derived using a one-sided 95% Wilson score interval, 14 assuming an acceptance criterion of 85% success to achieve 95% power. For the index study, data collection strategies were implemented to cover all variants of the catheter characteristics, intended uses (indications), and age groups, ensuring that data for an adequate number of cases were collected for each variant. These strategies were successful, except for the number of pediatric cases; thus, additional cases were sought. No sample size calculation was conducted for the pediatrics-only second phase of the study. Instead, HCP panelists were asked to provide data for all pediatric cases completed within the past 60 days from the onset of the second phase. Despite the absence of a formal sample size calculation, expert judgment, prior knowledge, and the EU notified body determined that the additional pediatric cases collected would likely be adequate to meet MDR requirements, provided that inferences were not overstated.
Statistical methods
R Project and IBM SPSS (v31 software, Armonk, NY) were used for statistical analysis. Standard descriptive statistics are presented according to the type of data. Continuous variables were presented as mean, SD, minimum, maximum, or median and interquartile range (IQR). Categorical variables were presented as counts and proportions. No specific hypothesis testing was performed on the data; therefore, normality was not assessed before presenting the results. An exception was the device duration of use data, which was evaluated using the Kolmogorov–Smirnov test of normality, yielding a p-value <0.001 at 5% level of significance. Event rates for complications were defined as the number of events per 1000 device days and as the proportion of total PICCs that experienced a given complication. Apart from 13 cases in which the type of image-guided technology used for device placement was not recorded, there were no missing data. These cases were acknowledged but retained in the analyzed dataset.
Results
Participants and patients
For the pediatric patient subgroup analysis, 38 healthcare panel members participated in the study, including clinicians from the United States (42%), European Union (42%), and United Kingdom (16%). Among them, 74% were physicians, and the remainder were nurses.
Data were collected for 178 pediatric cases; however, during initial analysis, 3 cases were excluded because they lacked information to determine the reason for device removal, which was needed for primary outcome endpoint analysis. Figure 1 presents the details regarding the final sample included in the analysis.

Inclusion and exclusion of cases, beginning with the original retrospective observational study of AM/AT PICCs in adult and pediatric patients requiring PICC access and ending with the final sample size of pediatric patients. AM/AT: antimicrobial/antithrombotic; PICC: peripherally inserted central catheter.
Descriptive data
Among the 175 patients receiving AM/AT PICCs, 54.3% were male, and 47.4% were aged 10–18 years. The device was used for intravenous therapy in 68.6% of cases (Table 1).
Patient demographics and clinical indications for device use (N = 175).

For some cases, more than one clinical indication for device usage was reported.
N: number of patients.
AM/AT PICC was most frequently inserted in the right basilic vein (29.1% of cases). Among catheter variations, double-lumen catheters (52.8%), catheters with a diameter of 4.5 Fr (61.7%), and catheters with a length of 40 cm (56.6%) were used most frequently. Among the image-guiding technologies used to facilitate PICC insertion, ultrasound was used most often (41.7% of cases). The VPS TipTracker™ Stylet (Teleflex Medical, Morrisville, NC, USA) was used during device placement and navigation through the vein in 29 cases, with successful use in each case. For cases in which AM/AT PICC was used for pressure injection of contrast media (n = 27), the mean number of injections per case was 3 (SD = 2), and all injections were successful without extravasation or other AEs. The mean maximum pressure applied was 145 psi (SD = 96) (Table 2).
PICC insertion site, catheter characteristics, and technologies used to aid insertion and venous navigation (N = 175).

Fr: French; N: number of patients; PICC: peripherally inserted central catheter.
Outcome data: main results: performance and safety
The primary outcome endpoint was successful device use without elective removal due to device-related AEs. Among the 175 pediatric cases included in the analysis, the success rate was 90.3%. In total, the 175 PICCs were in place for 4200 days. The device was removed due to lumen occlusion and/or suspected infection (as determined by the HCP) in 17 cases (9.7%), corresponding to 4.1 removals per 1000 catheter days. In 18 cases (10.3%), patients experienced AEs, with a total of 21 device-related AEs reported (5.0 per 1000 catheter days). None of the AEs were deemed serious or unexpected. Among AEs, occlusion occurred most frequently, i.e. in 6.3% of total cases (2.6 per 1000 catheter days). There were no reports of CLABSI, thrombosis, or anaphylaxis (Table 3). All data presented are unadjusted.
