Abstract
Introduction
Intravitreal dexamethasone (DEX) implant is indicated for the treatment of macular oedema due to diabetic retinopathy, retinal vein occlusion and uveitis. The most common complications are cataract and elevated intraocular pressure (IOP). Accidental injection of DEX implant into the lens is a rare complication and only few papers presented it.
Case Presentation
A 40-year-old man was treated with DEX implant for diabetic macular oedema in both eyes. At 1 week follow-up visit, slit lamp examination showed the DEX implant was located in the crystalline lens of the right eye (RE) without any sign of inflammation, cataract or elevated IOP, so we decided to plan a normal follow-up schedule. Macular oedema relapsed 5 months after the injection in the left eye (LE), whereas the RE did not show any sing of intraretinal or subretinal fluid. Six months after DEX implantation an uneventful phacoemulsification and intraocular lens placement were performed in the RE because of IOP elevation.
Conclusions
The therapeutic effect of DEX implant can be maintained for a longer period of time than intravitreal implant, determining complete reabsorption of macular oedema. Intralenticular implant can be maintained inside the lens until either IOP increases, cataract progresses, or other complications occur.
Keywords
Introduction
Ozurdex® (Allergan Inc., Irvine, CA, USA) is a sustained-release implant containing 700 µg of dexamethasone (DEX). The biodegradable rod-shaped capsule (Novadur®) is a solid polymer drug delivery system composed of lactic acid and glycolytic acid polymers. The device is implanted in the vitreous chamber through a 22-gauge needle, and then slowly dissolves while releasing dexamethasone, over a period of 3–6 months. DEX implant is indicated for the treatment of adults with diabetic macular oedema, macular oedema following central or branch retinal vein occlusion and inflammation of the posterior segment of the eye due to non-infectious uveitis. 1
According to literature, cataract and elevated intraocular pressure (IOP) are the most common adverse reactions after the administration of DEX implant, followed by anterior chamber migration, conjunctival haemorrhage and endophthalmitis. 2
During the ZERO study, designed to test the safety and efficacy of DEX intravitreal injections, damage to the lens was not reported. 3 To our knowledge, there are only a few papers presenting accidental injection of DEX implant into the lens. This extremely rare complication may occur due to a sudden movement of the patient's head during the injection, surgeon's inexperience, or an unsuitable surgical technique. 4
Case presentation
A 40-year-old man presented with severe visual acuity reduction in both eyes. His past medical history included acromegaly, caused by secreting pituitary adenoma, and secondary diabetes mellitus. Best-corrected visual acuity (BCVA) was 20/800 in the right eye (RE) and 20/63 in the left eye (LE). IOP was 16 mmHg in both eyes. Ophthalmic examination showed transparent cornea, early cortical sclerosis of the lens and moderate non-proliferative diabetic retinopathy, along with biliteral cystoid macular oedema and subretinal fluid. Optical coherence tomography (OCT) (RTVue XR 100 AVANTI, Inc. Fremont, USA) was performed, showing a central retinal thickness (CRT) of 801 μm in the RE (Figure 1a) and 621 μm in the LE (Figure 1d). We decided to administrate DEX implant, starting with the LE, while injection in the RE was performed 2 weeks later.
