Abstract
Introduction
We describe a case of acute exudative polymorphous vitelliform maculopathy (AEPVM) that recurred 9 years after the initial event. To the best of our knowledge, this is the first report of recurrent AEPVM showing recovery of retinal and retinal pigment epithelium (RPE) function and good visual outcome following treatment with intravitreal corticosteroid.
Case description
A 45-year-old Caucasian woman first presented with AEVPM in 2009. Her condition spontaneously resolved and she remained stable over several years. 9 years later, her condition recurred with bilateral reduction in visual acuity. Fundus examination revealed multiple small yellowish subretinal lesions across the posterior pole in both eyes. Optical coherence tomography (OCT) showed bilateral cystoid macular oedema (CMO). She was referred for electrophysiology and her electrooculogram findings were in keeping with severe generalised RPE dysfunction bilaterally, with a light peak to dark trough ratio (Arden index) of 110%, comparable to her initial presentation 9 years earlier. She was initially treated with oral steroids with some improvement. However, the maculopathy in the left eye recurred on cessation of oral treatment. A sustained-release 700ug dexamethasone intravitreal implant (Ozurdex®) was inserted in the left eye to which she responded remarkably, with improvement in visual acuity and complete resolution of the CMO. A year later, at her most recent clinic visit in March 2021, there was no evidence of any further recurrence.
Conclusion
Our case demonstrates clinical and imaging findings consistent with recurrence of AEPVM with CMO that has been successfully treated with Ozurdex®.
Introduction
Acute exudative polymorphous vitelliform maculopathy (AEPVM) was first described in 2 patients in 1988. 1 The condition has been described as accumulation of yellowish material in the subretinal space and serous retinal detachments with a tendency to accumulate in the form of a meniscus as well as round or oval clumps in a ring-like configuration.1,2 The aetiology remains uncertain but recent reports have suggested a paraneoplastic or viral cause.2–4 There is little information on the natural course and treatment options. 5 Recurrence of AEPVM is rare and only 1 previous case of recurrent AEPVM has been published. 5 We describe a case of AEPVM that recurred 9 years after the initial event. The patient was treated successfully with oral prednisolone followed by intravitreal dexamethasone implant, and was monitored with international standard pattern and full-field electroretinogram (PERG; ERG) and electro-oculogram (EOG) testing. 6 To our knowledge, this is the first report of recurrent AEPVM showing recovery of retinal and retinal pigment epithelium (RPE) function and good visual outcome following treatment.
Case description
A 45-year-old Caucasian woman with a history of left amblyopia presented in November 2009 with blurring of vision in both eyes, five days after a road traffic accident. 7 On examination, bilateral serous detachments and multiple small yellow subretinal lesions were observed in the posterior pole of both eyes. 7 The initial electrophysiological assessments were performed in April 2010. The full-field ERGs were normal but the left eye PERG P50 component was reduced (mean P50 amplitude 2.3µV on the right; 1.7µV on the left), in keeping with left macular dysfunction (Figure 1a, rows 1 and 2). 7 The EOG light peak to dark trough (LP:DT) ratio was 105% on the right and 110% on the left (Figure 1c), consistent with severe generalised RPE dysfunction. 7 Nine months after presentation she subjectively reported improvement in vision and her OCT showed first signs of improvement with less subretinal fluid and vitelliform material. 42 months after presentation, PERGs showed no significant change (P50 amplitude 2.4µV on the right; 1.3µV on the left) but the EOG LP:LT ratio had normalised (180% on the right; 190% on the left; Figure 1c), indicating improved generalised RPE function. 7 There was complete resolution on her macular OCT 48 months later and her best-corrected visual acuity (BCVA) had improved to 6/6 in the right eye and 6/9 in the left eye. Her condition remained stable and she was discharged 84 months after her initial presentation.
