Abstract
Objectives:
To evaluate the corneal effects of the intravitreal dexamethasone implantation using corneal topography and specular microscopy.
Material and methods:
27 eyes of the 27 patients who received a single intravitreal dexamethasone implantation dose for diabetic macular edema were enrolled in this study. Sirius topography and EM-3000 specular microscopic examinations were performed at the initial examination (baseline), and then on the first day, during the first week, and 1 month after IDI. Changes in corneal parameters were investigated.
Results:
The mean age was 58.66 ± 6.59 years. 15 patients were men, and 12 were women. The mean disease duration was 12.2 ± 2.4 months, and mean glycosylated hemoglobin (HbA1c) was 7.2 ± 1.1. After dexamethasone injection, the mean central corneal thickness, endothelial cell density, and coefficient variation of cell area presented a statistically significant decrease (
Conclusion:
Intravitreal dexamethasone implantation affects corneal endothelial cell structure but does not appear to affect corneal topography parameters.
Introduction
Nowadays, intravitreal injection is frequently used in the treatment of many ophthalmic diseases. The increase in the number of patients undergoing this treatment, expectations for good visual outcomes, and possible induction of corneal astigmatism may have become clinically important.
The intravitreal dexamethasone implant (IDI) (Ozurdex, Allergan Inc., Irvine, CA, USA) has been used widely for a variety of conditions, including diabetic macular edema (DME), retinal vein occlusion (RVO), and non-infectious posterior uveitis among others. This injectable implant represents an important innovation in the management of chronic ocular disease using a sustained-delivery technology. Ozurdex® is an intravitreal implant containing 700 μg preservative-free dexamethasone in a slow-release drug delivery system. It is a small rod-shaped implant with dimensions of 0.46 mm diameter and 6 mm length. The OZURDEX® applicator needle is 22 gauge (G) and features a coating designed to facilitate needle gliding through the sclera and into the posterior chamber.1,2
Several studies have investigated the effects of the dexamethasone implant on corneal endothelium.3–5 However, there are no studies in which the effects of the dexamethasone implant on anterior segment parameters have been investigated. We speculated that such posterior segmental interventions would have an effect on the anterior segment, which could lead to a direct effect on visual outcome. In this study, we aimed to evaluate corneal topographic changes and post-injection astigmatism after the IDI injection using the Sirius–Scheimpflug and Placido imaging system. This method is reproducible and measures almost all anterior segment parameters in patients with macular edema secondary to DME.
Methods
In this clinical study, 27 eyes of 27 patients who underwent IDI treatment due to DME were included in this study. All of the study patients were pseudophakic in order to eliminate the effects of cataracts/cataract surgery.
This study was conducted in accordance with the Declaration of Helsinki. All necessary authorizations were obtained from the Institutional Review Board of Okmeydanı Research and Training Hospital, İstanbul, Turkey, with number 894 at 08/05/2018. Written informed consent was obtained from all of the participating patients. All participants underwent a total ophthalmic examination. Best-corrected visual acuity, slit lamp examination, intraocular pressure (IOP) measurements with pneumotonometer, fundus examination, refraction measurements with an auto-keratorefractometer were performed. Patients <18 years, contact lens users and those with previous ocular trauma, history of ocular surgery, ocular inflammation, corneal diseases, cataract, endothelial cell count <1000/mm2, and/or Fuchs endothelial dystrophy were excluded from the study. Measurements of endothelial morphology, such as endothelial cell density (ECD) and coefficient of variation (CV) of cell area, were obtained by noncontact specular microscopy using an EM-3000 Specular Microscope (CBD/Tomey, Phoenix, AZ, USA). Anterior segment parameters, such as central corneal thickness (CCT), anterior chamber depth (ACD), iridocorneal angle (ICA), sim K1 and K2, pupillary diameter (PD), horizontal visible iris diameter (HVID), and corneal astigmatism were examined by Sirius Scheimpflug topography (Costruzione Strumenti Oftalmici, Florence, Italy). Sirius topography and specular microscopy examinations were performed at the initial examination, on the first day, during the first week, and 1 month after dexamethasone treatment.
Before dexamethasone injection, the eye was anesthetized with proparacaine hydrochloride (Alcaine 0.5%, Alcon Pharmaceuticals, Couver, Belgium), and the eyelids and ocular surface were disinfected with 5% povidone iodine. The IDI was then delivered through a 22-G needle with a preloaded dexamethasone implant applicator (Ozurdex) and then inserted into the vitreous cavity through the pars plana (4 mm behind the limbus).
