Abstract
Purpose:
To report patient demographics, the clinical course, and visual outcomes across a case series of patients who experienced dexamethasone (DEX) intravitreal implant (Ozurdex, Allergan, Inc) migration into the anterior chamber (AC), with a focus on the corneal transplantation rate.
Methods:
In this retrospective cross-sectional study, a chart review of all cases was performed. For numerical responses, means and SDs were calculated. Percentages and absolute numbers were used to report the proportion of patients who experienced various outcomes of interest.
Results:
The study comprised 32 cases. All cases occurred in pseudophakic eyes; of those, 8 (25.0%) had posterior chamber intraocular lenses in the capsular bag with no known capsular or zonular issues. The mean duration from DEX implant injection to detection of migration was 19.4 ± 14.5 days. The DEX implant was explanted in 21 patients (65.6%) and repositioned in the vitreous cavity or subconjunctival space in 6 patients (18.8%). Twelve patients (37.5%) ultimately required corneal transplantation.
Conclusions:
To our knowledge, this is the largest case series of DEX intravitreal implant migration into the AC compiled to date. Cases of migration occurred in individuals with no known history of significant prior zonule disruption. This potential complication should be discussed with all patients undergoing DEX implant injection, which may aid in earlier presentation and improved visual outcomes.
Introduction
Dexamethasone (DEX) intravitreal implants (Ozurdex, Allergan, Inc) are a sustained drug-delivery system consisting of 700 µg of dexamethasone in poly(lactic-co-glycolic acid
Over the past decade, there have been an increasing number of case reports describing the migration of DEX implants into the anterior chamber (AC).2–13 Some of these cases have been effectively managed with a combination of patient positioning, dilation, and gentle tapping on the eye, permitting repositioning of the implant back into the vitreous cavity.3,5 Others have described urgent surgical removal of the implant from the AC.10,13 The effect of these migrated implants on vision and corneal endothelium integrity is variable, ranging from minimal changes to nonresolving corneal edema. These corneal changes can progress despite removal of the implant, necessitating corneal surgery.10,13
Several individual case reports of migration have been published. The largest case series described 18 episodes across 15 patients. 13 Our goal was to collect cases of DEX implant migration into the AC across multiple institutions to compile a large, comprehensive case series, with a focus on the proportion of patients who required corneal transplantation. We compare our dataset to the existing body of literature as well as highlight risk factors for migration so that ophthalmologists working with DEX implants may be better able to identify patients at increased risk of this potential complication.
Methods
This study was performed in accordance with the principles outlined in the Declaration of Helsinki. Ethical approval for this study was obtained by the Nova Scotia Health Authority Research Ethics Board (approval number: 1026609). Informed consent was not sought for the present study as a waiver of consent was approved during the ethics board submission based on the possibility of a lack of a continued or existing relationship with the data holder at the time of the retrospective chart review.
This retrospective cross-sectional study comprised patients who experienced DEX implant migration into the AC. Consecutive cases from participating centers were included. If the case involved uneventful intravitreal injection of a DEX implant that migrated into the AC and there was enough information available to complete the circulated spreadsheet, the case was included. A consolidated document containing all cases from contributing sites was reviewed to ensure no duplicate cases were included. Data collected for each case included, but were not limited to, patient age, indication for DEX intravitreal implant injection, symptoms at the time of migration, duration from detection to treatment, type of intervention performed, and visual acuity (VA) before and after implant migration.
For numerical responses, means and SDs were calculated, and t tests were used to compare means, with significance defined as P < .05. Chi-square tests were used to compare distributions of categorical variables. Percentages and absolute numbers were used to report the proportion of patients who experienced the outcomes of interest, such as corneal transplantation, implant explantation, and conservative management.
Results
A total of 32 cases of DEX implant migration into the AC were included. Thirty-three cases from 20 sites were submitted to the study, with 1 to 3 cases contributed by each site. One case was excluded as the DEX implant was erroneously injected into the lens and did not truly migrate from the vitreous cavity to the AC.