Outcome endpoint results: performance and safety outcomes (N = 175).

Cases that required device removal due to AEs.
CLABSI: central line–associated bloodstream infection; N: number of patients; PICC: peripherally inserted central catheter; AE: adverse event.
Other analyses: duration of use and reason for removal
In total, the 175 PICCs were in place for 4200 days. The mean duration of PICC use was 24 days (SD = 42), and the median was 10 days (IQR = 5–24). The maximum duration of device use in any one case was 253 days. In the majority of cases (60.6%), the device was in place for ≤15 days. In 82.9% of cases, the device was removed due to planned termination of therapy; however, in 17 cases, the device was electively removed due to a device-related AE. There was a trend toward fewer device removals due to AEs in older pediatric patients. Odds ratio analysis comparing children aged <10 years with those aged ≥10 years showed that younger children were nearly four times more likely to have their PICC removed due to an AE (3.89 (95% confidence interval (CI), 1.24–12.25)). Children aged <1 year were over five times more likely to have the device removed due to an AE compared with older children (5.32 (95% CI, 1.95–14.51)). It should be noted that for these analyses, there was no adjustment for confounders, and the reported age effect may be perceived as biased (Tables 4 and 5).
Device duration of use/dwell time and reason for removal (N = 175).

N: number of patients.
AM/AT PICC removal due to device-related AEs (n = 17), by age group, with frequency (proportion).
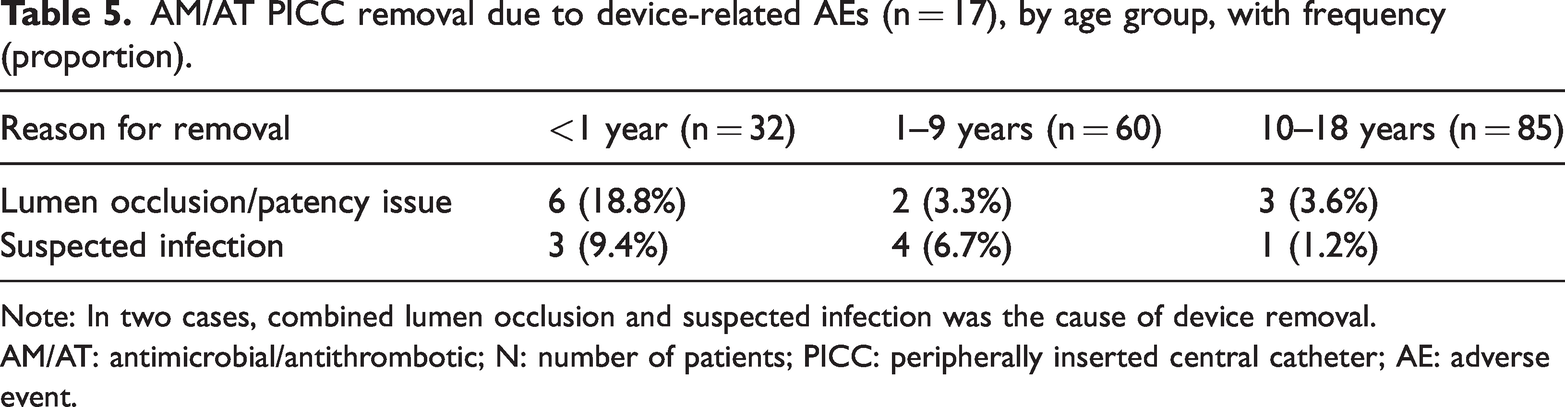
Note: In two cases, combined lumen occlusion and suspected infection was the cause of device removal.
AM/AT: antimicrobial/antithrombotic; N: number of patients; PICC: peripherally inserted central catheter; AE: adverse event.
Discussion
Although there is abundant clinical data reporting PICC use in the general population and moderate data describing its use in the pediatric population, only a few studies have described the use of PICCs with AM/AT properties in pediatrics—the population of interest for this report. During the study period, qualifying data from 175 consecutive cases of AM/AT PICC placement at 38 hospitals were collected. We showed that in pediatric patients, the device can be used safely and effectively as intended, without removal due to AEs in 90% of cases. In 69% of cases, the device was used for intravenous therapy, although the diagnosis and acuity of the patient were not recorded. Furthermore, we showed that the device can be used in pediatrics with a relatively low incidence of AEs, including one case in which the device remained in place for 253 days.