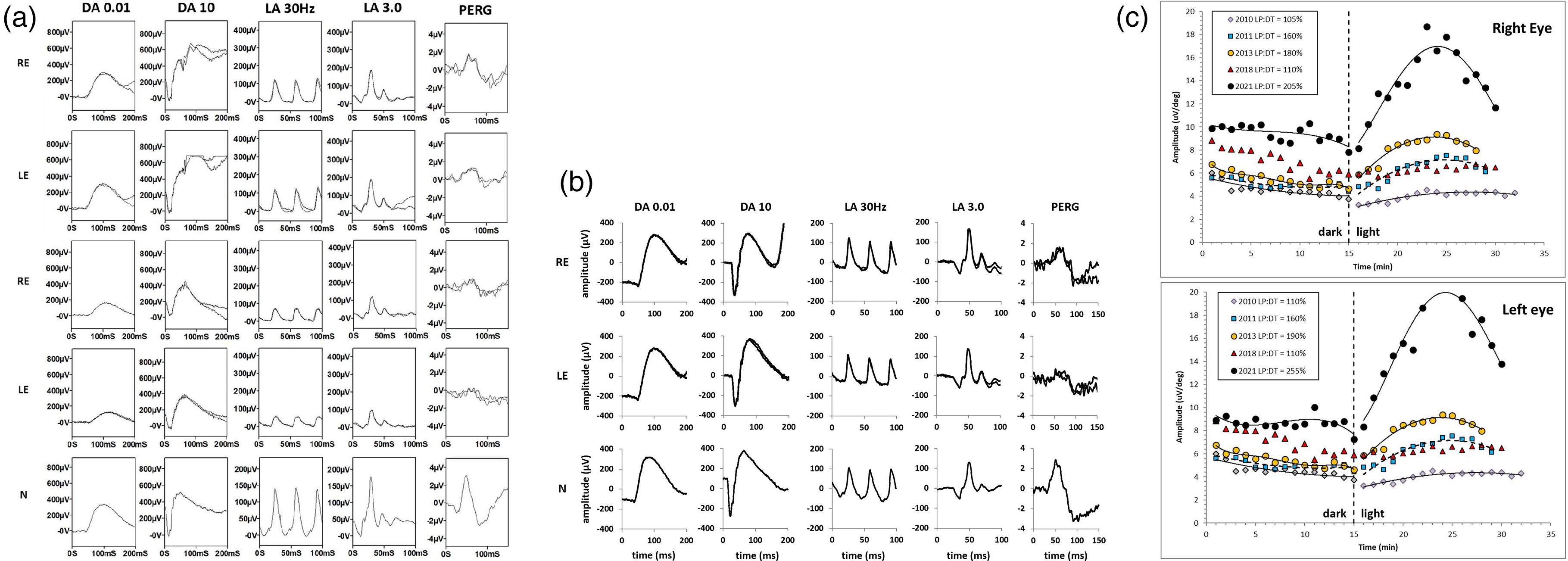
ISCEV-standard full-field and pattern electroretinogram (ERG; PERG) from the right eye (RE) and left eye (LE) of the patient, obtained at the initial visit in April 2010 (a: rows 1-2), in June 2018 (a: rows 3-4) and in April 2021 (b: rows 1-2) and representative control examples (N), recorded using gold foil corneal electrodes. The ERGs were initially normal but showed reductions without delay in June 2018, consistent with a bilateral loss of both rod photoreceptor and cone system function. Pattern ERGs were initially subnormal in the left eye only, indicating macular dysfunction, but worsened bilaterally. At final follow up (b) ERGs and the right PERG P50 normalised, but there was left P50 reduction, indicating persistent macular dysfunction. ISCEV-standard EOGs (c). Dark and light-adapted curves for each visit are fitted with solid or broken lines, according to polynomial functions, except for data from June 2018 (symbols only, for clarity). The light peak to dark trough (LP:DT) ratios are shown within the key. Note the gradual improvement between April 2010 and January 2013. Following recurrence of AEPVM there was severe reduction in the LP:DT ratio, with normalisation at final follow up.
