Abstract
Objectives
The aims of this study were to assess surgical biopsy of abdominal lymph nodes (LNs) in cats taken at the time of exploratory laparotomy and to evaluate any additional benefit of histopathology compared with LN cytology.
Methods
We carried out a retrospective study from a single institution of cats that had abdominal ultrasound and LN biopsies as part of an exploratory laparotomy (2014–2018). Clinical records were reviewed for presenting signs, medical investigations, ultrasound, cytology, surgical findings and histopathology.
Results
Fifty-one cats were included. In total, 60 LNs were biopsied. Forty-four cats had evident LN enlargement at the time of surgery. Nine of those cases were reported to have normal-sized LNs on ultrasound, including one cat that was subsequently diagnosed with lymphoma. Ultrasound-guided LN aspiration was performed in 19 cases. Five of these cats had a histological diagnosis of neoplasia following biopsy, but only one was correctly identified with cytology. Histopathology results were reactive hyperplasia (n = 41), tumour metastasis (n = 11), lymphadenitis (n = 6), inconclusive (n = 1) and normal (n = 1). The most common final diagnoses were inflammatory bowel disease (n = 17/51) and gastrointestinal lymphoma (n = 10/51). There were 15 cats with neoplastic disease; LN biopsies confirmed metastatic disease in 10 and ruled out nodal involvement in five.
Conclusions and relevance
Surgical biopsy of grossly enlarged or normal abdominal LNs provides additional information to LN cytology in cats and in the majority of cases is associated with a positive histological diagnosis. There appears to be the potential to underdiagnose neoplasia with cytology alone.
Introduction
Biopsy sampling of tissues during abdominal exploratory laparotomy and subsequent histological examination is a common technique that provides valuable diagnostic information.1,2 Intra-abdominal lymph node (LN) enlargement is commonly seen in both inflammatory and neoplastic conditions in feline patients, and it is recognised that differentiation between benign and malignant lymphadenopathy requires LN evaluation by either cytology or histopathology. 3
Abdominal LNs can be assessed with diagnostic imaging techniques, and methods of sampling include ultrasound-guided fine-needle aspiration, needle core biopsy and surgical biopsy.4–6 Several studies have evaluated ultrasonographic LN changes as a way to differentiate benign and malignant processes, with conflicting results being found among studies.3,7–10 Cytological examination of fine-needle aspirates (FNAs) of LNs is a valuable technique in human oncology and similar good results have been identified in dogs and cats with solid tumours, with correlation between cytology and histopathology ranging from 92% to 100%.4,11–13 However, these veterinary studies have not specifically looked at abdominal LN cytology where results may differ owing to the challenges of ultrasound-guided aspiration.
Abdominal LN biopsies are frequently performed during exploratory laparotomy in cats as either a primary or additional procedure. Nevertheless, to our knowledge, no study has specifically assessed their clinical relevance and additional benefit when compared with ultrasound and cytology.
The goal of this study was to assess the clinical relevance of abdominal LN biopsies during abdominal surgery in cats and to evaluate whether additional information was gained in comparison with cytology of FNAs of LNs.
Materials and methods
Case selection
The clinical records of the authors’ specialist referral hospital between 2014 and 2018 were reviewed. Cats were included if they had surgical biopsies of abdominal LNs at the time of exploratory surgery for investigation and treatment of other abdominal pathologies and if medical records were complete.
Medical record review
Signalment, history, presenting clinical signs and blood work abnormalities were evaluated. Results of diagnostic work-up, including imaging, cytology findings (when applicable), surgical reports and histological evaluation of LNs and other tissues submitted were recorded. The final diagnosis and time to discharge were noted. LNs were classified as ‘enlarged’ or ‘not grossly enlarged’ based on both ultrasound and surgical reports. The specific LNs affected were noted.
All imaging and surgeries were performed by European board specialists or supervised residents. The data were analysed using SPSS (IBM) descriptive statistics, frequencies and cross tabulations. The sensitivity, specificity and accuracy of LN cytology were calculated using the histology results as the basis for comparison.
Results
Fifty-one cats that had surgical abdominal LN biopsies were included. Breeds included domestic shorthair (n = 30), domestic longhair (n = 7), Persian (n = 3), Ragdoll (n = 3), British Shorthair (n = 3), Birman (n = 2), Bengal (n = 1), Russian Blue (n = 1) and Siamese (n = 1). Median age at presentation was 7.4 years (range 7 months to 16 years) and the majority were male, with 37 neutered males (72%), 13 neutered females (25%) and one entire female (2%).