Among all patient populations, PICC failure definitions and rates vary. In a 2015 systematic review, failure was defined as the inability to complete treatment with the device, and a rate of 12.4 failures per 1000 catheter days was reported. 15 In a 2018 feasibility randomized controlled trial (RCT) reported by Kleidon et al., 150 pediatric participants were randomized to receive either a polyurethane or BioFlo (AngioDynamics Inc, Queensbury, NY, USA) PICC. Patients were followed for up to 60 days and an additional 48 h post-catheter removal, and catheter failure was assessed for each group. Completion of therapy without catheter removal due to complication was accomplished in 89% of patients in the BioFlo group, compared with 78% in the polyurethane group, corresponding to 7.3 and 12.6 failures per 1000 catheter days, respectively. 16 The same year, in a retrospective study, Chenoweth et al. reported PICC use in 202 neonates and found that 16% of standard PICCs and 11% of BioFlo PICCs failed before completion of therapy. 17 This corresponding to incidence rates of 12.6 and 7.3 per 1000 catheter days, respectively. In our study, 9.7% of PICCs were removed due to catheter-related AEs, including lumen occlusion/patency issues (n = 11) and suspected infection (n = 8), yielding a rate of 4.1 removals per 1000 catheter days. Although this rate was somewhat lower than generally reported for pediatrics, interpretation should be made with caution due to the moderate sample size and other study limitations.
Among other published studies, the main outcomes of interest have been the incidence of thrombus, CLABSI, and occlusion. Compared with traditional CVCs, the use of PICC lines is widely believed to be a major cause of thrombosis, possibly due to the smaller diameter and longer length of the catheter, which may cause endothelial damage and blood clot formation. 18 The reported prevalence of catheter-related thrombosis varies from 1% to 9% in critically ill children,19,20 but relatively low rates of thrombosis from pediatric PICC use have been reported in most cases. In Zhang et al.’s 2022 study of PICCs placed in children for chemotherapy (N = 48), the thrombosis rate was 2.0%, 21 similar to the 1.4% rate observed by Kamata et al. (N = 73), in which PICCs were placed for the same indication. 22 In a larger study (N = 2553), Badheka et al. examined PICC use in hospitalized children and observed a 1.0% rate of thrombosis. 23 In an RCT (N = 1098) conducted by Ullman et al., who compared the performance and safety of three PICC types in the general population that was followed up for up to 8 weeks, there were 172 pediatric cases. Among those, thrombosis was observed in 3% of cases in which chlorhexidine PICCs were used, compared with 2% of cases in which both BioFlo hydrophobic and standard polyurethane PICCs were used. 24 An outlier to the low rate is Azzam et al.’s 77-patient study involving pediatric intensive care unit (ICU) patients with PICCs, in which an 18% thrombus rate was reported. 25 Apart from Ullman et al.’s study, the type of PICC in these studies was not reported but was presumed to be conventional polyurethane. In our study, in which AM/AT PICCs were used, there were no reports of thrombosis; however, our sample size was only moderate.
The reported incidence of CLABSI associated with PICC use is generally low. For example, Chenoweth et al. 17 and Badheka et al. 23 reported CLABSI rates of 0.7% and 1.9%, respectively. In our study, although eight catheters (4.6%) were removed because of suspected infection, no cases of CLABSI were identified. Occlusion is another complication that is not infrequently reported with PICC use in the pediatric population and may be attributed to the smaller diameter and longer length of these catheters, compared with those of other vascular access devices. In our study, 11 occlusions (6.3%) were observed. This rate is higher than the 2.7% reported in the 73-patient study by Kamata et al. 22 but lower than the 11% reported by Greencorn et al., 26 which included 3205 pediatric patients in a tertiary-care center who were followed up until PICC removal. In that study, 706 PICCs (22%) were associated with mechanical complications, including catheter occlusion, infiltration, migration, fracture, or disconnection, corresponding to 14 mechanical complications per 1000 catheter days. In our substantially smaller study, we observed 11 mechanical complications, all of which were lumen occlusions. This corresponds to a rate of 6.3% or 2.6 events per 1000 catheter days, both of which are lower than those reported in the larger and more definitive study by Greencorn et al. Rates ranging from 4.3 to 19.3 occlusions per 1000 catheter days have been reported in other studies.27–30 However, direct comparison of our findings with those of the Greencorn group and other studies is inappropriate because definitions of mechanical complications vary across studies and because of our moderate sample size. As reported by Greencorn et al. and other authors,26,30 our data suggest that the risk of complications associated with PICC use may decrease with increasing patient age. However, the absence of thrombosis and CLABSI in our study may be attributable, in part, to the lack of follow-up after device removal and the moderate sample size.
