Abstract
Objectives
The aim of this retrospective study was to determine the response to a single injection of pegylated asparaginase (‘pegaspargase’) and to assess the tolerability and outcome of prolonged incorporation of pegaspargase into a modified COP (cyclophosphamide, vincristine, prednisolone) regimen in pegaspargase sensitive cats.
Methods
Fifty-six client-owned cats with confirmed macroscopic high-grade lymphoma at any anatomical site were included. Treatment was commenced with a single pegaspargase injection. Cats showing an objective response were eligible to continue therapy with pegaspargase incorporated into a modified COP protocol and had their survival analysed using the Kaplan–Meier method and log-rank test.
Results
Objective response to pegaspargase was reported in 46 cats (82%), including 21 (38%) complete and 25 (44%) partial responses. Thirty-four responders continued therapy with pegaspargase-COP as the first-line treatment. Of these, 31 cats (92%) achieved complete remission with a median duration of the first remission (disease-free survival [DFS]) of 816 days. The median overall survival time (OST) for all 34 cats treated with pegaspargase-COP was 181 days. Response to the initial pegaspargase injection before COP initiation was significantly associated with DFS (P = 0.04) and OST (P = 0.001). Median DFS/OST for cats with complete response to initial pegaspargase injection was significantly longer compared with those with partial remission (>1273 days/>2066 days vs 77 days/108 days, respectively). Cats with gastric lymphoma showed a significantly longer survival (OST 854 days, 1- and 2-year survival rate 57.1%) compared with cats with intestinal lymphoma (OST 102 days, 1-year survival rate 0%). The pegaspargase-COP protocol was generally well tolerated, but two deaths were likely attributable to treatment-related toxicity during the maintenance phase. Importantly, none of the cats experienced hypersensitivity, despite multiple repeated treatments with pegaspargase.
Conclusion and relevance
Pegaspargase is an effective agent for feline lymphoma. Its incorporation into a COP chemotherapy protocol may confer a survival benefit, especially in cats with complete response to pegaspargase. Treatment is generally well tolerated, but careful monitoring is recommended. Further studies are required to assess the benefits of pegaspargase as monotherapy or as part of different multi-agent chemotherapy regimens.
Keywords
Introduction
Lymphoma is the most common haematopoietic neoplasia in cats, representing one-third of all feline tumours.1–8 Intermediate- to high-grade lymphomas have been most frequently identified,8–11 with 27–72% affecting the gastrointestinal tract.1,7,10,12–17
Chemotherapy remains a mainstay of therapy for lymphoma-bearing cats. Conventional treatment includes a backbone of multiagent chemotherapy consisting of cyclophosphamide, vincristine and prednisolone (COP), or cyclophosphamide, doxorubicin, vincristine and prednisolone (CHOP).1–8,10,18,19
The reported efficacy of both treatment protocols seems to be largely comparable, and therefore the addition of doxorubicin does not appear to offer a survival advantage in cats. Overall response rates for the COP-based protocol vary from 39% to 96%, with a median overall survival time (OST) of 45–388 days, whereas for CHOP-based protocols, response rates range from 38% to 95%, with an OST of 97–657 days.1–8,10,18,19
l-Asparaginase (ASP) is a bacterial enzyme, derived from either Escherichia coli or Erwinia chrysanthemi, that depletes serum asparagine. In contrast to most healthy cells, acute lymphoblastic leukaemia (ALL)/lymphoblastic lymphoma (LBL) cells lack or have low levels of the enzyme asparagine synthetase,20–23 and are therefore heavily dependent on exogenous asparagine circulating in the blood.21,23–25 At sufficient activity levels, ASP depletes serum asparagine, eventually resulting in apo-ptosis of the asparagine synthetase-deficient tumour cells. 25 Over the past several decades, ASP-based regimens have become a cornerstone of the successful treatment of ALL and non-Hodgkin/LBL in humans.20–30 Owing to therapeutic limitations and the toxicity spectrum in humans,20,28,31 native ASP is no longer commercially available and has more recently been replaced in many countries by its pegylated version, pegylated l-asparaginase (‘pegaspargase’). 23 Pegaspargase has many advantages, including decreased immunogenicity, resulting in lower rates of hypersensitivity reactions and a decrease in the development of antibodies to the asparaginase product. In addition, it has a considerably longer half-life than the native enzyme, allowing a less frequent administration of pegaspargase.21–24,26,32 In humans, the inclusion of pegaspargase in an intensive multiagent chemotherapy protocol has been proven beneficial, despite manageable toxicities, occurring mainly in adults and less commonly in children.23,24,28,31–33
Several studies have described the tolerability and efficacy of pegaspargase in dogs with non-Hodgkin’s lymphoma, either alone or in combination with standard chemotherapy.34–36 While ASP appears to be safe in cats, its survival benefit is poorly supported for both single- and multiagent chemotherapy protocols for feline lymphoma. However, the safety and efficacy of pegaspargase in cats with lymphoma have not been described.3,5–8,10,17,19,37,38
This retrospective study aimed to determine the response to a single injection of pegaspargase in asparaginase- and chemotherapy-naive lymphoma-bearing cats and to assess the tolerability and outcome of the prolonged incorporation of pegaspargase into a modified COP regimen in pegaspargase-sensitive cats.
