Abstract
Case series summary
Five domestic cats diagnosed with Bowenoid in situ carcinoma (BISC) via histopathology were included in this retrospective case series. All cats presented with several dermatologic lesions including but not limited to hyperkeratotic plaques or crusts, focal alopecia, pigmented papules, erosions and secondary pyoderma. The disease was gradually progressive in all cases, with a median time from lesion development to treatment of 365 days (range 84–1614). Of five cats, four had no other clinical signs at the time of presentation; one was pruritic. Each cat was treated with at least one session of carbon dioxide (CO2) laser ablation of the lesions under heavy sedation and local anesthesia or general anesthesia. No adverse events related to treatment were noted. Three cats had documented progressive disease (formation of recurrent or new lesions) at a median of 379 days (range 45–854) after first treatment (laser and/or surgery). Median time to progressive disease after first laser ablation was 269 days (range 158–379; n = 4 cats with follow-up available). Two cats were treated with CO2 laser ablation more than once. One was in complete remission 130 days after surgery and two laser ablation sessions. No cats developed metastatic disease.
Relevance and novel information
Feline BISC is a unique form of virally induced squamous cell carcinoma causing multifocal, superficial cutaneous lesions all over the body. This disease is often progressive, with cats developing regrowth of prior lesions or de novo ones, although deeper invasion and metastasis is rare. Historically, surgical excision and/or immunomodulation with imiquimod cream have been the mainstay of treatment. CO2 laser ablation is a new, well-tolerated, minimally invasive and effective alternative or adjunct treatment that can be repeated as new lesions arise.
Introduction
Bowenoid in situ carcinoma (BISC) is an uncommon, virally induced form of squamous cell carcinoma (SCC) in situ in cats1 –6 similar to human Bowen’s disease. 7 BISC has rarely been documented in dogs.8 –10
BISC lesions are superficial and do not penetrate the basement membrane. They are pre-neoplastic, being neither locally invasive nor metastatic, but have potential for either. In cats, BISC forms on haired or non-haired skin all over the body, often on the head and neck.6,11 Lesions are slow to develop, and were noticed from 1 month to over 12 months before biopsy in one study. 1 They are small (a few millimeters) to 3 cm, pigmented, proliferative or erosive and plaque-like, frequently causing crusts and are often multifocal. 1 BISC can be difficult to distinguish from infectious or autoimmune causes of crusting dermatoses, so skin scrape and cytology, along with fungal and/or bacterial culture, are recommended as baseline diagnostics. 12
SCCs in humans and animals arise from a variety of causes, including ultraviolet (UV) light exposure, immunosuppression and viral infections, especially by papillomaviruses. 6 Many studies have shown a strong association between feline papillomavirus infection and BISC development.1,11,13,14 Human papillomavirus infection is implicated in Bowen’s disease, 15 although there is also a solar component.
Feline BISCs are not induced by UV radiation, so light-coated cats and those with known sun exposure are not more commonly affected than others. Cats are middle-aged to older at the time of diagnosis, and there is no known sex or breed predilection.1,4 Biopsy and histopathologic evaluation are required for definitive diagnosis. BISC tissues should lack actinic changes, the presence of which would indicate solar damage. 6
If left untreated, BISC could progress to invasive SCC. This is very rare, reported in 3–5% of patients with Bowen’s disease, and inconsistently documented in veterinary studies. 4 Typically, cats develop new lesions in novel locations. 1 The disease is uncommonly painful or pruritic, although secondary infection can occur. One case series described concurrent Demodex cati infestation potentially due to local immunodeficiency. 3 When possible, the mainstay of treatment in human and veterinary medicine is surgery, as wide excision can be curative. In addition, because of the multifocal nature of the disease, future surgical procedures are recommended when new lesions develop. However, cat owners may be hesitant to pursue general anesthesia because of perceived risk in their aged animals, financial limitations or the large amount of skin to be removed due to the multifocal nature of this disease.