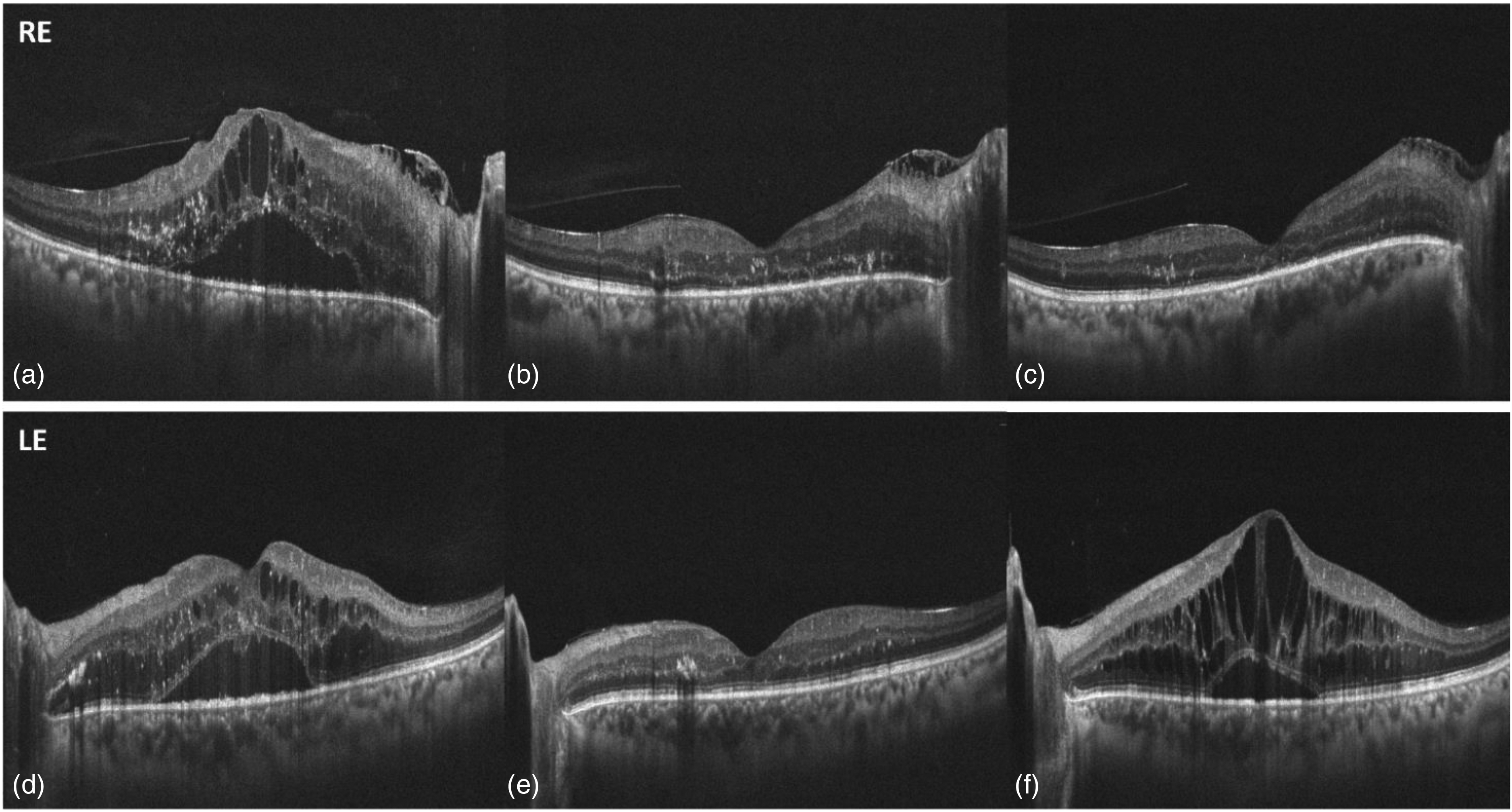
Horizontal crossline scan of the macula in the right eye, at patient presentation (a), at 2 months (b) and 5 months (c). Horizontal crossline scan of the macula in the left eye, at patient presentation (d), at 2 months (e) and 5 months (f).
One week after the injection in the RE, the patient underwent postoperative examination. BCVA was 20/400 in the RE and 20/50 in the LE. IOP was unchanged in both eyes. Slit lamp examination of the RE revealed DEX implant was positioned in the inferior sector of the lens (Figure 2). Surprisingly, the lens did not show any sign of opacity or dislocation.