Materials and methods
Study population
Medical records of client-owned cats with high-grade lymphoma presented to the Collaborative Veterinary Cancer Centers of the Netherlands and Belgium between January 2000 and January 2015, were retrospectively reviewed. Only cats with cytologically or histologically confirmed high-grade lymphoma affecting any anatomical site were eligible to enter the study. Cats with a prior history of pegaspargase, native ASP or chemotherapy administration were excluded. Previous corticosteroid therapy was only allowed if the treatment started at least 3 weeks before diagnosis and if bulky disease with clinical signs was present at the time of enrolment. Cats with an unknown feline leukaemia virus (FeLV) and/or feline immunodeficiency virus (FIV) status were not excluded. All cats had to commence their treatment with pegaspargase and have their response assessed 1 week post-pegaspargase administration. Cats with a complete or partial response (responders) were eligible to continue intended therapy with prolonged incorporation of pegaspargase into a modified COP protocol.
Data collection
Information obtained from the medical records included patient signalment, FeLV/FIV status (when available), anatomical location(s) of lymphoma, immunophenotype (when available), previous steroid therapy, response to first pegaspargase injection and related adverse events (AEs). For cats treated with pegaspargase-COP, additional information was collected: vincristine and cyclophosphamide dosage, number of administrations, number of pegaspargase injections, duration of therapy, documented toxicity, response to treatment, disease-free survival (DFS), OST, and date and cause of death.
Diagnosis and staging
The diagnosis of lymphoma was based on cytological or histological evaluation of enlarged lymph nodes, extranodal lesions or both. Staging was not consistently performed in all cats. Pretreatment evaluation included haematology and a complete serum biochemistry profile. Thoracic radiographs, abdominal ultrasound and endoscopic examination were performed at the clinician’s discretion. Lymphoma was classified according to the following anatomical categories: gastrointestinal (further subdivided into gastric and intestinal), abdominal extra-alimentary, nasal and other lymphoma. When available, immunophenotype was evaluated by immunohistochemistry or PCR for antigen receptor rearrangement.
Treatment protocol
Treatment was initiated with a pegaspargase injection. Cats with a complete or partial response, evaluated 7 days post-administration, were eligible to pursue therapy with the continued incorporation of pegaspargase into a modified COP protocol (pegaspargase-COP; Table 1). Pegaspargase (Oncaspar, ready to use preparation in 5 ml vials with a strength of 750 IU/ml) was administered intramuscularly at a dose of 40 IU/kg. The 5-week induction phase was followed by a maintenance phase, with the intended treatment duration of 9 months. During the maintenance phase vincristine, cyclophosphamide and pegaspargase were given at 3-week intervals, in separate administrations. Prednisolone was continued daily at a dose of 1 mg/kg until the end of the treatment protocol; thereafter, it was gradually tapered over a 3-week period. Treatment-associated toxicities were graded using Veterinary Cooperative Oncology Group - Common Terminology Criteria for Adverse Events (VCOG-CTCAE) v1.1 39
Pegaspargase-COP regimen for feline high-grade lymphoma

Pegaspargase effect evaluation
Response evaluation to a single pegaspargase injection and pegaspargase-COP
Response to pegaspargase was evaluated 7 days post-injection. Patients that continued treatment with pegaspargase-COP were additionally evaluated for their final response to this protocol. Prior to administration of vincristine and at each appointment during the treatment period, the response was determined by means of physical examination and measurement of palpable lesions or enlarged lymph nodes. When indicated, abdominal ultrasonography, thoracic radiographs and/or rhinoscopy were also performed. Alleviation of clinical signs gave an additional indication of response. All cats were reassessed when disease progression was suspected.
The response was defined as follows: complete remission (CR; resolution of clinical signs and disappearance of all measurable disease); partial remission (PR; at least partial improvement of clinical signs and <100% but >50% reduction in total tumour volume); stable disease (SD; a decrease <50% or <25% increase in tumour burden); and progressive disease (PD; >25% increase in overall measurable disease or development of new lesions).2,3,17
The overall response to the initial pegaspargase injection was defined as the percentage of cats with CR and PR (responders). The remaining cats were classified as non-responders.
Outcome assessment and survival analysis
Only patients treated with pegaspargase-COP were included in the response duration and survival analysis. OST was calculated from the start of therapy until lymphoma-/therapy-related death. DFS was defined as the interval from confirmation of CR to disease progression. Data censoring occurred accordingly in the survival analysis for cats that were lost to follow-up, alive at the end of the study period, died unrelated to therapy or before the occurrence of relapse and in the DFS analysis for cats without evidence of disease progression at the time of the last follow-up or the time of death.