Detailed history was available for all cats. Thirty-two cats (63%) had no relevant medical history prior to presentation, whereas 17 cats (33%) had chronic gastrointestinal (GI) signs. There was one diabetic cat (2%) and one cat (2%) that had a previous surgery to remove a cystolith. The most common clinical signs at presentation were vomiting (n = 22), weight loss (n = 16), anorexia (n = 15), diarrhoea (n =13) and lethargy (n = 12). Abdominal distension (n = 5), jaundice (n = 4), polyuria/polydipsia (n = 2) and tachypnoea (n = 2) were also reported. Haematology and biochemistry panels were performed in 48 cats. No haematology abnormalities were seen in 65% (n = 31/48) of the cats. Thirteen cats (27%) were anaemic and neutrophilia was identified in eight cases (17%). No biochemistry changes were seen in 38% of cats (n = 18/48). Increased liver enzymes (n = 11), hypoalbuminaemia (n = 8), azotaemia (n = 6) and hypocobalaminaemia (n = 5/17) were the most common abnormalities seen.
Imaging findings
Abdominal ultrasound was performed in 50 cases. One of these cats had thoracic radiographs in addition to abdominal ultrasound, whereas in another case thoracic radiography was the only preoperative imaging modality used. The most common imaging finding (n = 25) was the presence of GI thickening (Table 1). GI masses (n = 8), septic peritonitis (n = 3), pancreatitis (n = 2), ascites (n = 2) and extrahepatic biliary obstruction (n = 2) were also identified. Two cats (4%) had generalised abdominal lymphadenomegaly with no other abnormalities. Other findings that were identified less frequently (n = 1) included jejunal stenosis, a ureterolith, a renal mass, hepatomegaly with hyperechoic liver and biliary system, hepatomegaly with hypoechoic liver, hepatomegaly and splenomegaly, a hepatic cyst, a diaphragmatic hernia, megacolon and ileus. Ultrasonographic abdominal LN enlargement was reported in 74% of cats (n = 37). In total, 47 LNs were considered enlarged and the most commonly affected nodes were the mesenteric (n = 32), ileocolic (n = 5), pancreaticoduodenal (n = 4), hepatic (n = 3), medial iliac (n = 2) and gastric (n = 1).
Summarised imaging findings of 51 cats that underwent abdominal lymph node (LN) biopsies

GI = gastrointestinal
Cytology
Nineteen cats (37%) had ultrasound-guided FNAs of LNs: eight were classified as inconclusive, eight were reactive, one was consistent with lymphoma, one revealed a neutrophilic inflammation and the other showed pyogranulomatous inflammation. Of the eight LNs classified as inconclusive on cytology, two (25%) were metastatic on histology. Similarly, two cats with LNs classified as reactive hyperplasia (25%) had intestinal FNAs consistent with high-grade lymphoma and the LNs were found to be metastatic on histology. Lymphoma was confirmed on histology for the cat that had preoperative LN FNAs consistent with a haematopoietic tumour. In total, from five cases with metastatic LNs that had preoperative FNAs, only one was detected with this technique.
Surgical findings
The LNs of 44 cats (86%) were considered to be enlarged at the time of surgery. Four cats (8%) were classified as having normal-sized LNs, whereas in three cases (6%) there was no mention of LN size in the surgical report. In total, 53 LNs were classified as being enlarged. The mesenteric LN was the most commonly affected (n = 40), followed by colonic (n = 5), pancreaticoduodenal (n = 5), medial iliac (n = 2) and hepatic (n = 1). Sixty LN biopsies were performed: mesenteric (n = 45), colonic (n = 7), pancreaticoduodenal (n = 5), medial iliac (n = 2) and hepatic (n = 1).
All cats had abdominal LN biopsies at the same time as other surgical procedures. GI biopsies (n = 25), liver biopsies (n = 17), enterectomies (n = 13) and colectomies (n = 3) were the most common surgeries (Table 2). Other procedures included omentectomy, Billroth type 1 procedure, liver lobectomy, partial gastrectomy, subcutaneous urethral bypass placement, ureteronephrectomy, mesentery and peritoneum biopsies, revision for dehiscence of previous GI biopsy sites, diaphragmatic hernia repair, cholecystectomy and biliary cyst aspiration (all n = 1). One cat with septic peritonitis was euthanased at the time of surgery owing to the severity of intrabdominal findings. LN biopsy was not reported to be directly associated with any intraoperative complications.