Medical management with imiquimod 5% topical cream has been described in humans 16 and cats. 4 Imiquimod stimulates Toll-like receptor 7 of the innate immune system, resulting in direct antiviral effects. In one case series of 12 cats, all had an initial response to treatment. 4 Nine cats developed new lesions, but all re-treated lesions again improved or resolved. Side effects reported in 25% or less included local erythema, increased liver enzymes, neutropenia, hyporexia and vomiting. The median time to development of new lesions was 50 days (range 30–122) and the median survival time calculated using a time-to-failure model was 243 days (range not reported). 4 Other medical treatments with corticosteroids or retinoids have not been proven effective; however, photodynamic therapy and radiation show promise. 6
More recently in veterinary dermatology, carbon dioxide (CO2) laser has been utilized in the management of many benign and neoplastic conditions including BISC.17,18 CO2 lasers penetrate tissue to a depth of approximately 0.03 mm; however, once the laser energy is absorbed, the beam exerts an ablative effect by heating intracellular and extracellular water, resulting in vaporization of the tissue. 18 This process is painful, so cats must be under a plane of heavy sedation or general anesthesia. Lesions heal within 3–4 weeks, with minimally noticeable scars and hair regrowth. Procedures are recommended to be repeated up to every 12–18 months as needed. 17
This case series aims to describe the presentations and outcomes of cats with BISC treated with at least one session of CO2 laser ablation.
Case series description
Medical records from between January 2021 and September 2025 from a single academic institution were reviewed. Data collected included signalment, history, presenting complaint/clinical signs, physical examination, clinicopathologic and imaging findings, histologic diagnosis, treatments, adverse events (AEs) and outcomes. For inclusion, cats had to be diagnosed with at least one BISC lesion via histopathology and treated with CO2 laser. Prior or concurrent medical or surgical treatments were allowed. The initial and re-staging diagnostics and recheck timeline were based on clinician discretion. Five cats met the inclusion criteria; specific information about each is detailed in Table 1. Because of the small sample size, only descriptive analysis was performed.
Signalment, history, lifestyle, diagnostic methods and treatment outcomes
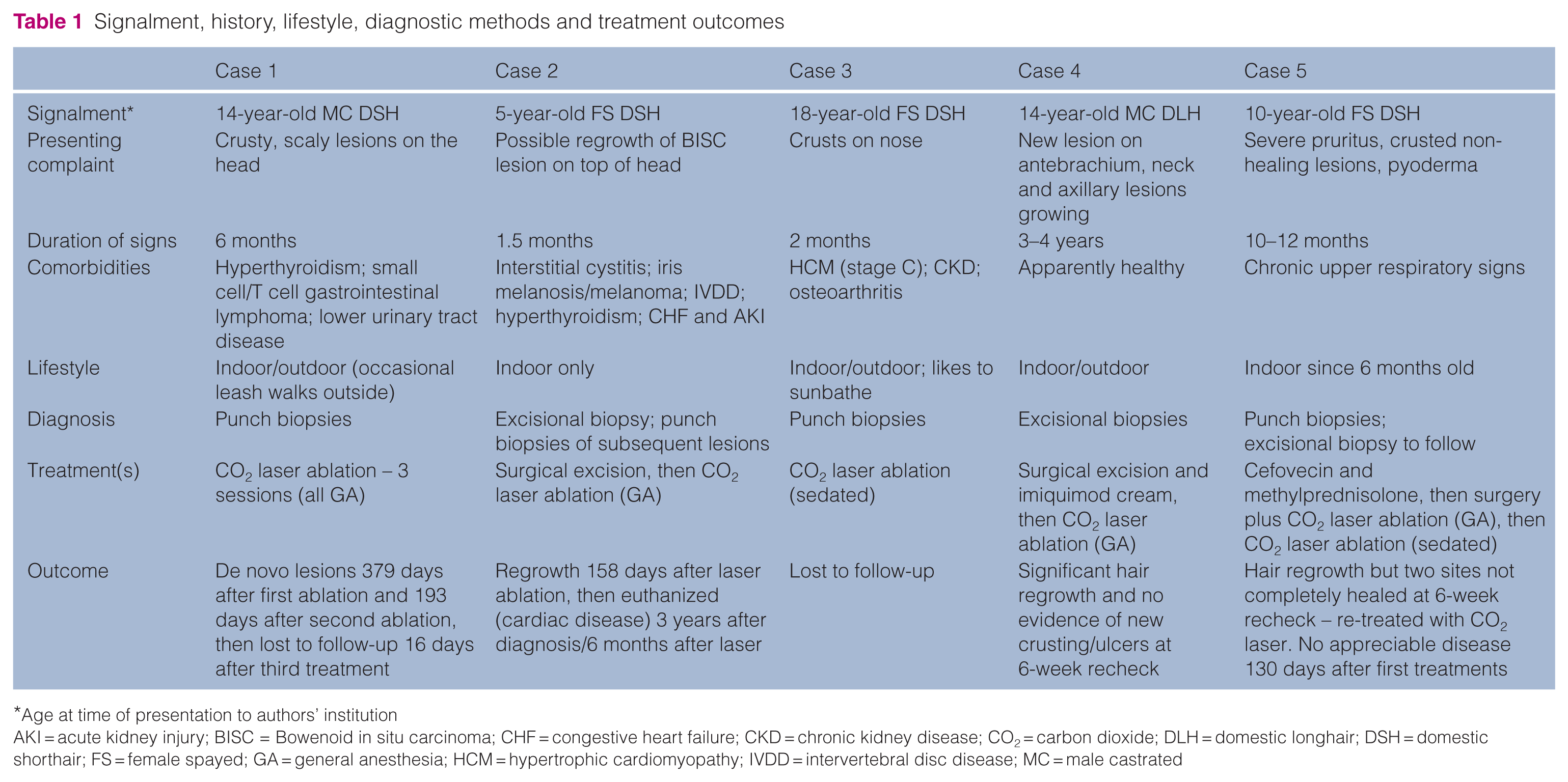
Age at time of presentation to authors’ institution