Statistical analysis
The normality of continuous data was assessed by the Kolmogorov–Smirnov test. Normally distributed data were expressed as mean ± SD; otherwise, median (range) was used. Categorical data were expressed as frequencies and percentages. Variables, including age, sex, breed, body weight, anatomical location of lymphoma and pretreatment with steroids, were compared across groups stratified based on response to a single injection of pegaspargase, responders vs non-responders, and separately across cats with complete response, partial response or stable/progressive disease. A one-way ANOVA was used for continuous variables. The χ2/Fisher’s exact test was used, as appropriate, to compare the proportions of categorical variables. The Kaplan–Meier product–limit method was used to estimate median DFS, OST and 1- and 2-year survival rates for the population of cats treated with pegaspargase-COP. The effect of potential prognostic factors (ie, age, sex, breed, body weight, anatomical location and pretreatment with steroids) on outcome measures (DFS and OST) was evaluated with log-rank tests and the Cox proportional hazard model. These tests were also used to assess DFS and OST between the different anatomical locations of lymphoma. Each variable was incorporated in univariate analysis, either as categorical or continuous. Owing to the small number of cats, multivariable analysis was not performed. A P value <0.05 was considered statistically significant; for post-hoc pairwise comparison, a significance threshold (P <0.01) was applied for individual comparisons. Statistical analysis was performed using SPSS 16.0.
Results
Response to a single pegaspargase injection
Fifty-six cats were objectively evaluated for response to a single pegaspargase injection. The patient characteristics are summarised in Table 2.
Summary of patient characteristics and clinical data in all 56 cats

FeLV = feline immunodeficiency virus; FIV = feline leukaemia virus
Overall, 46 cats (82%) responded to a single injection of pegaspargase (responders). Twenty-one cats (38%) achieved CR and 25 cats (44%) PR. The remaining 10 cats (18%) had SD (non-responders; Figure 1).

Responders to pegaspargase and patient selection for therapy with pegaspargase-COP. CR = complete response; PR = partial response; SD = stable disease; COP = cyclophosphamide, vincristine, prednisolone; CP = cyclophosphamide, prednisolone; P = prednisolone
Detailed information on response to pegaspargase including the anatomical location is provided in Table 3.
Distribution of response to pegaspargase in 56 cats with high-grade lymphoma

Data are n (%)
CR = complete response; PR = partial response; SD = stable disease
Statistical significance was not achieved for any of the variables (age, sex, neuter status, breed, body weight, anatomical location of lymphoma or pretreatment with steroids) when comparing responders with non-responders. However, male sex (P = 0.032), anatomical location of the lymphoma (P = 0.013) and pretreatment with steroids (P = 0.013) were found to be positively associated with the degree of response to a single pegaspargase injection when the responses were evaluated separately, as CR, PR or SD. Post-hoc pairwise comparison failed to reveal statistically significant differences in the magnitude of response to a single pegaspargase injection between different anatomical locations of lymphoma. Age, breed, neuter status and body weight were not significantly associated with the degree of response to a single pegaspargase injection.
Pegaspargase was well tolerated in all cats and none developed clinical signs compatible with drug-related toxicity.
Response to pegaspargase-COP
Of the 46 responders to the initial pegaspargase injection, 13 cats with CR and 21 with PR continued further therapy with pegaspargase incorporated into a modified COP protocol (pegaspargase-COP). These 34 cats had their response and outcome analysed. Twelve remaining responders were excluded from the survival analysis after their owners opted for another type of treatment (Figure 1).
Of the 34 cats, 13 cats (38%) had gastrointestinal lymphoma, seven cats (21%) had abdominal extra-alimentary locations involved, 11 cats (32%) had nasal lymphoma and three cats (9%) had other forms of lymphoma.
In total, 31 cats (91%) treated with pegaspargase-COP achieved CR, of which 13 cats had already reached CR and 18 cats PR following the initial pegaspargase dose. Three cats (9%) remained in PR.
The median number of chemotherapy treatments as part of COP was 17 (range 2–28), with a median of nine vincristine (range 1–15) and nine cyclophosphamide (range 1–13) administrations. The median number of pegaspargase injections was 11 (range 1–17). Fourteen of the 34 cats (42%) completed the intended treatment.
Outcome assessment and survival analysis of pegaspargase-COP
All 34 cats treated with pegaspargase-COP were included in the response duration and survival analysis. Overall, the median OST for all cats was 181 days (range 25–3752). The estimated 1-year OST was 42% and the 2-year OST was 39%. The median DFS for the 31 cats that achieved CR was 816 days (range 12–3740), with an estimated 1- and 2-year DFS of 52% (Figure 2).
