Abstract
Objectives
The aim of the study was to review diagnoses and infectious disease testing of cats with neutrophilic or pyogranulomatous lymphadenitis, and describe the history, clinicopathological abnormalities, diagnostic imaging findings, lymph node cytological/histological appearance, treatment response and outcome of steroid-responsive lymphadenitis in cats.
Methods
The medical records of 72 cats documented to have pyogranulomatous, neutrophilic or mixed lymphadenitis between January 2015 and December 2023 at a single referral hospital were retrospectively reviewed.
Results
Feline infectious peritonitis (25/72, 35%) was the most common cause of lymphadenitis and frequently pyogranulomatous. Neoplasia (11/72, 15%) was the overall next most common cause and the most common cause of neutrophilic lymphadenitis. The remaining diagnoses included nine (12.5%) cats with steroid-responsive lymphadenitis, eight (11%) with inflammatory disease, seven (10%) with other suspected bacterial infections, five (7%) with mycobacterial infections, five (7%) with neutrophilic to pyogranulomatous lymphadenitis of unclear cause and two (3%) with toxoplasmosis with variable inflammatory lymphadenitis. Steroid-responsive lymphadenitis cases were often middle-aged (median 7 years 10 months) castrated males (6/9). Common signs included lethargy (7/9, 78%), hyporexia/anorexia (7/9, 78%), pyrexia (7/9, 78%), peripheral lymphadenopathy (6/9) and weight loss (5/9). Frequently affected lymph nodes were submandibular (5/9), popliteal (4/9) and jejunal (4/9). Cytology was more commonly neutrophilic (8/14, 57%) than pyogranulomatous (4/14, 29%). After variable infectious disease testing, all cats received prednisolone (median dose 1.25 mg/kg, range 0.5–2.3) for a median of 5.75 months (range 4–11). All improved initially; relapse occurred in 33% during treatment, after tapering or discontinuation. Two cats (22%) were euthanased after relapse, with one developing neurological signs.
Conclusions and relevance
Steroid-responsive lymphadenitis should be considered in cats presenting with inflammatory lymphadenopathy when no identifiable underlying infectious, inflammatory or neoplastic cause is identified, and most cases respond well to glucocorticoid treatment.
Plain language summary
This study looked at cats with a particular type of lymph node inflammation called neutrophilic or pyogranulomatous lymphadenitis. These types of inflammation are usually caused by infection or cancer, but in some cats, no obvious cause is found. The aim was to find out which diseases caused this type of inflammation and what infectious disease tests were performed in these cases. We also focused on a small group of cats who responded well to steroid treatment after an infectious or cancerous cause was excluded, which suggests their condition might have been immune-related. We reviewed 72 cats and found the most common diagnosis was feline infectious peritonitis (FIP), diagnosed in 25 cats, but other causes included cancer, bacterial infections and rare infections, such as mycobacterial infections and toxoplasmosis. In nine cats, no infection, inflammatory condition or cancer was found, but they improved with steroid treatment and were diagnosed with steroid-responsive lymphadenitis. These cats were often middle-aged, castrated males. Their most common signs included tiredness, fever, swollen lymph nodes and weight loss. The lymph nodes under the jaw or associated with the intestines were most frequently affected. Diagnostic testing was used to rule out potential infections like FIP, toxoplasmosis and mycobacterial infections. Most of these cats received a steroid called prednisolone for several months, and the majority improved. Two cats were euthanased after relapsing, and 33% of cats had signs return during treatment or after their dose of medication was reduced. This study shows that if no infection or cancer is found, some cats with this type of lymph node inflammation may benefit from steroid treatment.
Keywords
Introduction
Cats may present with peripheral, intrathoracic and/or intra-abdominal lymphadenomegaly, which can have a solitary, regional or generalised presentation. Lymphadenomegaly can be a presenting complaint or be found during a physical examination and is often identified in cats presenting with pyrexia and non-specific clinical signs, including hyporexia, lethargy and weight loss. Causes of lymphadenomegaly include neoplasia, metastatic neoplasia, reactive hyperplasia and lymphadenitis (neutrophilic, eosinophilic, pyogranulomatous or granulomatous). 1 Lymphadenitis is an uncommon cause, with two recent studies identifying it in only 10% of 60 abdominal lymph node biopsies obtained during exploratory laparotomy and in 8.5% of lymph node fine-needle aspirates from 199 cats.2,3 Pyogranulomatous lymphadenitis is a chronic inflammatory response to certain infectious pathogens or foreign material and is characterised by infiltration of the lymph nodes by neutrophils and epithelioid macrophages. 4 Neutrophilic lymphadenitis is characterised by the presence of more than 5% neutrophils and has been associated with bacterial (eg, Escherichia coli, Mycoplasma felis), neoplastic or immune-mediated conditions.4 –6 In contrast, granulomatous inflammation consists of macrophages and multinucleated giant cells.7,8
In cats, pyogranulomatous inflammation is most often associated with infectious disease, particularly feline infectious peritonitis (FIP), and is commonly generalised as a sequela of systemic feline coronavirus (FCoV) infection, including reported cases of necrogranulomatous mesenteric lymphadenitis.9,10 Pyogranulomatous lymphadenitis has also been reported in fungal and bacterial infections, including aspergillosis, disseminated mycobacterial infection, E coli, Listeria species, Actinomyces species, Rhodococcus equi, Anaerobiospirillum species and necrotising granulomatous lymphadenitis in toxoplasmosis.11 –22 Bartonella species infection has also been associated with pyogranulomatous myocarditis, pancarditis and diaphragmatic myositis in cats.23,24 In addition, a mild pyogranulomatous lymphadenitis can also be seen in lymph nodes that drain areas of chronic inflammation or neoplasia. 25
Case reports of sterile pyogranulomatous lymphadenitis in young Springer Spaniels have been reported with or without concurrent cutaneous lesions or nodular panniculitis.26,27 Two recent retrospective studies reported the characteristics and outcome of steroid-responsive pyogranulomatous lymphadenitis comprising over 100 dogs collectively. Diagnosis is made by excluding infectious, inflammatory or neoplastic causes. A possible immune-mediated aetiology is suspected, given the absence of an obvious infection, poor response to antibiotics and rapid response to glucocorticoids or immunosuppressive treatment.28,29
In cats, idiopathic sterile pyogranulomatous dermatitis has been reported, and a recent case series described three cats with sterile pyogranulomatous masses without cutaneous involvement, two of which improved with glucocorticoid treatment.30,31 To date, only a single case report has described steroid-responsive, generalised sterile necrotising pyogranulomatous lymphadenitis in a young cat. 32 Extensive infectious disease testing was negative. The cat remained pyrexic after 9 days of broad-spectrum antibiotics (amoxicillin-clavulanic acid) but showed resolution of peripheral lymphadenopathy and pyrexia within 1 week of initiating immunosuppressive doses of prednisolone, with no relapse 7 months after discontinuation. 32 This represents the first description of this condition in cats in the veterinary literature, highlighting that an immune-mediated cause of pyogranulomatous lymphadenitis may also occur in cats.
Diagnostic investigations of feline lymphadenitis can be challenging and costly, often requiring extensive infectious disease testing to reach a definitive diagnosis. However, there is a paucity of evidence-based guidance to support clinical decision-making in these cases and, to date, no comprehensive description of steroid-responsive or presumed sterile lymphadenitis in cats has been published.