Statistical analyses were performed using SPSS for Windows (Statistical Package for Social Sciences 21.0, SPSS Inc, Chicago, IL). General linear models with repeated-measures analysis of variance (ANOVA) were used to analyze differences in specular microscopy and corneal topography parameters. For all analyses,
Results
27 eyes of 27 patients were included in the study. The mean age was 58.66 ± 6.59. 15 patients were men, and 12 were women. The mean duration of the diabetic retinopathy was 12.2 ± 2.4 months, and mean HbA1c was 7.2 ± 1.1. Baseline characteristics of the patients are shown in Table 1.
The Baseline Characteristics of the Patients.

After IDI, the mean CCT, ECD, and CV showed a statistically significant decrease (
Changes of the corneal parameters.
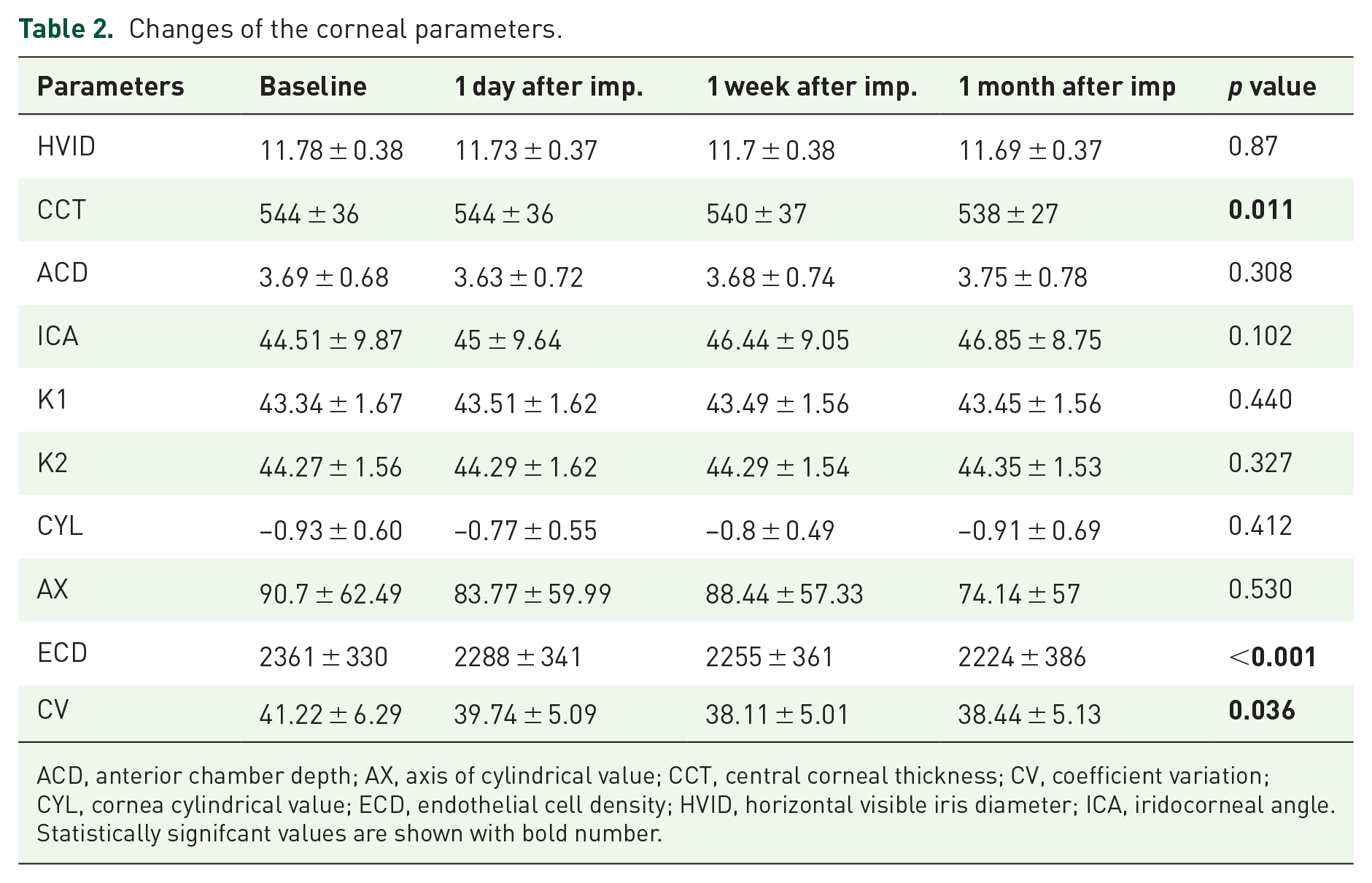
ACD, anterior chamber depth; AX, axis of cylindrical value; CCT, central corneal thickness; CV, coefficient variation; CYL, cornea cylindrical value; ECD, endothelial cell density; HVID, horizontal visible iris diameter; ICA, iridocorneal angle.