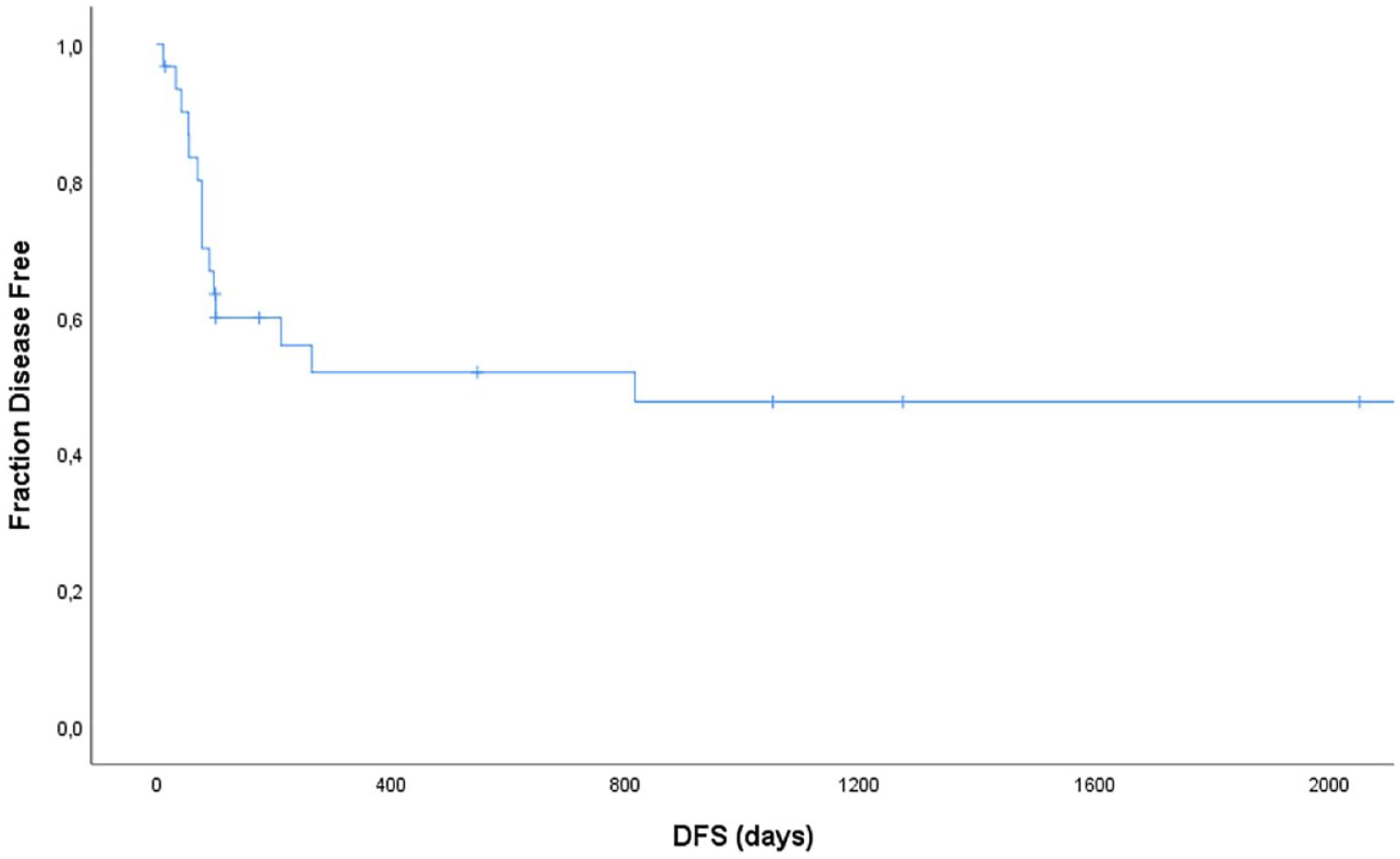
Kaplan–Meier curve depicting disease-free survival (DFS) for 31 cats treated with pegaspargase-COP that went into complete remission. The median DFS was 816 days. The overall estimated 1- and 2-year DFS was 52%. Vertical bars represent censored data
Response (CR vs PR) to the initial pegaspargase injection before COP commencement was significantly associated with OST (P = 0.001) and DFS (P = 0.04).
The median OST of the 13 cats with a complete response to the initial pegaspargase injection was significantly longer compared with 21 cats that showed only a partial response (not reached [>2066 days] vs 108 days), with an estimated 1-year OST of 76.9% and 2-year OST of 76.9% for cats with CR vs 1-year OST of 20% and 2-year OST of 15.0% for cats with PR (P <0.001).
Similarly, 13 cats with a complete response to the initial pegaspargase injection reached significantly longer DFS compared with the 18 cats with a partial response (not reached [>1273 days] vs 77 days), with the estimated 1- and 2-year DFS of 83.9% vs 24.8% (P = 0.001; Figure 3).