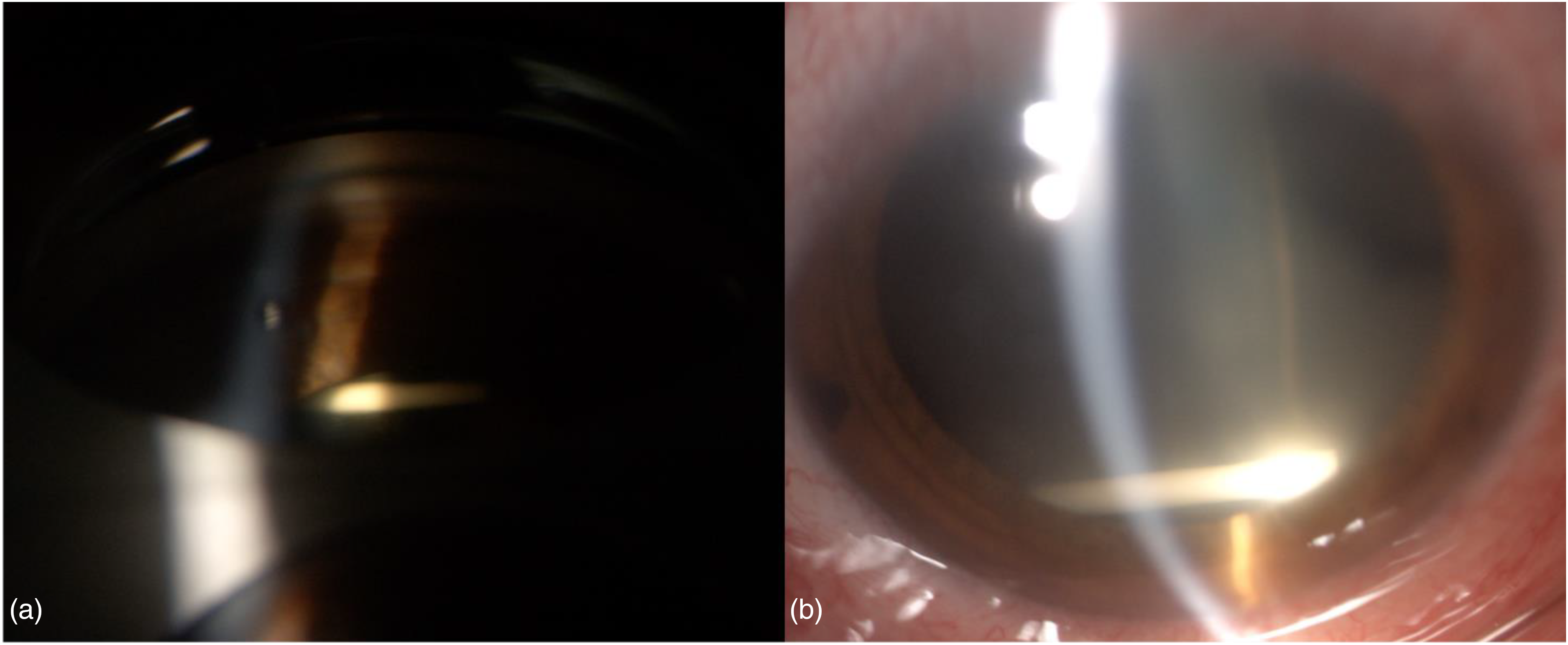
(a) Gonioscopic view of the intralenticular Ozurdex implant. (b) Slit lamp examination showing intralenticular Ozurdex implant.
As for the LE, DEX implant was correctly positioned in the vitreous chamber. Since macular oedema had reduced in both eyes and there was no evidence of inflammatory or mechanical lenticular complications, we planned a normal follow-up schedule.
At 2 months, OCT showed normalised values of CRT (RE 258 μm, LE 242 μm) as well as complete reabsorption of both intraretinal and subretinal fluid (Figure 1b and 1e). Only rare residual intraretinal hard exudates were identified.
At 5 months, BCVA was 20/250 in the RE while it reduced to 20/200 in the LE. There was no evidence of macular oedema in the RE (Figure 1c), while the reduced visual acuity in the LE was related to the relapse of intraretinal and subretinal fluid (Figure 1f) (CRT RE 213 μm, LE 596 μm). Because of this, a monthly cycle of 3 intravitreal injections of Aflibercept was performed in the LE.