She presented again 101 months after her initial presentation with a 1-week history of bilateral visual blurring. She denied other systemic symptoms. She had recently been diagnosed with grade 2 invasive ductal carcinoma of the right breast for which she had wide local excision seven months prior. She subsequently had combined epirubicin and cyclophosphamide chemotherapy and four-field adjuvant radiotherapy, completed three months and one month prior to re-presentation respectively. She was on anastrazole and zolendronic acid at the time of her review. On examination, BCVA was 6/9 in the right eye and 6/12 in the left eye. There was no inflammation on anterior or posterior segment examination. Fundus examination revealed multiple small yellowish subretinal lesions across the posterior pole in both eyes (Figure 2a). Optical coherence tomography (OCT) showed bilateral cystoid macular oedema, mild subretinal fluid and hyper-reflective deposits at the outer retinal surface (Spectralis HRA-OCT Heidelberg Engineering, Dossenheim, Germany), (Figure 2b). Fundus autofluorescence (FAF) in the right eye showed a low-density signal at the fovea with perifoveal stellate pattern of increased signal and in the left eye showed patches of macular hyper-autofluorescence. Background FAF was irregular with small discrete areas of increased and decreased signal bilaterally (Figure 2c).
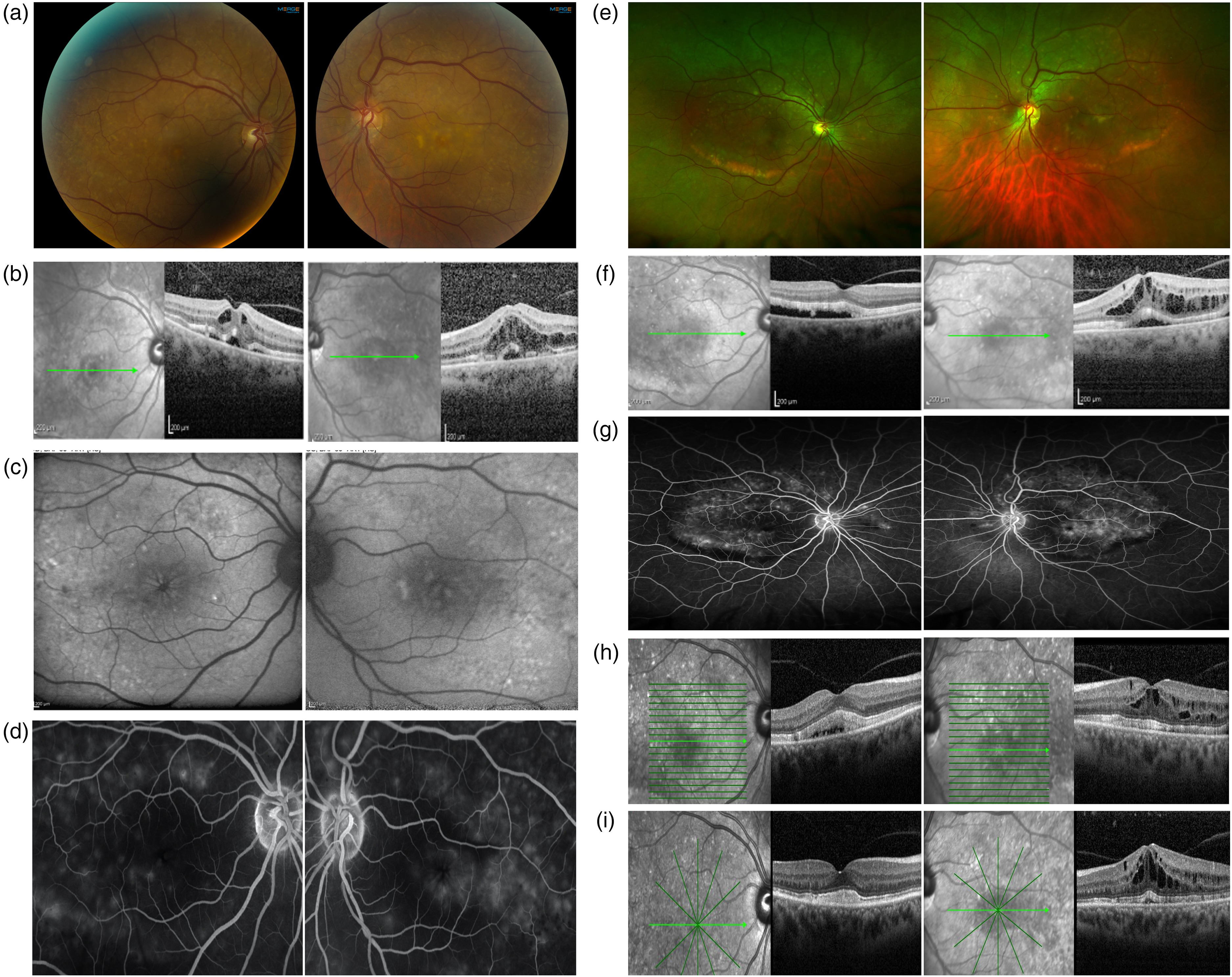
a. Colour fundus photos, b. OCTs and c. FAF at initial presentation of recurrent AEPVM. d. FFA showed early hyperfluorescence of the yellow lesions in both eyes and cystoid macular oedema pattern of leakage in the left eye. e. Colour fundus photograph f. OCT and g. FFA images (venous phase) at 6 months review. h. OCT in January 2019 while on oral prednisolone and i. OCT in August 2019 1-month after stopping oral prednisolone.