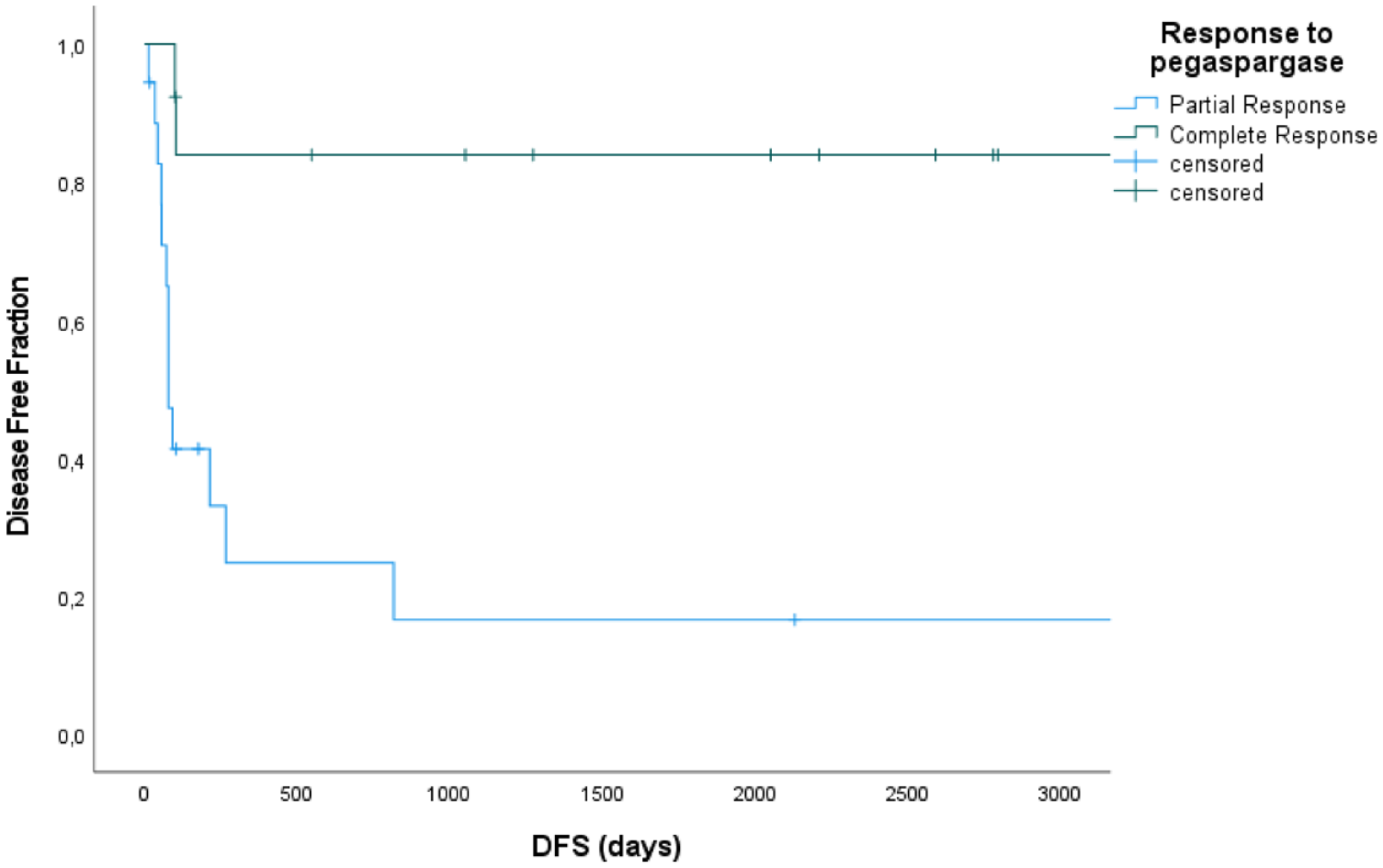
Kaplan–Meier curve depicting disease-free survival (DFS) for 31 cats treated with pegaspargase-COP regimen based on the initial response to pegaspargase. DFS for cats with complete response (CR) was not reached (>1273 days) vs 77 days for cats with a partial response (PR) to the initial pegaspargase. The overall estimated 1- and 2-year DFS was 83.9% for cats with CR vs 24.8% for cats with a PR (P = 0.001). Vertical bars represent censored data
Sex was significantly associated with OST (P = 0.047) but not DFS (P = 0.278). Age, breed, neuter status, weight, anatomical location of lymphoma (Table 4) and pretreatment with corticosteroids had no significant effect on either DFS or OST.
Summary of outcome including disease-free survival (DFS) in 31 cats with complete remission and overall survival time (OST) in all 34 cats with high-grade lymphoma treated with a pegaspargase-COP regimen stratified per different anatomical location

NR = not reached
Additionally, gastric and intestinal lymphomas were analysed separately. In total, seven cats with gastric lymphoma and six with intestinal lymphoma were treated with pegaspargase-COP. Overall, when comparing cats with gastric lymphomas to cats with intestinal lymphomas the median OST was 854 days (range 60–2805) vs 102 days (range 25–194; P = 0.009) and the estimated 1- and 2-year survival rate for cats with gastric lymphoma was 57.1% ± 18.7%, with no cat with intestinal lymphoma being alive at 1 year (Figure 4). The median DFS for gastric lymphoma was 816 days (range 77–2798) vs 70 days (range 33–175) for intestinal lymphoma, but the difference was not statistically significant (P = 0.061).