AKI = acute kidney injury; BISC = Bowenoid in situ carcinoma; CHF = congestive heart failure; CKD = chronic kidney disease; CO2 = carbon dioxide; DLH = domestic longhair; DSH = domestic shorthair; FS = female spayed; GA = general anesthesia; HCM = hypertrophic cardiomyopathy; IVDD = intervertebral disc disease; MC = male castrated
The presenting complaint in all cats was dermatologic: hyperkeratotic plaques or crusts, focal areas of alopecia with underlying erosion, pigmented papules, scale and, in one (case 5), serosanguineous and purulent exudate from the lesions. This cat had a 10–12-month history of generalized pruritus, otitis and non-healing wounds and had received supportive treatments including, but not limited to, oral, otic and injectable antibiotics and corticosteroids. See Figure 1 for examples of pretreatment lesion appearances. Aside from case 5, owners reported that their pets were unbothered by the lesions. The median duration from when lesions were noted to treatment was 365 days (range 84–1614). Figure 2 outlines the timeline of lesion development, treatment(s) and progression of disease.

Lesion appearance before treatment, immediately after treatment and at 6 weeks after treatment in two cases

Timeline before and after treatments. PFI = progression-free interval
Comorbidities at the time of diagnosis included hyperthyroidism (cases 1 and 2), recurrent lower urinary tract disease (cases 1 and 2), small cell gastrointestinal lymphoma (case 1), chronic upper respiratory disease of unknown etiology (case 5) and osteoarthritis (case 3), as well as both hypertrophic cardiomyopathy (stage C) and chronic kidney disease that were present in one cat (case 3) and congestive heart failure and acute kidney injury in another cat (case 2). Affected cats received appropriate medical management of these conditions alongside treatment of BISC.
Aside from skin lesions, physical examinations were generally unremarkable at initial presentations. All cats were assessed to be euhydrated and had vital signs within normal parameters. In the cat with known cardiac disease (case 3), an arrhythmia was auscultated. Case 2 later developed congestive heart failure; at recheck examination, it was noted to be tachypneic and dehydrated, and a new gallop sound was auscultated.
Clinicopathologic findings at or near the time of diagnosis were available for review in four cats. Complete blood count revealed thrombocytopenia (n = 3, although clumping was noted on blood smears), anemia (n = 2) and lymphopenia (n = 2). Chemistry and total thyroid hormone values were generally unremarkable, although case 2 later developed an acute kidney injury suspected to be an adverse effect of furosemide administration. Retroviral (feline leukemia virus/feline immunodeficiency virus) status was known in four cats, and all were negative.