Fundus fluorescein angiography (FFA) showed early hyperfluorescence of the yellow lesions in both eyes and cystoid macular oedema pattern of leakage in the left eye (Figure 2d; RE and LE). She was followed up in the retinal clinic and started on topical steroid (g.maxidex 0.1% QDS) and non-steroidal anti-inflammatory therapy (g. ketorolac 0.5% QDS). These were continued four times and day and stopped 6 weeks later due to lack of efficacy.
Repeat EOG testing 103 months after initial presentation revealed severe generalised RPE dysfunction (LP:DT 110% bilaterally; Figure 1c), unexplained by worsening of the ERG, that indicated a bilateral but relatively mild loss of rod photoreceptor and cone system function (Figure 1a, rows 3 and 4). The PERG P50 components were mildly subnormal in the right eye and subnormal in the left eye (P50 amplitude 1.6µV on the right; 0.9µV on the left) consistent with bilateral worsening of macular function.
Six months later, her BCVA in the right eye had improved to 6/7.5 and dropped in the left eye to 6/24 improving to 6/9 with pinhole correction. Fundus examination demonstrated gravitational settling and coalescence of the yellowish material along the inferotemporal arcade in both eyes (Figure 2e). She was found to have ongoing subretinal fluid in the right eye increasing macular oedema and subretinal fluid in the left eye on OCT (Figure 2f). FFA demonstrated some window defects but no active maculopathy in the right eye and cystoid pattern of maculopathy in the left eye. FFA also showed masking of fluorescence from the areas of coalescing yellow subretinal material in both eyes (Figure 2g).
The patient was commenced on 50mg of oral prednisolone daily with a slow taper. There was slow improvement and by January 2019, while on 10 mg of oral prednisolone, her BCVA was 6/6 in the right eye and 6/12 improving to 6/9 with pinhole correction in the left eye. The OCT confirmed a reduction in the subretinal fluid in both eyes and reduced intraretinal fluid in her left eye (Figure 2h). She was continued on oral prednisolone tapering by 1 mg every 2 weeks to a dose of 4mg during which her condition remained stable.
116 months after her initial presentation, the patient took an informed decision to discontinue her treatment with oral prednisolone. A month after cessation, there was increased intraretinal fluid in the left eye (Figure 2i). Peribulbar triamcinolone resulted in marginal reduction of intraretinal fluid but with no improvement in her symptoms. 20 months following the recurrence, there was persistent left macular oedema (Figure 3a). Her BCVA was 6/6 in the right eye and 6/12 in the left eye. An intravitreal dexamethasone implant (Ozurdex, 700ug dexamethasone) was inserted in the left eye a month later, with marked improvement in BCVA to 6/6 in both eyes and resolution of the macular oedema, but with persistence of subfoveal subretinal hyperreflective material and slight disruption of the ellipsoid zone in both eyes (Figure 3b).