The primary aim of this retrospective study was to report the history, clinicopathological abnormalities, diagnostic imaging findings, lymph node cytological/histological appearance, treatment response and outcome of steroid-responsive lymphadenitis in cats. Further objectives were to report the prevalence of the various diagnoses in cats with neutrophilic or pyogranulomatous lymphadenitis identified at a single veterinary referral hospital in the UK, and to report the infectious disease testing performed in lymphadenitis cases. Therefore, the authors will first present the summary of findings for cats with neutrophilic/pyogranulomatous lymphadenitis of any included cause, followed by a more focused evaluation of a group of cats with steroid-responsive disease.
Materials and methods
Study design and case identification
The medical records of cats in which pyogranulomatous, neutrophilic or mixed (pyogranulomatous with eosinophilic) lymphadenitis was identified on cytology or histopathology between January 2015 and December 2023 at a single UK referral veterinary hospital were retrospectively evaluated. Case identification was performed by searching the hospital’s electronic practice management system using the keywords ‘pyogranulomatous’, ‘pyogranulomatous inflammation’, ‘pyogranulomatous lymphadenopathy’, ‘pyogranulomatous lymphadenitis’, ‘neutrophilic lymphadenitis’, ‘granulomatous lymphadenitis’, ‘sterile lymphadenitis’ and ‘sterile lymphadenopathy’ during the study period. All patients included were client-owned cats and all investigations performed were clinically indicated.
Cats were eligible for inclusion if cytology and/or histopathology identified neutrophilic, pyogranulomatous, granulomatous or mixed lymphadenitis in lymph node samples obtained by fine-needle aspiration (FNA) or biopsy. Cats with purely eosinophilic lymphadenitis were not included. Cytology and/or histopathology reports were required to have been written or reviewed by a board-certified clinical pathologist. Pyogranulomatous lymphadenitis was diagnosed cytologically based on a mixed inflammatory infiltrate with increased numbers of neutrophils and macrophages within the lymph node; granulomatous lymphadenitis was defined as the presence of more than 2% histiocytic cells; neutrophilic lymphadenitis as more than 5% neutrophils; and necrotising lymphadenitis was diagnosed when necrosis accompanied neutrophilic or histiocytic inflammation. Mixed lymphadenitis was diagnosed based on the presence of a mixed inflammatory infiltrate of neutrophils, macrophages and eosinophils.4,33 Cats were categorised into groups based on their final diagnoses: FIP, Toxoplasma gondii, mycobacterial infection, inflammatory disease (including feline gastrointestinal eosinophilic sclerosing fibroplasia [FGESF]), other bacterial infection, steroid-responsive disease and unclear diagnosis. FIP was either definitively diagnosed by detection of FCoV antigen by immunostaining in association with typical cytological or histopathological features of FIP, or presumptively diagnosed based on positive FCoV PCR, signalment, clinical history, clinicopathological abnormalities (including abdominal fluid analysis), imaging findings and exclusion of other disease processes or compatible post-mortem findings. 34 T gondii infection was tentatively diagnosed by a four-fold increase in IgG antibody titre or detection of oocysts on faecal analysis in combination with compatible clinical signs. Mycobacterial infection was diagnosed using Mycobacterium species PCR. Neoplasia was diagnosed by either cytology or histopathology of lymph nodes, mass lesions, nasal biopsies, or endoscopic or surgical gastrointestinal biopsies in two cases, supported by the presence of monoclonal lymphocytes with PCR for antigen receptor rearrangement, or was highly suspected based on diagnostic imaging findings. Cases were categorised as inflammatory based on the final primary diagnosis being a strongly suspected/diagnosed inflammatory condition (eg, lymphocytic cholangitis with evidence of a hepatopathy), with inflammation present in other locations in addition to the lymph node (eg, liver, gastrointestinal tract) with documented response to glucocorticoids if they were indicated. Cats with inflammation of the gastrointestinal tract largely consisted of cases of FGESF, with either marked gastrointestinal changes, including loss of intestinal wall layering, or histiocytic colitis confirmed on histopathology. In these cases, lymphadenitis was often mixed, with an eosinophilic component, with or without intralesional bacteria within the lymph node. Other bacterial infection was suspected if bacteria were identified on cytology or if there was a clear response to antibiotics, evidenced by improvement in clinical signs and/or lymphadenomegaly. This was also suspected in cats diagnosed with conditions commonly associated with a bacterial aetiology (eg, ascending neutrophilic cholangiohepatitis). Cats were assigned to the ‘unclear diagnosis’ category if a definitive diagnosis could not be achieved because of a short survival time, insufficient follow-up to assess response to treatment or failure to meet the inclusion criteria for steroid-responsive lymphadenitis. Diagnosis was determined by review of the clinical record. If the diagnosis remained unclear, cases were reviewed by the authors to establish the most likely diagnosis based on the available data.
Medical record review
Data extracted from medical records from all cases included signalment, history (including clinical signs, time to referral and pre-referral treatment and response), physical examination findings (including lymph node location and distribution [internal/external, multiple or single]), clinical pathology (haematology, serum biochemistry), lymph node cytology and/or histopathology, results of infectious disease testing, diagnostic imaging findings and modalities used, diagnosis, treatment (including drug, dose and duration), response including time to relapse (if applicable), requirement for retreatment and final outcome (alive/euthanased/died or lost to follow-up). The majority of cats underwent haematology, serum biochemistry, abdominal imaging (ultrasound or CT) and lymph node FNA. Additional investigations performed in some cases included urinalysis, urine culture, thoracic imaging (radiographs or CT), echocardiography, exploratory laparotomy, lymph node and/or liver, spleen and intestinal biopsies, oropharyngeal swab for infectious diseases PCRs, bronchoalveolar lavage (BAL) cytology, culture and infectious disease PCRs (including Chlamydia felis, Mycoplasma felis, feline herpes virus, calicivirus, Bordetella bronchiseptica, Mycoplasma gatae, Aelurostrongylus abstrusus). Infectious disease tests frequently performed included FCoV PCR, immunocytochemistry or immunohistochemistry, Toxoplasma serology, lymph node culture, Ziehl-Neelsen (ZN) and periodic acid Schiff staining of lymph node cytology specimens, Bartonella species PCR and Mycobacterium species PCR. A selective number of cats had additional infectious disease testing, including feline leukaemia virus (FeLV)/feline immunodeficiency virus (FIV) and M felis PCR. The selection of tests varied between cases and was at the discretion of the managing clinician. In some cases, testing was limited by financial constraints.
Steroid-responsive group
Inclusion criteria for the steroid-responsive cases included the following: (1) presence of lymphadenopathy involving a peripheral and/or internal lymph node(s); (2) cytological or histopathological evidence of neutrophilic, pyogranulomatous or granulomatous lymphadenitis; (3) absence of an identifiable infectious, inflammatory or neoplastic cause; and (4) clinical improvement after glucocorticoid treatment. Cats previously treated with anti-inflammatory doses of glucocorticoids (eg, <1 mg/kg q24h) for unrelated comorbidities unlikely to influence lymphadenopathy (eg, feline asthma) or for the lymphadenopathy itself within 1 month before referral, were included if lymphadenitis was still evident and there was a clear response to dose escalation. Exclusion criteria included cats who responded to antibiotic treatment, received prior glucocorticoid treatment at immunosuppressive doses (2 mg/kg q24h) within 4 weeks of referral, if neutrophilic or pyogranulomatous inflammation was confined to other organs (eg, liver, spleen, pancreas, intestines) without confirmed concurrent lymphadenitis, or if clinical records were incomplete.