At 8 months, IOP increased in the RE (25 mmHg). Therefore, the patient was first treated with carbonic anhydrase inhibitors (Brinzolamide), and then underwent phacoemulsification in the RE (Figure 3). Surgery was uneventful and IOL was correctly placed in the capsular bag.
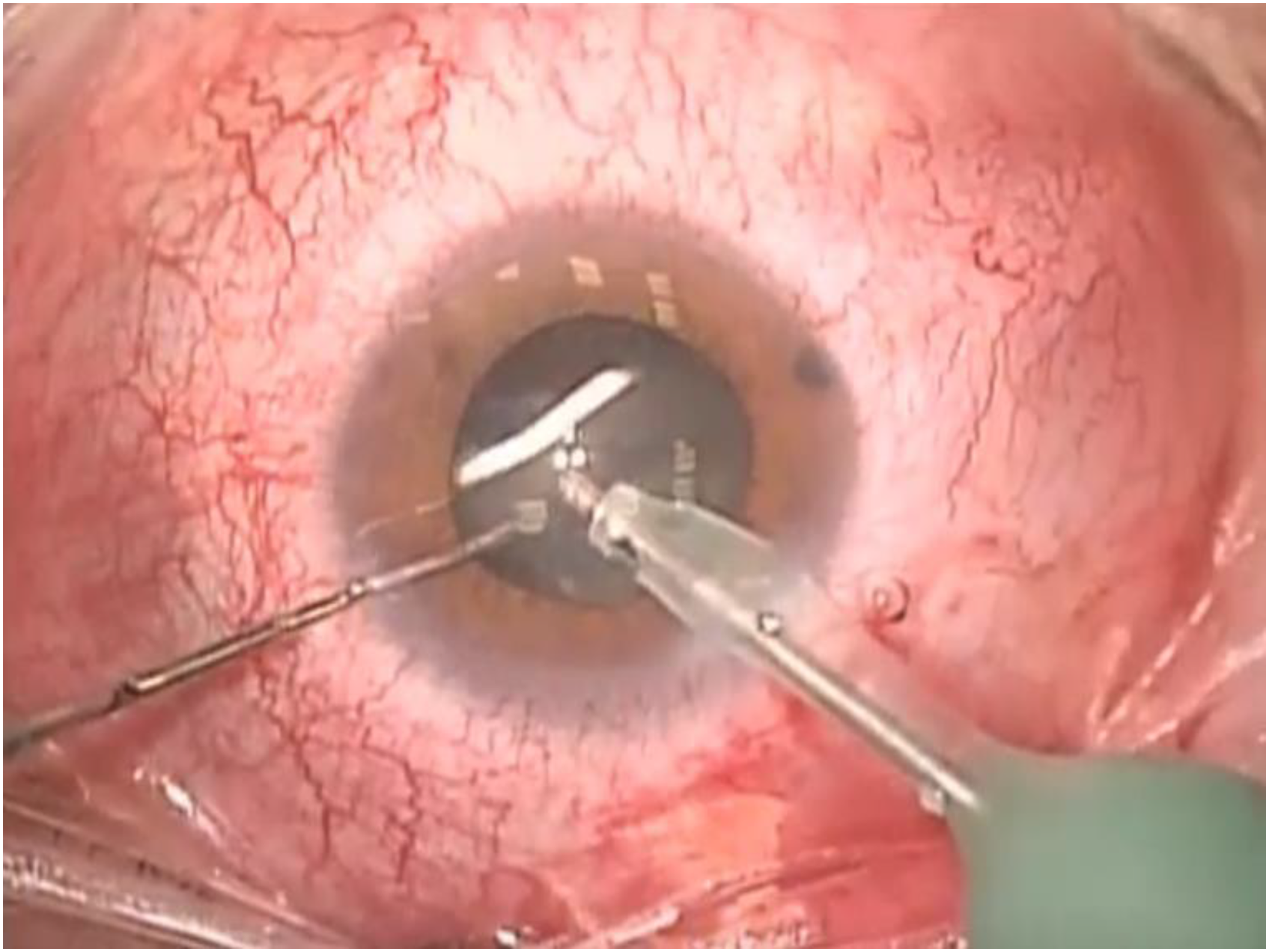
Intralenticular Ozurdex implant is being removed during phacoemulsification.