Initial cytology and/or culture of the lesions were consistent with infection in two cases (cases 1 and 3) and concerning for neoplasia in one case (case 5), with large, basophilic cells noted. The official diagnosis of BISC was obtained via punch (n = 3) or excisional (n = 2) biopsies of at least two representative lesions and histopathology. All tissues obtained were reviewed by board-certified veterinary pathologists. The median mitotic count in 10 high-power fields was 15 (range 4–87). Initial histopathology samples from case 5 also had characteristics of basal cell carcinoma, another probably virally induced neoplasm. After surgical excision of this cat’s largest lesion, final histopathology confirmed it was a basal cell carcinoma, with few basal cells invading the basement membrane. Additional biopsies of the smaller lesions were not obtained, so they were assumed to be BISC.
Initial treatment included surgical excision (case 2), a combination of surgery for the largest lesion and laser ablation for the smaller ones (case 5), and surgery followed by imiquimod cream (case 4). The owner of case 4 reported that the cat would hide and seemed uncomfortable when the imiquimod was applied, so it was inconsistently administered.
At the time of the CO2 laser ablation, the number of sites treated ranged from 2 to >10, and lesions were small (<1 cm). The median procedure duration was 24 mins (range 18–76). The CO2 laser (Aesculight; VetScalpel) was used in a continuous mode, with power settings in the range of 5–20 W. Using the paintbrush tip in a painting motion, the superficial epidermal layer of the lesions was ablated until all visible abnormal, pigmented tissue was removed. Silver sulfadiazine cream (SSD) was frequently applied to the treated areas immediately after the procedure. The same laser equipment was utilized each time by one of two board-certified veterinary dermatologists. Cats were heavily sedated or under general anesthesia and sometimes received additional local anesthetic (buffered lidocaine). All were discharged the same day.
Owners were instructed to apply a thin layer of SSD to the treated areas q24h to q12h to promote skin healing for at least several weeks or until the time of recheck. Oral analgesics, non-steroidal anti-inflammatory drugs, corticosteroids and antihistamines were intermittently prescribed. Recheck examinations were recommended 5–6 weeks after laser ablation to assess healing, then as needed if there was development of new lesions or concerns. In all cases, there were no reported immediate or long-term AEs. By the time of the first recheck examinations, clinical signs had improved greatly or resolved, with hair regrowth and decreased severity of pruritus in all evaluated cats (4/5). Figure 1 illustrates pretreatment, immediate post-laser and 6-week recheck lesion appearance for cases 4 and 5.
Three cats (cases 1, 2 and 4) developed progressive disease (PD), manifesting as skin lesions away from or near the previously treated sites. The median time to PD after first laser ablation was 269 days (range 158–379). Case 2 had recurrence (in the same anatomic region) of a narrowly excised BISC over 2 years later, confirmed with histopathology. This cat had suspected recurrent disease again 6 months after laser ablation but was not re-treated because of severe cardiac and renal disease. Histopathologic examination of new lesions was not performed before CO2 laser ablation in case 4.
Owing to this high frequency of PD, two cats underwent more than one CO2 laser procedure. Case 1 was treated again 12 and 18 months after the first ablation. Crusted lesions were noted on examinations but in different sites from those previously treated, so were considered de novo. Biopsy and histopathology of the new lesions were declined before the subsequent treatments. Case 5 had suspected incomplete treatment of two lesions, as mild crusting persisted at the sites 41 days later. Recheck histopathology was not pursued and a second CO2 laser ablation session was performed.
No cats had evidence of metastatic disease at diagnosis or rechecks, although staging and re-staging diagnostics were not consistently performed. However, after initial surgery and before adjunct treatment with CO2 laser, an irregular, cavitated pulmonary nodule was seen in the right caudal lung lobe on radiographs of case 4. Rule-outs included a single metastatic lesion, primary pulmonary neoplasia or a non-neoplastic mass; however, the latter was considered unlikely. The cat had no history of respiratory clinical signs, and additional imaging was not pursued. This case was lost to follow-up after the 6-week recheck. In case 5, thoracic radiographs and abdominal ultrasound were unremarkable 130 days after initial treatment.