Treatment and outcome
All steroid-responsive cases were treated with glucocorticoids, at various doses, with the majority receiving immunosuppressive doses (2 mg/kg q24h). A second immunosuppressive agent may have been used at the discretion of the clinician managing the case. Prednisolone doses were gradually tapered over the subsequent weeks to months and discontinued depending on the individual cat’s clinical response. A cat was considered steroid responsive if clinical improvement and lymphadenopathy improved or resolved (eg, pyrexia, lymphadenopathy or a 50% reduction in lymph node size if palpable) within 2 months of initiation of prednisolone treatment as reported by the owner or confirmed on physical examination and/or repeat imaging. Follow-up of the steroid-responsive cases was required for a minimum of 2 months to allow a documented response. If available, cases were followed for 3–6 months after diagnosis to evaluate ongoing treatment and relapse. Relapse was defined as a recurrence of clinical signs and/or lymphadenopathy after previous resolution. Patient outcomes were determined from medical records or by contacting the referring veterinary practice or owner using a client information sheet and consent form, which contained a link to a SurveyMonkey questionnaire (see S1 and S2 in the supplementary material). The study was approved by the Royal College of Veterinary Surgeons Ethics Review Panel (2024-074).
Statistical analysis
Categorical data are presented descriptively as percentages. Continuous variables (eg, age, drug dose, treatment duration) are reported as median (range). Age was recorded to the nearest month at the time of referral. Comparisons of age were made between all disease groups using the Mann–Whitney U-test. Fisher’s exact test was used to assess the association between breed and FIP diagnosis, and separately to evaluate sex predisposition between the steroid-responsive group and the remaining cats with other diagnoses. Comparison of purebred and domestic breed between the steroid-responsive group and the rest of the non-FIP groups was assessed using Fisher’s exact test. Comparisons of age and body weight between all groups were performed using the Kruskal–Wallis test. Statistical significance was set at P <0.05.
Results
Neutrophilic and pyogranulomatous lymphadenitis in cats
The initial search identified 118 cats, of which 46 were excluded, 11 had pyogranulomatous inflammation in the lungs and the remainder had pyogranulomatous inflammation confined to other organs (pancreas, skin, liver and kidney). Lymphadenomegaly was sometimes present in these cases but FNAs were either not obtained, or cytology was consistent with reactive hyperplasia (n = 3) or poorly cellular and not representative (n = 4) (Figure 1). Diagnoses in excluded cases included neoplasia (n = 14), FIP (n = 8), pyogranulomatous dermatitis (n = 5), mycobacterial infection (n = 3), M felis infection (n = 3), bacterial infection associated with oral lesions (n = 3), pancreatic cysts (n = 3), toxoplasmosis (n = 1), Nocardia species infection (n = 1), pyothorax (n = 1) and inflammation of unknown cause (n = 3). In total, 72 cats were identified as having pyogranulomatous, neutrophilic or mixed lymphadenitis (pyogranulomatous/neutrophilic and eosinophilic). Of these, nine cats met the inclusion criteria for steroid-responsive lymphadenitis.

Case selection and exclusion
Population demographics
Of the 72 cats, 45 (62.5%) were male (42 castrated, three intact) and 27 (37.5%) were female (22 spayed, five intact). The signalment and body weight by disease category are summarised in Tables S1–S3 (see supplementary material). Domestic cats comprised half of the population (n = 37, 51%): 30 (42%) domestic shorthair (DSH) cats and seven (10%) domestic longhair cats. The remaining 35 (50%) cats were purebred: British Shorthair was the most common purebred (n = 9, 13%). The median age for all cats was 4.04 years (range 0.3–16.75) and median body weight was 3.84 kg (range 0.91–6.75).
Final diagnoses
FIP was the most common category (25/72, 35%), followed by neoplasia (11/72, 15%) and steroid-responsive lymphadenitis (9/72, 12.5%). Final diagnoses by category are listed in Table 1.
Final diagnosis and disease category classification for 72 cats with pyogranulomatous and neutrophilic lymphadenitis

Data are n or n (%)
FGESF = feline gastrointestinal eosinophilic sclerosing fibroplasia; FIP = feline infectious peritonitis
Of the five cats with an unclear diagnosis, two died or were euthanased within 2 weeks of presentation, precluding further investigations. Two remained clinically well without glucocorticoid treatment (one was an asymptomatic cat who had a persistent pyogranulomatous cystic mesenteric lymph node and the other cat presented with lethargy, hyporexia and pyrexia and had generalised abdominal lymphadenomegaly [ileocolic, jejunal, gastric, hepatic and medial iliac with neutrophilic to pyogranulomatous ileocolic lymphadenitis]) and mild jejunal muscularis thickening). The last cat received both glucocorticoids and antibiotics and may have had concurrent cholangiohepatitis; however, diagnosis and treatment response could not be confirmed because of loss to follow-up.
British Shorthair cats were significantly over-represented in the FIP group compared with all other categories combined (8/25 vs 1/47; P <0.001). Cats with neoplasia were significantly older than those with FIP (P <0.001), inflammatory disease (P = 0.04) or an unclear diagnosis (P = 0.009). Cats with mycobacterial infection were significantly older than those with FIP (P = 0.019). FNA was performed on 92 lymph nodes and histopathology on 18 lymph nodes. Pyogranulomatous and granulomatous-based lymphadenitis was the most common cytological finding (n = 47, 51% of lymph nodes sampled) followed by neutrophilic/neutrophilic to pyogranulomatous (n = 31, 34%). Some lymph nodes documented reactive hyperplasia (n = 6, 6.5%) alongside pyogranulomatous/neutrophilic inflammation in another lymph node in the same cat. Cytology and histopathology results are summarised in Tables S4 and S5 (see supplementary material). FNAs from different lymph nodes from the same cat occasionally identified different pathologies (eg, inflammatory vs neoplastic). Histology identified pyogranulomatous in five lymph nodes, which was not identified cytologically.
Infectious disease testing in all cats
Details of the infectious disease testing performed by disease category are summarised in Table S6 (see supplementary material). The most common tests performed included ZN stain (58% of cats), FeLV/FIV SNAP test (56%), FCoV PCR (46%) and T gondii serology (42%). Bacterial culture was performed in less than one-third of cats on lymph node aspirates (n = 8), lymph node biopsy (n = 8), abdominal fluid (n = 3), liver (n = 3), BAL (n = 2) and colonic biopsy (n = 1). Culture was positive in three cats: E coli from a mesenteric lymph node in a cat with gastrointestinal large cell lymphoma, E coli from a colonic biopsy from a cat with histiocytic colitis and mixed anaerobes on extended culture of peritoneal fluid in a cat with bacteria detected on jejunal lymph node cytology. Two cats had bacteria documented on cytology but had negative culture: the first was the cat described above who had negative jejunal lymph node culture. The other cat had bacteria on lymph node aspirate obtained before referral after a cat bite; however, culture was subsequently negative on lymph node biopsy, although this was after 3 months of antibiotics before referral. Antibiotics before referral had been administered in 13/21 (62%) cats. Bartonella species PCR was performed in 25% of cats and was negative in all cases: one presumptive FIP case had positive Bartonella species serology. Four out of five cats (80%) with mycobacterial infection had negative staining rods on cytology; the remaining cat was ZN negative but diagnosed via Mycobacterium species PCR. Neoplasia was confirmed in 90% of cases by cytology or histology (45% lymph node/mass cytology, 27% gastrointestinal biopsies and 18% on nasal biopsies). One case was presumptively diagnosed via CT findings. Consequently, infectious disease testing was rarely performed in the neoplasia group.