Statistically signifcant values are shown with bold number.

The changes in CCT.
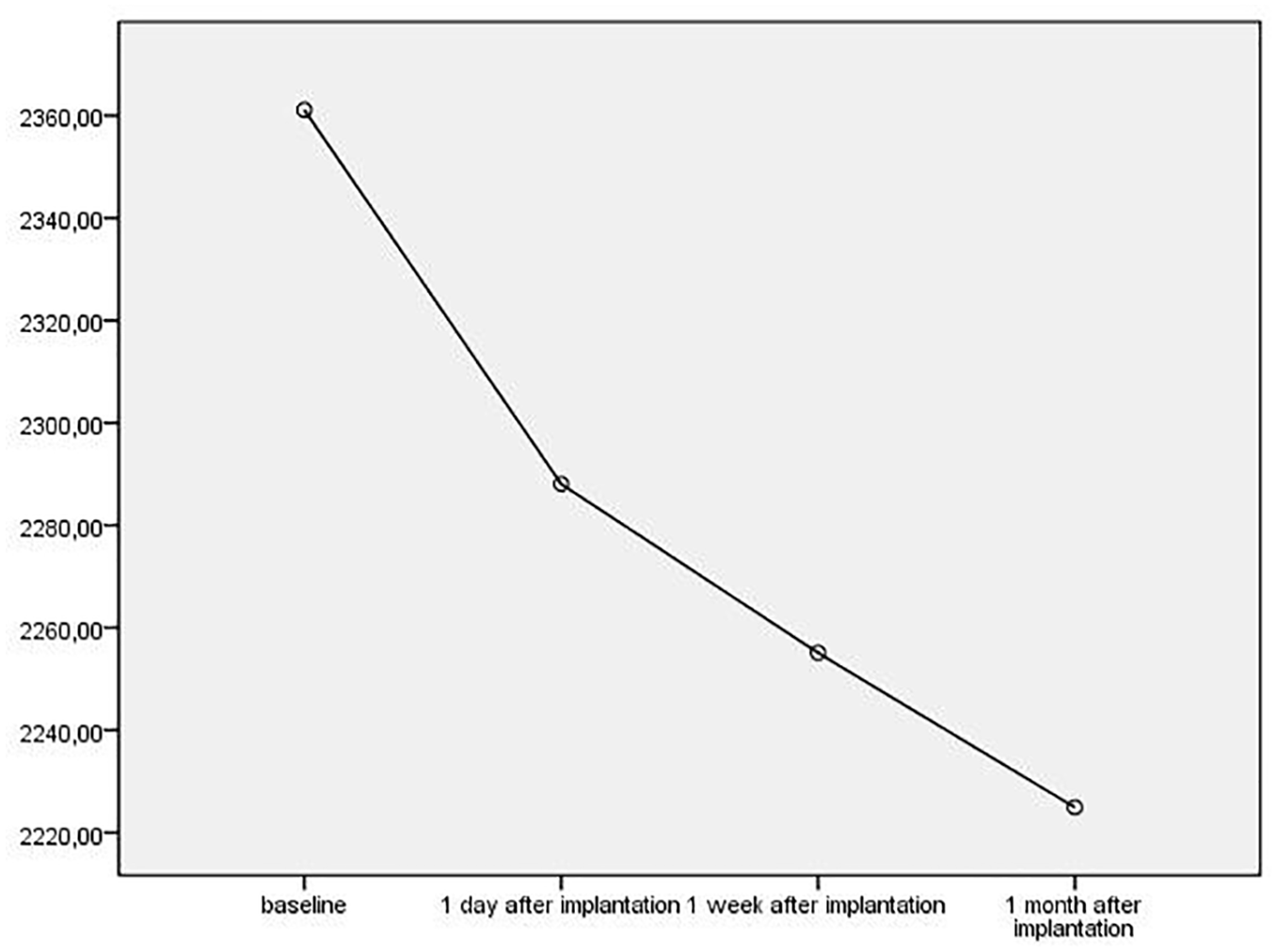
The changes in ECD.