Description of surgeries performed at time of abdominal lymph node biopsy in 51 cats

GI = gastrointestinal; CBD = common bile duct; FB = foreign body; SUB = subcutaneous ureteral bypass; ex-lap = exploratory laparotomy
Histology
Sixty LN biopsies were evaluated. Reactive hyperplasia was found in 41 LNs (68%). Evidence of metastatic disease was present in 11 LNs (10 cats): lymphoma (n = 7), mast cell tumour (MCT; n = 3) and jejunal adenocarcinoma (n = 1). Lymphadenitis was found in six cases (pyogranulomatous n = 3, eosinophilic n = 2, necrotising n = 1). The results are summarised in Table 3. There was one LN classified as non-diagnostic and one found to be normal on histology. The LNs of the four cats classified as not enlarged at the time of surgery were found to be reactive on histology.
Distribution of lymph node (LN) histology findings

MCT = mast cell tumour; WNL = within normal limits
The most common final diagnosis was inflammatory bowel disease (IBD; n = 17), followed by alimentary lymphoma (n = 10), cholangiohepatitis (n = 5), feline eosinophilic sclerosing fibroplasia (n = 4), septic peritonitis (n = 4), jejunal ulcers (n = 4) and abdominal MCT (n = 3). Intestinal MCTs were found in two cats and in another case the LN and mesentery were diagnosed with MCT, but a primary tumour was not identified (Table 4). Other diagnoses included (all n = 1) a biliary cyst, a cystic mass on the common bile duct, cholecystitis with cholangiohepatitis, a nephrolith and ureterolith, a colonic carcinoma and a renal carcinoma. One cat was found to have metastatic jejunal adenocarcinoma and small T-cell intestinal lymphoma. The two cats that had generalised abdominal lymphadenomegaly with no other findings on ultrasound had LN FNAs consistent with a reactive process and underwent surgical GI biopsies. Those were consistent with IBD with reactive LN changes.
Distribution of the final diagnosis based on histological reports (number of cats)

IBD = inflammatory bowel disease; GE = gastroenteritis; PARR = PCR for antigen receptor rearrangements; MCT = mast cell tumour; LN = lymph node; CBD = common bile duct
Clinical outcome
Forty-seven cats (92%) survived to discharge: intraoperative euthanasia was performed in one case; two cats were euthanased 1 day postoperatively owing to deterioration of clinical signs (nephrolith and intestinal MCT); one cat with lymphoplasmacytic gastroenteritis died 4 days postoperatively. The duration of hospitalisation ranged from 1 to 14 days (median 3 days). No postoperative complications could be associated with LN biopsy. In total, 15 cats (29%) had neoplastic disease: alimentary lymphoma was identified in 10 cats (seven with LN metastasis), abdominal MCT was found in three cats (LN metastasis in two) and one cat had metastatic jejunal adenocarcinoma and GI lymphoma. There was one cat with renal carcinoma and one with colonic carcinoma (both not metastatic) (Table 5).