Steroid-responsive lymphadenitis cases
Signalment
Nine cats fulfilled inclusion criteria for steroid-responsive lymphadenitis (Table S7a,b in the supplementary material). Six were males (67%) and three were females (33%); all were neutered. There was no significant sex predisposition in the steroid-responsive group compared with other disease categories (P = 1). The median age at presentation was 7 years 10 months (range 9 months to 12 years 1 month). Age differed significantly between all disease groups (P = 0.002). Cats with steroid-responsive lymphadenitis were significantly older than those with FIP (P = 0.027) but not significantly different from other disease categories. DSH represented one-third of the steroid-responsive lymphadenitis cases, with over half (56%) being purebred (Table S3). Steroid-responsive cats had higher odds of being purebred compared with cats in the other non-FIP categories (odds ratio 2.6, 95% confidence interval 0.59–11.49; P = 0.26), although this was not significant. The median body weight was 4.5 kg (range 2.6–6.75) with no significant difference between disease groups (P = 0.198).
Pre-referral treatment and response
Before referral, eight (89%) cats received antimicrobials and six received meloxicam. One cat received long-term anti-inflammatory doses of prednisolone (0.25 mg/kg q48h) for previously diagnosed rhinitis and feline lower airway disease. Antimicrobial treatment consisted of amoxicillin-clavulanic acid (n = 6), cefovecin (n = 2) and pradofloxacin (n = 1), with one cat receiving amoxicillin-clavulanic acid and pradofloxacin concurrently. The median antimicrobial course duration was 9.5 days (range 1–28). Treatment resulted in variable initial responses, with four cats showing transient improvement before deteriorating, five showing no improvement with antimicrobials alone (n = 1) or in combination with meloxicam (n = 1), or having insufficient time before referral to document a response (n = 3). In the latter cases, meloxicam injection (n = 2) and amoxicillin-clavulanic acid injection (n = 1) were administered within 24 h of referral. Persistent clinical signs including a combination of pyrexia, lethargy and lymphadenomegaly despite antibiotic treatment, prompted referral (Table S7b). The median time to referral was 8 days (range 1–70).
History and clinical signs
The most common clinical signs and physical examination findings reported before referral were lethargy (n = 7, 78%), hyporexia/anorexia (n = 7, 78%), pyrexia (n = 7, 78%), peripheral lymphadenomegaly (n = 6, 67%) and weight loss (n = 5, 56%). The median rectal temperature before referral was 40.3°C (range 39.6–40.6). Other signs are summarised in Table 2. One cat had an exploratory laparotomy to manage an intussusception 4 weeks before presentation during which abdominal lymphadenopathy was detected. One cat had a chronic history of sneezing and nasal discharge, and underwent dental extractions 3 weeks before developing cervical lymphadenopathy. One cat had an exploratory laparotomy performed to investigate abdominal masses 24 h before referral. One cat had previously documented abdominal lymphadenomegaly 4 years before presentation and another had neutrophilic lymphadenitis of unknown aetiology documented 17 months before referral. One cat was completely asymptomatic and presented with left-sided submandibular lymphadenomegaly.
Summary of presenting clinical signs in cats with steroid-responsive lymphadenitis

Physical examination findings
Physical examination on admission revealed pyrexia (range 39.4–41°C) in 4/9 (44%) cats, as three of the previously pyrexic cats were normothermic on admission. Peripheral lymphadenopathy was present in six (67%) cats, including submandibular (n = 5), popliteal (n = 4) and prescapular (n = 3). Additional findings included dehydration with tacky mucous membranes (n = 2), pallor (n = 1), moderate to severe gingival swelling associated with the right maxillary canine (n = 1), low-grade murmurs (grades I–II/VI) (n = 4), gallop sound (n = 1), abdominal lymphadenomegaly (n = 2), hepatosplenomegaly (n = 1), abdominal discomfort (n = 1) and poor body condition (n = 2).
Clinical pathology
Haematology and serum biochemistry abnormalities
Prominent clinicopathological findings are summarised in Tables 3 and S7b (in the supplementary material). Haematological abnormalities included mild to moderate non-regenerative/pre-regenerative anaemia (haematocrit 18.8–29.2 l/l, reference interval [RI] 30.3–52.3) in five (56%), cats, mild to severe neutrophilia (10.48–45.14 × 109/l, RI 2.3–10.29) in four (44%) cats, monocytosis (monocyte count 0.75–2.98 × 109/l, RI 0.05–0.67) in three (33%) cats, mild lymphocytosis (7.09–10.17 × 109/l, RI 0.92–6.88) in three (33%) cats and thrombocytopenia (32–61 × 109/l, RI 151–600) in two (22%) cats, although platelet clumps or macroplatelets were observed on blood smear examination. Lymphopenia and leukopenia were documented in one cat each. Biochemical abnormalities included mild to moderate hyperglobulinaemia (52–62 g/l, RI 28–51) in four (44%) cats and hyperbilirubinaemia (26–38 µmol/l, RI 0–15) in two (22%) cats. Two cats had no abnormalities on either haematology or serum biochemistry.
Most common haematology and biochemistry abnormalities detected in nine cats with steroid-responsive lymphadenitis

Diagnostic imaging investigations and findings
Diagnostic imaging performed included thoracic radiographs (n = 4), abdominal ultrasound (n = 7) and CT of the head, neck and thorax (n = 1) including the abdomen (n = 1). Echocardiography (n = 3) revealed no structural heart disease. CT of the head revealed marked cervical lymphadenomegaly in two cats, characterised by heterogeneous contrast enhancement and cavitation with a peripheral rim enhancement, consistent with necrosis or abscessation (Figures 2 and 3). One of these cats (case 1) had concurrent bilateral otitis externa (OE)/media and the other (case 6) had left-sided otitis media. The cat with bilateral OE had concurrent abdominal lymphadenomegaly and the other cat did not undergo abdominal imaging. The cat with bilateral OE/media had unilateral lymphadenomegaly (left parotid and medial retropharyngeal). In the cat with left-sided otitis media, the cervical lymphadenomegaly (right submandibular and medial retropharyngeal) was on the opposite side. Although the parotid lymph node drains the external ear canal and the retropharyngeal lymph node drains the middle ear, the submandibular lymph node does not typically drain the ear. Given the severity of the unilateral lymphadenomegaly in both cats and that it was contralateral to the unilateral otitis media, it was considered unlikely to be only a reactive change (Figures 2 and 3). Thoracic imaging documented cardiomegaly (n = 1) and mixed bronchointerstitial lung pattern (n = 2). No intrathoracic lymphadenomegaly was reported. Abdominal lymphadenopathy was detected in seven (78%) cats. Four out of six (67%) cats with peripheral lymphadenopathy had concurrent abdominal lymphadenopathy, but one case did not undergo abdominal imaging. Concurrent external and internal lymphadenopathy was documented in 4/9 (44%) cats. Other findings on abdominal imaging included mild hepatomegaly (n = 4), mild to moderate peritoneal effusion (n = 3), splenomegaly (n = 3), mild gastrointestinal wall changes (n = 4) including a prominent jejunal muscularis, duodenal and jejunal submucosal thickening, mildly thickened colonic wall, and a thickened and corrugated distal ileal wall. Other findings included a right renal mass (n = 1), bilateral renomegaly (n = 1), cholelithiasis (n = 1) and a prominent hypoechoic irregular pancreatic body and left limb (n = 1).