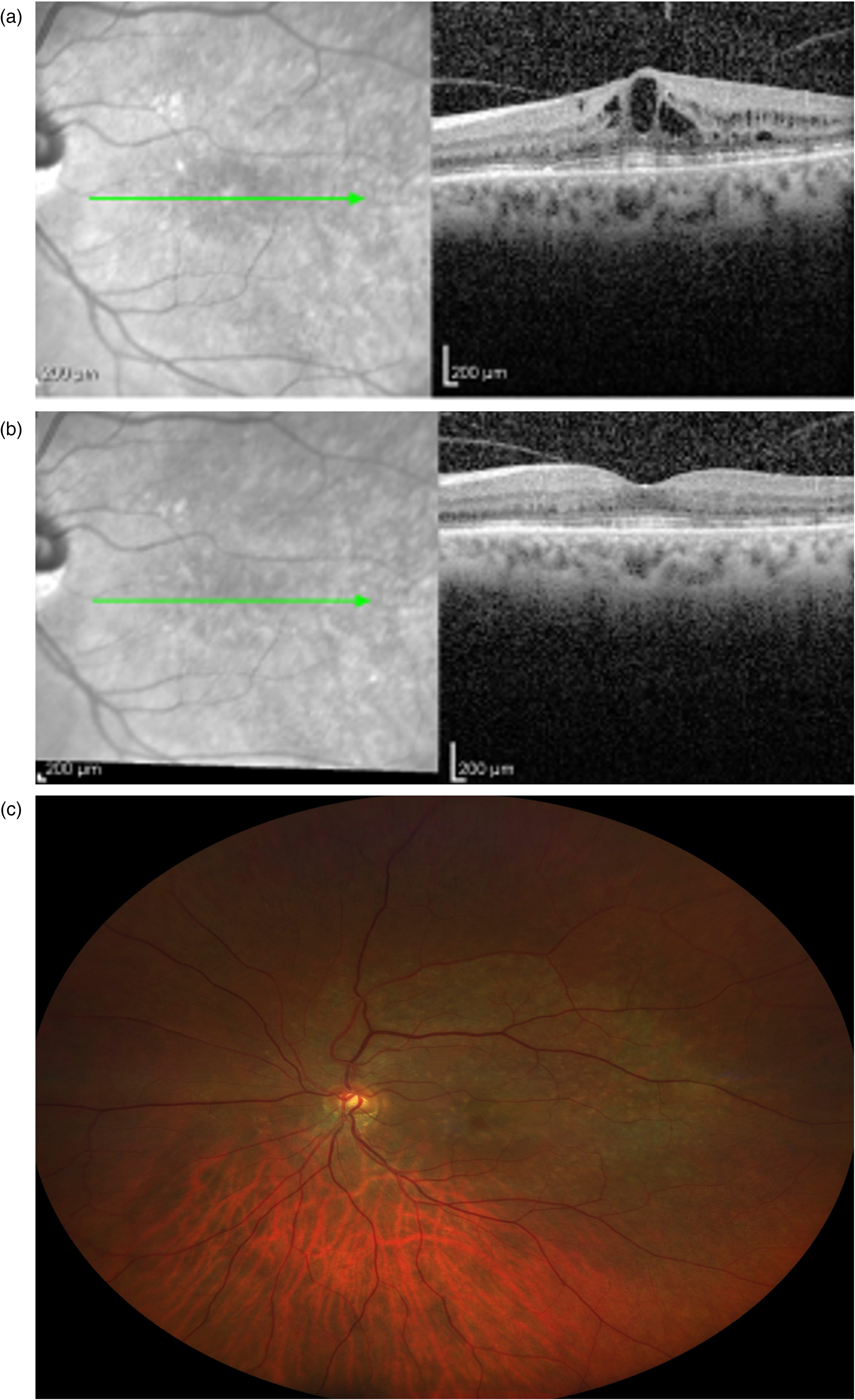
a. OCT of left eye pre-intravitreal steroid implant. b. OCT of left eye 1 months post intravitreal steroid implant. c. Fundal photo of left eye 13 months post intravitreal steroid implant.
137 months after initial presentation, there was no evidence of any further recurrence and there was partial reabsorption of the subretinal material (Figure 3c). Full-field ERG and EOG (LP:DT ratio 205% on the right; 255% on the left) were normal (Figure 1b and Figure 1c), consistent with recovery of retinal function and dramatic recovery of generalised RPE function. The right PERG P50 normalised, but there remained PERG evidence of left macular dysfunction (mean P50 amplitude 2.3µV on the right; 0.6µV on the left; Figure 1b, row 2), consistent with outer retinal disruption evident on OCT (Figure 3b).