Table 1 shows the patients’ characteristics. The mean age of the patients was 66.8 ± 12.5 years; most were men. The most common indications for DEX injection were macular edema secondary to uveitis and postoperative inflammation. The ocular history was significant for previous pars plana vitrectomy (PPV), trauma, and neodymium:YAG capsulotomy
Baseline Characteristics of 32 Patients Who Had DEX Intravitreal Implant Migration Into the Anterior Chamber.
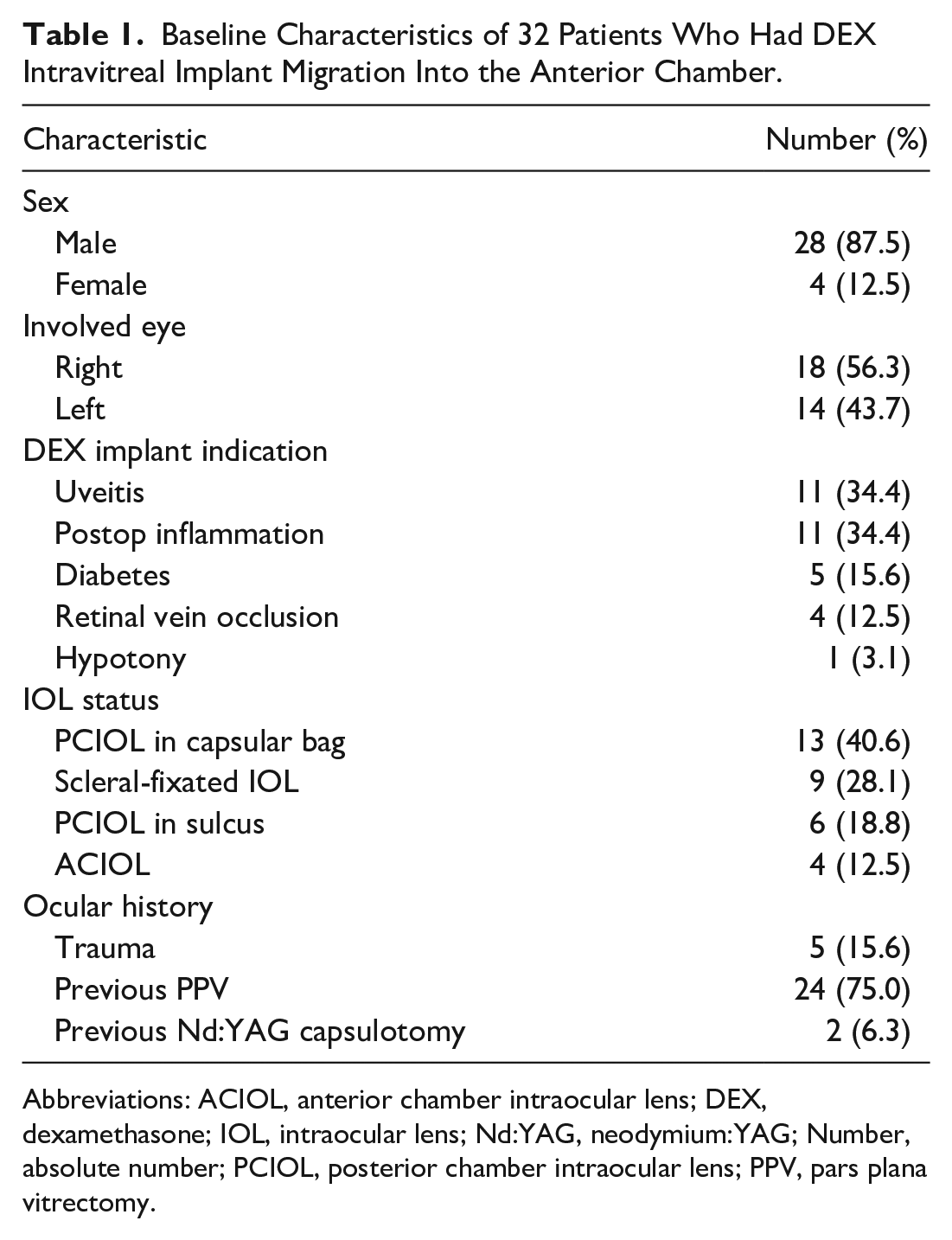
Abbreviations: ACIOL, anterior chamber intraocular lens; DEX, dexamethasone; IOL, intraocular lens; Nd:YAG, neodymium:YAG; Number, absolute number; PCIOL, posterior chamber intraocular lens; PPV, pars plana vitrectomy.