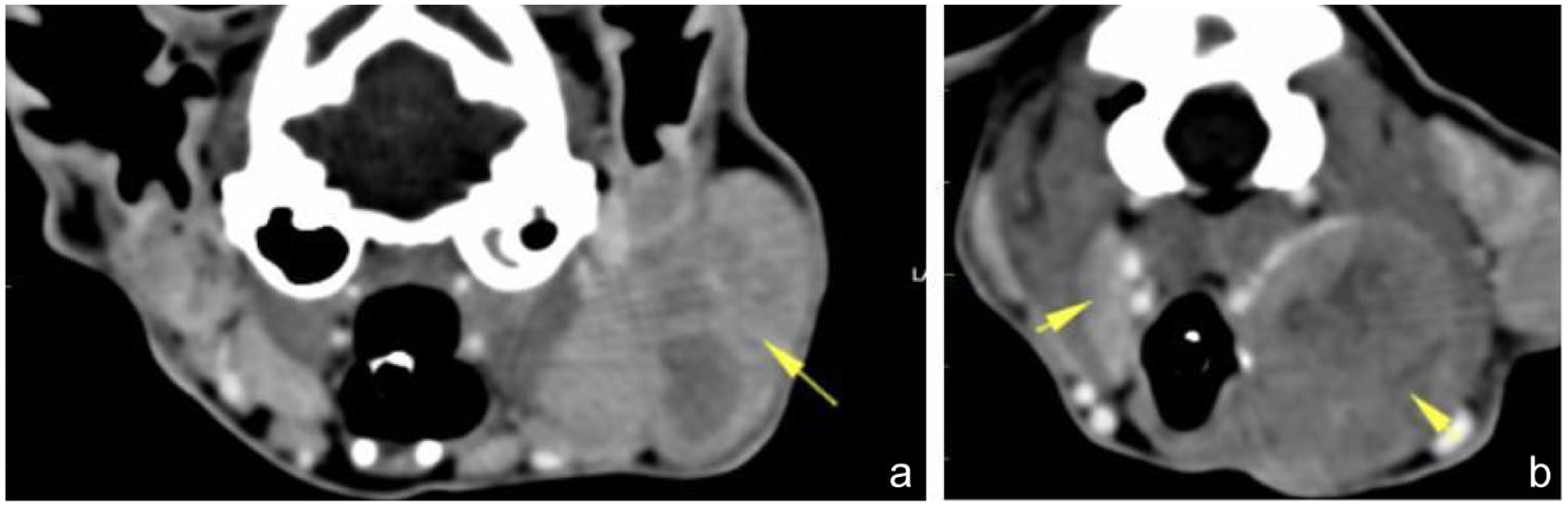
CT findings of a domestic shorthair cat aged 10 years 8 months (case 1) diagnosed with steroid-responsive neutrophilic lymphadenitis: (a) left parotid lymphadenomegaly (yellow arrow) (3.9 × 2 cm); and (b) left medial retropharyngeal lymphadenomegaly (2.5 cm) with heterogeneous contrast uptake and right medial retropharyngeal lymph node (0.5 cm thickness) (yellow arrows)

CT findings of a Singapura cat aged 12 years 1 month (case 6) diagnosed with steroid-responsive necrotising pyogranulomatous lymphadenitis: (a) well-defined oval cavitated right lateral submandibular lymphadenomegaly (2 × 1.5 cm); and (b) right medial retropharyngeal lymphadenomegaly (2.6 × 1.8 cm). Both lymph nodes are characterised by a peripheral parenchymal capsule, showing marked rim contrast enhancement and non-enhancing hypodense soft tissue/fluid attenuation centre consistent with necrotising lymphadenitis or abscessation
Distribution of lymphadenopathy
Based on clinical examination and diagnostic imaging, submandibular (n = 5, 56%), jejunal (n = 4, 44%) and popliteal (n = 4, 44%) lymph nodes were most commonly affected. Less commonly affected lymph nodes included colic, ileocecal and prescapular in 33% of cases. Peripheral lymphadenopathy was noted in 6/9 (67%) cases. Abdominal lymphadenopathy was detected in seven (78%) cats. Lymph node distribution is summarised in Table 4. The number of enlarged lymph nodes was in the range of 1–9, based on physical examination and diagnostic imaging. In some cats, the exact number could not be determined as individual nodes were not specified in the imaging report.
Distribution of lymphadenomegaly in nine cats diagnosed with steroid-responsive lymphadenitis

Lymph node sampling
In all steroid-responsive cats, a diagnosis of lymphadenitis was reached with cytology and/or histopathology. In total, 14 lymph nodes were sampled from nine cats: FNA cytology in 14 (100%), biopsy and histopathology in five (36%) and both in four cats. The predominant type of lymphadenitis diagnosed on cytology was neutrophilic (n = 8, 57% of the lymph nodes sampled), followed by pyogranulomatous (n = 4, 29%) and necrotising pyogranulomatous (n = 2, 14%). One lymph node aspirate was reactive and another non-diagnostic owing to poor cellularity. Conversely, the predominant type of lymphadenitis on histopathology was pyogranulomatous (n = 4, 80%), with one node identified as reactive hyperplasia. Agreement between cytology and histology occurred in only 20% of samples, while three (60%) samples differed. Establishing presence of agreement could not be determined in one sample because of lack of documentation of the specific lymph node aspirated. Three lymph nodes identified as neutrophilic (n = 2) and reactive (n = 1) on cytology were subsequently classified as pyogranulomatous (n = 2) and necrotising pyogranulomatous (n = 1) on histopathology. The reactive lymph node FNA was likely non-representative as low cellularity was reported.
Infectious disease screening
The most common infectious disease testing performed included Bartonella species PCR in seven (78%) cats, ZN stain in six (67%), T gondii serology in five (56%), and FCoV PCR, Mycobacterium species PCR and FeLV/FIV testing in four (44%). Lymph node bacterial and fungal culture was performed in only two (22%) cats and abdominal fluid culture in one cat. All tests were negative; no infectious organism was identified in any of the nine cats (Table S8 in the supplementary material).
Concurrent diseases
Seven cats (78%) were diagnosed with concurrent diseases, including suspected concurrent chronic enteropathy (n = 4), of which one cat had chronic moderate to marked lymphoplasmacytic enteritis confirmed via histopathology of full-thickness duodenal and ileal biopsies, suspected chronic pancreatitis (n = 1), eosinophilic granuloma complex cutaneous lesions (n = 1), suspected cardiac disease (n = 1) and a hiatal hernia, cholelithiasis, otitis media and periodontal disease (n = 1). Four cats had either hepatosplenomegaly (n = 2) or hepatomegaly (n = 2), and one cat had a mottled spleen. Inflammation was identified in these organs and was most frequently neutrophilic (liver n = 4, spleen n = 2), with a mottled spleen documented to have pyogranulomatous splenitis on histopathology (n = 1) and minimal reactive portal hepatitis with multifocal microgranulomas on histopathology (n = 1). Clinically, these cases were not typical of cholangiohepatitis (with absence of increased liver enzyme activities) and the inflammation was suspected to be part of a systemic inflammatory process; however, a cholangiohepatopathy could not be completely excluded in all cats.