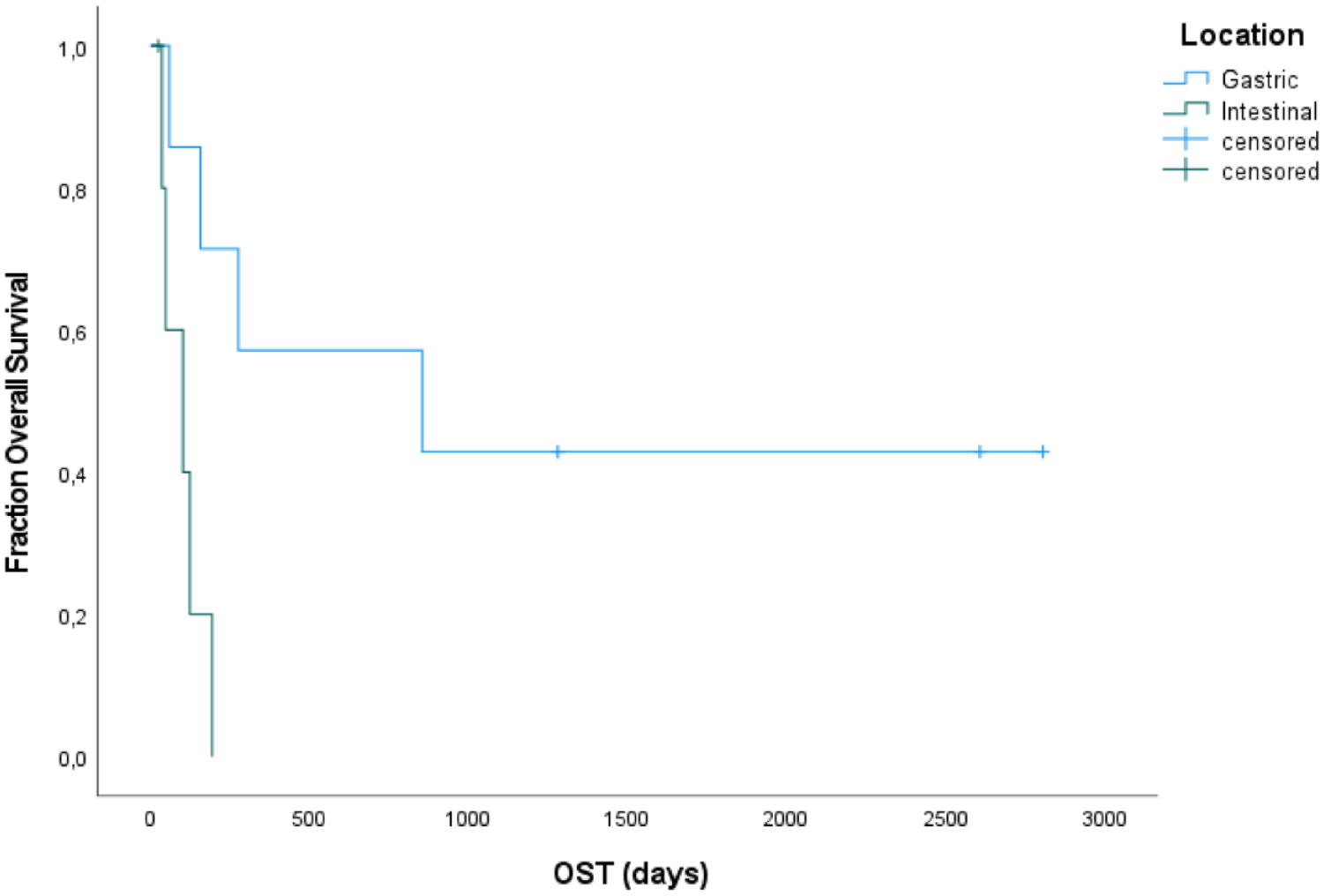
Kaplan–Meier curve depicting overall survival (OST) for seven cats with gastric and six with intestinal lymphoma treated with the pegaspargase-COP regimen. The median OST for cats with gastric lymphoma (854 days) was significantly longer than for cats with intestinal lymphoma (102 days; P = 0.009). Vertical bars represent censored data
At the time of the data analysis 4/34 cats were still alive, one with gastric lymphoma (2607 days) and three with nasal lymphoma (2066, 2787 and 3752 days, respectively). Thirty cats died or were euthanased. Eighteen cats succumbed to lymphoma, two cats died owing to suspected treatment-related complications and 10 cats died from non-tumour- or non-therapy-related causes. In total, 14 cats were censored from the OST analysis, the four living cats and 10 cats that died from another reason than lymphoma or their treatment. No cats were lost to follow up.
Sixteen cats were censored from the DFS analysis. Four of the cats were still alive and had no documented disease progression at the end of the study period. The remaining 12 cats died or were euthanased without evidence of lymphoma progression before death.