All 32 cases occurred in pseudophakic eyes (Table 1). Of note, 8 patients (25.0%) had a posterior chamber intraocular lens (PCIOL) in the capsular bag with no known capsular trauma or zonular disruption, although 6 of these patients had a previous PPV and 1 had a history of uneventful Nd:YAG capsulotomy. Of the 9 patients with a scleral-fixated IOL, 3 (33.0%) did not have a capsular bag and 2 (22.2%) had documented zonular dehiscence; in 4 cases (44.4%), the status of the capsular bag/zonular fibers was not explicitly mentioned.
Table 2 shows the features and management of patients with DEX intravitreal implant migration into the AC. The majority of patients presented with blurred vision and had corneal edema on initial examination. In the 2 patients who were asymptomatic on presentation, the implant migration was discovered on regular follow-up. The mean time from DEX implant injection to detection of migration was 19.4 ± 14.5 days.
Presenting Features and Management of Patients With Dexamethasone Intravitreal Implant Migration Into the Anterior Chamber.
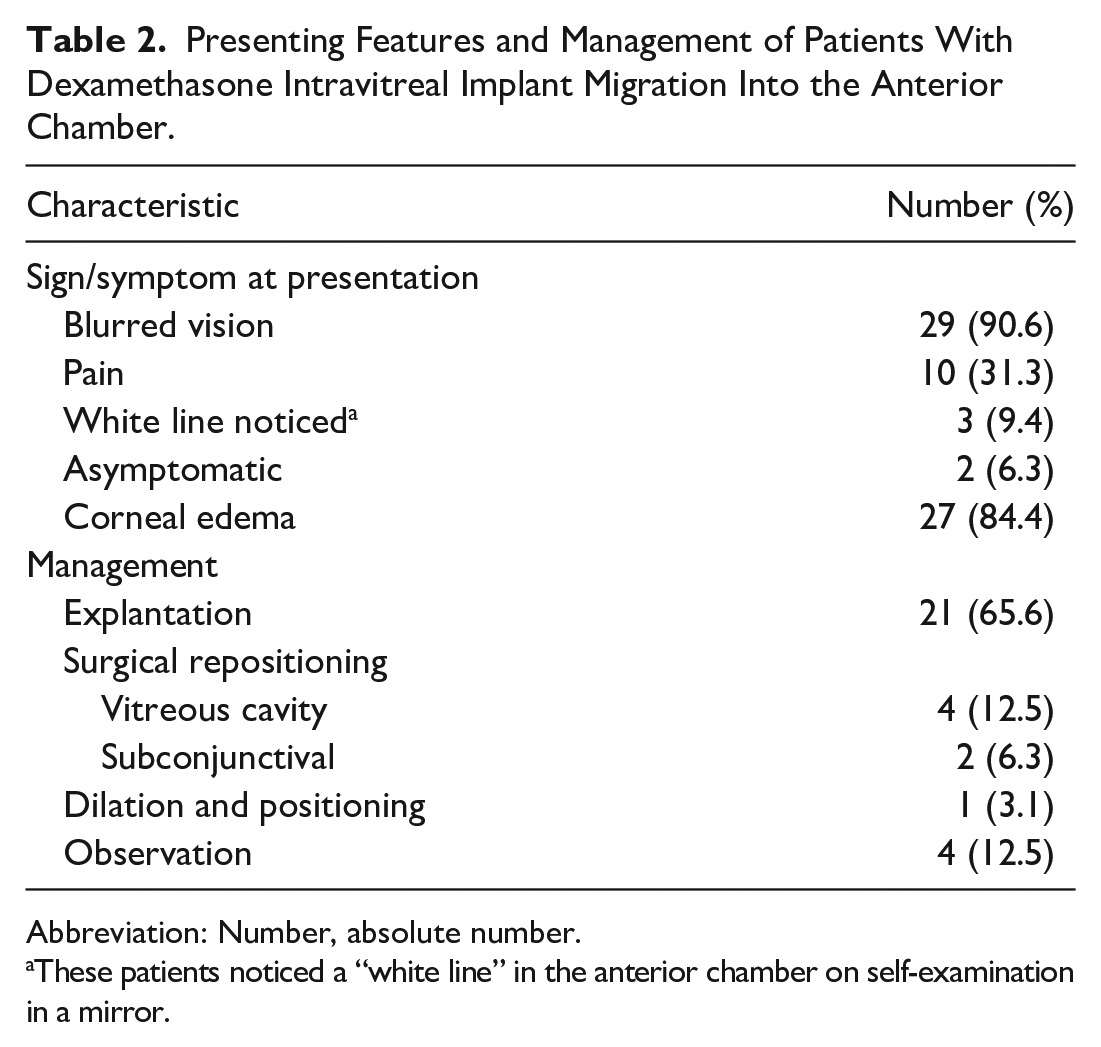
Abbreviation: Number, absolute number.