Treatment
All cats were treated with prednisolone. The median starting dose of prednisolone was 1.25 mg/kg q24h (dose range 0.5–2.3). Four cats (44%) required dose escalation after 1–3 weeks to a median dose of 2 mg/kg q24h or 1 mg/kg q12h (range 2–2.5 mg/kg q24h), with 67% of cats receiving immunosuppressive doses (2 mg/kg q24h). The reasons for increasing to immunosuppressive doses were because of persistent clinical signs (n = 2), relapse (n = 1), after receipt of negative PCR results (n = 1) or after monitoring for initial deterioration after introduction of prednisolone (n = 1).
Supportive treatment administered during hospitalisation included intravenous fluid therapy, ondansetron, maropitant, mirtazapine, buprenorphine, methadone, gabapentin and tramadol. These medications were administered to address dehydration, hyporexia, suspected pain and analgesia postoperatively after obtaining lymph node biopsies during exploratory laparotomy. Two cats had an oesophageal feeding tube placed to provide enteral nutrition. One cat received furosemide owing to the development of pleural effusion suspected to be due to fluid overload and the presence of underlying cardiac disease.
All cats had improvement in their clinical signs. Lethargy, pyrexia and hyporexia improved rapidly, often within 12–48 h of initiation of prednisolone treatment; however, the exact time frame for each cat could not be easily determined from clinical records. Lymphadenomegaly resolved or improved after a median of 4 weeks (range 2–10). For one cat, the exact time to resolution of lymphadenomegaly was not available because of the paucity of available follow-up. However, a relapse of clinical signs was reported 11 months later, indicating an initial response. The typical recommended tapering schedule was a dose reduction of 20–25% every 3–4 weeks but may have slightly varied between cats based on the individual cat’s initial response. The median prednisolone duration was 5.75 months (range 4–11) in six cats.
Antibiotic treatment was administered to four cats, either before or concurrently with glucocorticoid treatment, and included marbofloxacin, doxycycline, amoxicillin-clavulanic acid, clindamycin and cefovecin. The median duration of antibiotic treatment was 17.5 days (range 3–28). In all cases, antimicrobial treatment failed to resolve lymphadenomegaly, which persisted and only resolved after prednisolone was initiated or after a dose increase. Antibiotics were prescribed while infectious disease test results (T gondii serology, Bartonella species PCR) were pending or due to concerns about commencing immunosuppressive doses of prednisolone while an infectious process had not been entirely excluded. Antibiotics were discontinued after receipt of negative test results or because of lack of response with persistent lymphadenomegaly. One cat received concurrent chlorambucil treatment owing to concerns for possible emerging underlying small cell lymphoma alongside a chronic inflammatory enteropathy on gastrointestinal biopsies. Treatment details for each cat are summarised in Table S7a (see supplementary material).
Relapse and outcome
Relapse was suspected in 3/9 (33%) cats, with recurrence of lethargy, hyporexia, pyrexia and peripheral lymphadenomegaly. Two cats (22%) were euthanased after relapse of their clinical signs, with documented recurrence of lymphadenopathy in one of these cats. Euthanasia was elected because of neurological signs 5 months into treatment (n = 1) and persistent anorexia 3.5 months into treatment (n = 1). In the third cat who relapsed, pyrexia resolved with supportive treatment; however, it was unknown whether prednisolone was restarted because of lack of follow-up available from the primary veterinary clinic. Details are provided in Table S7a (see supplementary material). The median follow-up was 18 months (range 3.5 months–5.5 years). Among the five cats who were still alive or not lost to follow-up after 5.5–10 months, all were off medication and remained clinically well, with a median period off treatment of 37 months (range 14–60.5). Of the five cats who remained clinically well after prednisolone discontinuation, initial cytological inflammation had consisted of pyogranulomatous (n = 2), necrotising pyogranulomatous (n = 1), necrotising granulomatous (n = 1) and neutrophilic lymphadenitis (n = 1). These five cats initially presented with abdominal lymphadenomegaly (n = 2) and both abdominal and peripheral lymphadenomegaly (n = 3). Of the two cats who were euthanased, one had neutrophilic lymphadenitis with both abdominal and peripheral lymphadenomegaly, and one had pyogranulomatous lymphadenitis with abdominal lymphadenomegaly.
Discussion
This retrospective study is the first to investigate the causes of pyogranulomatous and neutrophilic lymphadenitis in a cohort of referred cats and provides the first description of steroid-responsive lymphadenitis in multiple cats. Pyogranulomatous inflammation was most frequently caused by FIP, accounting for over one-third of cases. This aligns with the well-recognised tendency of FCoV infection to induce systemic pyogranulomatous inflammation in cats with FIP. The challenging nature of diagnosing the ‘dry’ form of FIP likely contributed to the frequency of referral of these cases. Despite the ubiquitous nature of FCoV infection, only 25 FIP cases were identified over an 8-year period, although we did not search specifically for FIP cases. This reinforces the low prevalence of FIP at approximately 0.5%, with only 5–12% of seropositive cats developing FIP.34 –36 Some FIP cases were excluded from this study because of pyogranulomatous inflammation being confined to extra-nodal tissues.
Neutrophilic lymphadenitis was most frequently associated with neoplasia, particularly lymphoma, which accounted for 64% of cases. Cytology confirmed the diagnosis in 36% of neoplastic cases, whereas histopathology was required for the remainder. This is consistent with a recent study evaluating mesenteric lymph node biopsies in feline gastrointestinal lymphoma, with nodal involvement in 43% of cases and reactive changes in 58%. 37 Tumour-associated necrosis, mucosal ulceration and cytokine release likely provoked neutrophilic infiltration of the draining or metastatic lymph nodes. A recent MRI-based study of medial retropharyngeal lymphadenopathy in dogs and cats found that neoplastic lymph nodes were often larger than inflammatory ones, suggesting that size could aid in differentiation. However, as most cases in that study were canine, these findings may not extrapolate reliably to cats. 38
Steroid-responsive lymphadenitis was the second most common cause of neutrophilic lymphadenitis. The underlying pathophysiology in these cats remains uncertain; it is possible that this group encompasses cats with differing mechanisms, including primary immune dysfunction, low-grade occult infection or persistent inflammation. Therefore, response to corticosteroids may have been for variable reasons.
In our study, bacterial lymph node abscessation was rare. These findings align with the sparse veterinary literature, which to date includes only a single case of steroid-responsive sterile necrotising pyogranulomatous lymphadenitis. There are also rare reports of cervical and mesenteric lymph node abscesses due to E coli, methicillin-resistant Staphylococcus species and generalised neutrophilic lymphadenitis associated with a M felis abscess.5,6,32,39 These observations highlight that neoplasia or an immune-mediated, presumed sterile inflammatory process should be prioritised as differential diagnoses in cats with neutrophilic lymphadenitis rather than overt bacterial infection, particularly in older cats unresponsive to appropriate antimicrobial treatment. Mesenteric abscesses are likely under-represented in referral populations, as many resolve with antimicrobial treatment without the need for specialist referral.