However, for six patients (19.3%), IOP increased to >24 mmHg. They were all successfully treated with antiglaucoma drops. IOPs did not rise above 28 mmHg, and no patients exhibited corneal edema. No other complications, such as intraoperative lens injuries, endophthalmitis, retinal detachment, and/or migration of the implant to the anterior chamber, were observed.
Discussion
Diabetic retinopathy is the most frequent retinal vascular disease. 6 Several options are available for DME treatment. These include focal/grid laser, photocoagulation, subthreshold micropulse laser photocoagulation, intravitreal corticosteroids, intravitreal anti-vascular endothelial growth factor (VEGF), and pars plana vitrectomy (PPV). 7 In many studies, the dexamethasone implant was found to be effective in the treatment of DME. 8 Many studies have shown the effects of intravitreal anti-VEGF on the cornea.9–11 We wanted to examine the effects of IDI, one of the effective treatments for DME, on the cornea.
In previous studies, 29.7% of the implanted patients had IOPs >25 mmHg, 66% had cataract-related side effects, and most of them required cataract surgery in addition to presenting vitreous hemorrhage (10%), and injection-related side effects (<2%). 12 In our study, we took pseudophakic patients in order to avoid cataract progression. IOPs were controlled with antiglaucomatous treatment in cases of increased IOP, and none of patients needed glaucoma surgery. Six of the patients had increased IOP. Although, compared to other patients, the patients with increased IOP did not have any additional decrease in ECD, but we did not know whether increased IOP had effects on ECD.
There was no significant difference in CCT in previous studies after IDI.
3
However, in our study, we found that CCT decreased significantly one after injection (
In the literature, there are studies showing that intracameral dexamethasone applied after cataract surgery and after penetrating keratoplasty is not toxic to the corneal endothelium.5,13 However, in our study, we observed that ECD showed a statistically significant decrease (
Effects of IDI on corneal changes have been previously studied. Kwak and colleagues 16 reported no toxic effect on cornea, retina, and lens in a rabbit model following 400-μg IDI injection. Ilhan and colleagues 3 reported that 0.7-mg IDI application probably had no side effects on the corneal endothelium at 6 months in patients with macular edema caused by RVO. In another study, Güler and colleagues 17 show that IDI application had decreased ECD in RVO patients third month after injections. They did not find any decrease in ECD at first month, while we found decrease at first month. Güler and colleagues studied RVO patients, while we examined the diabetic eyes. It is well known that diabetic patients has lower ECD than healthy subject. 18 These rapid decrease in ECD could be related to combination effects of dexamethasone and DM on ECD.
There are many studies about corneal changes related to post-20-23-25-27-G microincision vitrectomy (especially corneal astigmatism and corneal endothelium).19–21 Many authors have reported that the corneal contour is significantly changed after 20-G pars plana vitrectomy (PPV), inducing surgically induced astigmatism.22–25 In a recent study in which the corneal topographic changes following PPV with the 23- and 25-G transconjunctival sutureless vitrectomy (TSV) in addition to the standard 20-G PPV were compared, it was found that 23- and 25-G TSV did not induce changes in corneal topographic parameters following surgery, whereas 20-G PPV was found to have induced transient topographic corneal changes that returned to pre-operative levels at third month post-operatively. 22
In this study, IDI had no effects on anterior corneal parameters, as ACD, iridocorneal ICA, sim K1 and K2 keratometry, PD, HVID, and corneal astigmatism. These findings appear to confirm the safety of IDI on the anterior cornea.
The short follow-up period and the small sample size were limitations of our study. However, we believe that our study is valuable because it is the one of the few studies showing the effects of IDI on the cornea in patients with DME. However, it should be supported by long-term, large sample-size studies.
Footnotes
Conflict of interest statement
The authors declare no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethics statement
All necessary authorizations were obtained from the Institutional Review Board of Okmeydanı Research and Training Hospital, İstanbul, Turkey with number 894 at 08/05/2018. All patients provided a written informed consent.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