Long-term outcomes were favorable in cases where data were available for review (4/5 cats). The median time to last follow-up after initial laser ablation was 144 days (range 35–572). Three cats (cases 1, 4 and 5) had significantly improved to resolved lesions at these follow-ups. Case 2 developed clinical cardiac and renal disease and was euthanized owing to recurrent owner-described syncopal events approximately 3 years after initial diagnosis and surgical excision, and 6 months after laser ablation. The cause of death was assumed to be unrelated to BISC, although a post-mortem examination was not elected. Case 5 is alive at the time of writing, more than 6 months after the initial ablation.
Discussion
Feline BISC is an uncommon dermatologic disease, primarily characterized by slow progression of multifocal plaques and crusts. Secondary infection and pruritus may occur. Affected cats are aged and have variable UV-light exposure (Table 1). Combination therapies including surgery, medications and CO2 laser ablation appear to provide the best outcomes.
The cats in this case series had similar signalments and presenting characteristics to those in prior studies,1 –5 indicating that this cohort is likely representative of the larger population of affected cats. The median age at presentation to the authors’ institution was 14 years, although one cat (case 2) was first diagnosed at approximately 1.5 years. All were domestic shorthair or domestic longhair breeds and neutered. Lesions were commonly crusted, pigmented plaques causing cosmetic concerns, with only one cat (case 5) clinically impacted. This cat also had a documented skin infection before treatment. No cats were suspected of being immunosuppressed due to retroviral infection, although one had concurrent neoplasia.
All cats tolerated CO2 laser ablation procedures well. Treatments were performed quickly and on an outpatient basis, arguing for their use by many practitioners. There were no documented acute or chronic AEs. When evaluated 6 weeks after ablation, lesions were more than 90% healed in all rechecked cats (4/5).
PD manifested as de novo skin lesions or regrowth at a previously treated site in three cases, at a median of 269 days (range 158–379) after the first laser treatment. None had clearly documented metastatic disease, although case 4 had a suspicious pulmonary lesion. This pattern of progression is like that noted in previous studies, so it is probably representative of BISC biologic behavior. Although CO2 laser ablation appears effective in the short-term management of BISC, owners should be counseled that additional procedures are likely needed for long-term control. Median overall survival time after CO2 laser treatment could not be quantified in this case series and should be a focus of future studies.
Limitations are inherent in a case series because of the small sample size, which prohibited inferential statistical analysis. Staging diagnostics were inconsistently recommended and performed, so the exact timeline of disease progression may not match that reported and metastatic disease may have been underappreciated. Tissue samples were examined by multiple pathologists and were not tested for papillomaviral infection. Not all initial lesions were sampled, new or recurrent similar-appearing skin lesions suspected to be BISC were not always histologically confirmed and no cases had post-mortem pathology performed. Although histopathologic examination of every lesion would have been ideal, this is often not practical nor widely practiced. Finally, there was no control group untreated with CO2 laser to compare lesion progression and clinical signs.
Ongoing investigation should include elucidation of prognostic factors for recurrence and long-term side effects of therapies, especially when used in combination or sequentially.
Conclusions
Surgical excision and/or medical management with imiquimod cream have been the historical mainstay treatments for feline BISC. Transformation into invasive SCC or development of metastatic lesions are rare. However, formation of new lesions at other sites on the skin is common. This case series illustrates that CO2 laser ablation is a well-tolerated, minimally invasive and effective alternative or adjunct treatment for feline BISC that can be repeated as new lesions arise, although it is not expected to be curative. Larger, prospective case-control studies are necessary to determine the best management strategies for feline BISC and the role of CO2 laser ablation.
Footnotes
Conflict of interest
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
The work described in this manuscript involved the use of non-experimental (owned or unowned) animals. Established internationally recognized high standards (‘best practice’) of veterinary clinical care for the individual patient were always followed and/or this work involved the use of cadavers. Ethical approval from a committee was therefore not specifically required for publication in JFMS. Although not required, where ethical approval was still obtained, it is stated in the manuscript.
Informed consent
Informed consent (verbal or written) was obtained from the owner or legal custodian of all animal(s) described in this work (experimental or non-experimental animals, including cadavers, tissues and samples) for all procedure(s) undertaken (prospective or retrospective studies). No animals or people are identifiable within this publication, and therefore additional informed consent for publication was not required.