Male cats appeared over-represented among steroid-responsive cases, although case numbers were low. One potential reason for an increased prevalence in males could reflect an increased risk of bite-related injuries from fighting. 39 It is possible that in some cats, an initial bacterial infection from a cat bite may have triggered a secondary inflammatory or immune-mediated lymphadenomegaly. One steroid-responsive cat had a recent bite wound that initially improved with a 5-day antimicrobial course but relapsed and ultimately required prednisolone. Two additional cats had bacteria identified cytologically within their lymph node and therefore did not meet the inclusion criteria; nonetheless, both required anti-inflammatory prednisolone treatment for complete resolution. Of these, one had a history of a cat bite and developed persistent lymphadenomegaly despite 7 weeks of amoxicillin-clavulanic acid and marbofloxacin. The other cat required prednisolone after only 3 days of the same antimicrobial combination due to marked systemic inflammation. Lymphadenomegaly resolved after 6 weeks but relapsed during prednisolone tapering. However, many cats in the steroid-responsive group did not have a history of known cat bite.
This pattern of partial or transient improvement with antimicrobials in some cats followed by complete resolution only with glucocorticoids supports the hypothesis of an unidentified infectious trigger inciting a disproportionate inflammatory or immune response. An inadequate response to antimicrobials may also reflect bacterial resistance, insufficient course duration, incorrect antibiotic choice, inappropriate host immune response or presence of an unusual pathogen. However, it is notable that the cats improved with steroid therapy rather than alteration of antimicrobial coverage.
A prior undetected bacterial abscess could also account for diffuse abdominal lymphadenopathy in some cats, as illustrated by a recent case of M felis-associated peripheral and abdominal lymphadenomegaly secondary to a cat bite abscess on the left antebrachium. 6
The median age at initial presentation was 7 years 10 months (range 9 months–12 years), similar to the median age of 6 years reported in dogs with sterile lymphadenitis and a previous report of feline idiopathic sterile pyogranulomas.29,31 This parallels the age distribution of inflammatory bowel disease, intestinal lymphoma and other immune-mediated diseases, such as immune-mediated polyarthritis, which are more prevalent in middle-aged cats.40,41 However, this is in contrast to the young age (median 3 years) reported in a different study in dogs with sterile steroid-responsive lymphadenitis. 28 One-third of our cats were young (<2.5 years old), consistent with a recent report of sterile lymphadenitis in a 1.5-year-old cat, suggesting that steroid-responsive lymphadenitis can occur across all ages.5,32
Most cases of steroid-responsive lymphadenitis presented with systemic illness, including lethargy, hyporexia/anorexia, pyrexia, peripheral lymphadenopathy and weight loss. A recent study assessing cervical lymphadenopathy in dogs and cats found inflammatory lymphadenitis cases were more likely to be pyrexic than neoplastic cases, likely due to extension of inflammation beyond the lymph node capsule and associated inflammatory cytokine release. 38 Overall, steroid-responsive cats shared many clinical features with dogs with sterile or idiopathic pyogranulomatous lymphadenitis but differed from the previously described asymptomatic idiopathic sterile pyogranulomas in cats, in that they were often systemically ill.27 –29,31 Only one cat in our cohort presented asymptomatically with unilateral submandibular lymphadenopathy.
The most common clinicopathological abnormalities were non-regenerative anaemia, neutrophilia and hyperglobulinaemia, consistent with chronic systemic inflammation. Two cats also had mild hyperbilirubinaemia with hepatomegaly and neutrophilic inflammation documented on liver aspirates. Hyperbilirubinaemia has been recently reported in a cat with sterile lymphadenitis. 32 In our cases, this likely reflected systemic inflammatory response syndrome (SIRS)-associated cholestasis, as both met SIRS criteria and one developed subsequent hypoalbuminaemia. 42 Concurrent cholangiohepatitis could not be excluded but was considered unlikely given imaging findings in addition to the absence of other supporting biochemical abnormalities.
Diagnostic imaging revealed that abdominal lymphadenopathy was more common than peripheral, with nearly half of affected cats having both peripheral and internal lymphadenomegaly. This distribution suggests these cats are affected by a widespread systemic inflammatory or immune-mediated process. Interestingly, all cats with abdominal lymphadenopathy alone had ultrasonographic evidence of intestinal wall abnormalities, and one had histopathological confirmation of an inflammatory enteropathy. Another cat had undergone exploratory celiotomy for an intussusception 4 weeks prior. In such cases, diffuse inflammatory enteropathy or ischaemic injury with bacterial translocation could be an inciting cause of diffuse intra-abdominal lymphadenitis, although this would be difficult to prove.29,43,44 There are rare reports in the literature of mesenteric abscessation secondary to suspected gastrointestinal translocation in cats.5,16 However, this remains an unusual finding, as abdominal lymphadenopathy associated with enteropathy is typically characterised by reactive hyperplasia rather than neutrophilic or pyogranulomatous lymphadenitis. 2
The submandibular, popliteal and jejunal lymph nodes were most frequently affected, similar to canine cases where submandibular and popliteal nodes predominate.28,29 This likely reflects their accessibility for palpation on physical examination. Interestingly, in our study, there were slightly more cats affected by abdominal lymphadenomegaly than peripheral lymphadenomegaly. Although numbers were relatively small, this could reflect a higher incidence of occult enteropathy in this population of cats than in canine studies or a different disease distribution in cats.
The diagnosis of lymphadenitis was made via cytology and/or histopathology. Neutrophilic lymphadenitis was the predominant type on cytology, whereas pyogranulomatous lymphadenitis was the predominant type on histopathology, consistent with a previous study in dogs. 28 Importantly, the type of inflammation detected on cytology occasionally varied between nodes sampled within the same cat. For example, one case had neutrophilic lymphadenitis in a submandibular node but lymphoma in the medial retropharyngeal node aspirate. This illustrates the heterogeneity of lymph node cytology and underscores the importance of sampling all enlarged accessible lymph nodes. A recent study reported that non-diagnostic samples were obtained in 55% of feline mesenteric lymph node aspirates, highlighting the limitations of ultrasound-guided sampling and the risk that cytological samples may not be fully representative of the lymph node pathology. 45 In our study, cytology and histopathology agreed in only 20% of cases, consistent with discrepancies previously described in feline and canine sterile lymphadenitis.28,32 These findings reinforce the recommendation that histopathology should be pursued whenever feasible to ensure an accurate diagnosis, as it is more representative with preservation of lymph node architecture. It would also allow further infectious disease testing, such as FCoV immunohistochemistry, Mycobacterium PCR and culture. In the cats in our study who initially responded to glucocorticoids but later deteriorated after tapering, an underlying infectious or neoplastic process may have been missed.