After 4 weeks since the removal of the DEX implant, macula oedema relapsed in the RE. Consequently, we scheduled a loading phase of 3 intravitreal injections of Aflibercept.
Discussion
Accidental injection into the lens rarely occurs during Ozurdex implantation and only few authors published articles about this uncommon complication. Sborgia et al. reviewed a total of 1066 eyes that underwent DEX implantation between 2015 and 2018, reporting mayor ocular complications in 4 eyes (0.37%). Specifically, they reported migration of the implant into the anterior chamber (2 eyes), acute endophthalmitis (1 eye) and accidental intralenticular implant (1 eye). 5 During a retrospective multicentric case series study of 21,653 patients, Meyer et al. found an incidence of 0,009% (2/21,653 eyes) for lens damage in phakic eyes. 6
In our case, the patient was affected by acromegaly. We wondered if anatomical changes of the crystalline lens are possible in this disease, increasing the risk of accidental intralenticular injection, but only few authors assessed ocular modifications in acromegaly. Altinkaynak et al. evaluated topographic and biomechanical corneal properties in 35 patients with acromegaly, reporting significantly higher central corneal thickness, corneal volume, corneal hysteresis and corneal resistance factor. IOP was also higher compared to the control study group. 7 Chang YH et al. reported a possible association between late bilateral IOL subluxation and acromegaly. 8 Anyway, further clinical evidence is necessary to address the topic in a more complete way.
The most common approach in the management of intralenticular DEX implant is early phacoemulsification of the crystalline lens, followed by repositioning of the implant in the vitreous chamber. If there are no complications, such as cataract progression, IOP increase, dislocation of the lens or infections, a wait-and-see strategy may be possible. In previous case reports, DEX implant was kept inside the lens for a period ranging from 7 to 20 months, retaining its therapeutic effect, and phacoemulsification was only performed when adverse events occurred. 9
In our case, we opted for the wait-and-see strategy, because the patient did not show any of the aforementioned complications. The intralenticular DEX implant of the RE determined the remission of macular oedema within the first 2 months, similarly to the intravitreal implant of the LE. Macular oedema relapsed in the LE after 6 months, while surprisingly, the RE did not show any meaningful anatomical change. After 8 months, IOP increased in the RE, requiring removal of the intralenticular implant, through phacoemulsification.
To the best of our knowledge, this is the first reported case of a patient treated bilaterally with Ozurdex exhibiting this rare complication, making it possible to compare the therapeutic effect of the intralenticular and intravitreal implant in the same subject.
Similarly to previous case reports, our evidence suggests that intralenticular implant effect lasts longer compared to the intravitreal implant. The reason behind this unexpected behaviour is unclear.
Our case is also atypical because posterior capsular rupture did not occur. There are several explanations that could account for this finding. It is possible that the injection was performed anteriorly to the lens equator, without injuring the posterior lens capsule; posterior capsule tear could be present, but too small to be detected; finally, according to recent studies, it may be possible that posterior capsule tear had spontaneously repaired through fibrotic process. 10
Conclusions
Intralenticular DEX implant management requires careful evaluation, because both therapeutic effect and complications differ considerably on an individual base. Cataract onset or progression should be assessed, as well as lens stability, posterior capsular integrity and macular oedema evolution.
Our case demonstrated that the therapeutic effect of intralenticular DEX can be maintained for a longer period of time than intravitreal implant, determining complete reabsorption of macular oedema. Therefore, in selected cases, early phacoemulsification may not be needed, and intralenticular implant may be kept in the lens until either IOP increases, cataract progresses, or other complications occur. Anyway, a strict follow-up schedule is advisable for the management of these patients. Further scientific evidence is needed to understand the reason behind the unexpected therapeutic effect of the intralenticular DEX implant.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article
Informed consent
Written informed consent for patient information and images to be published was provided by the patient.
Research ethics
This study followed the tenets of the 2013 Declaration of Helsinki