Conclusions
This report describes a rare case of recurrent AEPVM. Evolution, recurrence and efficacy of treatment were characterised by serial fundus imaging, spectral-domain OCT and detailed international standard electrophysiological assessments over more than a decade. The study highlights reduction in subretinal material on the fundus, recovery of retinal and RPE function, and the potential for successful treatment of persistent AEPVM-related macular oedema.
The clinical and imaging findings at presentation included typical features of AEPVM, with multiple yellow subretinal deposits scattered around the macula accompanied by elevation of neurosensory retina and fluid accumulation in the subretinal space. 8 Gravitational settling of the yellow subretinal material in a curvilinear fashion inferior and temporal to the retina was seen at follow-up, consistent with previous reports.1,2,8 The FFA features show early hyperfluorescence of the lesions due to choroidal fluorescence through the affected RPE. 8 At follow-up, the masking of fluorescence from the accumulation of material inferiorly is also consistent with previous published reports. 2 It has been hypothesised that the composition of the lesions changes with time, resulting from RPE dysfunction and accumulation of lipofuscin and A2E precursors derived from photoreceptor outer segment shedding. 2 The study corroborates earlier reports suggesting tendency towards reabsorption and regression of the subretinal material with time.2,7 The ERGs were consistent with a loss of photoreceptor function, but insufficient to explain the severely abnormal EOG findings, indicating severe generalised RPE involvement. The subsequent normalisation of ERG and EOG highlights the reversible nature of dysfunction in AEPVM, and the potential for recovery of retinal and RPE function.
The value of corticosteroid therapy in AEPVM remains to be established, with limited evidence in the literature to show the benefits of systemic steroids.1,8,9 Most patients recover spontaneously but some develop chronic retinal changes with poor visual recovery. 2 The presence of cystoid maculopathy in AEPVM has been observed, but with no corresponding leakage on FFA reported.2,8 Our case differed in that inner and outer retinal cystic changes were present, associated with a petaloid pattern of leakage on FFA particularly in the left eye, largely resistant to systemic steroid treatment and prompting further therapy. Only one published report to date has described the use of intravitreal dexamethasone implant in a case of AEPVM, but without clinical improvement. 10 To our knowledge, this is the first case showing efficacy associated with a combination of systemic and intravitreal treatments, for recurrent AEPVM-associated macular oedema. Barbazetto et al. 2 highlight 2 groups of AEPVM patients; those who show resolution of clinical signs and symptoms within a few months of onset and those who have visible retinal changes for years with poor visual recovery. Our patient showed improved visual acuity, but may have fallen into the second category should she not have been treated with steroids. It is possible that treatment may have also contributed to the improved generalised (mainly peripheral) retinal and RPE function, although the high degree of inter-ocular full-field ERG and EOG symmetry suggests this may have occurred spontaneously, or independently of the intravitreal implant. The study highlights that intravitreal dexamethasone implant may be an effective option for patients with idiopathic AEPVM in the presence of persistent macular oedema.
The importance of excluding a paraneoplastic process in patients presenting with features of AEPVM is highlighted, given an overlap of clinical features with paraneoplastic polymorphous retinopathy. An immune- mediated process has been postulated in the latter, whereby antibodies are generated against retinal pigment epithelium cells in response to the neoplastic process. 3 Although our patient had a history of breast cancer, the recurrence of AEPVM occurred 3 months after completion of chemotherapy and a month after cessation of radiotherapy. Furthermore, given that at 5 years of follow up following her surgery she had had no signs of metastatic disease, we postulate the recurrent AEPVM is unlikely to be of paraneoplastic origin, although it remains a possibility.
This report describes the evolution, recurrence and extent of structural and functional recovery in a patient with AEPVM, monitored over a period of more than a decade. Treatment of chronic AEPVM-related macular oedema is demonstrated, following both systemic and intravitreal steroids. The study highlights the value of serial electrophysiological testing, revealing dramatic recovery of RPE and retinal function and providing objective evidence of treatment efficacy for AEPVM-related macular oedema.
Patient consent
Patient has provided signed consent to the publication of this material.
Footnotes
Acknowledgements
AGR is supported by the National Institute for Health Research Biomedical Research centre at Moorfields Eye Hospital NHS Foundation Trust and UCL Institute of Ophthalmology, London, UK.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