These patients noticed a “white line” in the anterior chamber on self-examination in a mirror.
Twenty-one patients ultimately had DEX implant explantation, and 6 had their implants surgically repositioned, either in the vitreous cavity or subconjunctival space. Four patients were observed and monitored until the implant dissolved; of those, 2 ultimately required corneal transplantation. One patient was treated conservatively with pupil dilation and positioning alone and did not require corneal transplantation. In 3 patients, the implant re-migrated into the AC after initial repositioning. One of these patients went on to have surgical explantation of the DEX implant, and 2 underwent surgical repositioning procedures (Table 2).
Twelve patients (37.5%) required corneal transplantation for nonresolving corneal edema, and 2 (6.3%) required multiple transplants as a result of graft failure. Two patients (6.3%) requiring corneal transplantation had a history of prior corneal transplantation in the same eye for reasons unrelated to DEX implant migration. The time between management of the migrated implant to the decision to pursue corneal transplantation ranged from 1 to 16 months.
Of the patients who required corneal transplantation, the most common indication for initial DEX implant injection was postoperative cystoid macular edema (6/12 [50.0%]), followed by uveitis (3/12 [25.0%]). One person each had hypotony, branch retinal vein occlusion with macular edema, and diabetic macular edema as their underlying diagnosis. Of the 2 patients who required multiple corneal transplants, 1 had uveitis and the other had hypotony.
Table 3 shows the statistical likelihood of outcomes of interest in patients with DEX implant migration into the AC. Of those who had surgical intervention, the time from symptom onset to surgery was statistically significantly longer in patients who ultimately required corneal transplantation than in those who did not (P = .007). Those who required corneal transplantation were statistically significantly more likely than those who did not have transplantation to have a final Snellen VA of 20/200 or worse at their most recent follow-up (P = .02).
Statistical Likelihood of Outcomes of Interest in Patients With Dexamethasone Implant Migration Into the Anterior Chamber.

Abbreviation: VA, visual acuity.
Statistically significant difference between groups (P < .05).
The time from DEX implant injection to detection of migration was not significantly different between the group that required corneal transplantation and the group that did not (P = .283). There was no statistically significant difference in the corneal transplantation rate among those who were initially managed without surgery and those who had surgery (P = .48) or in those who had the DEX implant left in the eye and those whose implant was explanted (P = .50) (Table 3).
Sixteen cases of migration (50%) occurred with first DEX intravitreal implant injection. Two cases (6.3%) occurred in eyes with more than 10 previous uncomplicated DEX implant injections. Only 2 patients (6.3%) have received repeat treatment with DEX intravitreal implants after the injection complicated by AC migration.
Conclusions
To our knowledge, this is the largest case series of DEX implant migration into the AC to date. A case series published by Khurana et al 13 described 18 episodes of DEX implant migration in 15 patients. Of those, 6 (40.0%) required corneal transplantation, which is fairly consistent with the 37.5% transplantation rate in our case series.
PPV as a risk factor for intravitreal implant migration has been well documented and is thought to be caused by a lack of scaffolding or support for the implant, allowing more opportunity for anterior egress. All 15 patients in the study by Khurana et al 13 had a previous PPV, 13 as was the case in several other previously published case reports.2,11,12 Compounded risk occurs in patients who are concurrently aphakic.2,13,14 However, only 75% of our cases had a history of PPV, and none was aphakic. This suggests that although previous vitrectomy and aphakia increase the likelihood of DEX implant migration, patients lacking one or both of these risk factors should be counseled on the possibility of implant migration.