Adverse events
Of the cats treated with pegaspargase-COP, 22 (64%) developed AEs, mostly low grade. Four episodes of grade 4 bone marrow toxicity were documented in 2/34 (6%) cats. In total, 6/34 (18%) cats required a dosage reduction and 3/34 (9%) cats required treatment delays. Two cats died due to suspected treatment-related complications, one due to acute pancreatitis and one due to grade 4 neutropenia and acute kidney injury. Both deaths occurred during the less intensive maintenance phase. The most common toxicities experienced during the treatment are listed in Table 5.
Summary of the most common treatment-related toxicities in 34 cats treated with pegaspargase-COP, graded according to VCOG-CTCAE v1.1 39

Discussion
The use of pegaspargase in veterinary medicine remains extremely sparse in contrast to human oncology where for decades pegaspargase has been a key component of successful therapy in patients with ALL and certain non-Hodgkin’s lymphoma.20–29 The objective of the current study was two-fold: first, to determine the response to pegaspargase in lymphoma-bearing cats; and second, to assess the tolerability and outcome of the prolonged incorporation of pegaspargase into a modified COP regimen in pegaspargase sensitive cats.
Our results show that 82% of cats with high-grade lymphoma can achieve an objective response following a single injection of pegaspargase. In the presented population of 56 cats, 38% obtained CR and 44% PR. These results can only be compared to the findings stated by Van Vliet et al, 40 where 12 cats with nasal lymphoma were treated with pegaspargase. From a clinical perspective, all cats attained CR with a median time of 25 days. It is important to bear in mind that in these cats CR was reached often after multiple treatments, in contrast to the single injection used in our study.
Although, while comparing the results, the limitations arising through differences between study populations, inclusion criteria and treatment protocols should be considered.
According to the authors’ experience, a maximum response in cats is seen after a single injection of pegaspargase and additional pegaspargase doses only result in further improvement in a minority of patients. It is well known that nasal lymphomas may respond more favourably to treatment and carry a better prognosis compared with other locations. 2 Similarly, in the present study, the anatomical location appeared to be a positive predictive factor for response to pegaspargase. Although the highest objective response rate (ORR) was seen in cats with extra-alimentary abdominal lymphoma (ORR 100%, CR 20%, PR 80%) followed by the gastrointestinal form (ORR 84%, CR 26%, PR 58%), cats with nasal lymphoma had the highest CR rate (64%).
Pretreatment with steroids appeared to be a positive prognostic factor for response to pegaspargase in our study. This is an interesting observation; however, given that only 10/56 (18%) cats received previous steroids, further investigation in a larger group of patients is warranted to confirm its truly positive effect. Similar to humans,23,26 steroids might decrease the immune response to pegaspargase and the development of neutralising antibodies in cats; however, this has not been studied to date.
It appears striking that pegaspargase seems much more effective than native ASP. LeBlanc et al 38 reported an ORR of 30% in cats with various anatomical forms of lymphoma following a single injection of native ASP.
So far, pharmacokinetic and dose-finding studies on pegaspargase in cats are lacking and the current dosage has been mostly based on empirical data. All cats in the present study were administered a dose of 40 IU/kg, which was analogously calculated using one-tenth of the proportion between native ASP and pegaspargase in human patients. The dose in the present study is similar to those reported in the treatment of canine lymphoma.34–36 Owing to the longer half-life of pegaspargase vs native ASP, fewer injections are required.22,23,25,26 In line with the treatment recommendations in humans, pegaspargase was not administered more often than every 14 days in the present study. To date, the plasma concentration of pegaspargase, necessary to produce a clinical response in cats, remains unknown. An unpublished pilot study in cats, executed in one of the author’s institutions, showed that despite inter-individual differences, complete asparagine depletion can be observed until at least day 21 post-injection, and as long as plasma pegaspargase activity can be measured (de Vos and Krupa, unpublished data). Similar to the authors’ experience in cats, others have found clinical responses even at lower doses of 10–30 IU/kg in canine patients.34–36 However, for optimal efficacy and safety, further pegaspargase pharmacokinetic/pharmacodynamic studies are warranted.
The second objective of the present study was to investigate the tolerability and outcome of the prolonged incorporation of pegaspargase into a modified COP regimen in pegaspargase sensitive cats. Lymphoma in cats is typically treated with chemotherapy, either COP- or CHOP-based protocols, which are generally well tolerated. While the comparison in survival outcomes across different reports is challenging, both seem to have largely comparable results.1–5,8,10,14,37 The COP protocol remains one of the most commonly utilised protocols in daily practice, given its satisfactory efficacy (CR rate 75–79% with a median CR duration of 153–421 days, median OST of 266–388 days) and low toxicity.2,5,18 According to the literature, the addition of native ASP does not seem to improve the remission rate,4,5 although the effect of native ASP was not specifically investigated. 5 In the present study, therapy with pegaspargase-COP resulted in a CR rate of 91%. However, this result must be interpreted with caution, as only cats sensitive to pegaspargase were eligible to continue therapy with pegaspargase-COP. It is worth noting that the median duration of the first remission for cats with CR attained with the presented protocol was 816 days, which compares favourably with the results of the COP or CHOP regimen, in which reported remission duration ranges from 150 to 654 days.1–6,8,10,18 In the present study, response to initial pegaspargase injection was the only variable that significantly influenced both DFS and OST, which were significantly longer in cats with CR compared with cats with PR to pegaspargase (median DFS >1273 days vs 77 days, and median OST >2066 days vs 108 days, respectively). These results support the conclusion that pegaspargase-COP may be an excellent treatment option, especially in cats sensitive to pegaspargase.