Investigation into the underlying cause of lymphadenomegaly is inherently challenging and often expensive. It usually requires extensive infectious disease testing. In the steroid-responsive cases, no definitive infectious agent was identified. However, not all cats underwent comprehensive infectious disease testing owing to financial constraints and variation in case management between clinicians. Lymph node culture was only performed in 22% of cats. False-negative results are also possible as prior antimicrobial treatment may have suppressed bacterial culture growth, and fastidious organisms such as Mycoplasma species, Bartonella species or Mycobacterium species are inherently difficult to culture. Molecular techniques such as PCR amplification of a 900 base pair fragment of the 16S rRNA gene and 16S RNA probe fluorescence in situ hybridisation analysis for eubacteria are more sensitive but were not performed in this cohort, unlike in previous sterile/steroid-responsive lymphadenitis reported in cats.31,32 Because lymph node culture has limited sensitivity, with only 50% sensitivity reported in dogs with mesenteric lymph node abscesses, some cases could still have represented chronic E coli abscesses or prior infection.15,29,39,43 Such lesions can show an encapsulating pyogranulomatous inflammatory response and may explain the initial partial or transient responses to antimicrobials observed in four cats.15,29,39,43 This parallels the findings in human medicine, where a specific aetiology is lacking in 12–36% of granulomas. 46 Nevertheless, active bacterial infection was considered unlikely given the lack of clinical response to antimicrobial treatment given to most cats before referral, the absence of bacteria on cytology and the consistent response to glucocorticoids, with 71% receiving immunosuppressive doses. Feline mycobacteriosis is often paucibacillary, and ZN staining has low sensitivity, with approximately two-thirds of biopsies ZN negative despite histopathological features indicative of mycobacterial infection.47,48 Therefore, mycobacterial infection remains possible, as despite six having ZN staining only four cats underwent PCR. FCoV PCR on lymph node aspirates and/or immunohistochemistry on a mesenteric lymph node was negative in 55% of cats. Given the reported 90% sensitivity of mesenteric lymph node FCoV PCR, these results make FIP an unlikely diagnosis in those cats. 49 Prolonged survival provides further support, as 57% of non-euthanased cats in our study survived for more than 2.5 years, whereas survival of up to 1–2 years with glucocorticoid or supportive treatment has only rarely been reported in FIP.50,51 However, FIP cannot be definitively ruled out in untested cats.
T gondii serology was not performed in four cats; however, infection was considered unlikely given the response to initial immunomodulatory glucocorticoid treatment in all cases. Based on these findings, we recommend future cases have aerobic and anaerobic culture, FCoV PCR, ZN staining, Mycobacterium species PCR and T gondii serology as a minimum infectious disease screen if feasible.
All cats in the steroid-responsive group received prednisolone at a median starting dosage of 1.25 mg/kg q24h (range 0.5–2.3). Lethargy, pyrexia and hyporexia improved rapidly after treatment, with lymphadenomegaly resolving or improving within 1 month on average. Nearly half of the cats required dose escalation, with two-thirds of cats ultimately receiving immunosuppressive doses (2 mg/kg q24h). Dose escalation was prompted by waxing and waning clinical signs, persistent lymphadenomegaly or after receipt of negative infectious disease test results. The median course duration was nearly 6 months, with relapses occurring in 33% of cats. Relapse was generally associated with a poorer outcome, as 2/3 relapsed cats were subsequently euthanased. This may reflect a more treatment-resistant subset of the same disease process, emphasising the need for cautious tapering. These findings are comparable to retrospective studies of sterile lymphadenitis in dogs, where most dogs were successfully treated with immunosuppressive prednisolone, with 26–36% of dogs experiencing a relapse.28,29
Two cats classified in the unclear diagnosis group did not require any steroid treatment. One was suspected of having neutrophilic to pyogranulomatous lymphadenitis secondary to a self-limiting viral infection and responded to supportive treatment, although follow-up abdominal ultrasound to reassess his abdominal lymphadenopathy was not performed. The other remained asymptomatic for 3.5 years despite persistent large cystic pyogranulomatous mesenteric lymphadenomegaly, resembling a previously reported sterile pyogranuloma case in cats. 31 In contrast, most of our steroid-responsive cases were systemically ill, suggesting this may represent a distinct presentation of similar disease. Whether all cases require prednisolone treatment and at what dose remains uncertain and warrants further research.
The limitations of this study include the small sample size of the steroid-responsive group, inconsistent diagnostic investigations, including lack of histopathology in nearly half of the steroid-responsive cats, and incomplete infectious disease screening performed owing to the retrospective nature of our study. In some instances, retrospective interpretation of records was required to determine the likely final diagnosis, and this may have led to misclassification. Further studies using histopathology and standardised infectious disease screening in a larger cohort are needed to address the limitations of this retrospective study and to better define whether steroid-responsive lymphadenitis represents a genuine, distinct condition and to help optimise management of these cases. Such work can hopefully further address if this represents a primary autoimmune dysregulation vs steroid-responsive inflammation secondary to another disease process such as occult infection.
Conclusions
To the authors’ knowledge, there are no published studies describing steroid-responsive lymphadenitis in cats and only a single recent case report. We wanted to raise awareness of this condition and highlight that steroid-responsive neutrophilic or pyogranulomatous lymphadenitis should be considered in cats with peripheral and/or internal lymphadenopathy and pyrexia, particularly in middle-aged cats, following exclusion of an infectious, inflammatory or neoplastic aetiology. Although the underlying aetiology remains poorly understood, the absence of an identifiable infectious trigger, favourable response to glucocorticoid therapy and the tendency for relapse in some cases during tapering are suggestive of either an immune-mediated or inflammatory aetiology, similar to that previously reported in dogs.
Supplemental Material
Table S1
Signalment and body weight of the different disease group.
Supplemental Material
Table S2
Signalment and body weight of the different disease group.
Supplemental Material
Table S3
Signalment and body weight of the different disease group.
Supplemental Material
Table S4
Signalment and body weight of the different disease group.
Supplemental Material
Table S5
Signalment and body weight of the different disease group.
Supplemental Material
Table S6
Signalment and body weight of the different disease group.
Supplemental Material
Table S7a,b
Signalment and body weight of the different disease group.
Supplemental Material
Table S8
Signalment and body weight of the different disease group.
Supplemental Material
S1
Signalment and body weight of the different disease group.
Supplemental Material
S2
Signalment and body weight of the different disease group.
Footnotes
Author note
This paper was presented as an oral abstract at the 35th European College of Veterinary Internal Medicine – Companion Animals congress in Maastricht, the Netherlands, September 2025.
Supplementary material
The following files are available as supplementary material:
Table S1: Signalment and body weight of the different disease group.
Table S2: Median age of 72 cats with lymphadenitis, as classified by disease category.
Table S3: Breed distribution between the different disease groups.
Table S4: Cytological diagnosis of all cats.
Table S5: Histological diagnosis of all cats.
Table S6: Comparison of infectious disease testing performed on all cats and steroid-responsive cats during investigations into lymphadenitis.
Table S7a,b: Case information.
Table S8: Infectious disease testing performed in the steroid-responsive cat.
S1: Client information sheet.
S2: Consent form and questionaire.
Conflict of interest
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
Linnaeus Veterinary supported the costs of the open access publication charges.
Ethical approval
The work described in this manuscript involved the use of non-experimental (owned or unowned) animals. Established internationally recognised high standards (‘best practice’) of veterinary clinical care for the individual patient were always followed and/or this work involved the use of cadavers. Ethical approval from a committee was therefore not specifically required for publication in JFMS. Although not required, where ethical approval was still obtained, it is stated in the manuscript.
Informed consent
Informed consent (verbal or written) was obtained from the owner or legal custodian of all animal(s) described in this work (experimental or non-experimental animals, including cadavers, tissues and samples) for all procedure(s) undertaken (prospective or retrospective studies). No animals or people are identifiable within this publication, and therefore additional informed consent for publication was not required.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