All patients in the study by Khurana et al had a disrupted capsular bag, zonular dialysis, or trauma, similar to several other reported cases that describe DEX implant migration in the setting of documented posterior capsule rupture.11,13,15,16 There have been published cases of implant migration in patients with intact lens capsules and zonular stability, although with patent iridectomies as the suspected route of entry into the AC. 3 To our knowledge, there are only 4 published cases of AC migration in patients who were pseudophakic but had no documented capsule or zonular deficiencies.5,6,10,12 We had several patients with a PCIOL in the capsular bag and no documented trauma, surgical complications, zonular fiber disruption, or iridectomies. This makes it difficult to anticipate those who might experience this potentially vision-threatening complication, and warrants widespread and comprehensive counseling on this risk before DEX implant injection.
Various management options were used across the cases we collected, including monitoring for absorption, dilation and positioning, tapping on the globe, minor procedures using needles or a balanced salt solution for posterior repositioning, and a wide array of techniques for surgical removal. Sixty-seven percent of those in the study by Khurana et al 13 had the migrated DEX implant removed, which is similar to the 65.6% rate in our study. Observation or conservative management alone was effective in mitigating the need for corneal transplantation in only 3 of our patients.
Only three patients in our study were documented as noticing a “white line” in their eye on gross examination (looking in a mirror), while all patients would have had this finding to some degree. Before implant injection, it may be helpful to show patients what a DEX implant looks like, provide clear visuals of the implant in the AC, and highlight this as an abnormal finding that warrants urgent ophthalmologic assessment. Discussion about the importance of symptoms associated with migration, such as decreased vision, redness, and pain, is also needed given that patients with a longer duration between symptom onset and surgical intervention were more likely to require corneal transplantation in the current study. Improved patient education can lead to better outcomes, as demonstrated in prior studies that have shown those undergoing surgical removal of the implant have a reduced likelihood of permanent corneal edema with earlier intervention. 13
We acknowledge that not all cases of implant migration present for medical attention, given that some patients could remain asymptomatic and the migration might spontaneously resolve. Therefore, this study might underestimate the true incidence of DEX implant migration into the AC.
In conclusion, our large case series highlights the variety of patients who may present with DEX implant migration into the AC. Although previous vitrectomy, zonular or capsular bag disruption, aphakia, and iridectomy increase risk and provide a route for entry into the AC, it is not altogether uncommon for this complication to occur in patients with none of these risk factors. This potential complication, as well as its associated signs and symptoms, should be discussed with all patients having DEX intravitreal implant injection; such discussion might aid in earlier presentation and improved visual outcomes.
Footnotes
Acknowledgements
We would like to thank Dr. Corey Smith for his assistance with statistical analysis.