In contrast to other studies, in our population of cats treated with pegaspargase-COP, the anatomical location did not appear to be significantly associated with outcome. High-grade gastric and intestinal lymphomas in cats are most often described as one entity.1–8,14,16–19,41 Reported survival data vary widely and specific location has not been shown to be prognostic.4,11,15,37 So far, only one study has described the treatment outcome of feline gastric lymphoma treated with multiagent chemotherapy. Twelve of 16 cats had high-grade lymphoma. Nine cats with CR appeared to have a longer median remission duration than the three cats with PR, which was 189 days vs 125 days, respectively. 42 In addition, the present study analysed gastric and intestinal lymphoma separately. Interestingly, gastric lymphoma appeared to have a significantly better prognosis (DFS 816 days, OST 854 days) compared with the intestinal form (DFS 70 days, OST 102 days). This difference could theoretically result from variations in immunophenotype and pathogenesis of gastric and intestinal lymphoma.15,41,43,44
Up to 25–30% of human patients treated with pegaspargase may experience toxicity, the most notable of these being hypersensitivity reactions. Other toxicities include pancreatitis, thrombosis, liver dysfunction, osteonecrosis and dyslipidaemia. Despite this, concurrent use of pegaspargase and chemotherapy in humans has been proven to be safe and beneficial.23,24,26,28,31–33 In the present study, to avoid overlapping toxicity, treatments were spread over time during the maintenance phase. None of the cats developed hypersensitivity reactions following a single or repeated administration of pegaspargase. This is very similar to results in dogs exposed to pegaspargase.34–36 No specific follow-up blood work or testing for pancreatitis in our study was performed unless there was a clinical indication. During the pegaspargase-COP treatment protocol, one of the cats died as a result of acute pancreatitis. However, given the paucity of data on the toxicity profile of pegaspargase in cats, the cause of the acute pancreatitis remains undetermined.
This study had several limitations, foremost being its small size and retrospective character. Staging was not consistently performed in all cats, and therefore the influence of disease stage on prognosis could not be evaluated. Thirty of 56 cats lacked information on FeLV/FIV status but were included, given the overall low (<0.3% in the Netherlands) incidence of FeLV infection and its decreasing significance in the pathogenesis of feline lymphoma.2,5,8,13,18 Discerning treatment-related toxicities to a specific drug was found to be challenging owing to the multidrug protocol. No standardised criteria for pegaspargase toxicity monitoring and management were followed owing to the absence of specific guidelines. The sustained efficacy of repeated pegaspargase injections during concurrent chemotherapy might have been overestimated given possible inter-individual variations in response to pegaspargase and the risk of ‘silent inactivation’, known in humans.21,25,30 Lastly, although still under debate, the effectiveness of pegaspargase in asparagine depletion in cats could be influenced by diet owing to the high demand for dietary protein, and the fact that a high rate of hepatic protein metabolism affects the plasma concentrations of amino acids.38,45–47 A standardised diet was not prescribed in our study.
Conclusions
Pegaspargase appears to be safe and effective in cats with high-grade lymphoma. Its incorporation into a COP chemotherapy protocol is generally well tolerated and may confer a survival benefit, especially in cats with complete response to pegaspargase. Further modifications and finding the optimal dosing strategy are required to improve the benefits of pegaspargase as monotherapy or as part of different multiagent chemotherapy regimens.
Footnotes
Acknowledgements
This manuscript is dedicated to Johan de Vos, one of the Dutch pioneers in the field of veterinary clinical oncology, and the founder of Collaborative Veterinary Cancer Centers (SDK) of The Netherlands and Belgium. Johan sadly passed away during this research, but his contribution to the advancements in cancer treatment for pets will never be forgotten.
Author note
The results of this study were partially presented at the 2013 Annual Veterinary Cancer Society Conference and the 2014 Annual European Society of Veterinary Oncology Congress.
Conflict of interest
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
The work described in this manuscript involved the use of non-experimental (owned or unowned) animals. Established internationally recognised high standards (‘best practice’) of veterinary clinical care for the individual patient were always followed and/or this work involved the use of cadavers. Ethical approval from a committee was therefore not specifically required for publication in JFMS. Although not required, where ethical approval was still obtained, it is stated in the manuscript.
Informed consent
Informed consent (verbal or written) was obtained from the owner or legal custodian of all animal(s) described in this work (experimental or non-experimental animals, including cadavers) for all procedure(s) undertaken (prospective or retrospective studies). For any animals or people individually identifiable within this publication, informed consent (either verbal or written) for their use in the publication was obtained from the people involved.