Our study also reports the use of a dedicated stylet, known as the VPS TipTracker Stylet, which provides PICC navigation through the veins using electromagnetic technology. Although this accessory is not indicated for use in pediatric patients, it was used in 29 cases, and in all cases, the stylet successfully facilitated navigation of the PICC during placement. It should be noted that the manufacturer does not recommend, suggest, or promote the use of this product for any indication not listed in the Food and Drug Administration (FDA)-approved labeling. We also report the use of AM/AT PICC for pressure injection of contrast media in the pediatric population. For this indication, the device was used in 27 cases, all of which were successful and were not associated with extravasation or other AEs. Additional research is required to confirm the safety and performance of AM/AT PICCs when used with dedicated stylets and for pressure injection.
Several limitations of this study should be acknowledged. First, the retrospective design precluded active monitoring of data accuracy and completeness, as would be possible in a prospective study. In addition, consecutiveness of eligible cases could not be verified. Chart reviews were conducted by clinicians who were also device users, introducing the potential for observer bias and underreporting. Furthermore, data on patient comorbidities, diagnoses, and severity of illness were not available, which may limit interpretation of the findings. No multivariate analysis was performed to adjust for potential confounders, such as patient age, insertion site, or indication. Detailed information regarding reported infections was lacking, and there was no independent adjudication or blinding of outcome assessment. The absence of adjustment for confounders, including patient diagnosis, comorbidities, and site-level practice variation, limits causal inference and the robustness of the conclusions. Additionally, event rates reported per catheter day lack time-to-event data and CIs; therefore, these rates should be interpreted with caution. Finally, the study was not powered to detect rare or unknown events in specific subpopulations. Despite these limitations, this study provides initial observational data on the use of AM/AT PICCs in pediatric patients.
Conclusions
This study describing the use of the Arrowg+ard Blue Advance Antithrombotic/Antimicrobial Catheter in pediatric patients suggest the potential for safe use in this population. The results are favorably consistent with those of other studies in which PICCs composed of various materials were used. No cases of CLABSI, thrombosis, or anaphylaxis were reported; however, these findings should be interpreted with caution because of the moderate sample size and the overall rarity of these events in clinical practice. In addition, this study provides initial observations on the use of specialized stylets during PICC placement in pediatric patients and presents the first description of AM/AT PICC used for pressure injection of contrast media in this population. Given the limitations of this study, prospective, controlled trials with rigorous outcome assessment are required to confirm these observations.
Footnotes
Acknowledgments
The authors acknowledge and appreciate the assistance of Michael Bishop in providing biostatistical support and consultation.
Author contributions
Thomas E Philbeck, Caroline Ryan, and Amy Bardin contributed to the study design, data analysis, and manuscript writing and editing.
Consent to participate
This study had a retrospective design and was exempt from informed consent by the Advarra Institutional Review Board (review number Pro00076111, Advarra, Columbia, MD, USA). The need for informed consent was waived.
Consent for publication
Not applicable.
Data availability
The original data are available for review upon reasonable request.
Declaration of conflicting interest
Thomas E Philbeck, Caroline Ryan, and Amy Bardin as well as the biostatistician Bishop are employees of Teleflex Incorporated, the manufacturer of the study device described in this report.
Ethical considerations
The study was approved by the Advarra Institutional Review Board (review number Pro00076111, Advarra, Columbia, MD, USA), with an exemption from informed consent and continued review, and was conducted in accordance with the Declaration of Helsinki.
Funding
All aspects of the study described in this report were funded by Teleflex Incorporated, the manufacturer of the study device described in this report.