Distribution of ultrasound and surgical findings and lymph node (LN) cytology and histology results in cats found to have neoplasia on histology
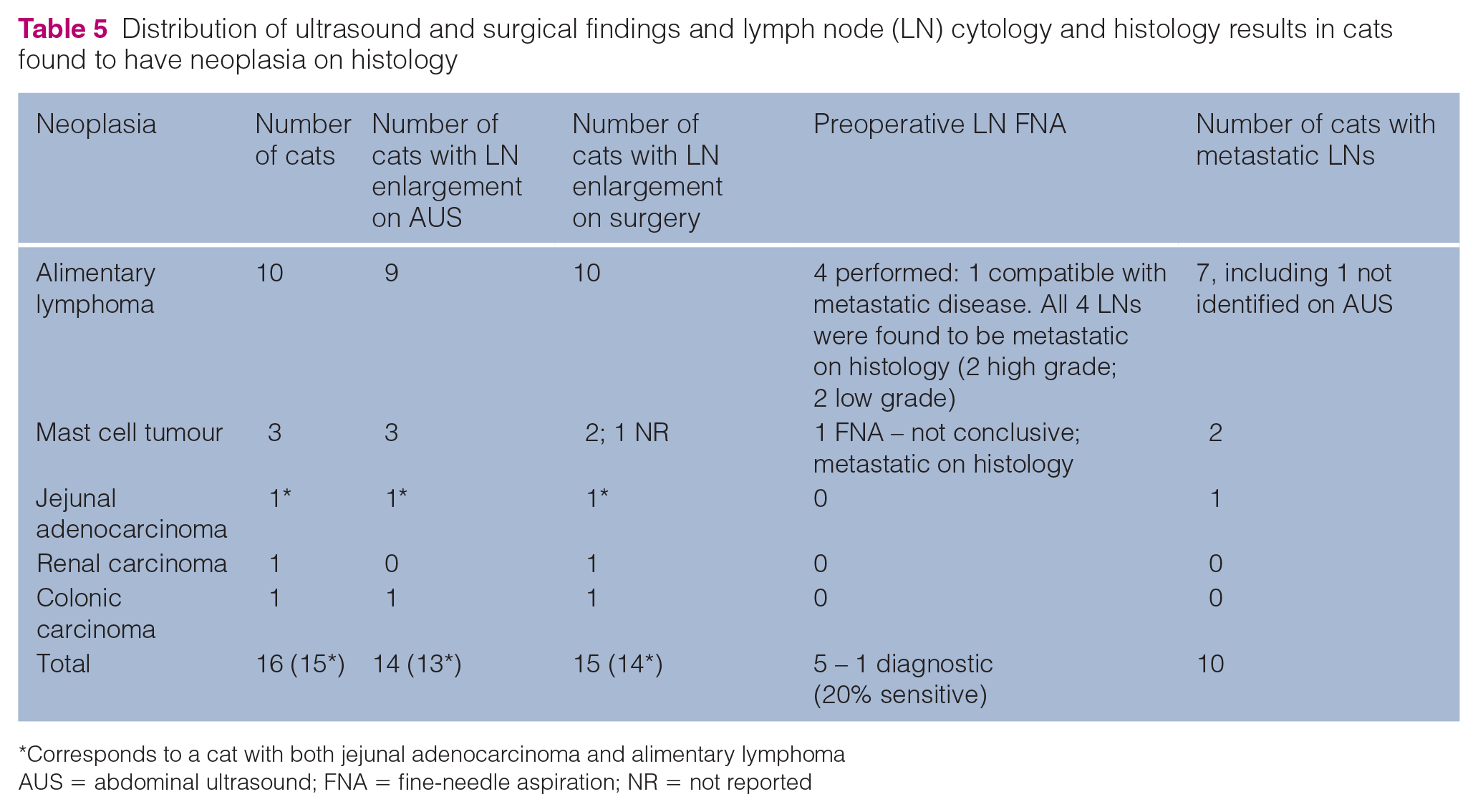
Corresponds to a cat with both jejunal adenocarcinoma and alimentary lymphoma
AUS = abdominal ultrasound; FNA = fine-needle aspiration; NR = not reported
Comparison between investigation, surgery and histology findings
Forty-four cats (86%) had LN enlargement at surgery. Of these, nine were classified as having normal sized LNs on ultrasound (Table 6). Interestingly, in one of those cases the LN histopathology was found to be consistent with metastatic small T-cell alimentary lymphoma on histology.
Comparison between the presence of abdominal lymphadenomegaly on abdominal ultrasound and surgery

Cat did not have preoperative ultrasound
AUS = abdominal ultrasound; LN = lymph node; NA = not available
FNAs were taken from 19 LNs before surgery, including five LNs that were later found to be metastatic on histology. Only one of those cases was correctly identified on cytology (sensitivity 20%). The specificity and accuracy of LN cytology to detect metastatic LN disease in our study were 100% and 79%, respectively.
Discussion
The results of the current study show that surgical abdominal LN biopsy is a useful technique in cats. From 60 LNs assessed, 11 were found to be metastatic (eight GI lymphoma, two MCT, one jejunal adenocarcinoma) and six biopsies allowed exclusion of metastatic LN involvement in the presence of intra-abdominal neoplasia.Surprisingly, in this study, the sensitivity of LN cytology in detecting metastatic disease was only 20% (1/5 cases), which is lower than previous studies, and particularly relevant given the fact that two of the cases were from cats with alimentary high-grade lymphoma.4,7,14 The two other misdiagnosed cases were from a cat with metastatic small T-cell lymphoma and a cat with intestinal MCT. While obtaining cytology samples is simple and non-invasive, results may be unreliable, particularly for low-grade alimentary lymphoma, where the reported rate of false-negative results for mesenteric LNs can reach 50%, which is, in part, due to the difficulty in distinguishing low-grade malignancy from reactive hyperplasia.14,15 The sensitivity and specificity of cytology for diagnosis of metastatic LNs in dogs and cats with solid tumours has been reported to be 100% and 96%, respectively. 4 The lower sensitivity in the present study could potentiality be attributed to the fact that only abdominal LNs of cats were assessed, whose location could have affected the ability to obtain adequate samples. Besides, the low number of cases that had LN cytology (19 in total and only five cases with metastatic disease) could have contributed for a possible underestimation of sensitivity. A study with larger number would be required to clarify this.