*Study group authors:
Landon J. Rohowetz, MD2, Nicolas A. Yannuzzi, MD2, Audina M. Berrocal, MD2, Nimesh A. Patel, MD2, Jayanth Sridhar, MD2, Jorge A. Fortun, MD2, Eric Tourville, MD3, Tom Sheidow, MD4, Serge Bourgault, MD5, Steve Levasseur, MD6, Efrem Mandelcorn, MD7, Ananda Kalevar, MD8, Mark Seamone, MD9, Mark Greve, MD9, Geoff Williams, MD10, Amin Kherani, MD10, Arif Samad, MD1, John Galic, MD11, Alan R. Berger, MD12, Karim Hammamji, MD13, Emmanuel Chang, MD14,15,16, Andrew Kirker, MD17, Flavio Rezende, MD18, Nathan Steinle, MD19, Renaud Duval, MD20
1 Department of Ophthalmology & Visual Sciences, QEII Health Sciences Centre, 2035-2 West Victoria Building, 1276 South Park Street, Halifax, NS, Canada
2 Department of Ophthalmology, Bascom Palmer Eye Institute, University of Miami Miller School of Medicine, 900 NW 17th Street, Miami, Florida, USA
3 Department of Ophthalmology, Centre Universitaire d’Ophtalmologie, CHU de Québec-Université Laval, QC, Canada
4 Ivey Eye Institute, Department of Ophthalmology, University of Western Ontario, ON, Canada
5 Université Laval, Hôpital du Saint-Sacrement, CHU de Québec, QC, Canada
6 The University of British Columbia Department of Ophthalmology & Visual Sciences Vancouver, BC, Canada
7 Department of Ophthalmology, University Health Network/Toronto Western Hospital, Toronto, ON, Canada
8 Department of Ophthalmology, University of Sherbrooke, Sherbrooke, QC, Canada
9 Department of Ophthalmology & Visual Sciences, University of Alberta, Edmonton, AB, Canada
10 Department of Surgery, Section Ophthalmology, University of Calgary, Calgary, AB, Canada
11 McGill University; McGill Academic Eye Centre, Montreal, QC, Canada
12 Department of Ophthalmology, St. Michael’s Hospital, Toronto, and Department of Ophthalmology & Vision Sciences, University of Toronto, Toronto, ON, Canada
13 Department of Ophthalmology, Centre hospitalier universitaire de Montréal, Montreal, QC, Canada
14 Retina and Vitreous of Texas, Houston, TX, USA
15 Blanton Eye Institute, Methodist Hospital, Houston, TX, USA
16 Cullen Eye Institute, Baylor College of Medicine, Houston, TX, USA
17 Department of Ophthalmology & Visual Sciences, Faculty of Medicine, Eye Care Centre, Vancouver General Hospital, University of British Columbia, 2550 Willow Street, Vancouver, BC, Canada
18 CUO-HMR, CIUSS de l’Est-de-l’Île-de-Montréal (University of Montreal), 5415 Assumption Blvd, Montreal, QC, Canada
19 California Retina Consultants, Santa Barbara, California, USA
20 Department of Ophthalmology, CUO-Hôpital Maisonneuve-Rosemont, Montréal, QC, Canada
Ethical Approval
Ethical approval for this study was obtained by the Nova Scotia Health Authority Research Ethics Board (Approval number: 1026609).
Statement of Informed Consent
Informed consent was not sought for the present study as a Waiver of Consent was approved during the REB submission due to possibility of a lack of continued or existing relationship with the data holder at the time of retrospective chart review.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: The following authors declare conflicts of interest:
Andy Kalevar: Bayer/Novartis/Allergan/Alcon (Consulting fees, honoraria for lectures/educational events, leadership role)
John Galic: Bayer (honoraria for lectures/educational events)
Nimesh Patel: Allergan/Eye Point/Alimera/Genentech (participation on Data Safety Monitoring or Advisory Board)
Tom Sheidow: Bayer (lecture honoraria), Roche/Novartis (Advisory panel)
Renaud Duval: Bayer (educational and research grant), Bayer/Novartis/Roche (consulting fees), Bayer/Novartis (honoraria for lectures/educational events), Bayer/Novartis/Roche (participation on Data Safety Monitoring or Advisory Board)
Landon Rohowetz: NIH Center Core Grant, Research to Prevent Blindness Grant
Emmanuel Chang: Genentech/Regeneron/Allergan (Advisory board)
Nicholas Yannuzzi: Genentech/Regenxbio (advisory/consulting)
Mark Seamone: Novartis (Advisory board, consulting fees, honoraria for lectures)
Arif Samad: Ozurdex (Review of Ozurdex submission to Health Canada)
Geoff Williams: Bayer/Novartis/Roche (Advisory board), Bayer/Novartis/Roche/Allergan/AbbVie/Chengdu Kanghong (Clinical trials), Bayer (travel grant)
Amin Kherani: Abbvie/Alcon/Bayer/Bausch+Lomb/Novartis/Allergan/Roche/Appellis (Advisory board), Bayer/Novartis/Roche/Genentech/Allergan/Abbvie/Chengdu Kanghong Biotechnology/Ophthotech/Regeneron/Regenexbio/Iveris Bio INC/Opthea/Alexion (Clinical trials)
Serge Bourgault: AbbVie/Bayer/Novartis (Advisory board)
All other authors declare that there is no conflict of interest.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