In this study, surgical inspection identified 53 enlarged LNs, whereas only 47 were defined as enlarged on ultrasound (44 vs 37 cats). This difference could be explained by the fact that surgical assessment of LN size is a subjective evaluation, especially when considering abdominal LNs located in areas with perinodal fat, whereas specific guidelines for ultrasound classification of abdominal LN enlargement have been proposed in cats.16,17
Several studies have looked at the ultrasonographic appearance of LNs to differentiate between benign and malignant processes with a lack of completely reliable features being found in both humans and animals.7–10,18 Both in dogs and humans, LN shape, echogenicity and hilar flow measured with Doppler have been attributed predictive value. In our study, only LN size was available on the ultrasound report and so, unfortunately, this is likely to have limited further assessment.
Interestingly, even though increased size has constantly been associated with metastatic LNs, one LN classified as normal sized on ultrasound was found to be metastatic for small T-cell alimentary lymphoma on histology.7,14 Moreover, no cats without considered surgical LN enlargement had metastatic LNs, which could suggest that surgical inspection is a valuable technique with acceptable sensitivity, in line with previous reports. 7
Most cats had clinical signs consistent with GI disease and the most common ultrasound finding was the presence of intestinal thickening, especially of the muscularis layer; therefore, surgical GI biopsy was the procedure most performed. IBD and GI lymphoma were the most common diagnoses, which are conditions that cause chronic GI tract disease in cats.19–22 IBD is characterised by diffuse inflammatory cell infiltration of the mucosal layer, commonly presenting as a chronic, lymphoplasmacytic enteritis.19,20,22,23 GI lymphoma is the most common clinical presentation of lymphoma in cats and is the most frequent intestinal neoplasia of this species. 21 Most are histologically classified as low-grade small T-cell lymphoma. 21 Definitive diagnosis of IBD and GI lymphoma requires histologic evaluation of GI biopsy specimens and further testing, such as immunohistochemistry analysis and clonality testing by PCR for antigen receptor rearrangements (PARR), may be necessary.20,23,24 In our study, PARR was performed when the pathologist felt it was necessary. It ruled out lymphoma in four cases and confirmed it in one case.
Whereas most enlarged LNs in cats with neoplastic disease were metastatic (66%), only LN sampling allows accurate differentiation and staging, which can provide useful prognostic information. In high-grade alimentary lymphoma, a small case series has suggested that the absence of LN metastasis has positive prognostic information.14,25 According to this study, the risk of having progressive disease and dying increased 2.4 and two times, respectively, for each increase in clinical stage. 25 Other studies have found no prognostic information on clinical staging of cats with alimentary lymphoma.26,27 For cats with intestinal MCT and intestinal adenocarcinoma, the absence of LN metastasis is associated with a better prognosis.28–30
No intraoperative and postoperative complications associated with LN biopsies were reported in our study. Complication rates associated with surgical procedures are best assessed by prospective evaluation so limited conclusions can be made in this study owing to possible under-reporting of true biopsy-related complications. However, from the surgical reports evaluated, biopsy was not associated with any significant haemorrhage, which may be the primary reason why surgeons avoid LN biopsy. Another limitation of this study being retrospective and dependent on accurate medical reports relates to the method by which LN size was assessed, particularly intraoperatively, which is a subjective estimation and would lead to both under- and overestimation of LN size.
The fact that multiple ultrasound operators were used and that the sampling technique was not standardised is a further limitation of this study. The same applies to interpretation of cytology and histology samples, which were performed by different pathologists and not reviewed. Given the challenges of interpretation of GI biopsies regarding IBD and lymphoma, having used the same pathologist could have allowed a better standardisation, as previously suggested. 31
Conclusions
This study demonstrates that abdominal LN biopsy is a useful and valuable technique in cats that provides complementary information to cytology of LN FNAs.
Footnotes
Acknowledgements
The authors would like to acknowledge Sara Verganti for her contribution as a medical oncologist to the lymphoma section.
Author note
The abstract of this paper has been submitted for presentation at the ECVS 2020 congress.
Conflict of interest
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
This work involved the use of non-experimental animals only (owned or unowned), and followed established internationally recognised high standards (‘best practice’) of individual veterinary clinical patient care. Ethical approval from a committee was therefore not necessarily required
Informed consent
Informed consent (either verbal or written) was obtained from the owner or legal custodian of all animal(s) described in this work for the procedure(s) undertaken. No animals or humans are identifiable within this publication, and therefore additional informed consent for publication was not required.
